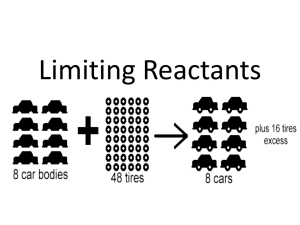
concept presentation
advertisement

HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Presenter: Namrta Bhardwaj Limiting Reactants Background Information Prerequisite knowledge for this section should include nomenclature and balancing chemical equations. It is also essential that students understand mole to mole and mass to mole calculation, which incorporates their knowledge of the relationship between number of particles, moles and mass. It is important that students understand the law of conservation of mass. Students should also be skilled in using ratios and conversion factors in calculations. Curriculum Expectations Overall expectations: D2. Investigate quantitative relationships in chemical reactions, and solve related problems D3. Demonstrate an understanding of the mole concept and its significance to the quantitative analysis of chemical reactions Specific expectations: D2.1 Use appropriate terminology related to quantities in chemical reactions, including, but not limited to: stoichiometry, percentage yield, limiting reagent, mole, and atomic mass [C] D2.5 Calculate the corresponding mass, or quantity in moles or molecules, for any given reactant or product in a balanced chemical equation as well as for any other reactant or product in the chemical reaction [AI] D2.6 Solve problems related to quantities in chemical reactions by performing calculations involving percentage yield and limiting reagents [AI] D3.4 Explain the quantitative relationships expressed in a balanced chemical equation, using appropriate units of measure (e.g., moles, grams, atoms, ions, molecules) Place in the Curriculum Strand B- Matter, Chemical Trends, And Chemical Bonding Strand C- Chemical Reactions Strand D - Quantities in Chemical Reactions Strand E- Solutions and Solubility Strand F- Gases and Atmospheric Chemistry Placement of the Concept Within the Unit Chemical Quantities Counting Atoms and Molecules: The Mole The Avogadro Constant and the Mole Molar Mass Chemical Proportions in Compounds Percentage Composition Empirical Formula of a Compound Molecular Formula of a Compound Quantities in Chemical Reactions Stoichiometry Limiting Reactant Percentage Yield HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Presenter: Namrta Bhardwaj Misconceptions / Learning Difficulties 1) When the mass of both reactants is given students are required to recognize that one of the masses is limiting and the other mass is in excess. Students sometimes randomly select one of the given masses as limiting or excess (or they assume that the masses are in the correct mole ratios). Also, students assume the smaller mass is the limiting reagent. Need to point out these are not trick questions Important to stress the common steps i.e. in converting masses into amounts in moles in order to relate to mole ratios in balanced equations. Use demonstrations such as copper chloride reacting with different amounts of aluminum to illustrate a mass to mole relationship 2) If reactants are in the same physical state the reaction reaches completion when both reactants are totally converted Sodium hydroxide solution and copper sulphate solution are combined in a beaker. Copper hydroxide precipitate is formed. Carry out this reaction in different mole ratios the presence of copper sulphate can be detected by its blue colour 3) If the reactants are in two different physical states (solid and liquid) the reaction reaches completion when the entire solid is consumed. Demonstrate this using different amounts of Mg reacting with HCl. Students will see that as the amount of Mg is increase it no longer reacts since all of the HCl has been consumed. 4) Students have a limited view of the chemical system since they have been taught to consider a reaction system as: reactants -> products Need to rewrite equation so that excess reactants appear on the right hand side of the equation. Reactants -> Products + excess reactant. Gizmo activity can be used to illustrate this. Roadblocks: 1. Too much math 2. Ratios are a difficult concept 3. Each problem requires many steps which can be time consuming Caution: this stuff is difficult to follow at first. Be patient. HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Lesson Sequence Day 1 Teaching Strategies Presenter: Namrta Bhardwaj Learning Styles (MI) Take up homework Review mass to mass calculation Teacher directed – student participate Visual, Auditory (Logical/ Mathematical) Use hot dogs and hot dog bun analogy to introduce concept of limiting reactants Discussion Auditory (Verbal/ Linguistic Logical/ Mathematical) Demo- Na reacting with large amount of water Demo Visual, Auditory (visual/ spatial) Assessment For learning Homework check, assess student’s answers as homework is being taken up Exit pass: For the reaction of copper chloride and aluminum which of the statements is true and explain why: Have students provide alternative analogies Think-pair-share Auditory (Verbal/ Linguistic, Intrapersonal, Interpersonal) Equal masses of reactants will react completely in a chemical equation. Copper chloride and Aluminum lab Demo Visual, Auditory (visual/ spatial, Logical/ Mathematical) Chemicals react based on mole ratios found in a chemical equation. Have students brainstorm practical reasons for having limiting reactants Discussion Auditory (Verbal/ Linguistic) Chemicals react based on mass ratios found in a chemical equation. Have students conduct baking soda and vinegar lab (or Mg and HCl) lab Day 2 Solve sample problems Provide students with a flow chart to follow Guided inquiry Kinesthetic (kinesthetic, visual/spatial) Direct instruction Visual, Auditory (visual/ spatial, logical/ mathematical) Start with balanced chemical reaction that has reactants in a 1:1 mole ratio Progress to more difficult sample problems Equal moles of reactants will react completely in a chemical reaction. For learning 3...2...1 3 things I know about stoichiometry..... 