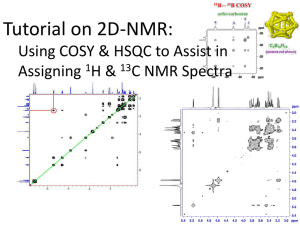
Heme Synthesis and 2D COSY NMR
advertisement

True or false? Blood is red because of the iron in hemoglobin. “Red blood cells” A close-up of red blood cells within a capillary. Over 99 percent of the blood vessels in the body are capillaries, even though they only hold 5 percent of the body’s blood. False por·phy·rin Function: noun Etymology: from Greek porphyra purple -any of various compounds with a macrocyclic structure that consists essentially of four pyrrole rings joined by four =CH- groups; especially : one (as chlorophyll or hemoglobin) containing a central metal atom and usually exhibiting biological activity Wikipedia’s dynamic picture There are many different hemes used in biochemistry as: • Electron transfer cofactors as Fe(3+) / Fe(2+) • Catalytic Cofactors as cytochrome P450 • Besides O2 transport as hemoglobin One example: Cytochrome c Oxidase Where dioxygen is reduced to water during respiration Note all the hemes! How to draw a porphyrin: The Adler synthesis of simple, symmetrically substituted porphyrins: Meso position R = phenyl (TPP) toluyl (TTP) p-chlorophenyl (TClPP) Metallation of porphyrin Anything change at Fe? Porphyrin substituents on heme in hemoglobin How do you know when the reaction is done? Absorption Spectrum of a Porphyrin Figure 1. Typical UV-Vis absorption spectrum of a porphyrin. The highly conjugated porphyrin macrocycle shows intense absorption (extinction coefficient > 200,000 M-1cm-1) at around 400 nm (the "Soret" band), followed by several weaker absorption bands (Q Bands) at higher wavelengths (450 to 700 nm). 1H NMR Characterization: Challenges…. and how to meet them L- keto Ru(L-amino) 9.6 9.1 8.6 8.1 7.6 7.1 10.1 9.6 9.1 8.6 8.1 7.6 7.1 10.1 9.6 9.1 8.6 8.1 7.6 7.1 Ru(L-allox) Ru(L-keto) 10.1 ppm Complicated spectrum? Try 2-D COSY NMR (correlation spectroscopy) from Ben Williams from Ben Williams from Ben Williams from Ben Williams ,aʹ b a,bʹ c cʹ 1 2 10.1 9.6 9.1 ppm 8.6 8.1 7.6 7.1 from Alex Gaudette. BMC’12 Ru(L-amino) d c 10.1 a,b,bʹ 3 cʹ 9.6 aʹ 9.1 8.6 8.1 Ru(L-keto) d 7.6 7.1 7.6 7.1 7.6 7.1 ,aʹ a b,bʹ c cʹ 10.1 9.6 9.1 8.6 8.1 Ru(L-allox) ,aʹ a c cʹ 10.1 9.6 9.1 8.6 ppm b,bʹ 8.1 For Thursday, Sept. 27: • Do heme synthesis using your prophyrin, TPP, TTP or TClPP, determining reaction completion by fluorescence • Filter Ru complex (if necessary); obtain MS of product • Observe 2D COSY NMR process • Obtain UV/vis spectrum of your Fe(porphyrin)Cl complex Writing Assignments • Goal = writing a full synthesis and characterization manuscript on the Ru complex you made • Structure like those we discussed on first day • Includes, synthesis, 1D NMR and 2D COSY, and CV • Due dates • Draft 1 on Wednesday Oct.3 of the Experimental Section with outline form of introduction • Draft 2 on Wednesday Oct. 10: Introduction, revised Expermental and Results section with NMR Results ,aʹ cʹ Ru(L-aap) c b a,bʹ 1 2 d Ru(L-amino) 10.1 c 9.6 cʹ a,b,bʹ 9.1 8.6 3 9.6 7.6 aʹ d 10.1 8.1 ,aʹ 9.1 a 8.6 b,bʹ8.1 7.6 7.1 9.1 8.6 8.1 7.6 7.1 Ru(L-keto) c cʹ 10.1 9.6 7.1 Ru(L-aap) ,aʹ c a,bʹ cʹ 1 2 10.1 9.6 9.1 8.6 8.1 Ru(L-amino) d c 10.1 3 cʹ 9.6 Ru(L-keto) 7.6 7.1 7.6 7.1 7.6 7.1 a,b,bʹ aʹ 9.1 8.6 8.1 d ,aʹ a b,bʹ c cʹ 10.1 9.6 9.1 8.6 8.1 ,aʹ -allox) b c cʹ a