生物化學概要
Atom (原子)
Atoms are the basic unit of chemistry.
They consist of 3 smaller things:
• Protons (質子) - these are positively
charged (+)
• Electrons (電子) - these are negatively
charged (-)
• Neutrons (中子) - these have no charge
• These 3 smaller particles are arranged in
a particular way. In the center is the
Nucleus (原子核) where you find the
positive Protons and neutral Neutrons.
• In orbit (軌道) around the nucleus are the
Electrons. These are found in a series of
orbits (depending on the atom) with
differing numbers of electrons as seen
below.
原子的相互作用
• It's the electrons in orbit around the nucleus that
allow one atom to interact with other atoms so
they can be linked together.
• For example, H2O consists of an Oxygen atom
linked to 2 Hydrogen atoms. The linkage or
interaction between the electrons of the
Hydrogen and Oxygen atoms is called a
Chemical Bond (化學鍵結).
Chemical Bond (化學鍵結)
• Covalent (共價鍵):Two atoms share
electrons. Bonding of Oxygen and
Hydrogen in H2O.
• Ionic (離子鍵):Oppositely charged ions
are attracted to each other. Bond between
Na+ and Cl- in salt.
• Hydrogen (氫鍵):Forms between
oppositely charges portions of covalently
bonded hydrogen atoms.
極性和非極性物質
• A Polar substance has charges that can
interact with the charges in water.
Therefore Polar molecules are said to be
Hydrophilic (親水性) too (water loving).
• A Nonpolar substance then lacks any
charges and will not be able to interact
with water. Nonpolar molecules are said to
be Hydrophobic (厭水性) (water hating).
Dissolve (溶解)
pH (酸鹼值)
• Measure of Hydrogen Ions (H+) in solution:
The more Hydrogen Ions (H+) there are
the more acidic a solution is.
• Where do Hydrogen Ions (H+) come from?
Well, it's all part of water really...
• Water (H2O) splits into Hydrogen Ions (H+)
and Hydroxyl Ions (OH-).
• Anything that's too acidic or too basic will
degrade organic matter.
Oxidation (氧化) 和
Reduction (還原)
• The original view of oxidation and
reduction is that of adding or removing
oxygen. An alternative view is to describe
oxidation as the losing of electrons and
reduction as the gaining of electrons.
• One example in which this approach is of
value is in the high temperature reaction of
lead dioxide .
2Pb4+O2-2 -> 2Pb2+O2- + O02
In this reaction the lead atoms gain an
electron (reduction) while the oxygen loses
electrons (oxidation).
• If one reagent in a reaction contributes
oxygen, extracts hydrogen, or extracts
electrons, it is said to be an oxidizing
agent. Of course, it is reduced in the
process. Since oxidation and reduction are
symmetric processes, always occurring
together, there is always an oxidizing
agent and a reducing agent in the reaction.
• Oxidation agent (氧化劑)
Useful oxidizing agents are bleaches and
antiseptics.
Fluorine (氟) is the strongest oxidizing
agent.
• Reduction agent (還原劑)
Hydrogen gas is a very useful reducing
agent, used widely in the isolation of pure
metals by reduction.
• Important tasks are accomplished by
reducing agents as photographic
developers and antioxidants (抗氧化劑).
And in photosynthesis (光合作用), the
reducing agents accompish a task which is
essential to our survival.
• Lithium (鋰) is the strongest reducing
agent.
有機和無機化合物
• Organic molecules contain Carbon,
inorganic molecules do not.
• There are 4 basic organic compounds
used to build cells:
• Carbohydrates (醣類)
• Lipids (脂質)
• Proteins (蛋白質) are used to make
Enzymes
• Nucleic Acids (核酸)
混合物和化合物
• A mixture (混合物) is formed when two or
more different substances are mixed
together, while a compound (化合物) is
formed when two or more elements
become chemically combined together.
• Compounds react in definite amounts but
mixtures can be present in any amounts.
While it may be possible to separate
components of a mixture, it is often not
possible to convert back the elements of a
compound.
生物化合物主要類別
•
•
•
•
•
Major categories of bio-compounds:
Carbohydrates (醣類):
Lipids (脂質):
Proteins (蛋白質):
Nucleic acids (核酸):
Carbohydrates
• composed of carbon (C), hydrogen (H)
and oxygen (O).
• monosaccharides (單糖)
• disaccharide (雙糖)
• polysaccharide (多糖)
monosaccharides (單糖)
• the simplest form of sugar and are usually
colorless, water-soluble, crystalline solids, that
are absorbed directly into the bloodstream
during digestion.
• glucose (葡萄糖):
one of the main products of photosynthesis (光合
作用) and fuels for cellular respiration (呼吸作用).
Starch and cellulose (纖維素) are polymers (聚合
物) derived from the dehydration of D-glucose.
