The work is described in more detail here
advertisement

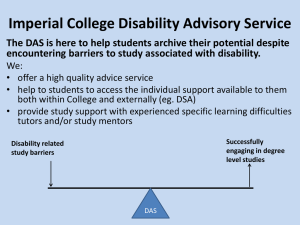
Gypsum Waste Minimisation Presented at the 3rd Annual WARMNET Conference “Tackling Waste 2006”, Nottingham, 6th and 7th July 2006 Rosemary Greaves Department of Materials Prof. Alan Atkinson Dr. Mark Tyrer Page 1 of 19 © Imperial College London Presentation Outline • The gypsum waste minimisation project • Titanogypsum by-products • Alternative approaches • The ammonia process • Experimental & analysis • Conclusions & further work Page 2 © Imperial College London Gypsum Calcium Sulfate Dihydrate CaSO4.2(H2O) • Vital component of many construction materials • In the UK demand outstrips domestic supply [1] Page 3 © Imperial College London Impetus For Work • Sponsors landfill ~75kt of by-product gypsum per annum – Red titanogypsum – Construction site waste • Disposal costs have escalated – EU landfill directive [2] – Increased landfill tax Minimise and reuse gypsum wastes Page 4 © Imperial College London The Gypsum Waste Minimisation Project Research Partners Sponsors Imperial College London Huntsman Tioxide • • • Professor Alan Atkinson Dr Mark Tyrer Rosemary Greaves Lafarge Plasterboard Coventry University • • • Dr Peter Claisse Dr Esmaiel Ganjian Seema Karami Funding EPSRC University of Birmingham • • Page 5 Dr Gurmel Ghataora Waliur Rahman Miniwaste Faraday Partnership © Imperial College London Titanogypsum By-products By-product of TiO2 production from ilmenite (FeTiO3) Waste Acid Calcium Oxide Calcium Hydroxide Partial Neutralisation Complete Neutralisation Carbon Dioxide White Gypsum Red Gypsum Effluent to Drain Page 6 © Imperial College London Red Titanogypsum Fe(OH)3 Mass per gram (mg/g) Fe(OH)2 Element Page 7 © Imperial College London Routes to Red Gypsum Minimisation Precipitate metal salts prior to gypsum formation • Wastewater contains a complex combination of impurities • Focus on colour • Use simplified solution based on sponsor’s analysis Page 8 [H2SO4]g/l [Fe2+] pH 142.10 36.12 ~2.5 © Imperial College London Eh (Volts) Pourbaix Diagram [3] pH Page 9 © Imperial College London Eh (Volts) Oxidise pH Page 10 © Imperial College London Eh (Volts) Increase pH pH Page 11 © Imperial College London Ammonia Addition Process [4] H 2 SO 4 NH 4 OH ( NH 4 ) 2 SO 4 H 2 O • Regeneration of NH3 • Iron content 160g/kg Page 12 0.42g/kg © Imperial College London Titration Experiments How does pH effect precipitation? 1. Produce Analogue: – H2SO4 – FeSO4.7(H2O) 2. Titrate aqueous ammonia (NH4OH) Page 13 © Imperial College London Precipitation of Iron pH 4.7 6.3 6.8 7.0 7.6 8.2 8.7 Samples taken at various pH • Centrifuged • Solids filtered from solution • Iron concentration of solution analysed Page 14 © Imperial College London Spectrophotometry 1. Reduce to iron (II) 2. Chelate – 1,10-phenanthroline 3. Illuminate – – – l=515nm Visible monochromatic Intensity a concentration Page 15 © Imperial College London Data Analysis Page 16 © Imperial College London Effect of pH on Iron Removal Page 17 © Imperial College London Conclusions and Future Work • Alkalizing the solution causes iron to precipitate • Concentration of iron decreases with pH • Precipitates must be analysed • Perform a Comparison with alternative techniques • Analyse resultant gypsum samples Page 18 © Imperial College London References 1. Hillier, J. A., L. E. Taylor, et al. (2004). United Kingdom Minerals Yearbook 2004, British Geological Survey. 2. European Council Directive 1999/31/EC on the Landfill of Waste. 1999. 3. Roine, A., HSC Chemistry 5.1 for Windows, Outokumpu Research Oy. 4. Hyvrard, F. and P. Muller, Method for Treating and Upgrading Effluents Containing Metalic Sulphates Using an Ammonia Addition Step. 2004: US. p. 5. Page 19 © Imperial College London