Ambion, Inc.
advertisement

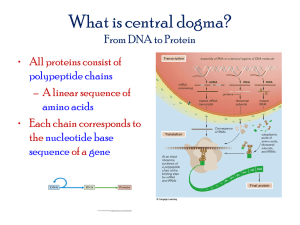
NAT Testing in Solid Tissues and Organs: A Pre-analytical Solution for Nucleic Acid Stabilization Cindy WalkerPeach, Ph.D. cwalkerpeach@asuragen.com Copyright © 2007 Asuragen, Inc Ambion Diagnostics Becomes Asuragen! Ambion, Inc. Formed 1989 Ambion Research Products Division Purchased by Applied Biosystems Group (NYSE: ABI) for $273M March 2, 2006 Copyright © 2007 Asuragen, Inc Ambion Diagnostics Division Ambion Services Division Formed 2000 Formed 2005 Asuragen, Inc. Ambion Diagnostics Ambion Services Key Ambion R&D Scientists and Executive Team RNARetain™ Pre-analytical Solution • Overview • Applications • Examples • Conclusions Copyright © 2007 Asuragen, Inc RNARetain Overview Copyright © 2007 Asuragen, Inc • Versatile, non-toxic, non-organic solution for preservation of nucleic acids by inactivating nucleases to prevent degradation of RNA/DNA • Stabilizes nucleic acids at room temperature, without requirement of dry ice or liquid nitrogen, thereby allowing biological samples to be shipped on wet ice or at ambient temperatures • Compatible with all downstream RNA extraction methods • Based on formulation widely used in life science research (RNAlater - Ambion, Inc.) • Covered by US patent: 6,204,375 and WW equivalents RNARetain - cGMP Traceability • Manufactured under QSR 21 CFR Part 820 guidelines to ensure lot-to-lot consistency critical for reagents used in clinical specimen collection and storage for downstream RNA extraction and molecular analysis • Compatibility with major extraction procedures and molecular analysis techniques will allow end-user to qualify reagent for use in specific downstream applications • cGMP traceability provides consistent performance in enduser’s hands Copyright © 2007 Asuragen, Inc Selected Publications • Development and validation of a method for using breast core needle biopsies for gene expression microrarray analyses Matthew Ellis, et al. Clin Cancer Res. 2002, 8:1155-1166 • The effect of sample degradation and RNA stabilization on classical swine fever virus RT-PCR and ELISA methods Stuart D. Blacksell, Syseng Khounsy and Harvey A. Westbury. J. Vir Methods. 2004, 118(1):33-37 • Viral infectivity is maintained by an RNA protection buffer C. Uhlenhaut and M. Kracht. J. Virol Methods. 2005, 128(1-2):189-91 • Prognostic gene expression signatures can be measured in tissues collected in RNAlater preservative Dondapati Chowdary, et al. JMD. 2006, Vol. 8, No. 1 • Currently there are over 1,000 citations since Jan 2001 in HighWire Press: http://Highwire.Stanford.edu Copyright © 2007 Asuragen, Inc Unmet Clinical Need………… Easy and Safe Sample Acquisition Long-Term Cell/ Tissue Banking “Freezes” mRNA and microRNA Expression Profiles Clinical Molecular Isolation (RNA/DNA) Virus Preservation Convenient Sample Transport Clinical Research, Tissue/Cell Banking, Expression Profiling Services, Molecular Diagnostics Copyright © 2007 Asuragen, Inc Intracellular RNA Preservation mirVana Sample Trizol 5E+5 MCF-7 Breast Tissue Cells 2.02 1.75 10.00 Flash Frozen 2.01 1.20 9.65 RNARetain 1.72 1.30 9.15 Flash Frozen 1.87 1.45 8.90 Nanodrop® Agilent Bioanalyzer Copyright © 2007 Asuragen, Inc mirVana RNARetain Trizol mirVana™ or Trizol® Extraction x4 RIN RNARetain Flash Freeze Flash Freeze Or RNARetain 260/280 28S:18S RNARetain preserves the quality of intracellular RNA to the same extent as the currently established standard of flash-freezing qRT-PCR Reproducibility x5 40 1E+3 HCV1b ARQ Process Control 45 43 35 41 TBP Median Ct 39 mirVana Extraction 30 37 35 25 33 31 20 HCV1b Median Ct 5E+5 MCF-7 Cells In RNARetain Sample # 260/280 28S:18S RIN 1.96 1.73 9.87 1 1.99 1.83 9.97 2 2.09 1.77 9.90 3 1.98 1.63 9.90 4 1.97 1.77 9.93 5 29 27 HCV1b (process con) and TBP (internal con) qRT-PCR at 40ng sample input Copyright © 2007 Asuragen, Inc 15 25 1 x3 2 3 4 5 Sample Number Reproducible RNA quality and qRT-PCR amplification efficiency is achieved for intracellular RNA extracted from RNARetain preserved cells TBP ARQ HCV1b Armored RNA® Recovery from RNARetain 1E+7 HCV1b ARQ + or - RNARetain 40 Median Ct 35 RNA Isolation (Trizol or mirVana Kit) x3 30 25 20 15 + - mirVana HCV1b qRT-PCR x3 Process Rep 1 + - RNARetain Trizol Process Rep 2 Process Rep 3 Pseudo-viral particles can be preserved in and extracted from RNARetain in a reproducible manner Copyright © 2007 Asuragen, Inc RNARetain Data Summary • Preserves quality of intracellular RNA to same extent as flash-freezing • Does not interfere with reproducibility of intracellular RNA extraction and analysis methods • Effective for storage and preservation of viral RNA as indicated by compatibility with Armored RNA Copyright © 2007 Asuragen, Inc Case Study: C p=1.77E-13 14 12 10 miRNAs 6 4 2 0 Normal Chronic Cancer Cell Line 196Ct - 217Ct 8 10 8 6 4 2 0 -2 -4 -6 -8 N N Ca Ch Ca Ch n=7 n=10 n=7 Combination of two microRNAs can be used to distinguish normal, chronic pancreatitis, and carcinoma samples Copyright © 2007 Asuragen, Inc Analysis of FNA Specimens Signal ratios for two miRNAs showing differential expression specific to cancerous (ca) or normal (N) tissue are maintained in RNARetain (FNA samples) Endoscopic microdissection of pancreatic mass (EU-FNA) 6 RNA Isolation 2 196aCt- 217Ct Preservation in RNARetain 196aCt- 217Ct 12 -2 -6 Copyright © 2007 Asuragen, Inc 4 0 -10 -4 -14 -8 Ca FNA n=7 microRNA expression profiling (qRT-PCR) 8 Ca N Frozen n=2 n=2 RNARetain preserves miRNA signatures for downstream molecular profiling Ca N Frozen n=10 n=7 Frozen tissues from Szafranska et al. Closing Remarks • RNARetain is a pre-analytical solution which stabilizes intracellular RNA in cells and solid tissues for downstream extraction and molecular characterization • RNARetain is compatible with Armored RNA, illustrating potential utility in collection and storage of viral-infected tissues, thus eliminating the need for immediate processing or flash freezing • Next step will be to identify beta test sites to evaluate clinical utility of RNARetain for collection and transport of viral-infected tissue specimens • Agendia, B.V. Used in conjunction with MammaPrint® 70 gene expression signature which predicts the risk of breast cancer recurrence Received US FDA approval Feb 6, 2007 Copyright © 2007 Asuragen, Inc