Answers
advertisement

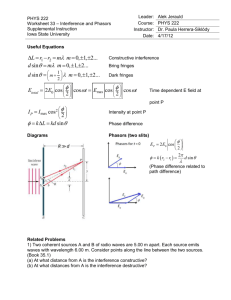
Day 2.1 Wave-Particle Duality (12.2) Watch the Challenge of Quantum Reality from 3:00 to 6:14 http://www.youtube.com/watch?v=wihrAjFXg3o Use the following diagram for the next five questions about double slits. Assume that the slits are narrow and close together. . A) . B) C) DS) 1) A water wave passes through two slits. Which matches the amplitude far from the slits? A double slit interference pattern will form. The last answer is a single slit interference pattern because its central maximum is twice as wide as the others. B is for close to the slits. C 2) Sound is directed toward two slits. Which matches the loudness of the sound far from the slits? Sound consists of longitudinal waves that don’t have crests and troughs. However, its intensity will show a double slit nodal pattern. C 3) A beam of tennis balls are being fired through two slits toward a wall. Sketch what it will look like if the balls leave a mark on the wall. Which pattern above can represent the density of marks? What you see will be a bunch of dots. They will show either of the first two patterns, depending on how far apart the slits are and the angular spread from each slit. 4) A beam of light passes through two slits toward a screen. Sketch what it will look like. Which pattern above can represent the brightness? You will see stripes whose intensity is like C 5) A beam of electrons passes through two slits and then hit a detector. Sketch what the distribution of electrons will look like. Which pattern above can represent this? The electron has a set mass and charge that is always the same and we should expect to see a pattern like the tennis balls and therefore A or B. Many students will already ‘know’ that electrons form an interference pattern and will pick C. However, can they explain this? The video shows clear evidence of an interference pattern. Watch the Challenge of Quantum Reality from 7:50 -13:51 http://www.youtube.com/watch?v=wihrAjFXg3o or Controlled Double-slit electron buildup http://www.youtube.com/watch?v=hv12oB_uyFs The second video is from the same group in Nebraska shown in The Challenge of Quantum Reality but is a clearer version. Details of the experiment can be found in the paper Controlled double-slit electron diffraction by Roger Bach, Damian Pope, Sy-Hwang Liou and Herman Batelaan. http://iopscience.iop.org/1367-2630/15/3/033018/ . 6) The double-slit experiment shows that electrons A) behave like waves and behave like particles. B) behave like waves, but are particles C) behave like particles, but are waves. D) are both waves and particles. This is pretty subtle and very important. The physicists are very careful in what they say. Electrons cannot be classical particles because they form an interference pattern. They can’t be classical waves because they arrive at one spot. A. 7) What is the equation for the wavelength of an electron? Instructions: The students have not learnt about momentum yet, however it is a common term and has a simple equation p = mv. The details of impulse and momentum conservation will be left until later. = h/p, where p = momentum = mass x velocity and h = 6.63 x 10-34 Js 8) It is easier to see an interference pattern if the electrons A) travel slower B) travel faster C) speed has no effect on the pattern It is easier to see interference if the wavelength is large. Therefore the electrons must be moving slowly. 9) The photo below shows the interference pattern produced by an electron double-slit experiment. The electrons were sent through a double-slit apparatus with slit separation of 200 nm. The detector screen was 79.0 cm from the double slits. The image has been magnified by a factor of 100. a) Calculate the wavelength of the electrons. b) Calculate their momentum and velocity a) = x d/L. Watch out for units! Put everything in metres. A nm is 10-9 m. Reduce x by 100 because the image has been magnified by that amount. b) p = h/ and v = p/m (The mass of an electron is 9.11 x 10-31 kg.) This question gives students a break from the strange ideas. It establishes a common theme - that the quantum math works even if the ideas are hard to accept. Watch the Challenge of Quantum Reality from 14:05-15:23 http://www.youtube.com/watch?v=wihrAjFXg3o or Double-slit interference buildup https://www.youtube.com/watch?v=H11hJWIcUY0&feature=youtu.be More details about the photon experiment the video is based on can be found in the paper which is very interesting and surprisingly readable. You can download it at http://arxiv.org/abs/1304.4943 10) Sketch a light interference pattern. Sketch how it will change if the intensity of light is reduced. The interference pattern becomes a few random dots. Light behaves like a particle, called a photon. 11) The video shows light behaving like a particle. What behaviours show that light behaves like a wave and not like a particle? A) refraction, interference, polarization B) reflection, interference, polarization C) reflection, refraction, polarization D) reflection, refraction, interference Reflection can be modelled by both waves and particles if the particles are frictionless and elastic. Watch the Challenge of Quantum Reality from 15:23-16:48 or Single Molecules in a Quantum Movie http://www.youtube.com/watch?v=vCiOMQIRU7I&feature=player_embedded using phthalocyanine - C32 H18 N8. 12) The first experiment used buckminsterfullerene or buckyballs - C60 - moving at 210 m/s. Each molecule has an atomic mass of 720 atomic units and a diameter of 1 nm. The molecules passed through the slits with widths of 50 nm and separations of 100 nm. After the slits, the molecules travelled 1.25 m before being detected. a) What is the mass of one buckyball? (1 amu = 1.660538922 x 10-27 kg.) m = 720 x 1.660538922 x 10-27 kg = 1.20 x 10-24 kg. (Note: There are exactly 720 nucleons but there are not exactly 720 amu because protons and neutrons have slightly different masses. (1.007 vs. 1.008 amu, but 1.001 for each.) The answer should only have three significant digits. b) What is the momentum? p = 1.20 x 10-24 kg x 210 m/s = 2.52 x 10-22 kg m/s c) What is its wavelength? = h/p = 2.63 x 10-12 m d) Draw a scale diagram of the buckyball, its wavelength and the slit widths and separations. The buckyball’s size is 1/50th that of the slit width and 380 bigger than its wavelength. On the diagram below a buckyball would be the size of the period at the end of this sentence. The wavelength can’t be seen at this scale. 13) It is harder to see interference with buckyballs than electrons because buckyballs A) are neutral and harder to accelerate B) are bigger and need bigger slits C) have smaller wavelengths D) have bigger wavelengths As we saw from the question above the buckyballs are much smaller than the slits. The problem is that they have much more mass and therefore a much smaller wavelength. They need to be made to move slowly in order to have a reasonable wavelength. Photons from a laser are use to get them moving by bumping into them. 14) Why have interference effects with tennis balls not been observed? A) The de Broglie wavelength equation, = h/p is only for sub-microscopic objects. B) The experiment has not been done yet. C) The de Broglie wavelength for a tennis ball will be much smaller than for an atom. D) The de Broglie wavelength for a tennis ball will be larger than for an atom. We don’t know if there is a limit for quantum effects or where it would be. It is true that the experiment hasn’t been done, but it hard to imagine how it could be done. A tennis ball is so massive that its wavelength will be really really small even if it is hardly moving. Suppose it has a mass of 0.1 kg and moves at 1 mm a second. The wavelength is 6.63 x 10-30 m which is 1/1000th the spacing between atoms in a crystal. 15) An electron microscope can produce clearer images of significantly smaller objects than a light microscope can because the electrons have a A) larger frequency B) smaller size. C) slower speed. D) shorter wavelength. Electrons have a wavelength but they don’t have a frequency. The sizes of electrons and photons are not known. Electrons do go slower but that doesn’t explain why they can produce clearer images. The smaller the wavelength - the smaller the objects that can be resolved. Resolution is limited by diffraction. Textbook: 12.2 p. 614 # 1, 6, p. 620 # 1, 2, 3 (an eV is a unit of energy)