• fructose (果糖):
From plant sources, is found in honey, tree
and vine fruits, flowers, berries, and most
root vegetables. Commercially, fructose is
frequently derived from sugar cane (甘蔗),
sugar beets (甜菜), and maize (玉米).
Sucrose is a compound (化合物) with one
molecule of glucose covalently linked to
one molecule of fructose.
• galactose (半乳糖):
When combined with glucose, through a
condensation reaction, the result is the
disaccharide lactose (乳糖). In nature,
lactose is found primarily in milk and milk
products. #
disaccharide (雙糖)
• formed when two monosaccharides
undergo a condensation (濃縮) reaction
which involves the elimination of a small
molecule, such as water, from the
functional groups only.
• Three common examples are sucrose (蔗
糖), lactose (乳糖), and maltose (麥芽糖).
• sucrose (蔗糖):
The sugar from sugar cane and sugar
beets (sucrose) is made from glucose and
fructose.
• lactose (乳糖): milk sugar (lactose) is
made from glucose and galactose.
• maltose (麥芽糖):is made up of two
glucose molecules. #
polysaccharide (多糖)
• composed of long chains of
monosaccharide units bound together by
glycosidic bonds (糖苷鍵).
• Examples include storage polysaccharides
such as starch and glycogen, and
structural polysaccharides such as
cellulose and chitin (幾丁質).
• starch:
This polysaccharide is produced by most
green plants as an energy store. It is the
most common carbohydrate in human
diets and is contained in large amounts in
such staple foods (主食) as potatoes,
wheat, maize (corn), rice, and cassava (樹
薯).
• Cassava is the third largest source of food
carbohydrates in the tropics, after rice and
maize. Nigeria is the world's largest
producer of cassava, while Thailand is the
largest exporting country of dried cassava.
• Glycogen:
The polysaccharide structure represents
the main storage form of glucose in the
body, that serves as a form of energy
storage in animals and fungi (真菌).
• Glycogen is the analogue (類似物) of
starch, a glucose polymer in plants, having
a similar structure to amylopectin (支鏈澱
粉)(a component of starch) but more
extensively branched and compact than
starch.
• In humans, glycogen is made and stored
primarily in the cells of the liver and the
muscles, and functions as the secondary
long-term energy storage (with the primary
energy stores being fats held in adipose
tissue脂肪).
• Muscle glycogen is converted into glucose
by muscle cells and liver glycogen
converts to glucose for use throughout the
body including the central nervous system.
• Only the glycogen stored in the liver can
be made accessible to other organs. #
• Cellulose:
is an organic compound with the formula
(C6H10O5)n, consisting of a linear chain of
several hundred to over ten thousand Dglucose units. Cellulose is an important
structural component of the primary cell
wall of green plants, many forms of algae
and the oomycetes (卵菌).
•
•
•
Some species of bacteria secrete it to form
biofilms (生物膜). Cellulose is the most
abundant organic polymer on Earth.
Cellulose is mainly used to produce
paperboard (硬紙板) and paper.
Some animals, particularly ruminants (反芻)
and termites (白蟻), can digest cellulose with
the help of symbiotic micro-organisms that live
in their guts, such as Trichonympha (原生生物).
#
• Chitin:
It is the main component of the cell walls
of fungi, the exoskeletons of arthropods
(節肢動物) such as crustaceans (甲殼類動
物) (e.g., crabs, lobsters and shrimps) and
insects, the radulas (齒舌) of mollusks (軟
體動物), and the beaks and internal shells
of cephalopods (頭足類動物), including
squid (烏賊) and octopuses (章魚). ##
Lipids :
•
•
•
•
•
fats (脂肪)
oils (油)
essential oils (精油)
waxes (蠟)
cholesterol (膽固醇)
fats (脂肪)
• are generally soluble in organic solvents
and generally insoluble in water.
Chemically, fats are triglycerides (三酸甘油
酯): triesters of glycerol (甘油) and any of
several fatty acids. Fats may be either
solid or liquid at room temperature,
depending on their structure and
composition. fat is a subset of lipid.[
• Fats or lipids are broken down in the body by
enzymes called lipases (脂肪分解酵素)
produced in the pancreas (胰臟).
• fats can be categorized into saturated (飽和) fats
and unsaturated fats. Unsaturated fats can be
further divided into cis (順式) fats, which are the
most common in nature, and trans (反式) fats,
which are rare in nature but present in partially
hydrogenated (氫化) vegetable oils.
順式和反式
•
順式 (直線)
反式 (彎曲)
oils (油)
• "Oils" is usually used to refer to fats that
are liquids at normal room temperature,
while "fats" is usually used to refer to fats
that are solids at normal room temperature.
essential oils (精油)
• An essential oil is a concentrated
hydrophobic liquid containing volatile
aroma compounds from plants.
