BondingWebquestReview.doc
advertisement

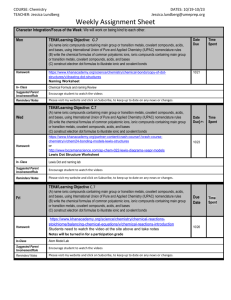
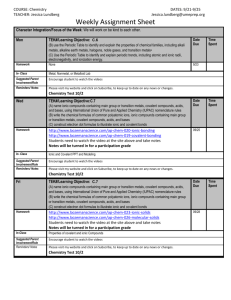
Webquest Review Go to http://sites.google.com/site/ionicandcovalentbonding/ Part One: View the Concepts and Skills page. 1. When looking at an ionic compound formula, what should I be able to do? 2. Lewis dot structures will be needed to answer many test questions. List a few concepts that require a Lewis dot structure. Part Two: View “How can I tell the difference between ionic and covalent compounds?” 3. Summarize the difference between ionic and covalent compound composition. Part Three: Ionic Compounds A: View “Formation of Ions and Ionic Bonds.” 4. Explain how Mg3(PO4)2 is a neutral compound. 5. How and why does aluminum form a cation? B: View “Writing and Naming Ionic Compounds.” 6. Write formulas for the following: a. magnesium nitride b. iron (II) hydroxide c. ammonium sulfide d. calcium chlorate 7. Name the following: a. KNO3 b. CuSO4 c. AgBr d. CoPO4 C: View “Analyzing Ionic Compounds: Higher-level Thinking.” 8. How many carbonate ions are in CaCO3? 9. If GdO exists and Dy(NO3)4 exists, what is the formula for gadolinium nitrate? Part Four: Covalent Compounds A: View “Writing and Naming Covalent Compounds.” 10. Name the following: a. NF3 b. SiO2 c. N2O5 d. C3H8 11. Write formulas for the following: a. dichlorine heptasulfide b. carbon monoselenide c. silicon tetrabromide d. phosphorus trichloride B: View “Drawing Lewis dot structures.” 12. Draw all of the resonance structures for the nitrate ion. 13. How many lone pairs of electrons exist in sulfur dioxide? C: View “VSEPR – Valence Shell Electron Pair Repulsion.” 14. How many areas of high electron density surround the central atom in NH3? 15. What is the molecular shape of SO3 molecule? 16. Why is water a bent molecule? D: View “Electronegativity Difference and Polarity.” 17. Is silicon tetrafluoride a polar molecule? Draw the structure. 18. Is ammonia (NH3) a polar molecule? Draw the structure. 19. Is carbon dioxide a polar molecule? Draw the structure. E: View “Intermolecular Forces.” 20. Explain why magnesium chloride should dissolve in water. 21. Which of the following would be the most difficult to boil: H2O, CH3Cl, CO2 Why? 22. Carbon tetrachloride experiences dispersion forces. Why? F: View “Naming and Writing Formulas for Acids.” 23. Name the following acids: a. H2CO3 b. H2S c. H3PO3 d. H3P 24. Write the formula for the following acids. a. Acetic acid b. Hydrochloric acid c. Nitrous acid d. Nitric acid Part Five: Comments/Suggestions Rate the website according the following areas. 1 = unhelpful; 5 = very helpful Difference between ionic and covalent page Forming ions and ionic bonds page Naming and writing formulas for ionic page Analyzing ionic compounds page 1 1 1 1 2 2 2 2 3 3 3 3 4 4 4 4 5 5 5 5 Writing and naming covalent page Drawing Lewis dot structures page VSEPR page Electronegativity Difference & Polarity page Intermolecular Forces page 1 1 1 1 1 2 2 2 2 2 3 3 3 3 3 4 4 4 4 4 5 5 5 5 5 Naming and Writing Formulas for Acids page 1 2 3 4 5 Comments or suggestions for improvement of website: