(-)-Histrionicotoxin

Synthesis of Spirocyclic
Compounds
Yi He
• Introduction
• Synthetic methodologies
• Syntheses of spirocyclics in natural products
– Gelsemine
– Spirotryprostatin
– Histrionicotoxin
– Ginkgolide B
• Conclusion
Introduction
• “Spirocyclane” introduced by Baeyer in
1900
• Widely present in natural compounds
• Synthetic challenging
– Chiral quaternary carbon center
– Synthesis of two fused rings
Methodologies for Constructing
Spirocenters
• Alkylations
• Rearrangement reactions
• Cycloadditions
• Transition metal catalyzed reactions
• Cleavage of bridged systems
Sannigrahi, M. Tetrahedron 1999 , 55 , 9007-9071.
Alkylation
X m n
Substitution m n
X m
X n
1,4 - Addition m n
Alkylation using Bis-acetal
MeO
MeO
MeO OMe
CH
2
OTs
TMSOTf
MeO
MeO
H
H
OTs
MeO
MeO
H
H
OTs
Solvent Yield%
77 CH
3
CN
THF 52
A%
71
26
B%
29
74
O
MeO
H
H
A
OTs
O
MeO
H
H
OTs
B
Tanaka, T.; Okuda, O; Murakami, K; Yoshino, H.; Mikamiyama, H;
Kanda, A; Iwata, C. Tetrahedron Lett. 1994 , 35 , 4125-4128.
Ph
Me
N
Alkylation using Ynamine
R
R
O
H
O n
MgBr, CH
3
CN, 70 ºC
60-70%
O
H O
C
N
Ph
Me n
O
R
Me
Ph
N R n
+
Me N O
Ph
A
1
1 n
2
O
B
O
R
CH
3
CH
3
A % B %
85
80 i -Pr 100
15
20
0 n
( ±)-Acoradiene
Ficini, J.; Revial, G.; Genêt, J. P.
Tetrahedron Lett. 1981 , 22 , 629-632.
Kaiser, R.; Naegeli, P. Tetrahedron Lett. 1972, 13 , 2009-2012.
Michael Addition
Ph
N
O
CO
2
Me
EtO
2
C
N
Boc
O a) DIEPA, CH
3
CN b) H
2
, Pd/C, CH
3
85% 2 steps
OH
Ph
N
EtO
2
C
O
N
Boc
O
H
CO
2
Me
Ph
H
EtO
2
C
O
H
N
N
H
O
Manzamine A
Brands, K. M.; DiMichele, L. M. Tetrahedron Lett . 1998 , 39 , 1677-1680.
Rearrangement Reactions for
Spiroannulation
• Vinylcyclopropanol/Vinylcyclobutanol rearrangement
• Pinacol-type rearrangement
• Sigmatropic rearrangement
Vinylcyclobutanol/Vinylcyclopropanol
Rearrangement n
OR m
OCH
3
OCH
3
Lewis acid n
OR m
OCH
3 n m
O
OCH
3 n = 1 or 2 m = 1 or 3
Trost, B. M.; Lee, D. C. J. Am. Chem. Soc. 1988 , 110 , 6556-6558.
Trost, B. M.; Chen, D. W. D. J. Am. Chem. Soc. 1996 , 118 , 12541-12554.
Vinylcyclopropane Rearrangement
OTMS
OCH
3
OCH
3
1 eq. TMSOTf, 0.7 eq. pyr
-40 °C, 0.01M CH
2
H OTMS
O
CH
3
H favored
OTMS
O CH
3 disfavored
H
Cl
2
, 85%
O
OCH
3
85.2% de
O
OCH
3
O
OCH
3
Trost, B. M.; Lee, D. C. J. Am. Chem. Soc. 1988 , 110 , 6556-6558.
