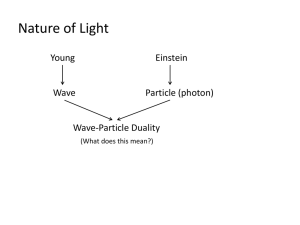
Chapter 41 Problems
electron occupies and (b) the precise energy
of the electron.
1, 2, 3 = straightforward, intermediate,
challenging
Section 41.1 An Interpretation of
Quantum Mechanics
1.
A free electron has a wave function
x Aei 5.00 10 x
10
where x is in meters. Find (a) its de Broglie
wavelength, (b) its momentum, and (c) its
kinetic energy in electron volts.
2.
The wave function for a particle is
x
a
x a2
2
for a > 0 and –∞ < x < +∞. Determine the
probability that the particle is located
somewhere between x = –a and x = +a.
Section 41.2 A Particle in a Box
3.
An electron is confined to a onedimensional region in which its groundstate (n = 1) energy is 2.00 eV. (a) What is
the length of the region? (b) How much
energy is required to promote the electron
to its first excited state?
4.
An electron that has an energy of
approximately 6 eV moves between rigid
walls 1.00 nm apart. Find (a) the quantum
number n for the energy state that the
5.
An electron is contained in a onedimensional box of length 0.100 nm. (a)
Draw an energy-level diagram for the
electron for levels up to n = 4. (b) Find the
wavelengths of all photons that can be
emitted by the electron in making
downward transitions that could
eventually carry it from the n = 4 state to the
n = 1 state.
6.
An alpha particle in a nucleus can be
modeled as a particle moving in a box of
length 1.00 × 10–14 m (the approximate
diameter of a nucleus). Using this model,
estimate the energy and momentum of an
alpha particle in its lowest energy state. (mα
= 4 × 1.66 × 10–27 kg)
7.
A ruby laser emits 694.3-nm light.
Assume light of this wavelength is due to a
transition of an electron in a box from its n
= 2 state to its n = 1 state. Find the length of
the box.
8.
A laser emits light of wavelength λ.
Assume this light is due to a transition of
an electron in a box from its n = 2 state to its
n = 1 state. Find the length of the box.
9.
The nuclear potential energy that
binds protons and neutrons in a nucleus is
often approximated by a square well.
Imagine a proton confined in an infinitely
high square well of length 10.0 fm, a typical
nuclear diameter. Calculate the wavelength
and energy associated with the photon
emitted when the proton moves from the n
= 2 state to the ground state. In what region
of the electromagnetic spectrum does this
wavelength belong?
10.
A proton is confined to move in a
one-dimensional box of length 0.200 nm. (a)
Find the lowest possible energy of the
proton. (b) What If? What is the lowest
possible energy of an electron confined to
the same box? (c) How do you account for
the great difference in your results for (a)
and (b)?
11.
Use the particle-in-a-box model to
calculate the first three energy levels of a
neutron trapped in a nucleus of diameter
20.0 fm. Do the energy-level differences
have a realistic order of magnitude?
12.
A photon with wavelength λ is
absorbed by an electron confined to a box.
As a result, the electron moves from state n
= 1 to n = 4. (a) Find the length of the box.
(b) What is the wavelength of the photon
emitted in the transition of that electron
from the state n = 4 to the state n = 2?
Section 41.3 The Particle Under Boundary
Conditions
Section 41.4 The Schrödinger Equation
13.
Show that the wave function ψ =
i(kx
–
ωt)
Ae
is a solution to the Schrödinger
equation (Eq. 41.13) where k = 2π/λ and U =
0.
14.
The wave function of a particle is
given by
x A coskx B sin kx
where A, B, and k are constants. Show that
ψ is a solution of the Schrödinger equation
(Eq. 41.13), assuming the particle is free (U
= 0), and find the corresponding energy E of
the particle.
15.
Prove that the first term in the
Schrödinger equation, –(ћ2/2m)(d2ψ/dx2),
reduces to the kinetic energy of the particle
multiplied by the wave function (a) for a
freely moving particle, with the wave
function given by Equation 41.3 and (b) for
a particle in a box, with the wave function
given by Equation 41.17.
