20140817022329567
advertisement

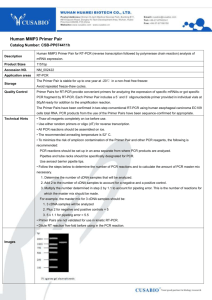
实验13 cDNA的合成及RT-PCR法研究基因的表达 RT-PCR (reverse transcription-polymerase chain reaction) is the most sensitive technique for mRNA detection and quantitation currently available. Compared to the two other commonly used techniques for quantifying mRNA levels, Northern blot analysis and RNase protection assay, RT-PCR can be used to quantify mRNA levels from much smaller samples. In fact, this technique is sensitive enough to enable quantitation of RNA from a single cell. Two-step procedures In the first tube, first-strand cDNA synthesis is performed under optimal conditions, using either random hexamers, oligo(dT) primers (generating a cDNA pool), or sequence-specific primers. An aliquot of the RT reaction is then transferred to another tube (containing thermostable DNA polymerase, DNA polymerase buffer, and PCR primers) for PCR. Advantages of this approach: This method is useful for experiments where multiple transcripts have to be analyzed from the same RT reaction or for specific applications such as Differential Display Reverse Transcription (DDRT) or Rapid Amplification of cDNA Ends (RACE). Also, since the RT reaction is performed under optimal conditions, this approach produces the longest RT-PCR products (up to 14 kb in length, if the appropriate enzymes are used). A two-step procedure has the following advantages Optimizes reaction conditions. The two-step format allows both reverse transcription and PCR to be performed under optimal conditions to ensure efficient and accurate amplification. Provides flexibility. Two-step procedures allow the product of a single cDNA synthesis reaction to be used for analysis of multiple transcripts. This flexibility is valuable for such specialized applications as rapid amplification of cDNA ends (RACE) and Differential Display Reverse Transcription (DDRT). Amplifies long sequences. With the right combination of reverse transcriptase and thermostable DNA polymerase, two-step RT-PCR can amplify RNA sequences up to 14 kb long. RT-PCR常见问题分析及其解决方案 二. RQ1 RNase-Free DNase 消化总RNA 去除DNA污染 1.建立如下反应体系: RNA in water or TE buffer 40ul 10×reaction buffer 10ul RQ1 RNase-Free DNase (1U/ug/RNA) 40ul DEPC处理水 10 ul 2. 37℃ 30min 3.加等体积水饱和酚:氯仿,12400rpm,15min 4.取上清到一新eppendorf tube中,加2.5倍体积的无水乙 醇,12400rpm,15min. 5.70%乙醇洗涤一次,加50ul DEPC处理水溶解,-20℃保存. 三. 快速mRNA纯化 一. 快速 mRNA纯化试剂盒说明(深圳依诺金生物科技有限公司) 60min可充分回收mRNA,回收率达到90%; 对各种剂量的起始总RNA均能灵活适应(50-500ug); 所得mRNA可用于所有下游实验: RT-PCR,cDNA合成,文库构建 消减杂交 体外翻译 差异显示 SAGE 技术 Northern, dot,slot blotting等 二. 注意事项: 试剂盒中的各种试剂和耗才都经过特殊处理,已灭活 RNAse,取用时应小心操作,避免污染! 操作过程中,必须戴一次性手套; mRNA吸附剂中含有0.1%的叠氮钠(NaN3).应戴手套小心 操作. 三. 分离纯化方法 一) 说明 1.总RNA样品质量直接影响到mRNA的分离效果和回收率,因此 在分离操作前需先进行变性agarose electrophoresis以确定 RNA的质量,如果质量合格(28S 和18S rRNA条带清晰,且亮度比 为1.5:1-2.5-:1)则可用于mRNA分离,否则建议不用. 