Justin epigenome
advertisement

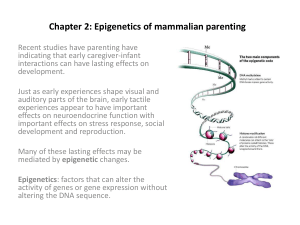
More than Base Pairs: The Impact of Environmental Factors on the Epigenome Justin Young | Fall 2011 Genetics Term Paper A growing body of research increasingly suggests that environmental exposures, stressors and deficiencies during critical periods of an organism’s development play a role in gene expression. Epigenetic modifications provide a plausible link between these environmental factors and alterations of phenotype. In a recent interview Michael Skinner, a prominent epigenticist, explained that a “useful analogy is to think of the epigenome as the software that directs the genomic hardware of a computer. All of an organisms cells contain the same DNA and genes, but it is the epigenome that decides how these genes are expressed and determines how a cell becomes a heart cell, a liver cell or even a hair cell” (Chadra 2011). It also appears that many of these changes can lead to relatively stable, transgenerational shifts in phenotype. Methods are now becoming available to investigate the relevance of these phenomena and their correlation to human disease (Jirtle & Skinner 2007). There are multiple epigenetic processes, but two of the most widely studied types of changes to the epigenome are DNA methylation and histone deacetylation. Both of these processes serve to inhibit gene expression. It is important to highlight one important aspect of methylation which occurs in DNA sequences rich in the CG sequence. These areas are commonly called “CpG islands”; the “p” signifying the phosphodiester bond between the two base pairs. These “islands” are often located around gene promoters and they are often methylated in inactive genes. DNA methylation can affect transcription of genes in two ways. First, the methylation of DNA may physically impede the binding of transcriptional proteins to the gene, and second, methylated DNA may be bound by proteins known as methyl-CpG- binding domain proteins (MBDs). MBD proteins then recruit additional proteins to the locus, such as histone deacetylases that cause changes in chromatin structure. Acetylation of histones causes DNA to “unwind” and genes to be transcribed. Deacetylation has the opposite effect causing histones to be tightly bound thus reducing gene expression. Both of these processes occur naturally during embryonic development and cell differentiation but environmental exposures, nutritional deficiencies and other stressors can alter both DNA methylation and histone acetylation. Recent epigenetic studies have demonstrated the effects of maternal diet on an offspring’s genome. For example, a diet supplemented with methyl donors (B12, choline and betaine) during pregnancy could alter the coat color of the offspring by increasing methylation in key genes (Waterland & Jirtle 2003). Expounding on this study, Dolinoy et al. (2007) examined the effects of dietary genistein (found in soy) on agouti mice. Genistein also had the effect of shifting the phenotype from yellow to pseudoagouti (50% vs 23% in control). Analysis of site-specific methylation at nine individual CpG sites revealed significantly different methylation patterns between the unsupplemented and genistein-supplemented diet groups. More specifically, it showed that a transposable element upstream of the Agouti gene rather than a change in the base pair sequence was responsible for the difference (Dolinoy et al. 2007). Statistical analysis also demonstrated that week-60 mean pseudoagouti body weight is significantly reduced when compared with each of the four other coat-color classes picture below (p = 0.0001) supporting the link between neonatal nutrition, epigenetic pathways and disease. Sometimes epigenetic changes are sex-linked (parent of origin) in a process known as genomic imprinting. This results in mono-allelic gene expression. These changes are established in the germline and carried out through the development of the organism. Genomic imprinting was most convincingly demonstrated in 2005 in an experiment that examined the effects of endocrine disruptor (a substance which interferes with the hormone system) exposure on gestating rats and the effect on fertility of subsequent generations (Dolinoy et al. 2005). Researchers introduced two different chemicals: a fungicide (vinclozolin) and a pesticide (methoxychlor) during embryonic development. Transient exposure to these chemicals led to an abnormally high occurrence of male infertility (through reduced sperm count, impaired sperm motility and apoptosis) in subsequent generations. To demonstrate that male infertility was the result of epigenetic factors two outcross experiments were conducted. The first was between wild type females and males exposed to Vinclozolin, which resulted in male offspring with an increased rate of infertility. The second was between wild type males and females exposed to Vinclozolin – no effect on male fertility was observed in the offspring. These two outcrossing experiments suggest that the phenotype was transmitted through the male germ line and was the result of exposure to the endocrine disruptor. Similar results were found for methoxychlor exposure (Anway et al. 2005). The frequency observed for the phenotype (>90% of all males in all generations) cannot be explained through classical Mendelian gentics. “A high-frequency genetic (DNA sequence) mutation hotspot event would be an order of magnitude less that observed.” (Anway et al. 2005).” The results of PCR-based methyl-sensitive HpaII restriction enzyme digest analysis also