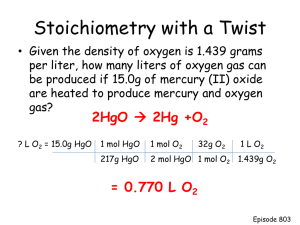UCL_Strathclyde meeting
advertisement

Hydrogen Generation Using a Photoelectrochemical Reactor: Materials Assessment and Reactor Development Steve Dennison & Chris Carver Chemical Engineering Imperial College Photoelectrolysis of water absorption Semiconductor h Semiconductor (eCB , hVB ) 2H2O 4hVB O2 4H 2H2O 2eCB H2 2OH Requires 1.23 V: thermodynamic value from G0 = -237 kJmol-1. Equivalent to a photon of wavelength ~1000 nm The semiconductor-electrolyte interface Conduction Band Redox Electroyte Conduction Band EF EF Valence Band Semiconductor Metal Valence Band Semiconductor Metal The semiconductor-electrolyte interface 2 Band Bending eSeparation between Fermi energy and Conduction band edge H+ / H2 Ef Thermodynamic Potential of Water: h O2 / H2O h+ Overpotential for O2 evolution e- The semiconductor-electrolyte interface 3 0.4V 0.3V Ef An ideal semiconductor for water-splitting has band gap of: ~2.6eV H+ / H2 1.23V O2 / H2O 0.4V Choosing the semiconductor • It must be an OXIDE – Stability/insolubility in aggressive media – Stability under conditions of oxygen evolution Candidate Materials – TiO2: Eg ~ 3.0-3.2 eV (410-385 nm) – Fe2O3: Eg ~ 2.2 eV (>565 nm) – WO3: Eg ~ 2.6 eV (475 nm) Match to Solar Spectrum TiO2 WO3 Fe2O3 Fe2O3: typical photoresponse 3.5E-06 Normalised Photocurrent 3.0E-06 2.5E-06 2.0E-06 1.5E-06 1.0E-06 5.0E-07 0.0E+00 300 350 400 450 500 Wavelength / nm 550 600 650 700 Fe2O3: voltammetry under illumination 2.000E-05 5.0E-05 1.500E-05 1.000E-05 cd / Acm -2 Water-MeOH 5.000E-06 0.000E+00 4.8 4.0E-05 4.9 5 5.1 5.2 5.3 5.4 5.5 -5.000E-06 Water -1.000E-05 -1.500E-05 t/s Water Water/MeOH 3.0E-05 1.00E-05 6.00E-06 4.00E-06 2.0E-05 2.00E-06 cd / Acm -2 0.00E+00 4.8 4.9 5 5.1 5.2 5.3 5.4 5.5 -2.00E-06 -4.00E-06 -6.00E-06 -8.00E-06 1.0E-05 -1.00E-05 t/s Water Water-MeOH 2.000E-05 1.500E-05 0.0E+00 1.000E-05 cd / Acm -2 cd / Acm -2 8.00E-06 5.000E-06 0.000E+00 4.8 4.9 5 5.1 5.2 5.3 5.4 5.5 -5.000E-06 -1.0E-05 -0.5 -1.000E-05 -0.25 0 0.25 0.5 -1.500E-05 0.75 Potential vs qre / Volt 1 t/s Water Water/MeOH Fe2O3: photocurrent transients @ +0.6V 2.50E-05 cd / Acm -2 2.00E-05 1.50E-05 1.00E-05 5.00E-06 0.00E+00 -5.00E-06 4.8 4.9 5 5.1 5.2 t/s Water Water/MeOH 5.3 5.4 5.5 Fe2O3: photocurrent transients @ +0.6V 2.50E-05 2.00E-05 cd / Acm -2 1.50E-05 1.00E-05 5.00E-06 0.00E+00 -5.00E-06 4.80 4.90 5.00 5.10 5.20 5.30 Time / s Water/MeOH Water 5.40 5.50 5.60 5.70 Fe2O3: photocurrent transients @ +0.1V 8.0E-06 6.0E-06 cd / Acm -2 4.0E-06 2.0E-06 0.0E+00 -2.0E-06 -4.0E-06 -6.0E-06 -8.0E-06 4.8 4.9 5 5.1 5.2 t/s Water Water-MeOH 5.3 5.4 5.5 Findings for Fe2O3 • Preliminary (and not concluded yet) – In the absence of MeOH see cathodic “dark” current, even at 0.6 V. – As applied potential is decreased, the