Crystal Shapes
advertisement

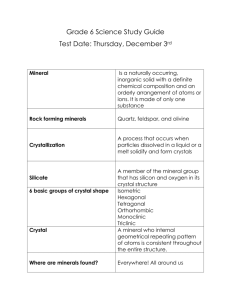
Minerals A Mineral is… • Naturally Occurring – made by nature – not by man A Mineral is… • Inorganic - not alive, never was alive, not made from a living process A Mineral is… • Solid – not a liquid or a gas A Mineral is… • Unique Chemical Composition – every mineral is different A Mineral is… • Crystalline Structure – atoms repeat in patterns Crystal Shapes Basics of Crystals • Crystals are defined by the number of sides (faces) and the angles of these sides (geometry) 1. Cubic • cubic – all sides are square 2. Tetragonal • tetragonal – 4 rectangles, 2 squares 3. Orthorhombic • orthorhombic – all rectangles, 3 different sized rectangles 4. Monoclinic • monoclinic – 4 rectangles, 2 parallelograms 5. Triclinic • triclinic – all parallelograms 6. Hexagonal • hexagonal - six-sided prisms. When you look at the crystal on-end, the cross section is a hexagon Mineral Groups • Minerals are often grouped together by their composition (what they are made of) • There are over 4,000 known minerals and only 7 groups 1. Silicates (most common) • Contain silicon and oxygen • Ex. Quartz, Olivine, Augite 2. Carbonates • Contain CO3 • Ex. Calcite, Dolomite 3. Oxides • Contain oxygen bonded with 1 or more elements • Ex. Corundum, Hematite 4. Sulfides and 5. Sulfates • Sulfides (contain Sulfur), Sulfates contain SO4 • Ex. Gypsum, Galena, Pyrite 6. Halides • Contain halogens (Fluorine, Chlorine, etc.) • Ex. Halite, Fluorite 7. Native Elements • Mostly metals, found as individual elements • Ex. Copper, Gold, Silver How do Minerals form? • • • • 1. Cooling magma or lava 2. Hydrothermal 3. Evaporation of liquid solutions 4. Precipitate out of solution 1. Cooling magma or lava 2. Hydrothermal 3. Evaporation of liquid solutions 4. Precipitate out of solution 5 Characteristics Used to Identify Minerals • • • • • 1) Hardness 2) Break Tendency 3) Luster (metallic/non-metallic) 4) Streak 5) Color 1) Hardness (Moh’s Scale) • - how easily a mineral can be scratched 2) Break Tendency • Fracture - breaks along rough, jagged edges • Cleavage – breaks along smooth, flat surfaces Conchoidal fracture – curved breakage 3) Luster (metallic/non-metallic) • - how light shines off of its surface • Metallic Non-metallic 4) Streak Color of a mineral when broken or powdered 5) Color Other ways to identify minerals • • • • Reactivity with acids Density/specific gravity Magnetism More special properties to come Other Properties of Minerals • • • • • • • Reactivity with Acid Magnetic Fluorescence Phosphouresence Density Crystal Shape Triboluminesence Reactivity with acid • Carbonate minerals fizz when acid is applied Magnetic • Some iron rich minerals are magnetic Fluorescence Phosphorescence Triboluminesence