1 - Rosshall Academy

2.2 Hydrocarbons Learning Outcomes
In all cases pupils should be able to:
1 State that a Homologous Series is a set of compounds with the same general formula and similar chemical properties.
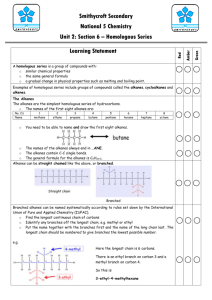
2 State that the alkanes are a subset of the set of hydrocarbons.
3 State that the general formula for the alkanes is C n
H
2n+2
4 State that an alkane can be identified from the “-ane” ending.
5 Name the straight chain alkanes C1 to C8 from molecular formulae, shortened and full structural formulae.
6 Write molecular formulae and draw full and shortened structural formulae given the names of straight-chain alkanes C1 to C8
7 Give the systematic names of branched chain alkanes from shortened and full structural formulae C4 to C8.
8 Write molecular formulae and draw full and shortened structural formulae given the names of branched-chain alkanes C4 to C8
9 State that alkanes are saturated hydrocarbons. They contain carbon to carbon single bonds only.
10 State that alkanes are fairly stable molecules and really only readily undergo combustion reactions.
11 State that Fractional Distillation of crude oil yields more long-chain hydrocarbons than are useful for present-day industrial purposes.
12 State that cracking is an industrial method for producing a mixture of smaller, more useful molecules, some of which are unsaturated.
13 State that the catalyst allows the reaction to take place at a lower temperature.
14 State that cracking can be carried out in the laboratory using an aluminium oxide or silicate catalyst.
15 State that the alkenes are a subset of the set of hydrocarbons.
16 State that the general formula for the alkenes is C n
H
2n
17 State that an alkene can be identified from the carbon to carbon double bond and the “-ene” ending.
18 Name straight chain alkenes, incorporating the position of the double bond, from shortened and full structural formulae C2 to C8.
19 Write molecular formulae and draw full and shortened structural formulae given the names of straight-chain alkenes C2 to C8
20 State that alkenes are unsaturated hydrocarbons. They contain at least one carbon to carbon double bond.
21 State that alkenes readily undergo addition reactions.
22 State that an alkene reacts with hydrogen to form the corresponding alkane.
23 State that an alkene reacts with bromine to produce a dibromoalkane and that this is the chemical test for unsaturation.
24 State that an alkene reacts with water to form the corresponding alkanol.
25 State that the reactions of an alkene with hydrogen, bromine and water are addition reactions.
26 State that the cycloalkanes are a subset of the set of hydrocarbons.
27 State that the general formula for the cycloalkanes is
C n
H
2n
28 Identify a cycloalkane from the name.
29 Name the cycloalkanes C3 to C8 from molecular formulae, shortened and full structural formulae.
30 Write molecular formulae and draw full and shortened structural formulae given the names of cycloalkanes C3 to
C8
31 State that isomers are compounds with the same molecular formula but different structural formula.
32 State that straight chain alkanes C4 to C8 have branched chain isomers
33 State that alkenes C4 to C8 have