• Essential oils are generally extracted by
distillation, often by using steam. Other
processes include expression or solvent
extraction.
•
•
They are used in perfumes (香水),
cosmetics (化妝品), soaps and other
products, for flavoring food and drink,
and for adding scents to incense (香) and
household cleaning products.
Interest in essential oils has revived in
recent decades with the popularity of
aromatherapy (芳香療法).#
waxes (蠟)
• are a class of chemical compounds that
are plastic (malleable可塑性) near ambient
temperatures. They are also a type of lipid.
Characteristically, they melt above 45 °C
(113 °F) to give a low viscosity (黏性) liquid.
Waxes are insoluble in water but soluble in
organic, nonpolar solvents. All waxes are
organic compounds, both synthetic and
naturally occurring.
• Waxes are biosynthesized by many plants
and animals.
• Because they are mixtures (混合物),
naturally produced waxes are softer and
melt at lower temperatures than the pure
components.
Animal waxes
• The most commonly known animal wax is
beeswax, but other insects secrete waxes.
A major component of beeswax is the
ester myricyl palmitate (軟脂酸蜜蠟酯)
which is used in constructing their
honeycombs. Its melting point is 62-65 °C.
•
Spermaceti (鯨蠟油,鯨腦) occurs in large
amounts in the head oil of the sperm
whale (抹香鯨). One of its main
constituents is cetyl palmitate (鯨蠟棕櫚
酯), another ester of a fatty acid and a
fatty alcohol. Lanolin (羊毛脂) is a wax
obtained from wool, consisting of esters
of sterols (固醇).#
Plant waxes
• Especially in warm climates, plants
secrete waxes as a way to control
evaporation (蒸發) and hydration. From
the commercial perspective, the most
important wax is Carnauba wax (巴西棕櫚
蠟), a hard wax obtained from the Brazilian
palm Copernicia prunifera, Containing the
ester myricyl cerotate (軟脂酸蜂花酯).
• Other more specialized vegetable waxes
include candelilla wax (小燭樹蠟), ouricury
wax (小冠巴西棕櫚蠟), sugarcane wax,
retamo wax (阿根廷波尼西亞灌木蠟).
• The epicuticular (外表皮) waxes of plants
are mixtures of substituted long-chain
aliphatic (脂肪族) hydrocarbons,
containing alkanes (烷), fatty acids,
primary (CH2OH) and secondary (CHROH)
alcohols, diols (二元醇), ketones (酮),
aldehydes (醛).#
cholesterol (膽固醇)
• It is a sterol (固醇) (or modified steroid類固
醇), and an essential structural component
of animal cell membranes that is required
to establish proper membrane permeability
(通透性) and fluidity (流動性).
• also serves as a precursor (前驅物) for the
biosynthesis of steroid hormones, bile (膽)
acids, and vitamin D.
• Cholesterol is the principal sterol
synthesized by animals, all cells; in
vertebrates the liver typically produces
greater amounts than other cells. #
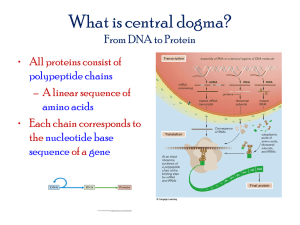
Proteins (蛋白質)
• consisting of one or more chains of amino
acid (氨基酸) residues.
• Proteins differ from one another primarily
in their sequence of amino acids, which is
dictated by the nucleotide (核苷酸)
sequence of their genes, and which
usually results in folding of the protein into
a specific three-dimensional structure that
determines its activity.
• A polypeptide (多胜鏈) is a single linear
polymer chain derived from the
condensation of amino acids. The
individual amino acid residues are bonded
together by peptide bonds (胜鍵) and
adjacent amino acid residues.
• The sequence of amino acid residues in a
protein is defined by the sequence of a
gene, which is encoded in the genetic
code (遺傳密碼).
• In general, the genetic code specifies 20
standard amino acids.
• Many proteins are enzymes that catalyze
biochemical reactions and are vital to
metabolism. Proteins also have structural
or mechanical functions, such as actin (肌
動蛋白) and myosin (肌凝蛋白) in muscle
and the proteins in the cytoskeleton (細胞
骨架), which form a system of scaffolding
that maintains cell shape.
• Other proteins are important in cell
signaling, immune responses, cell
adhesion, and the cell cycle.
• Proteins are also necessary in animals'
diets, since animals cannot synthesize all
the amino acids they need and must
obtain 10 essential amino acids from food.
• Through the process of digestion, animals
break down ingested protein into free
amino acids that are then used in
metabolism.##
Nucleic acids (核酸)
• Are essential for all known forms of life,
which include DNA (deoxyribonucleic acid,
去氧核糖核酸) and RNA (ribonucleic acid,
核糖核酸), are made from monomers
known as nucleotides (核苷酸).