HO
Prins – Pinacol Rearrangement
Y
R slow
Y XR fast
Y
XR
HO
O
XR
XR
XR
O
XR
TMSO TMSO
O
Prins-Pinacol Spiroannulation t -Bu
OTMS TMSOTf, DTBMP
CH
2
Cl
2
, RT, 70%
MeO OMe t -Bu
O
OMe
RuCl
3
•3H
2
O
NaIO
4
CH
3
, CCl
4
CN, H
2
O t -Bu
O
O t -Bu
TMSO
MeO
TMSOTf, DTBMP
OMe
CH
2
Cl
2
, RT, 28% t -Bu
O
OMe RuCl
3
•3H
2
O
NaIO
4
CH
3
, CCl
CN, H
4
2
O t -Bu
O
Me
DTBMP = t -Bu N t -Bu
O
Minor, K. P; Overman, L. E. Tetrahedron 1997 , 53 , 8927-8940.
t -Bu
Mechanism for Regioselectivity
XR' fast
OR
H
X
R' t -Bu OR
XR' t -Bu
O slow
XR'
XR' t -Bu
RO t -Bu
O
XR' t -Bu OR
X
R'
H t -Bu t -Bu
RO
OR
XR' t -Bu slow
XR' t -Bu
O
O
XR'
Mechanism for Low Yield of Axially-
Tethers Electrophile t -Bu
X
R' t -Bu
OR OR neither bond anti to the carbocation!
XR'
2,3-Sigmatropic Rearrangement
SPh
O
N
2
Rh
2
(OAc)
4
CO
2
Et
PhH, reflux
PhS CO
2
Et
O
Ph
S
CO
2
Et
O
EtO
2
C
Ph
S O
O
(+)-Acorenone
Kido, F.; Abiko, T.; Kato M. J. J. Chem. Soc. Perkin Trans . 1 1992 , 229-233.
• [4+2]
• [3+2]
• [2+2]
• [2+1]
Cycloaddition
X n n n
X
X n
+
+
X
Y n n n n
X
Diels-Alder Approach to Shizuka-
Acoradienol m
+
MeO
2
C
O
O
CHCl
3
, 50 °C
300 MPa, 57%
-CPBA
MeO
2
C
O O O
MeO
2
C xylene, reflux
45%
O O
MeO
2
C
O
HO
Shizuka-acordienol
Hatsui, T.; Hashiguchi, T.; Takeshita. H. Chemistry Express 1993 , 8 , 581-584.
[2+1]Cycloaddition in the Total
Synthesis of (-)-Acorenone
N
2
Cu powder cyclohexane
O
O O
(-)-Acorenone
HCl
CHCl
3
Ruppert, J. F.; Avery, M. A.; White, J. D. J. Chem., Soc. Chem. Commun. 1976 , 978.
Gelsemine
• Major alkaloid component of Gelsemium sempervirens
• Novel hexacyclic cage structure
• Unique spiro-oxindole center
O
H
N
Me
N
O
Retrosynthesis of Gelsemine by
Johnson
O
H
N
N
Me
O
N
Me
O
O
+
O
Me
N
O
Me
N
TMS
N
N
N
OMe
O
O
Sheikh, Z.; Steel, R.; Tasker, A. S.; Johnson, A. P.
J. Chem. Soc., Chem. Commun. 1994 , 763-766.
Johnson’s Total Synthesis of Gelsemine
O
Me
N
O
O
+
TMS
N
N
N
OMe n -BuLi (2 equiv.)
N
N
N
OMe
MeO
N
N N
O
Me
N
O
+
O
Me
N
O major minor
h v , CH
3
CN
Pyrex
MeO N
O
Me
N
O
+ O
Me
N
1 : 2
N
O
OMe
Sheikh, Z.; Steel, R.; Tasker, A. S.; Johnson, A. P.
J. Chem. Soc., Chem. Commun. 1994 , 763-766.
Mechanism of Radical Cyclization
O
Me
N
O
Me
N
O
N
N
N
OCH
3
N
O
OMe
O
Me
N
•
N
•
OCH
3
O
Me
N
MeO
N
O
O
•
•
N
OCH
3
O
Me
N
O rotation
O
Me
N
MeO
•
•
N
O
Unexpected Formation of Oxetane
O
Me
N
•
•
N
O
H
H
H
O
O
Me
N
•
N
O
CH
•
2
O
O
Me
N
Ph N
O
O
Me
N
Retrosynthesis of Gelsemine by
Speckamp
O
H
N
O
O
Me
N
O
N
SEM Br
OTDS
O
Me
N
OTf
OTDS
O
Me
N
OH
TIPSO
O
H
N
CH
3
OEt
+
O
N
CH
3
O
OH
Newcombe, N. J.; Ya, F.; Vijn, R. J.; Hiemstra, H.; Speckamp, W. N.
