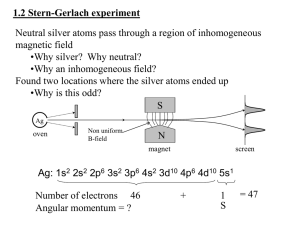
The Pauli Principle
advertisement

Solid State Chemistry
Chapter 3
Atomic Structure and Spectra
AGENDA
The structure and spectra of Hydrogenic atoms
The structure of many-Electron atoms
1. Pauli’s principle
2. Penetration and shielding
3. Building up principle
The spectra of complex atoms
1. Quantum defects
2. Singlet and triplets
3. Spin-orbit coupling
4. The total angular momentum
5. Term symbols and selection rules
The Structure of Many-electron Atoms
The Orbital Approximation
r1 , r2 , (r1 ) (r2 )
Justification 10.5
T heindividual orbitalsas resemblingthehydrogenicorbitals,
but corresponding to nuclear chargesmodifiedby thepresenceof all theotherelectrons
Helium atom:1s 2
The Pauli Principle
Pauliexclusionprinciple:
No more than twoelectronsmay occupyany givenorbitaland,
if two do occupyoneorbital,then their spinsmust be paired.
Pauli principle:
When the labels of any two identical fermions are
exchanged, the total wavefunction changes sign. When the
labels of any two identical bosons are exchanged, the total
wavefunction retains the same sign
(2,1) = -(1,2) for two electrons
Penetration and Shielding
s orbitalsgenerallylielowerin energy than p orbitalsof a given
shell,and p orbitalslielower thand orbitals
Effectivenuclear charge:
electronexperiences a shielded nuclear chargeshielding constant:
Z eff Z
Penetration and Shielding
A s electronhas a greaterpenetration
throughinnershell thana p electron:
s electronexperiences less shielding thana p electron
s pd f
Li atom:1s 2 2s1
Valence electrons: theelectronsin theoutermost
shell of an atomin it s ground state
The Building-up (Aufbou) Principle
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s
- Elect ronsoccupydifferentorbit als
of a given subshell before
doubly occupyingany one of t hem
Hund's maximummult iplicit y rule :
- An at omin it s ground st at eadopt s
a configuration wit h he
t great est
number of unpairedelect rons
- Spin correlat ion
- St rongelect ron- elect ronrepulsionsin 3d orbit als
Ionization Energies and Electron Affinities
- The first ionizationenergy,
I1
- The secondionizationenergy,I 2
Ionization Energies and Electron Affinities
- St andard ent halpyof ionization
ion H (T ) I
5
RT
2
- Elect ronaffinit yE ea : theenergy released
when an electronat t aches
t oa gas - phase at om
E ea 0 implies t hatelect ronat t achmentis
exot hermic
.
St andard ent halpyof elect rongain, eg H
5
RT
2
eg H ( X ) ion H ( X )
eg H (T ) Eea
Self-consistent Field Orbitals
Ze2
1
e2
V i
i
40 ri 2
40 rij
H (1)2 p (1) V (otherelectrons)2 p (1) V (exchangecorrection)2 p (1) E2 p 2 p (1)
•
•
•
The first term on the left is the
contribution of the kinetic energy and
the attraction of the electron to the
nucleus, just as in a hydrogenic atom
The second takes into account the
potential energy of the electron of
interest due to the electrons in the other
occupied orbitals
The third term takes into account the
spin correlation effects.
The Spectra of Complex Atoms
Quantum defects and ionization limits
Det ermination of ionizat ionenergies: at omicspect roscopy
Quant umdefect , : a purely empiricalquant it y
hcR
E ~
(n ) 2
Rydberg st at e: some excit edst at est hatare so diffuse t hat
1 n 2 variat ionis valid
I
R
~
v
2
hc n
Singlet and Triplet States
Hund's maxinummultiplicity rule
singlet
_(1, 2) (1 21 2 ) (1) (2) (1) (2)
(1) (2),
(1, 2) (1 21 2 ) (1) (2) (1) (2),
(1) (2)
The tripletstatelieslowerin energy that thesingletstate.
The originof energydifference : Spin correlation
The Spectrum of Atomic Helium
- Only excitedconfiguration :1s1nl1
- No radiativetransitions between singlet and tripletstates
Spin-Orbit Coupling
Spin- orbitcoupling: theinteraction of spinmagneticmoment
with themagneticfield arisingfrom the orbitalangularmomentum
The Total Angular Momentum
When thespin and orbitalangular momentaare nealy parallel,the totalangular momentumis high;
when thetwo angular momentaare opposed,the totalangular momentumis low
1
(when thetwo angular moment aare in thesame direction)
2
1
j l (when theyare opposed)
2
j l
Fine Structure
Spin - orbit couplingconstant,A
1
El , s , j hcA j ( j 1) l (l 1) s( s 1)
2
Strengthof thespin - orbit coupling
depends on thenuclear charge
- thecouplingincreasessharply
with atomicnumber (as Z4 )
Term Symbols and Selection Rules
1. T helet t er(for example,P or D in t heexamples)
indicat est he t ot alorbit alangular moment um
quant um number,L.
2. T heleft superscript in t het ermsymbol
(for example,t he2 in 2 P3 2 ) gives
t he mult iplicit y of t he t erm.
3. T heright subscript on t het ermsymbol
3
(for example,t he in 2 P3 2 ) is t he value of
2
t he t ot alangular moment umquant um number,J.
Total Orbital Angular Momentum
Total angular momentum quantum
number L
Angular momentum =
{L(L + 1)}1/2h
L = l1+l2, l1+l2-1….,|l1-l2|
Clebsch-Gordan series
L: 0,1,2,3,4,… (S,P,D,F,G…)
Example:
d2 electron
Multiplicity
Total spin angular momentum
quantum number S
Spin angular momentum =
{S(S + 1)}1/2h
S = s1+s2, s1+s2-1….,|s1-s2|
Multiplicity: 2S + 1
Example:
Two unpaired electrons
Total Angular Momentum
Total angular momentum quantum number
J
J = j where j = l + ½, |l – ½|
Example:
[Ne]3s1
[Ne]3p1
Russell-Saunders coupling:
If spin-orbit coupling is weak, then it is
effective only when all the orbital momenta
are operating cooperatively
J = L + S, L + S – 1,….., |L – S|
Example:
[Ne]2p13p1
Selection rules
S = 0 L = 0,±1 l = ±1 J = 0,±1
Short Summary
The Structures and spectra of many
electron atoms
1. The Pauli principle
2. Penetration and shielding
3. Singlet and triplet states
4. Spin-orbit coupling
5. Term symbols and selection rules
HW#2: 10.3d, 10.7d, Exe: 10.4b, 10.6a, 10.8a, 10.12b, 10.18b, 10.19a