2 things I am getting better at...... 1 thing I still need help with...... HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Presenter: Namrta Bhardwaj Day 3 Extra practice As learning and for learning Cooperative grouping Have groups display answers on white boards – gallery walk Visual, Auditory (visual /spatial, logical/ mathematical) Day 4 Quiz on limiting and excess reactants Assess student’s cooperative skills, participation skills and answers to questions Of learning – mark quiz Percent Yield Calculations Day 5 Direct Instruction Conduct Limiting and excess lab with percent yield calculation Guided inquiry Visual, Auditory (Logical/ Mathematical) Of learning- collect and mark lab Visual, Kinesthetic (visual / spatial, kinesthetic) Supporting Diverse Student Needs Challenge gifted students with questions such as: How many grams of a particular starting material will remain at the end of a reaction? Gifted students may figure stoichiometry out for themselves, given some appropriate starter questions. Encourage them to develop their own methods for solving these problems rather than following a given format. ELL students who have difficulty understanding verbal explanations in class may benefit from writing out the thought process used in solving sample problems. Students that required more support may be giving more scaffolding and support in questions. Have students make common sense prediction when solving problems. HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Presenter: Namrta Bhardwaj Advanced Preparation Day 1 Refer to Interactive Demonstrations for Mole Ratios and Limiting Reagents (annotated reference 1) and prepare material for the lab accordingly. Refer to How Big Is the Balloon? Stoichiometry Using Baking Soda and Vinegar (annotated reference 6) and prepare material for the lab accordingly. Day 5 Refer to pg 165 of the Nelson teacher’s resource for percentage yield in a chemical reaction lab and prepare material for the lab accordingly. Safety Considerations Remember to always wear safety goggles. Reaction of Copper (II) chloride with Aluminum Copper(II) chloride is toxic by ingestion and irritating to skin Aluminum will also react with the solution to produce hydrogen gas. Flames should not be used in this investigation. Reaction of baking soda with vinegar Avoid getting baking soda or vinegar in eyes Reaction of Na with water demo Never use a piece of sodium larger than a ½ pea r. The reaction should be carried out behind a clear safety barrier or at a distance from the students. Applications and Societal Implications Careers in chemistry – Chemical Engineer Cooking recipes, industrial reactions, prescription drug dosages, manufacturing, construction The amount of carbon on earth is the ultimate limiting reagent for the number of people the earth can have and in the sense that carbon is our "food"... the land area of the earth is a "limiting reagent" that will limit our population before carbon does..... L. H. Holmes Jr. HS – Chemistry Instructors: Janine Extavour and Marty Zatzman Presenter: Namrta Bhardwaj Annotated References: Breyfogle, Bryan. (2006) Interactive Demonstrations for Mole Ratios and Limiting Reagents. Journal of Chemical Education, 85 (5), 741. The demonstration of the mole and mass ratios for the reaction of CuCl2 and Al were obtained from this article. Clyde, Dillard R. (1972) Two Lecture Experiments Demonstrating Limiting Quantities. Journal of Chemical Education, 49 (12), A694. Resource used for conducting limiting reactant experiment using Mg and HCl. Gauchon, Laure, et al. (2007) Learning About Stoichiometry: From Students’ Preconceptions to the Concept of Limiting Reactant . Chemistry Education Research and Practice, 8 (4), 362-375. Article discusses many misconceptions that student encounter with studying limiting reactants. Holmes, L. H. Jr. (1998) Limiting Reagent and Kinetics: Social Implications and Malthus' Prediction. Journal of Chemical Education, 75 (8), 1004. Interesting article that discusses food supply and land area in terms of limiting reagents. Jenkins, Frank, et al. (2002). Chemistry 11. Toronto: Nelson. (course textbook) Journal’s Editorial Staff (1997). How Big Is the Balloon? Stoichiometry Using Baking Soda and Vinegar. Journal of Chemical Education, 74 (2), 1328A-1328B. Article used for the Baking soda and vinegar lab. Resource used for conducting limiting reactant experiment using vinegar and baking soda. Kalanter, A.H. (1985) Limiting Reagent Problems Made Simple for Students. Journal of Chemical Education, 62 (2), 106. Article used to for the ICE method for solving limiting reactants. Mustoe, Frank, et al. (2201). Chemistry 11. Toronto: McGraw-Hill Ryerson. (course textbook) Websites: http://www.chalkbored.com/lessons/chemistry-11.htm good power point presentation on limiting/ excess reactants, also the chart method for solving limiting reactant problems was taken from this site http://www.youtube.com/watch?v=xk7PSpFC7NI&feature=related demonstration of the flaming table and whoosh bottle http://www.chemprofessor.com/ flow chart method was obtained from this site