16.
A particle in an infinitely deep
square well has a wave function given by
2 x
2
2x
sin
L L
for 0 ≤ x ≤ L and zero otherwise. (a)
Determine the expectation value of x. (b)
Determine the probability of finding the
particle near L/2, by calculating the
probability that the particle lies in the range
0.490L ≤ x ≤ 0.510L. (c) What If? Determine
the probability of finding the particle near
L/4, by calculating the probability that the
particle lies in the range 0.240L ≤ x ≤ 0.260L.
(d) Argue that the result of part (a) does not
contradict the results of parts (b) and (c).
17.
The wave function for a particle
confined to moving in a one-dimensional
box is
nx
L
x A sin
Use the normalization condition on ψ to
show that
A
2
L
Suggestion: Because the box length is L, the
wave function is zero for x < 0 and for x > L,
so the normalization condition (Eq. 41.6)
reduces to
18.
L
0
dx 1
2
2x
sin
L L
Calculate the probability of finding the
electron between x = 0 and x = L/4.
19.
An electron in an infinitely deep
square well has a wave function that is
given by
2 x
21.
A particle in an infinite square well
has a wave function that is given by
2
The wave function of an electron is
x
x = 0 and x = L. (a) Find an expression for
the probability, as a function of ℓ, that the
particle will be found between x = 0 and x =
ℓ. (b) Sketch the probability as a function of
ℓ/L. Choose values of ℓ/L ranging from 0 to
1.00 in steps of 0.100. (c) Find the value of ℓ
for which the probability of finding the
particle between x = 0 and x = ℓ is twice the
probability of finding the particle between x
= ℓ and x = L. You can solve the
transcendental equation for ℓ/L
numerically.
2
2x
sin
L L
for 0 ≤ x ≤ L and is zero otherwise. What are
the most probable positions of the electron?
20.
A particle is in the n = 1 state of an
infinite square well with walls at x = 0 and x
= L. Let ℓ be an arbitrary value of x between
1 x
2
x
sin
L L
for 0 ≤ x ≤ L and is zero otherwise. (a)
Determine the probability of finding the
particle between x = 0 and x = L/3. (b) Use
the result of this calculation and symmetry
arguments to find the probability of finding
the particle between x = L/3 and x = 2L/3. Do
not re-evaluate the integral. (c) What If?
Compare the result of part (a) with the
classical probability.
22.
Consider a particle moving in a onedimensional box for which the walls are at
x = –L/2 and x = L/2. (a) Write the wave
functions and probability densities for n = 1,
n = 2, and n = 3. (b) Sketch the wave
functions and probability densities.
(Suggestion: Make an analogy to the case of
a particle in a box for which the walls are at
x = 0 and x = L.)
23.
A particle of mass m moves in a
potential well of length 2L. Its potential
energy is infinite for x < –L and for x > +L.
Inside the region –L < x < L, its potential
energy is given by
U x
2 x2
mL2 L2 x 2
In addition, the particle is in a stationary
state that is described by the wave function
ψ(x) = A(1 – x2/L2) for –L < x < +L, and by
ψ(x) = 0 elsewhere. (a) Determine the
energy of the particle in terms of ħ, m, and
L. (Suggestion: Use the Schrödinger
equation, Eq. 41.13.) (b) Show that A =
(15/16L)1/2. (c) Determine the probability
that the particle is located between x = –L/3
and x = +L/3.
26.
Sketch the wave function ψ(x) and
the probability density x for the n = 4
2
state of a particle in a finite potential well.
(See Fig. 41.8.)
Section 41.6 Tunneling Through a
Potential Energy Barrier
27.
An electron with kinetic energy E =
5.00 eV is incident on a barrier with
thickness L = 0.200 nm and height U = 10.0
eV (Fig. P41.27). What is the probability
that the electron (a) will tunnel through the
barrier? (b) will be reflected?
24.
In a region of space, a particle with
zero total energy has a wave function (a)
Find the potential energy U as a function of
x. (b) Make a sketch of U(x) versus x.
x2
x Axe
L2
Section 41.5 A Particle in a Well of Finite
Height
25.