J. Chem. Soc., Chem. Commun. 1994 , 767-768.
Speckamp’s Total Synthesis of Gelsemine
O
O
Me
N
Me
N
OTf
1) Pd(OAc)
2
, PPh
3
, Et
CO, 2-bromoaniline
3
N
DMF, RT, 24 h, 79%
OTDS
2) NaH, SEMCl, THF
O
Me
N
O
N
SEM
OTDS de? 60:30
1) Bu
4
NF, THF, RT., 2 h
2) HgO, Tf
MeNO
2
O, N,N -dimethylaniline
2
, RT, 3 d, 60%, 3 steps
3) NaBH
4
4) Bu
4
, NaOH, CH
2
Cl
2
, EtOH, 80%
NF, THF, 4 Å MS, reflux, 4 h, 90%;
O
H
N
O
N
SEM Br
OTDS
O
Me
N
Pd
2
(dba)
3
, Et
3
N
PhCH
3
, refulx, 4h
60%
O
H
N
O
TDS = thexyldimethylsilyl
AlH
3
, THF, -65 - 0 °C
4 h, 50%
Me
N
O
Newcombe, N. J.; Ya, F.; Vijn, R. J.; Hiemstra, H.; Speckamp, W. N.
J. Chem. Soc., Chem. Commun. 1994 , 767-768.
Retrosynthesis of Gelsemine by
Fukuyama
O
H
N
O
H
N
Me
N
O
X
O
NH
RO
2
C
Me
N
OH
X
O
H
N
RO
2
C
O
H
N
X
HN
Me
CO
2
R
CO
2
R
X
O
N
H
Fukuyama, T.; Liu G. Pure & Appl. Chem. 1997 , 69(3) , 501-505.
Fukuyama’s Total Synthesis of Gelesmine
HO
OAc
CO
2
CHO
CH
3
1) 4-iodooxindole
cat. piperidine
MeOH, RT, 89%
2) DCC, DMSO
pyridium trifluoroacetate
Et
3
N, CH
2
Cl
2
, RT, 91%
O
O
NH 1) PhCH
3
/CH
3
CN, 1:1
90 ºC, 45 min, 98%
2) n -Bu
3
SnH, cat. AIBN
95 ºC, 1 h, 85%
H
3
CO
2
C
O
NH
O
CO
2
CH
3 I
O
H
N
Me
N
O
O
NH
O
CO
2
CH
3 I
O
O
H
N
I
H
3
CO
2
C
O
O
NH
I
H
3
CO
2
C
Fukuyama, T.; Liu G. Pure & Appl. Chem. 1997 , 69(3) , 501-505.
Spirotryprostatins
• Isolated from the fermentation broth of
Aspergillus fumigatus
• Inhibits cell cycle at G2/M phase
• Spiro-oxindole rings
O
O
HN
N
N
H
3
CO
O
HN
O
N
O
O
N
Spirotryprostatin A Spirotryprostatin B
Cui. C. B.; Kakeya, H.; Osasa, H. Tetrahedron 1996 , 51 , 12651-12666.
Cui. C. B.; Kakeya, H. l.; Osasa, H. Tetrahedron 1997 , 53 , 59-72.
Danishefsky’s Pinacol-type
Rearrangement Approach
N
H
CO
2
CH
3
NBS, THF
NBoc
H
2
O, AcOH
SPh
HN
O
CO
2
CH
3
NBoc
SPh
N
H O
H
Br
CO
2
CH
3
NBoc
SPh
H
3
CO
HN
O
H
O
N
N
O
Spirotryprostatin A
Edmonson, S. D.; Danishefsky, S. J. Angew. Chem. Int. Ed. Engl. 1998 , 37 , 1138-1140.
Williams’ 1,3-Dipolar Cycloaddition
Approach
Ph
HN
Ph
O
O
OHC
Me
OMe
Me
4Å MS, PhCH
3
EtO
2
C
N
H
O
Ph
Me
MeO
Me
O
HN
N
Ph
O
O
CO
2
Et
[1,3]-dipolar cycloaddition
82%
MeO
Me Ph
Me
O
N
Ph
HN
O
H
O
CO
2
Et
HN
O
O
N
O
N
Spirotryprostatin B
Sebahar, P. R.; Williams. R. M. J. Am. Chem. Soc. 2000 , 122 , 5666-5667.