photocurrent becomes more transient – As applied potential is decreased the cathodic “dark” current increases (relative to the photocurrent) WO3: recent work • Photocurrent observed (poor efficiency) • Enhancement with oxygen evolution catalyst (electrodeposited IrO2) not realised • Further detailed electrochemical analysis underway (plus SEM/TEM, XRD, etc.) Christopher Carver Dr Klaus Hellgardt Design flexible test-bed reactor Hydrogen production experiments - 10 x 10cm photoanode - Photon absorption - Semiconductor material - Quantum efficiency - Electrode configuration - High mass transfer rate coefficients - Separate hydrogen and oxygen titanium Good absorption Stable in alkali Recombination Stable in acid/alkali UV absorption only Good efficiency Stable in acid PVDF quartz Current density (A/m2) Butler-Volmer equation Kinetic control Increasing mass transport rate Transport control Overpotential (V) PC SOLAR SIMULATOR PEC REACTOR POTENTIOMETER PUMP RESERVOIR / H2 COLLECTION quartz window PHOTOANODE membrane cathode PHOTOANODE electrolyte Photo-Response of Fe2O3 90 80 70 Current Density (A/m2) 60 50 Dark 40 Illum 30 20 0.5 V 1.0 V 1.5 V 2.0 V 2.5 V 3.0 V 10 0 0 -10 20 40 60 80 100 Time (s) 120 140 160 180 200 Averaged Photo-Response of Fe2O3 90 80 70 Current Density (A/m2) 60 50 Dark 40 Illum 30 20 0.5 V 1.0 V 1.5 V 2.0 V 2.5 V 3.0 V 10 0 0 -10 20 40 60 80 100 Time (s) 120 140 160 180 200 Photoresponse of Fe2O3: Configuration A 2.5 Current Density (mA/cm2) 2 1.5 1 0.5 0 0.8 -0.5 0.9 1 1.1 1.2 Voltage (V) 1.3 1.4 1.5 1.6 Photoresponse of Fe2O3: Configuration B 2.5 Current Density (mA/cm2) 2 1.5 1 0.5 0 0.8 -0.5 0.9 1 1.1 1.2 Voltage (V) 1.3 1.4 1.5 1.6 quartz window membrane cathode PHOTOANODE electrolyte Set Up Reactor Experiments Analyse Results Electrical connections Semiconductors / electrolyte Quantify H2 production Hydrogen extraction system Electrode configuration Optimise reactor design Evaluate reactor design Multiple electrodes Temperature Deliver report QUESTIONS? Electrolyte Flow (with H2 or O2) Electrolyte Inflow (2) Diffusion (3) Kinetics (1) Fluid Flow+ Diffusion (4?) Diffusion (1) Fluid Flow+ Diffusion H2 Electrolyte Flow (if laminar) 2H+ Membrane O2 + (3) Mesh Cathode (Conductor) 2e 2H+ H2O Kinetics Absorption, (2) Diffusion (2), Band Bending + Absorption, α Photo-Anode h+ e- Choosing the semiconductor • Absolute levels of the electronic levels in the semiconductor: – Defined by the electron affinity – Require EA ~ 3.7 eV Fe2O3: NaOH-H2O 5.0E-05 Dark 450 nm 4.0E-05 cd / Acm -2 3.0E-05 2.0E-05 1.0E-05 0.0E+00 -1.0E-05 -0.5 -0.25 0 0.25 Potential vs qre / Volts 0.5 0.75 1 Fe2O3: NaOH-H2O/MeOH (80:20) 5.0E-05 Dark 450 nm 4.0E-05 cd / Acm -2 3.0E-05 2.0E-05 1.0E-05 0.0E+00 -1.0E-05 -0.50 -0.25 0.00 0.25 Potential vs qre/ Volt 0.50 0.75 1.00