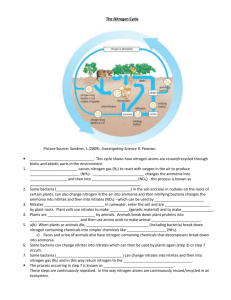
Nitrogen Cycle Model.
advertisement

Name: ____________________________________________ Per. _____ Date: ______________________________ Nitrogen Cycle Model N2 in Atmosphere Lightning Organic N in Plants (proteins) Death Organic N in Animals (proteins) Digestion and Biosynthesis Nitrogen-fixing Bacteria Nitrogen fixation Decomposers Inorganic N in Soil 1. This model illustrates how nature recycles what natural resource? 2. Lightning only removes a very small amount of nitrogen from the atmosphere. What is the main process that converts N2 gas into inorganic nitrogen in the soil? 3. What type of organism is required for the process you named above? 4. Can plants use nitrogen from the atmosphere directly? Explain why or why not, based on what you see in this model. Read This! Nitrogen-fixing bacteria live in the root nodules of legumes. Legumes are a category of plants that produce seed pods, like peas and beans. Nitrogen-fixing bacteria can take nitrogen gas from the atmosphere and convert it into inorganic nitrogen compounds that are available for plants to use. Plants then absorb these inorganic compounds through their roots, and incorporate the nitrogen into organic compounds like proteins and amino acids. 5. How do animals obtain the nitrogen they need? 6. What is the role of decomposers in the nitrogen cycle? Summary and Additional Information – Read This! Nitrogen makes up 78 percent of Earth’s atmosphere. It’s also an important part of living things. Nitrogen is found in proteins, nucleic acids, and chlorophyll. The nitrogen cycle moves nitrogen through the abiotic and biotic parts of ecosystems. The figure below shows how nitrogen cycles through a terrestrial ecosystem. Nitrogen passes through a similar cycle in aquatic ecosystems. Nitrogen Cycle in a Terrestrial Ecosystem. Nitrogen cycles between the atmosphere and living things. Even though nitrogen gas makes up most of Earth's atmosphere, plants cannot use this nitrogen gas to make organic compounds for themselves and other organisms. The two nitrogen atoms in a molecule of nitrogen gas are held together by a very stable triple bond. This bond must be broken for the nitrogen to be used. The nitrogen gas must be changed to a form called nitrates, which plants can absorb through their roots. The process of changing nitrogen gas to nitrates is called nitrogen fixation. It is carried out by nitrogen-fixing bacteria. The bacteria live in soil and roots of legumes, such as peas. When plants and other organisms die, decomposers break down their remains. In the process, they release nitrogen in the form of ammonium ions. This process is called ammonification. Nitrifying bacteria change the ammonium ions into nitrites and nitrates. Some of the nitrates are used by plants. The process of converting ammonium ions to nitrites or nitrates is called nitrification. Still other bacteria, called denitrifying bacteria, convert some of the nitrates in soil back into nitrogen gas in a process called denitrification. The process is the opposite of nitrogen fixation. Denitrification returns nitrogen gas back to the atmosphere, where it can continue the nitrogen cycle.