verified that there were no changes in in the underlying DNA sequences that would account for this change, but were rather the result of increased levels of methylation (Anway et al. 2005). Even more surprising was the fact that the male phenotype persisted through the 4th generation which suggests the possibility for stable, transgenerational, epigenetic inheritance. Recent research has demonstrated that epigenetic changes can even be induced by emotional stress. In an experiment designed to simulate PSTD in lab rats, researchers demonstrated how predatory exposure increased levels of methylation in the Bdnf IV exon, which is responsible for the production of brain-derived neurotropic factor (Roth et al. 2011) – a protein known to be critical in the formation of new neurons. Prior research had shown that methylation of this gene leads to lower levels of the protein. Based on these observations, Anway et al. restrained the rats and repeatedly exposed them to predators and unstable living conditions (to further replicate PTSD experiences) for one month. Following this period, they examined methylation levels on several exons in the hippocampus, amygdala and prefrontal cortex. The charts below show the differences in hippocampal methylation levels between the control and “PTSD” rats. As predicted, there were decreased levels of Bdnf mRNA in both the dorsal and ventral CA1 in rats exposed to PTSD-like stress. No changes were observed in the amygdala and prefrontal cortex where methylation levels remained unchanged (Anway et al. 2005). This research reinforces the view that DNA methylation remains an active process in the mature CNS that is modifiable by the emotional stress. Though more tenuous, other research has attempted to show the role that epigenetics plays in human development. Similar to the study above, researchers at Mount Sinai School of Medicine and the University of Edinburgh (Yehuda et al. 2005) studied the relationship between mothers who experienced posttraumatic stress syndrome disorder (PTSD) symptoms in response to the 9/11 terrorist attacks and cortisol levels in their infants. Mothers who experienced symptoms of PTSD had lower cortisol levels compared to mothers who did not develop this condition. Lower cortisol levels are common in individuals who suffer from PTSD. Potential confounding factors such as maternal age, ethnicity, body mass index, hours of sleep and wakefulness, and breastfeeding were tested for associations with cortisol. Only mother’s age was correlated with maternal and baby cortisol levels and was used as a covariate (Yehuda et al. 2005).The research revealed that infants of mothers who developed PTSD had significantly lower levels of cortisol even one year after birth, while babies of mothers without PTSD experienced no such decrease. Lower cortisol levels were most apparent in babies born to mothers with PTSD during their third trimesters on September 11, yet PTSD symptom severity in the entire sample was correlated with infant cortisol levels regardless of trimester. These data suggest there maybe a critical, sensitive period in human infant development which has the capacity to significantly alter gene expression years after birth (Yehuda et al.l 2005). A 2005 study in the European Journal of Human Genetics (Pembry et al. 2005) highlighted a statistical link between fathers who smoked during a slow growth period (just prior to puberty) and obesity rates in their sons. This same paper also examined 200 years of meticulously-maintained records from a small Swedish village. The data showed a strong correlation between paternal nutrition in the same slow growth period and (during pregnancy for mothers as egg formation occurs at birth) and future disease susceptibility of same-sex offspring (Pembry et al. 2005). No work has been done to provide molecular explanations for these correlations. Therefore, it is clear that there is a need for more research to understand the delicate interplay between the environment, stress and diet and the role they play in gene expression in humans. Sources Anway, Matthew D, Andrea S. Cupp, Mehmet Uzumcu and Michael K. Skinner (2005) Epigenetic Transgenerational Actions of Endocrine Disruptors and Male Fertility Science , New Series, Vol. 308, No. 5727 pp. 1466-1469 Chadra. Rima (2011) “A Tale of Two Mice.” Nova Science Now. Web www.pbs.org/wgbh/nova/body/epigenetic-mice.html Jirtle, Randy & Michael Skinner (2007). “Environmental Epigenomics and disease Susceptibility.” Nature Reviews Vol 8 Pembrey, Marcus E, Lars Olov Bygren, Gunnar Kaati, Sören Edvinsson, Kate Northstone, Michael Sjöström, Jean Golding, and The ALSPAC Study Team (2005) “Sex-specific, male-line transgenerational responses in humans.” European Journal of Human Genetics p159-166 Roth, T.L., Zoladz, P.R., Sweatt, J.D., & Diamond, D.M "Epigenetic modification of hippocampal BDNF DNA in adult rats in an animal model of post-traumatic stress disorder." Journal of Psychiatric Research, 45, 919-926. Waterland, Robert and Randy L. Jirtle (2003). “Transposable Elements: Targets for Early Nutritional Effects on Epigenetic Gene Regulation Robert” Mol. Cell. Biol. Vol 23 No. 15 Wikipedia: The Free Encyclopedia (2011) “Epigenetics”: Wikimedia Foundation, Inc. Yehuda, Rachel, Stephanie Mulherin Engel, Sarah R. Brand, Jonathan Seckl, Sue M. Marcus and Gertrud S. Berkowitz (2005) “Transgenerational Effects of Posttraumatic Stress Disorder in Babies of Mothers Exposed to the World Trade Center Attacks during Pregnancy.” The Journal of Clinical Endocrinology & Metabolism Vol 90 No 7