Solid
advertisement

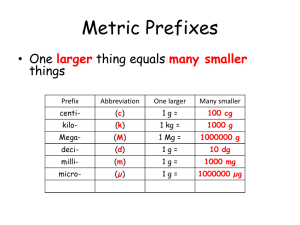
Descriptive Abbreviations • • • • (s) Solid (l) Liquid (g) Gas (aq) Aqueous – dissolved in water • How do we know the states of matter in a reaction? – Word problem tells us – States of matter from the periodic table – Common knowledge Episode 605 Determining State at Room Temperature • All metals are solids except for mercury, which is a liquid. • Most non-metals are gases with these exceptions: – Liquid – bromine – Solids – carbon, phosphorus, sulfur, selenium, tellurium, and astatine • All metalloids are solids • Ionic compounds are solid, unless told otherwise. • Covalent compounds are liquid, unless told otherwise. Episode 605 When heated, solid mercury (II) oxide yields mercury and oxygen gas. • Write the balanced chemical equation including the descriptive abbreviations for the state of matter. 2 HgO(s) → 2 Hg(l) + O2(g) Episode 605 Classifying Reactions http://video.about.com/chemistry/What-Are-Types-of-Chemical-Reactions.htm • Synthesis • Two or more substances combine to form a more complex substance A + B AB Fe(s) + S(s) FeS(s) H20(l) + SO3(g) H2SO4(aq) (acid rain) http://www.natgeoeducationvideo.com/film/1233/acid-rain Episode 605 Classifying Reactions • Decomposition • A single substance is broken down into two or more simpler substances AB A + B H2O (l) H2(g) + O2(g) 2HgO 2Hg(l) + O2↑ Episode 605 Classifying Reactions • Single Replacement • A free element replaces a less reactive element in a compound A + BY AY + B Zn (s) + 2HCl(aq) ZnCl2(aq) + H2(g) 2Al(s) + 3CuCl2(aq) 2AlCl3(aq) + 3Cu(s) Cu(s) + MgCl2(aq) NR (no reaction) Activity Series: an arrangement of elements in the order of their tendency to react Episode 605 Decreasing activity Activity Series Lithium Potassium Barium Calcium Sodium Magnesium Aluminum Manganese Zinc Chromium Nickel Tin Lead (Hydrogen) Copper Mercury Silver Gold Episode 605 Classifying Reactions • Double Replacement • The elements of a reacting compounds replace each other • Normally takes place in an aqueous solution • Also called metathesis reaction AX + BY AY + BX 2KI(aq) + Pb(NO3)2(aq) PBI2(s) + KNO3(g) Precipitate An insoluble solid May be represented in an equation by (s) or ↓ NaCl(aq) + AgNO3(aq) NaNO3(aq) + AgCl ↓ Episode 605 Classifying Reactions • Combustion • Involves the reaction of a substance with oxygen • Often called burning • The complete combustion of a hydrocarbon produces CO2 and H2O CxHy + O2 CO2 + H20 C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(l) CH4(g) + 2O2(g) CO2(g) + 2H2O(l) Episode 605