Suppose a particle is trapped in its
ground state in a box that has infinitely
high walls (Fig. 41.4). Now suppose the lefthand wall is suddenly lowered to a finite
height and width. (a) Qualitatively sketch
the wave function for the particle a short
time later. (b) If the box has a length L, what
is the wavelength of the wave that
penetrates the left-hand wall?
Figure P41.27
28.
An electron having total energy E =
4.50 eV approaches a rectangular energy
barrier with U = 5.00 eV and L = 950 pm as
shown in Figure P41.27. Classically, the
electron cannot pass through the barrier
because E < U. However, quantummechanically the probability of tunneling is
not zero. Calculate this probability, which is
the transmission coefficient.
29.
What If? In Problem 28, by how
much would the width L of the potential
barrier have to be increased for the chance
of an incident 4.50-eV electron tunneling
through the barrier to be one in a million?
30.
An electron has a kinetic energy of
12.0 eV. The electron is incident upon a
rectangular barrier of height 20.0 eV and
thickness 1.00 nm. By what factor would
the electron’s probability of tunneling
through the barrier increase assuming that
the electron absorbs all the energy of a
photon with wavelength 546 nm (green
light)?
Section 41.7 The Scanning Tunneling
Microscope
31.
A scanning tunneling microscope
(STM) can precisely determine the depths
of surface features because the current
through its tip is very sensitive to
differences in the width of the gap between
the tip and the sample surface. Assume that
in this direction the electron wave function
falls off exponentially with a decay length
of 0.100 nm; that is, with C = 10.0/nm.
Determine the ratio of the current when the
STM tip is 0.500 nm above a surface feature
to the current when the tip is 0.515 nm
above the surface.
32.
The design criterion for a typical
scanning tunneling microscope specifies
that it must be able to detect, on the sample
below its tip, surface features that differ in
height by only 0.002 00 nm. What
percentage change in electron transmission
must the electronics of the STM be able to
detect, to achieve this resolution? Assume
that the electron transmission coefficient is
e–2CL with C = 10.0/nm.
Section 41.8 The Simple Harmonic
Oscillator
Note: Problem 43 in Chapter 16 can be
assigned with this section.
33.
Show that Equation 41.24 is a
solution of Equation 41.22 with energy E =
½ ħω.
34.
A one-dimensional harmonic
oscillator wave function is
Axebx
2
(a) Show that ψ satisfies Equation 41.22. (b)
Find b and the total energy E. (c) Is this a
ground state or a first excited state?
35.
A quantum simple harmonic
oscillator consists of an electron bound by a
restoring force proportional to its position
relative to a certain equilibrium point. The
proportionality constant is 8.99 N/m. What
is the longest wavelength of light that can
excite the oscillator?
36.
(a) Normalize the wave function for
the ground state of a simple harmonic
oscillator. That is, apply Equation 41.6 to
Equation 41.24 and find the required value
for the constant B, in terms of m, ω, and
fundamental constants. (b) Determine the
probability of finding the oscillator in a
narrow interval –δ/2 < x < δ/2 around its
equilibrium position.
37.
Two particles with masses m1 and m2
are joined by a light spring of force constant
k. They vibrate along a straight line with
their center of mass fixed. (a) Show that the
total energy
1
1
1
2
2
m1v1 m2 v 2 kx 2
2
2
2
can be written as ½ μv2 + ½ kx2 where
v v1 v2 is the relative speed of the
particles and μ = m1m2/(m1 + m2) is the
reduced mass of the system. This result
demonstrates that the pair of freely
vibrating particles can be precisely modeled
as a single particle vibrating on one end of a
spring that has its other end fixed. (b)
Differentiate the equation
½ μv2 + ½ kx2 = constant
with respect to x. Proceed to show that the
system executes simple harmonic motion.
Find its frequency.
38.
The total energy of a particle–spring
system in which the particle moves with
simple harmonic motion along the x axis is
(b) Show that the minimum energy of the
harmonic oscillator is
Emin K U
Additional Problems
39.