Overman’s Retrosynthesis of
Spirotryprostatin B
HN
O
O
N
O
N
3-allypalladium capture asymmetric
Heck cyclization
I
RN
O
O
N
HN
O
HN
L
2
Pd
I
O
HN
O
N
O
Overman, L. E.; Rosen, M. D. Angew. Chem. Int. Ed. Engl. 2000 , 39 , 4596-4599.
Overman’s Intramolecular Heck Approach
H
O
NH
I
N
H
O
OTBDPS
1) SEM-Cl, NaH
2) TBAF
3) Dess-Martin
4) A , t -BuOK
O
N
[Pd
2
(dba)
3
] •CHCl
3
(otol)
3
P, KOAc
THF, 70 °C
(72%, B:C = 1:1)
N
SEM
O
I
SEMN
O
O
N
O
N
Me
2
AlCl
DIEPA (93%)
HN
O
O
N
O
N
SEMN
O
B
O
N
N
O
Me
2
AlCl
DIEPA (93%)
HN
O
O
N
O
N
H
O
A =
O
N
NH
PO(OMe)
2
C
Overman, L. E.; Rosen, M. D. Angew. Chem. Int. Ed. Engl. 2000 , 39 , 4596-4599.
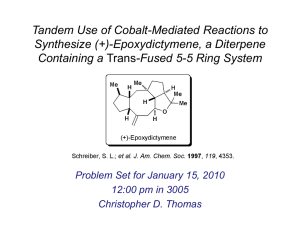
(-)-Histrionicotoxin
• Isolated from the skin of frog Dendrobates histrionicus
• Novel spiro-piperidine structure
OH
H
N
(-)-Histrionicotoxin
Daly, J. W.; Karle, I.; Myer, C. W.; Tokuyama, T.; Walters, J. A.; Witkop, B.
Proc. Natl. Acad. Soc. U. S. A. 1971 , 68 , 1870.
Retrosynthesis of (-)-Histrionicotoxin by
Stork
HO
HN
N
H
HO
O
O
R
OTBS
HO
CONH
2
Br
R
CO
2
Me OTBS
H
+
O H
Br
Stork, G.; Zhao, K. J. Am. Chem. Soc. 1990 , 112 , 5875-5876.
Stork’s Synthesis of (-)-Histrionicotoxin
CO
2
Me OTBS
O
O
OTBS
H
+
O H
O
O
Br
LDA
43%
I
1) (CF
3
CH
3
CO
2
)
2
CN, H
2
IPh
O, 3 d
2) Et
3
N, ClCH
2
CH
2
Cl
65-70 °C, 2h, 31%
OH
I
1) Ph
3
P, CBr
4
2) NH
4
, ether, 2h, 53%
Cl, AlMe
3
, PhH, 40 °C, 18h
3) Ac
2
O, Pry, DMAP, 70%
AcO
HN
I
I
1) O
3
, PPh
3
2) (Ph
3
P
+
CH
2
I)I
-
, NaN(TMS)
2
HMPA, THF, 52%
3) 5% HCl, THF
AcO
HO
HN
CONH
2
Br
I
I
Stork, G.; Zhao, K. J. Am. Chem. Soc. 1990 , 112 , 5875-5876.
Retrosynthesis of (-)-Histrionicotoxin by
Holmes
N
H
HO
N
R O R'
R
N
O
R'
NH
R OH
R'
Williams, G, M.; Roughley, S. D.; Davies, J. E.; Holmes, A. B.
J. Am. Chem. Soc. 1999 , 121 , 400-401.
Holmes’ Synthesis of (-)-Histrionicotoxin
O O
NaN(TMS)
2
1-chloro-1-nitrosocyclohexane
X
(CH
2
)
3
OTBDPS
X = (+)-10,2-camphorsultam
THF, then HCl (aq), 70%
X
NHOH
(CH
2
)
3
OTBDPS
CN
(CH
2
)
3
OTBDPS
1) PHCH
3
, 80 °C, 6 h
2) styrene, 75 °C
85%, 2 steps
X
O
N
O
Ph
BnO
N
O
Ph
TMS TMS
BnO
PHCH
3 sealed tube
BnO
N
O CN
N
O
CN
N
O
1) Zn, AcOH, 30 min, 98%
2) K
2
CO
3
, MeOH, 94%
N
H
HO
Williams, G, M.; Roughley, S. D.; Davies, J. E.; Holmes, A. B.