• Each nucleotide has three components: a
5-carbon sugar (五碳糖), a phosphate
group (磷酸), and a nitrogenous base (鹼
基).
• Together with proteins, nucleic acids are
the most important biological
macromolecules; each is found in
abundance in all living things, where they
function in encoding (印碼), transmitting
(傳送) and expressing (表現) genetic
information.
• genetic information is conveyed through
the nucleic acid sequence (序列), or the
order of nucleotides within a DNA or RNA
molecule. Strings of nucleotides strung
together in a specific sequence are the
mechanism for storing and transmitting
hereditary, or genetic, information via
protein synthesis.
• Nucleic acids are generally very large
molecules. Indeed, DNA molecules are
probably the largest individual molecules
known. Well-studied biological nucleic acid
molecules range in size from 21
nucleotides (small interfering RNA) to
large chromosomes (human chromosome
1 is a single molecule that contains 247
million base pairs).
• In most cases, naturally occurring DNA
molecules are double-stranded and RNA
molecules are single-stranded. There are
numerous exceptions, however—some
viruses have genomes made of doublestranded RNA and other viruses have
single-stranded DNA genomes, and, in
some circumstances, nucleic acid
structures with three or four strands can
form.
•
•
the nucleobases found in the two nucleic
acid types are different: adenine (腺嘌呤),
cytosine (胞嘧啶), and guanine (鳥糞嘌呤
)are found in both RNA and DNA, while
thymine (胸腺嘧啶) occurs in DNA and
uracil (尿嘧啶) occurs in RNA.
DNA
• Deoxyribonucleic acid (DNA) is a nucleic
acid containing the genetic instructions
used in the development and functioning
of all known living organisms (with the
exception of RNA viruses). The DNA
segments carrying this genetic information
are called genes.
• DNA consists of two long polymers of
simple units called nucleotides, with
backbones made of sugars and phosphate
groups joined by ester bonds.
• Attached to each sugar is one of four
types of molecules called nucleobases
(informally, bases).
• It is the sequence of these four
nucleobases along the backbone that
encodes information. This information is
read using the genetic code, which
specifies the sequence of the amino acids
within proteins.
• The code is read by copying stretches of
DNA into the related nucleic acid RNA in a
process called transcription (轉錄). Within
cells DNA is organized into long structures
called chromosomes (染色體). During cell
division these chromosomes are
duplicated in the process of DNA
replication, providing each cell its own
complete set of chromosomes.
• Eucaryotic (真核) organisms (animals,
plants, fungi, and protists) store most of
their DNA inside the cell nucleus and
some of their DNA in organelles (胞器),
such as mitochondria (粒腺體) or
chloroplasts (葉綠體).
• Prokaryotes (原核) (bacteria and archaea
古細菌) store their DNA only in the
cytoplasm (細胞質).
• Bacterial chromosomes, plasmids (質體),
mitochondrial DNA and chloroplast DNA
are usually circular double-stranded DNA
molecules, while chromosomes of the
eukaryotic nucleus are usually linear
double-stranded DNA molecules. #
Chromosome (染色體)
• A chromosome is an organized structure
of DNA, protein, and RNA found in cells. It
is a single piece of coiled DNA containing
many genes, regulatory elements and
other nucleotide sequences.
Chromosomes also contain DNA-bound
proteins, which serve to package the DNA
and control its functions.
Human chromosomes
• Human cells have 23 pairs of
chromosomes (22 pairs of autosomes (體)
and one pair of sex chromosomes), giving
a total of 46 per cell. In addition to these,
human cells have many hundreds of
copies of the mitochondrial genome.
Sequencing of the human genome has
provided a great deal of information about
each of the chromosomes.
• In most species, including humans,
mtDNA is inherited solely from the mother.
• An egg contains 100,000 to 1,000,000
mtDNA molecules, whereas a sperm
contains only 100 to 1000.
• Whatever the mechanism, this single
parent (uniparental) pattern of mtDNA
inheritance is found in most animals, most
plants and in fungi as well. #
RNA
• Ribonucleic acid (RNA) functions in
converting genetic information from genes
into the amino acid sequences of proteins.
The three universal types of RNA include
transfer () RNA (tRNA), messenger RNA
(mRNA), and ribosomal RNA (rRNA).
Messenger (訊息) RNA acts to carry
genetic sequence information between
DNA and ribosomes, directing protein
synthesis.
• Ribosomal (核糖體) RNA is a major
component of the ribosome, and catalyzes
peptide bond formation. Transfer (運送)
RNA serves as the carrier molecule for
amino acids to be used in protein
synthesis, and is responsible for decoding
the mRNA. In addition, many other classes
of RNA are now known.###