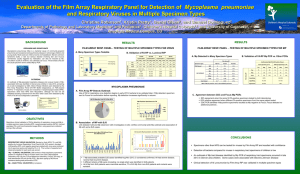
******** Biochemistry and Molecular biology
advertisement

Molecular Biology C Conception, theory, research, and application ——Logic and LIY (Learn It Yourself) Sheng Zhao (赵晟), Biochemistry and Molecular Department of Medical school in Southeast University Couse QQ Club: 112342994 (分子生物学C ) Web: http://teaching.ewindup.info/ Email: shengzhao@seu.edu.cn or windupzs@gmail.com QQ /MSN/Skype/gChat: windupzs@gmail.com Mobile:18551669724 or 13675130010 Chapter 4: Finding Scientific Nemo (From clinic samples to scientific research) Section 1: Trust it or trash it? ——Number, selection, and target (Establish sample library) Section 2: A colorful microscopic world ——Specific labeling (Visualization techniques) Case 4: Immortal clinic samples ——Primary culture and gene manipulation (Construction of the cell line) Sample library Setup your library Define your goal Question based Type of study Time frame Involved techniques Planning for specimen collection Specimen collection and processing Storage, packaging, and transport of specimens Performing experiments, analysis, and publication Request more specimen Planning for specimen collection Always plan in advance 1. Who 2. Where 3. When 4. What 5. How Define the possible questions you need to answer which clinical specimens are required to answer these questions Plan for the possible scale of the study Decide how to collect, process and transport the specimens Define the procedures necessary for specimen management Biosafety issues Specimen collection and processing Safety and decontamination procedures Basic safety precautions Gloves, clothing, mask, etc. Handle needles 1% household bleach for soak, 10% for clean up spills Awareness Labelling and identification of specimens Preprinted labels (at least five) should be used whenever possible. patient name unique identification number specimen type and date and place of collection name or initials of specimen collector. Label accompanying forms Aliquots! Storage, packaging, and transport Storage Aliquots!! Freezer (e.g. -80C) Liquid nitrogen Packaging Blue ice Dry ice Transport Multicenter clinical study Notify the receiving laboratory Car, High speed rail way, Flight, etc. Examples 1. Collection of Respiratory Tract Specimens • Upper Respiratory tract • Optimal timing are usually earlier • Swabs, Nasopharyngeal aspirates, Nasal washings, Oral cultures, etc. • Lower Respiratory Tract Specimens • Less time sensitive • Tracheal aspirate, sputum, pleural fluid, Open lung biopsy, etc. Examples 2. Blood and tissue specimens • Collection of Blood Components • Acute and convalescent serum specimens • Whole blood for Culture • Whole blood plasma for molecular biology • Aliquot sera • Collection of Tissue Specimens • Fixed tissues • Complete autopsy on all fatal cases • Non-fixed tissues Target population INPATIENTS Lower Respiratory OUTPATIENTS • Bronchoalveolar lavage, Upper Respiratory • Nasopharyngeal (NP) tracheal aspirate, pleural fluid and oropharyngeal • Sputum (OP) • Nasopharyngeal Upper Respiratory wash/aspirate • Nasopharyngeal (NP) and oropharyngeal (OP) swabs Lower Respiratory • Nasopharyngeal wash / • Sputum aspirate Blood Blood • Serum: Acute (at • Serum: Acute (at onset) onset) and and convalescent (3-6 weeks convalescent (3-6 post onset) weeks post onset) • Whole blood (plasma) • Blood (plasma) • Urine • Stool • Tissue (e.g., lung) • Urine • Stool FATAL CASES All available premortem Specimens Tissue • Fixed tissue from all major organs (e.g., lung, heart, spleen, liver, brain, kidney, adrenals) • Non-fixed tissue from lung and upper airways (e.g., trachea, bronchus) Lower Respiratory • Bronchoalveolar lavage, tracheal aspirate, pleural fluid • Sputum Blood • Serum • Blood (plasma) Deep lung swab for bacterial Experiments • Select criteria to choose specimen for analysis • Maximize the usage of the specimen Convert to re-generable library Multiple experiments • Normalize among samples • Feedback to collectors • Request more or refined samples • Or stop collect samples Convert to re-generable library • In vitro library Sequencing library cDNA library miRNA library Non-coding RNA library Genome library • Microbe library Fungus Bacteria Virus • Cell lines (Case 4) Maximize the usage of the specimen • High throughput methods • High throughput ELSA • 2D-gel and Mass mapping • Next generation sequencing • High content screening • Sensitivity of the technology • Real-time PCR • Western blot • Immunochemistry • Pathology staining • Reliability of the technology • Make sure you understand the principle! Maximize the usage of the specimen Maximize the usage of the specimen Question: How to detect the expression for my gene(s) of interest? 1) Translation level: • Western blot (density measurement, need good antibody, saturation) 2) Transcription level: • Northern blot (density, saturation); • RNase protection assay (density, saturation) • In situ hybridization (density, saturation); • Competitive RT-PCR (end point analysis) • Quantitative RT-PCR (Method of choice) PCR reaction 1 Double n cycles (2)n 2 3 Linear plot Ideal outcome AMOUNT OF DNA 1 2 4 8 16 32 64 128 256 512 1,024 2,048 4,096 8,192 16,384 32,768 65,536 131,072 262,144 524,288 1,048,576 2,097,152 4,194,304 8,388,608 16,777,216 33,554,432 67,108,864 134,217,728 268,435,456 536,870,912 1,073,741,824 1,400,000,000 1,500,000,000 1,550,000,000 1,580,000,000 1400000000 1200000000 1000000000 800000000 600000000 400000000 200000000 0 0 5 10 15 20 25 30 35 30 35 PCR CYCLE NUMBER Log plot AMOUNT OF DNA CYCLE NUMBER 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 AMOUNT OF DNA 1600000000 10000000000 1000000000 100000000 10000000 1000000 100000 10000 1000 100 10 1 0 5 10 15 20 25 PCR CYCLE NUMBER Only in the exponential phase, PCR product depends on template Why endpoint analysis doesn’t work Out of exponential phase SERIES OF 10-FOLD DILUTIONS False amplification (Blank) Need enough initial template 2500 2000 1500 1000 500 0 1 4 7 10 13 16 13 16 19 22 25 28 31 34 37 40 43 46 49 8 7 6 5 4 3 2 1 0 1 4 7 10 19 22 25 28 31 34 37 40 43 No enough reaction volume (=10 ul), CV%>3% 4.00E+03 3.50E+03 3.00E+03 2.50E+03 2.00E+03 1.50E+03 1.00E+03 5.00E+02 0.00E+00 1 4 7 10 13 16 19 22 25 28 31 34 37 40 43 46 49 Alignment of the machine Treat the machine as a baby, gently please. Example: Determine quality of experiments 7 2.8 R a w flu o re s c e n c e R a w flu o re s c e n c e 6 5 2.6 E=0.562 E = 0 .8 0 3 2.4 4 2.2 0 4 8 12 16 20 24 28 32 36 40 44 48 C ycle n u m b er 3 E = 0 .5 6 2 2 0 4 8 12 16 20 24 28 C y c le n u m b e r 32 36 40 44 48 Melting curve to valuate primer