NBOG F 2012-1
advertisement
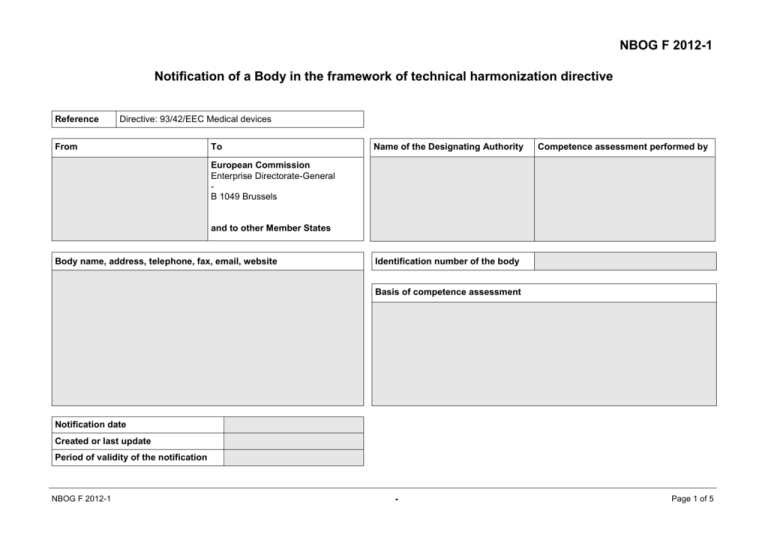
NBOG F 2012-1 Notification of a Body in the framework of technical harmonization directive Reference Directive: 93/42/EEC Medical devices From To Name of the Designating Authority Competence assessment performed by European Commission Enterprise Directorate-General B 1049 Brussels and to other Member States Body name, address, telephone, fax, email, website Identification number of the body Basis of competence assessment Notification date Created or last update Period of validity of the notification NBOG F 2012-1 - Page 1 of 5 1. Medical devices, non-active, 93/42/EEC, competence for the selected product(s) and procedure(s) 1Mark selected products and procedures with a cross (x) in the gray boxes. Annex II: Full quality assurance system; Annex III: EC type-examination; Annex IV: EC verification; Annex V: Production quality assurance; Annex VI: Product quality assurance 2Specify MD 0000 MD 0100 MD 0101 MD 0102 MD 0103 MD 0104 MD 0105 MD 0106 MD 0107 MD 0108 MD 0109 MD 0110 MD 0200 MD 0201 MD 0202 MD 0203 MD 0204 MD 0300 MD 0301 MD 0302 MD 0303 limitations (Annexes and/or products) where applicable Medical Devices, Non-Active General non-active, non-implantable medical devices Non-active devices for anaesthesia, emergency and intensive care Non-active devices for injection, infusion, transfusion and dialysis Non-active orthopaedic and rehabilitation devices Non-active medical devices with measuring function Non-active ophthalmologic devices Non-active instruments Contraceptive medical devices Non-active medical devices for disinfecting, cleaning, rinsing Non-active devices for in vitro fertilisation (IVF) and assisted reproductive technologies (ART) Non-active medical devices for ingestion Non-active implants Non-active cardiovascular implants Non-active orthopaedic implants Non-active functional implants Non-active soft tissue implants Devices for wound care Bandages and wound dressings Suture material and clamps Other medical devices for wound care NBOG F 2012-1 1 2 Annexes Limitations II III IV V VI II III IV V VI II III IV V VI - Page 2 of 5 MD 0000 MD 0400 MD 0401 MD 0402 MD 0403 2. Medical Devices, Non-Active Non-active dental devices and accessories Non-active dental equipment and instruments Dental materials Dental implants 1 Annexes II III 2 Limitations IV V VI Medical devices, active, 93/42/EEC, competence for the selected product(s) and procedure(s) 1Mark selected products and procedures with a cross (x) in the gray boxes. Annex II: Full quality assurance system; Annex III: EC type-examination; Annex IV: EC verification; Annex V: Production quality assurance; Annex VI: Product quality assurance 2Specify MD 1000 MD 1100 MD 1101 MD 1102 MD 1103 MD 1104 MD 1105 MD 1106 MD 1107 MD 1108 MD 1109 MD 1110 MD 1111 MD 1112 limitations (Annexes and/or products) where applicable Medical Devices, Active General active medical devices Devices for extra-corporal circulation, infusion and haemopheresis Respiratory devices, devices including hyperbaric chambers for oxygen therapy, inhalation anaesthesia Devices for stimulation or inhibition Active surgical devices Active ophthalmologic devices Active dental devices Active devices for disinfection and sterilisation Active rehabilitation devices and active prostheses Active devices for patient positioning and transport Active devices for in vitro fertilisation (IVF) and assisted reproductive technologies (ART) Software Medical gas supply systems and parts thereof NBOG F 2012-1 1 Annexes II III 2 Limitations IV V - VI Page 3 of 5 MD 1000 MD 1200 MD 1201 MD 1202 MD 1300 MD 1301 MD 1302 MD 1400 MD 1401 MD 1402 MD 1403 MD 1404 3. Medical Devices, Active Devices for imaging Imaging devices utilising ionizing radiation Imaging devices utilising non-ionizing radiation Monitoring devices Monitoring devices of non-vital physiological parameters Monitoring devices of vital physiological parameters Devices for radiation therapy and thermo therapy Devices utilising ionizing radiation Devices utilising non-ionizing radiation Devices for hyperthermia / hypothermia Devices for (extracorporal) shock-wave therapy (lithotripsy) 1 Annexes II III 2 Limitations IV V VI II III IV V VI II III IV V VI Medical devices, 93/42/EEC, competence for the selected specifics 1Mark selected specifics with a cross (x) in the gray boxes. limitations, when they are applicable. Without any limitation, each specific item is applicable to the chosen scopes under MD 0000 and MD 1000. 2Specify MDS 7000 MDS 7001 MDS 7002 MDS 7003 MDS 7004 MDS 7005 MDS 7006 1 1 Specifics of Medical Devices Medical devices incorporating medicinal substances, according to Directive 2001/83/EC Medical devices utilising tissues of animal origin, including Commission Regulation (EU) No 722/20121 Medical devices incorporating derivates of human blood, according to Directive 2000/70/EC, amended by Directive 2001/104/EC Medical devices referencing the Directive 2006/42/EC on machinery (currently not used) Medical devices in sterile condition Select 2 Limitations Including aseptic processing, ethylene oxide gas sterilisation (EOG), low temperature steam and formaldehyde sterilisation, moist heat sterilisation, radiation sterilisation (gamma, x-ray, electron beam), others (need to be specified) Until 28 August 2013 Directive 2003/32/EC NBOG F 2012-1 - Page 4 of 5 MDS 7000 MDS 7007 MDS 7008 MDS 7009 MDS 7010 1 Specifics of Medical Devices Medical devices utilising micromechanics Medical devices utilising nanomaterials Medical devices utilising biological active coatings and/or materials or being wholly or mainly absorbed Medical devices incorporating software / utilising software / controlled by software NBOG F 2012-1 - Select 2 Limitations Page 5 of 5