Keeping a constant speed of 0.8 m/s,
a marble rolls back and forth across a
shoebox. Make an order-of-magnitude
estimate of the probability of its escaping
through the wall of the box by quantum
tunneling. State the quantities you take as
data and the values you measure or
estimate for them.
40.
A particle of mass 2.00 × 10–28 kg is
confined to a onedimensional box of length
1.00 × 10–10 m. For n = 1, what are (a) the
particle’s wavelength, (b) its momentum,
and (c) its zero-point energy?
41.
An electron is represented by the
time-independent wave function
x
x Ae x
Ae
2
E
px
kx
2m
2
for x 0
for x 0
2
where px is the momentum of the particle
and k is the spring constant. (a) Using the
uncertainty principle, show that this
expression can also be written
2
1
k
4 m
4
2
p
k 2
E x
2m 8 p x 2
(a) Sketch the wave function as a function
of x. (b) Sketch the probability density
representing the likelihood that the electron
is found between x and x + dx. (c) Argue
that this can be a physically reasonable
wave function. (d) Normalize the wave
function. (e) Determine the probability of
finding the electron somewhere in the
range
x1
1
2
to
x2
1
2
42.
Particles incident from the left are
confronted with a step in potential energy
shown in Figure P41.42. Located at x = 0,
the step has a height U. The particles have
energy E > U. Classically, we would expect
all of the particles to continue on, although
with reduced speed. According to quantum
mechanics, a fraction of the particles are
reflected at the barrier. (a) Prove that the
reflection coefficient R for this case is
R
k1 k 2 2
k1 k 2 2
where k1 = 2π/λ1 and k2 = 2π/λ2 are the wave
numbers for the incident and transmitted
particles. Proceed as follows. Show that the
wave function ψ1 = Ae i k1 x + Be– i k1 x satisfies
the Schrödinger equation in region 1, for x <
0. Here Ae i k1 x represents the incident beam
and Be– i k1 x represents the reflected particles.
Show that ψ2 = Ce i k2 x satisfies the
Schrödinger equation in region 2, for x > 0.
Impose the boundary conditions ψ1 = ψ2
and dψ1 / dx = dψ2 / dx at x = 0, to find the
relationship between B and A. Then
evaluate R = B2/A2. (b) A particle that has
kinetic energy E = 7.00 eV is incident from a
region where the potential energy is zero
onto one in which U = 5.00 eV. Find its
probability of being reflected and its
probability of being transmitted.
Figure P41.42
43.
Particles incident from the left are
confronted with a step in potential energy
shown in Figure P41.42. The step has a
height U, and the particles have energy E =
2U. Classically, all the particles would pass
into the region of higher potential energy at
the right. However, according to quantum
mechanics, a fraction of the particles are
reflected at the barrier. Use the result of
Problem 42 to determine the fraction of the
incident particles that are reflected. (This
situation is analogous to the partial
reflection and transmission of light striking
an interface between two different media.)
44.
An electron is trapped in a quantum
dot. The quantum dot may be modeled as a
one-dimensional, rigid-walled box of length
1.00 nm. (a) Sketch the wave functions and
probability densities for the n = 1 and n = 2
states. (b) For the n = 1 state, calculate the
probability of finding the electron between
x1 = 0.150 nm and x2 = 0.350 nm, where x = 0
is the left side of the box. (c) Repeat part (b)
for the n = 2 state. (d) Calculate the energies
in electron volts of the n = 1 and n = 2 states.
Suggestion: For parts (b) and (c), use
Equation 41.5 and note that
sin
2
ax dx
1
1
x
sin 2ax
2
4a
45.
An atom in an excited state 1.80 eV
above the ground state remains in that
excited state 2.00 μs before moving to the
ground state. Find (a) the frequency and (b)
the wavelength of the emitted photon. (c)
Find the approximate uncertainty in energy
of the photon.
46.
An electron is confined to move in
the xy plane in a rectangle whose
dimensions are Lx and Ly. That is, the
electron is trapped in a two-dimensional
potential well having lengths of Lx and Ly.
