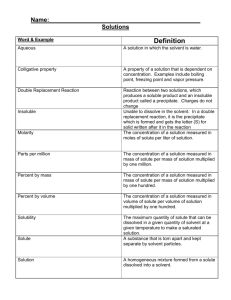
Factors Affecting Solubility
advertisement

Factors Affecting Solubility We have learned that all matter is made up of particles and that they are constantly rotating, vibrating, and moving about from one place to another. (Fluids) In a solution, this means that solute particles are always bumping against other solute particles as well as solvent particles. Rate of Dissolving How quickly a substance dissolves in a solvent is variable The rate of dissolving is affected by __________, ____________ and __________________ Stirring Stirring a solution increases the rate at which a solute dissolves in a solvent Stirring a mixture of water and drink crystals results in a more uniform arrangement of flavor crystals and water particles and makes dissolving occur more quickly Temperature Particles move more rapidly at higher temperatures Since the rate of dissolving depends on solute particles bumping into solvent particles, when the particles move more rapidly, more solvent and solute particles will bump into one another In addition, the solvent particles will more rapidly carry away the solute particles from the edge of the solute Particle Size Large particles take longer to dissolve than smaller particles of the same substance. Particles of a solvent will contact solute particles only at the surface of a clump or crystal of solute particles The more the solute is broken up the greater the surface area available to come into contact with the solvent. Answer questions #6 & #9 on pg. 236 (you will need the graph from #7 on pg. 226)