3.3
Conversion Factors
Conversion Factors
What happens when a measurement
is multiplied by a conversion factor?
3.3
Conversion Factors
A conversion factor is a ratio of equivalent
measurements.
The ratios 100 cm/1 m and 1 m/100 cm are
examples of conversion factors.
3.3
Conversion Factors
When a measurement is multiplied by
a conversion factor, the numerical
value is generally changed, but the
actual size of the quantity measured
remains the same.
3.3
Conversion Factors
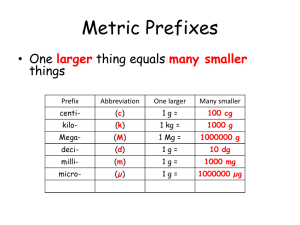
The scale of the micrograph is in nanometers.
Using the relationship 109 nm = 1 m, you can write
the following conversion factors.
3.3
Dimensional Analysis
Dimensional Analysis
Why is dimensional analysis useful?
3.3
Dimensional Analysis
Dimensional analysis is a way to analyze and
solve problems using the units, or dimensions,
of the measurements.
3.5
3.5
for Sample Problem 3.5
3.6
3.6
3.6
for Sample Problem 3.6
For Sample Problem 3.6
3.3
Converting Between Units
Converting Between Units
What types of problems are easily
solved by using dimensional
analysis?
3.3
Converting Between Units
Problems in which a measurement
with one unit is converted to an
equivalent measurement with another
unit are easily solved using
dimensional analysis.
3.7
3.7
3.7
for Sample Problem 3.7
3.3
Converting Between Units
Multistep Problems
When converting between units, it is often
necessary to use more than one conversion
factor.
3.8
3.8
3.8
for Sample Problem 3.8
3.3
Converting Between Units
Converting Complex Units
Many common measurements are expressed
as a ratio of two units. If you use dimensional
analysis, converting these complex units is just
as easy as converting single units. It will just
take multiple steps to arrive at an answer.
3.9
3.9
3.9
for Sample Problem 3.9
3.3 Section Quiz
1. 1 Mg = 1000 kg. Which of the following would
be a correct conversion factor for this
relationship?
a. 1000.
b. 1/1000.
c. ÷ 1000.
d. 1000 kg/1Mg.
3.3 Section Quiz
2. The conversion factor used to convert joules to
calories changes
a. the quantity of energy measured but not the
numerical value of the measurement.
b. neither the numerical value of the measurement
nor the quantity of energy measured.
c. the numerical value of the measurement but not
the quantity of energy measured.
d. both the numerical value of the measurement and
the quantity of energy measured.
3.3 Section Quiz
3. How many g are in 0.0134 g?
a. 1.34 10–4
b. 1.34 10–6
c. 1.34 106
d. 1.34 104
3.3 Section Quiz
4. Express the density 5.6 g/cm3 in kg/m3.
a. 5.6 106kg/m3
b. 5.6 103kg/m3
c. 0.56 kg/m3
d. 0.0056 kg/m3
Slide
33 of 43
© Copyright Pearson Prentice Hall
END OF SHOW