In this situation, the allowed energies of the
electron depend on two quantum numbers
nx and ny. The allowed energies are given
by
2
2
ny
h n x
E
2
2
8me Lx
L y
For a particle in a one-dimensional box
extending from x = 0 to x = L, show that
x2
L2
L2
2 2
3 2n
48.
A particle is described by the wave
function
(a) Determine the normalization constant A.
(b) What is the probability that the particle
will be found between x = 0 and x = L/8 if its
position is measured? (Suggestion: Use Eq.
41.5.)
49.
A particle has a wave function
2
(a) Assuming Lx = Ly = L, find the energies of
the lowest four energy levels for the
electron. (b) Construct an energy-level
diagram for the electron, and determine the
energy difference between the second
excited state and the ground state.
47.
For a particle described by a wave
function ψ(x), the expectation value of a
physical quantity f(x) associated with the
particle is defined by
f x * f x dx
(a) Find and sketch the probability density.
(b) Find the probability that the particle will
be at any point where x < 0. (c) Show that ψ
is normalized, and then find the probability
that the particle will be found between x = 0
and x = a.
50.
A particle of mass m is placed in a
one-dimensional box of length L. What If?
Assume the box is so small that the
particle’s motion is relativistic, so that K =
p2/2m is not valid. (a) Derive an expression
for the kinetic energy levels of the particle.
(b) Assume the particle is an electron in a
box of length L = 1.00 × 10–12 m. Find its
lowest possible kinetic energy. By what
percent is the nonrelativistic equation in
error? (Suggestion: See Eq. 39.23.)
51.
Consider a “crystal” consisting of
two nuclei and two electrons as shown in
Figure P41.51. (a) Taking into account all
the pairs of interactions, find the potential
energy of the system as a function of d. (b)
Assuming the electrons to be restricted to a
one-dimensional box of length 3d, find the
minimum kinetic energy of the two
electrons. (c) Find the value of d for which
the total energy is a minimum. (d) Compare
this value of d with the spacing of atoms in
lithium, which has a density of 0.530 g/cm3
and an atomic mass of 7 u. (This type of
calculation can be used to estimate the
density of crystals and certain stars.)
is a solution to the simple harmonic
oscillator problem. (a) Find the energy of
this state. (b) At what position are you least
likely to find the particle? (c) At what
positions are you most likely to find the
particle? (d) Determine the value of B
required to normalize the wave function.
(e) What If? Determine the classical
probability of finding the particle in an
interval of small width δ centered at the
position x = 2(ħ/mω)1/2. (f) What is the actual
probability of finding the particle in this
interval?
53.
Normalization of wave functions. (a)
Find the normalization constant A for a
wave function made up of the two lowest
states of a particle in a box:
x
2x
x Asin 4 sin
L
L
(b) A particle is described in the space –a ≤ x
≤ a by the wave function
x
x
B sin
2a
a
x A cos
Figure P41.51
52.
The simple harmonic oscillator excited.
The wave function
Determine the relationship between the
values of A and B required for
normalization. (Suggestion: Use the identity
sin 2θ = 2 sin θ cos θ.)
54.
The normalized wave functions for
the ground state, ψ0(x), and the first excited
state, ψ1(x), of a quantum harmonic
oscillator are
a
0 x
1/ 4
4a 3
1 x
e a x
2
/2
1/ 4
xe a x
2
/2
where a = mω/ħ. A mixed state, ψ01(x), is
constructed from these states:
01 x
1
2
0 x 1 x
The symbol <q>s denotes the expectation
value of the quantity q for the state ψs(x).
Calculate the following expectation values:
(a) <x>0 (b) <x>1 (c) <x>01.
© Copyright 2004 Thomson. All rights reserved.
55.
A two-slit electron diffraction
experiment is done with slits of unequal
widths. When only slit 1 is open, the
number of electrons reaching the screen per
second is 25.0 times the number of electrons
reaching the screen per second when only
slit 2 is open. When both slits are open, an
interference pattern results in which the
destructive interference is not complete.
Find the ratio of the probability of an
electron arriving at an interference
maximum to the probability of an electron
arriving at an adjacent interference
minimum. (Suggestion: Use the
superposition principle.)