Quantum Mechanics review WS
advertisement

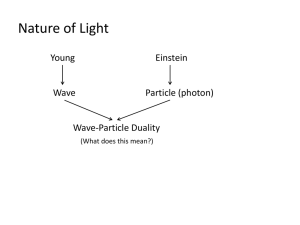
Quantum Mechanics review WS 1. According to the wave theory, the energy content of light corresponds to what characteristic? It corresponds to the INTENSITY of the light, which is related to the AMPLITUDE of the light wave. 2. Define quantum. A discrete bundle of energy 3. Describe the photoelectric effect. The emission of electrons from a metal when light strikes it. 4. Describe the inconsistency that existed between the photoelectric effect phenomenon and the wave theory of light. A cutoff frequency existed for each metal – the minimum frequency of incident light that would cause electron emission. According to the wave theory, the frequency of the light should have nothing to do with this since the energy of the light wave was not connected to the light frequency. 5. Define photon. A quantum of light energy 6. According to the photon theory, the energy content of light corresponds to what characteristic? The energy contained in a photon of light corresponds to the FREQUENCY of the light source. 7. Explain how the photon theory cleared up the inconsistency mentioned in #4. In order for an electron to escape, the photon that it absorbs must contain sufficient energy. The amount of energy in a photon is related to the frequency of the light. Therefore, the light must be at or above the cutoff frequency in order that the photons have enough energy to cause electron emission. 8. Who formulated the photon theory of light? Albert Einstein 9. What did the scientist from #8 say about the true nature of light? He said that light had a dual nature – it was both a wave and a particle (but not at the same time – it was what the conditions of the experiment required it to be). 10. In the Compton Effect, which X-rays had a lower frequency, the ones scattered at a small angle or a large angle from the original motion line? The ones scattered at a large angle have a lower frequency because those photons gave more energy to the crystal particles. 11. Louis DeBroglie created a mathematical link between particles and waves. Write down the equation that illustrates this link. h mv 12. If a stream of particles (like BB’s) is sent toward a barrier with two openings in it, what would the “strike pattern” be on a detector screen placed behind the barrier? Two collision points would be observed – one directly behind each opening. 13. If a light wave is sent toward a barrier with two openings in it (and the distance between the openings is close to the wavelength of the wave), what would the “strike pattern” be on a detector screen placed behind the barrier? An interference pattern would be observed – alternating areas of bright spots and dark patches. A bright spot would exist directly behind the central barrier while dark spots would exist directly behind each opening. 14. When a stream of electrons is sent toward a barrier with two openings in it (and the distance between the openings is close to the wavelength of the electron’s “matter wave”), what would the “strike pattern” be on a detector screen placed behind the barrier? It would be the same pattern as in #13, showing that particles do have a wave nature. 15. What did Max Born think these “matter waves” really were? They were probability functions governing the motions of the particles. 16. What does the wavelength of a particle’s psi wave represent? The MOMENTUM of the particle 17. What does the spatial extent (Δx) of a particle’s psi wave represent? All of the POSSIBLE LOCATIONS of the particle 18. What does the amplitude of a particle’s psi wave represent? The likelihood (probability) of the particle being at that location in space 19. If you know the exact momentum of a particle, what does quantum mechanics theory say about your knowledge of its position? Explain how this can be. You have no idea of its position. The psi wave for the particle would spread out infinitely, giving the particle an equal probability of being at any location. 20. What can you do to a particle’s psi wave in order to better pinpoint the location of a particle? What else occurs when you do this? You can add other wavelengths to it to create a ‘wave packet’. By doing this you cause the momentum of the particle to become more uncertain. 21. What is Werner Heisenberg’s claim to fame? He formulated the Heisenberg Uncertainty Principle, which states that it is impossible to know both the exact momentum and location of a particle simultaneously. The better you know one quantity, the more uncertain you must be of the other. 22. According to quantum mechanics theory, is it possible to track the motion of a particle from start to end? What does the theory say we can know about particle motion? No, that is impossible – the motion of a particle is random. The best we can do is to calculate the probability distribution for the motions of a group of such particles. 23. What is antimatter, how is it created, and what happens when it meets is corresponding matter particle? Antimatter has the same mass but opposite charge of regular matter. An antielectron (or positron) has the same mass as the electron but a charge of +1. Antimatter is created by pair production – the creation of a particle and its corresponding anti-particle during an energy fluctuation. When an anti-particle meets its corresponding particle both are completely destroyed, producing energy (pair annihilation) 24. Describe the phenomena of quantum tunneling and quantum entanglement. QUANTUM TUNNELING can occur when a subatomic particle is “trapped” in a region that is smaller than the Δx of its psi wave. This gives the particle a non-zero probability of existing outside of the trap. At some point in the future, the particle may disappear from inside the trap and reappear outside the trap, as if it tunneled through the barrier. QUANTUM ENTANGLEMENT involves two particles that were created out of a single particle (such as two photons created when one is sent through a beam splitter). These “twin” particles have psi waves that are related to each other. Alterations that are made to the characteristics of one particle can result in the same changes happening to the other particle simultaneously, even if the particles are separated by a large distance.