J. Am. Chem. Soc. 1999 , 121 , 400-401.
Ginkgolide B
• Isolated from Ginkgo biloba
• Complex structure
– Six ring in a confined space
– Eleven stereogenic carbons
– Four contiguous quaternary centers
– Ten oxygenated carbons
O O
HO
HO
Me
HO
O
O
O
O
O
CMe
3
Retrosynthesis of Ginkgolide B’s
Synthetic Intermediate by Corey
O
H
O
H t -Bu
MeO
O t -Bu
O
MeO
O t -Bu
•
O
OMe
OMe
O
MeO
O t -Bu
CO
2
H
Corey, E. J.; Kang, M.-C.; Desai, M. J.; Ghosh, A. K.; Houpis, I. N.
J. Am. Chem. Soc. 1988 , 110 , 648-651
Corey’s Total Synthesis of Ginkgolide B
O
OMe
OMe
1) t -Bu
2
Cu(CN)Li
2
, Et
2
O
-78 to -45 °C
2) TMSCl, Et
3
N, -45 to -10 °C
3) 1,3,5-trioxane TiCl
4
, CH
2
Cl
2
-78 °C, 65% 3 steps;
MeO
O
O t -Bu
1) LDA, DME, -78 to 0 °C
then PhNTf
2
, 0 to RT, 80%
2) A , Pd(PPh
3
)
4
, CuI, n -PrNH
PhH, 16 °C, 84%;
2
MeO
O
MeO
O t -Bu
1) (Cy-Hex)
2
BH, THF, 0 °C
2) AcOH, H
2
O
2
, pH 10
3) 1 N HCl, pH 3
4) pH 11, 4 h, pH 3
86% 4 steps t -Bu
O
O
O
1) (COCl
2
)
2
, PhH
2) n -Bu
3
N, PhCH
3
80% two steps
CO
2
H
Me
MeO
O
•
O t -Bu
O
H
O
H t -Bu
O
O O
HO
HO
Me
HO
O
O
O
O
CMe
3
A = Me
O
O
O
Corey, E. J.; Kang, M.-C.; Desai, M. J.; Ghosh, A. K.; Houpis, I. N.
J. Am. Chem. Soc. 1988 , 110 , 648-651
Retrosynthesis of Ginkgolide B’s
Synthetic Intermediate by Crimmins
Me
HO
O
O
O OMe
O
O
OMe
CMe
3
EtO
2
C
O
CO
2
Et
O
Et
3
SiO CMe
3
O
Et
3
SiO CMe
3
O
CO
2
Et
O
Et
3
SiO CMe
3
Crimmins, M. T.; Pace, J. M.; Nantermet, P. G.; Kim-Meade, A. S.; Thomas, J. B.
Watterson, S. H.; Wagman, A. S. J. Am. Chem. Soc. 1999 , 121, 10249-10250.
Crimmins’ Total Synthesis of Ginkgolide B
EtO
2
Et
3
C
O
SiO
Et
3
SiO CMe
3
CO
2
Et
O
CMe
3
O
(EtO
2
CCH
2
O
O
O
CH
2
)
2
H
O
ZnCu
O
Et
3
CO
2
Et
SiO dimethyldioxirane
CMe
3
H
2
O, p -TSA, 94%
O
h
, 366 nm
hexane. 100%
CMe
3
O
O
O
OH
O
OH
OH
CMe
3
1) MeOH, p -TSA
CH(OMe)
3
, 95%
2) CS
2
, MeI, DBU
3) Bu
3
SnH, AIBN
60 °C, 78% 2 steps
O
O
O
OMe
O
CMe
3
OMe
O O
HO
HO
Me
HO
O
O
O
O
O
CMe
3
Crimmins, M. T.; Pace, J. M.; Nantermet, P. G.; Kim-Meade, A. S.; Thomas, J. B.
Watterson, S. H.; Wagman, A. S. J. Am. Chem. Soc. 1999 , 121 . 10249-10250.
Conclusions
• Structure diversity of spirocyclic compounds
• Various approaches to spirocenters
Thanks
• Kiessling Group Members