2.建议用本试剂盒提供的RNA溶解液(预先37℃,10-20min,摇 匀),来溶解总RNA样品. 3.除特别说明外,操作在室温下进行. 二)操作前准备工作 1.使用前,须预处理mRNA吸附剂,去除NaN3. A.充分悬浮mRNA吸附剂,用吸头取出50 ul到1.5ml eppendorf tube 中,12000 rpm,1min,吸去上清; B. 加入200 ul平衡缓冲液,悬浮混匀,12000rpm,1min, 去上清,加入80ul平衡缓冲液,悬浮混匀. 2.RNA溶解液,吸附缓冲液,平衡缓冲液,洗涤缓冲 液,使用前在37℃水浴预热20 - 30min,摇动,放室温备用. 三)操作步骤 1. 将50-500ug总RNA用RNA溶解液调整到250-300ul,在65℃水 浴加热15min,然后立即置于冰中冷却至少5min. 2. 加入等体积的吸附缓冲液,混匀后转移到预处理好的含mRNA 吸附剂的管中. 3. 将mRNA吸附管平放在往复式摇床上,于80-100rpm震摇30min, 然后12000rpm,1min,吸去上清液(保留此上清液至获得mRNA为 止). 4. 加入250ul平衡缓冲液,悬浮混匀,用宽口吸头将悬浮液小心 转移至离心柱中,12000rpm,1min,去除收集液,重复洗涤1次. 5. 再加入250ul洗涤buffer悬浮混匀,12000rpm,1min,去除收集 液,重复洗涤共三次. 6.离心柱在12000rpm,1min,彻底去除柱中的洗涤buffer. 7.将离心柱转移到一新的eppendorf tube中,加入30-60ul 65 ℃ 预热的洗脱buffer,12000 rpm 1min洗脱mRNA. 8.收集洗脱液,-70℃保存备用.(为提高mRNA得率,可进行2次洗 脱) Reverse Transcriptase M-MLV (RNase H¯) TaKaRa Code:D2639A 1st-Strand cDNA合成的实验操作方法 1. Microtube管中配制下列模板RNA/引物混合液,全量 6 μl。 3ul 2ul * Total RNA的使用量一般为1 ng~1 μg;mRNA的使用量一般 为10 pg~1 μg。 2. 70℃保温10分钟后迅速在冰上急冷2分钟以上。 3. 离心数秒钟使模板RNA/引物的变性溶液聚集于Microtube管底 部。 4. 在上述Microtube管中配制下列反转录反应液。 0 ul 0.25ul 1.25ul * 当起始模板RNA量>500 ng时,RTase M-MLV(RNase Hˉ)的使用量应> 0.25 μl。 5. 42℃保温1小时*。 6. 70℃保温15分钟后冰上冷却,得到的cDNA溶液可直接用于2nd-Strand cDNA的合成或者PCR扩增等,PCR扩增时cDNA溶液的使用量建议使用1 μl~5 μl。 6) PCR check actin (0.6 kb)) dd water 10.625 ul 10×Taq buffer 1.5 ul dNTP mixture(2.5mM) 1.2 ul ACTINP1 (20uM) 0.3 ul ACTINP2 (20uM) 0.3 ul rTaq (5U/ul) 0.075 ul Template 1 ul (total volume: 15 ul) PCR program: 94℃ 5 min; 94℃ 1 min, 60℃,1 min, 72℃ 1 min, 35 cycles; 72℃ 7min; 12℃,30 min. 1 % agarose electrophoresis and photograph. 四.First-strand synthesis of cDNA under the instruction of M-MLV reverse transcriptase (promega). The following procedure uses 2 ug of RNA. 1) In a sterile RNase-free eppendorf tube, add 5 ul DEPctreated water, add 5 ul mRNA, add 1 ul (0.5 ug of) 15T (in a total volume < 15 ul ); 2) Heat the tube in 70℃ for 5 min to melt secondary structure within the templates; 3) cool the tube immediately on ice to prevent secondary structure from reforming; 4) then spin briefly to collect the solution at the bottom of the tube. 5) add the following components to the annealed primer/template in the order shown: Note: do not alter the ration of primer to mRNA M-MLV 5* reaction buffer 5 ul dATP, 10 mM 1.25 ul dTTP ,10mM 1.25 ul dCTP, 10mM 1.25 ul dGTP ,10mM 1.25 ul RNasin 1 ul M-MLV 1 ul Nuclease-free water 2 ul the final volume 25 ul mix gently by flicking(轻弹) the tube and incubate for 60 min at 42℃. 6) PCR check actin (0.6 kb)) dd water 13.4 ul 10*Taq buffer 2 ul BSA 0.8 ul dNTP mixture(2.5mM) 1 ul UBI-L (50uM) 0.25 ul UBI-R (50uM) 0.25 ul Taq (5U/ul) 0.2 ul Template 2 ul (total volume: 20 ul) PCR program: 94℃ 5 min; 94℃ 1 min, 60℃,1 min, 72℃ 1 min, 35 cycles; 72℃ 7min; 12℃,30 min. 0.9 % agarose electrophoresis and photograph.