What cleaning criteria are used?
advertisement

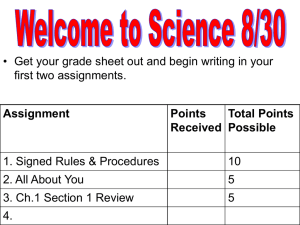
Implants, from cleaning to sterilization Dr. Abdess NAJI, Consultant, Auditor Medical Devices and Health Biotechnology abdess.naji@wanadoo.fr June 1st, 2012 IMPLANT-2012-Congress 1 What cleaning criteria are used? How can cleanliness be assessed? Validating the cleaning processes used New standards governing final cleaning: how will the intermediary cleaning stage be affected? June 1st, 2012 IMPLANT-2012-Congress 2 Definitions Cleaning : Action allowing to reach the lower organic and/or inorganic residues level. Cleanliness : Surface quality of product where the organic and/or inorganic residue level is lowest. Note : Do not confuse cleaning and disinfection micro-organisms elimination or inhibition Decontamination = cleaning + disinfection June 1st, 2012 IMPLANT-2012-Congress 3 ISO 13485 : 2003 requirements § 7.5.2.1 - The organization shall validate any processes for production and service provision where the resulting output cannot be verified by subsequent monitoring or measurement. This includes any processes where deficiencies become apparent only after the product is in use or the service has been delivered. Cleaning process validation is depending on its type, its purpose and its related risks. June 1st, 2012 IMPLANT-2012-Congress 4 Reference No specific standard available in Europe Standard published : XP S94-091 (September 2008) related to “Implants for surgery Validation of the cleaning of orthopaedic implants before final conditioning” Guideline/Recommendations for cleaning process validation. Some published guidance for pharmaceutical companies GMP Guidance - Europe FDA – 21 CFR 211 – Good Manufacturing Practices Document released by Health Canada : ‘’Directive on the validation of the cleaning processes’’ June 1st, 2012 IMPLANT-2012-Congress 5 Cleaning methods 1 - Decontamination = Cleaning + disinfection 2 - Rinsing 3 - Drying June 1st, 2012 IMPLANT-2012-Congress 6 1- Decontamination : Cleaning + disinfection Influence of : • Material surface • Irregularity, microscopic anfractuosities location • Chemical composition of material : polymer, plastic, rubber, metal, teflon • Capacity of germ adhesion • Biofilm formation • Using of chemicals reagents and solvents : • According to their activity spectrum • According to their not-aggressiveness and compatibility • Not containing aldehydes June 1st, 2012 IMPLANT-2012-Congress 7 Manual cleaning: • Steeping • Brushing • Ultrasounds using Automatic cleaning: • Avoid products handling • Giving regularity in cleaning quality Decontamination improves biocompatibility (non toxicity) and sterilization process June 1st, 2012 IMPLANT-2012-Congress 8 2- Rinsing Elimination of residues (organic substances, solvents ….) • Using of city water • Using of demineralised water • Using of ultra filtered water The quality of water must be controlled: • Maintenance of water installation • Microbiological control/endotoxins • Control water hardness (presence of limestone) • TOC control June 1st, 2012 IMPLANT-2012-Congress 9 3- Draining and drying • Avoid bacterial development • Avoid marks and imperfection • Avoid condensation during sterilization June 1st, 2012 IMPLANT-2012-Congress 10 June 1st, 2012 IMPLANT-2012-Congress 11 Osteoblast attachement on hostile surface June 1st, 2012 IMPLANT-2012-Congress 12 Cleaning process validation : Purpose To eliminate organic and/or inorganic residues present on medical device surface But also: To maintain cleaning process effectiveness all time To guarantee cleaning reproducibility and homogeneity for all manufactured batches To apply cleaning procedures easily To improve sterilization effectiveness June 1st, 2012 IMPLANT-2012-Congress 13 How to validate? Documented information : Identification of medical device physical and chemical characteristics (Geometry, constitution, porosity, used materials …) Identification of manufacturing processes (before the cleaning process) Identification of surface state before cleaning June 1st, 2012 IMPLANT-2012-Congress 14 General approach Product family Group of different product that can be given the same cleaning effectiveness - Definition of enclosure criteria : – Raw material – Surface state and geometric complexity – Components – Design and dimension of the product – Manufacturing process – Manufacturing environment – Production sites Taken into account all the variables that impact the cleaning effectiveness. - Designation of family representative product: Master product Equivalent product Simulated product June 1st, 2012 IMPLANT-2012-Congress 15 How to validate? Determination control method: Endotoxins rate Mineral pollution Organic pollution Visual aspect Residues toxicity … June 1st, 2012 IMPLANT-2012-Congress 16 How to validate? Cleaning process selected : Selection of adapted cleaning equipment Selection of adapted cleaning method Selection of active products or cleaning agents Determination of rinsing condition (to eliminate cleaning products and/or residues) June 1st, 2012 IMPLANT-2012-Congress 17 How to validate? Follow process validation methodology : Installation Qualification (IQ) Operational Qualification (OQ) Performance Qualification (PQ) June 1st, 2012 IMPLANT-2012-Congress 18 How to control products? Critical parameter measurement : Pyrogen test (Endotoxines control) Soluble and non soluble organic components TOC Rate (Total Organic Carbon), Heavy metal measurement Control water hardness (limestone presence) and conductivity Mineral residues Impact on passivation if needs Cytotoxicity tests … June 1st, 2012 IMPLANT-2012-Congress 19 How to control the process? Using adapted procedures to control: Equipments and infrastructure maintenance Staff qualification and training Environmental conditions June 1st, 2012 IMPLANT-2012-Congress 20 Cleaning process : Modification Every cleaning process modification should be validated: could affect process effectiveness and reproducibility Raw material changes Component or surface state change Manufacturing equipment change Manufacturing work environment change Manufacturing process change Cleaning product change June 1st, 2012 IMPLANT-2012-Congress 21 Cleaning process : Modification Methodology : Evaluation of any possible impact on risk management Determination if any new qualification must be done All re-qualifications must be documented If modification is not followed by any actions, the justification shall be documented June 1st, 2012 IMPLANT-2012-Congress 22 Thank you for your attention. Dr. Abdess NAJI Auditor / Expert Consultant MATIERE & SANTE 4, rue Paul Nizan 33140 VILLENAVE D'ORNON-FRANCE Tel/Fax : +33 5.56.87.88.91 Mobile : +33 6.16.41.25.01 E-mail : abdess.naji@wanadoo.fr June 1st, 2012 IMPLANT-2012-Congress 23