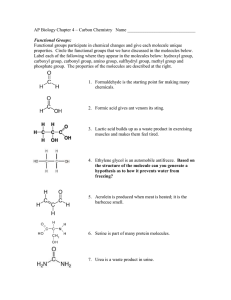
Water as a Solvent
• Water is a good solvent for charged and polar
substances:
– amino acids and peptides
– small alcohols
– carbohydrates
• Water is a poor solvent for nonpolar substances:
– nonpolar gases
– aromatic moieties
– aliphatic chains
Solubility of Polar and Nonpolar Solutes
Why are nonpolar molecules poorly soluble in water?
Dissolving Salts Involves Breaking
Ionic Interactions
Dissolving Salts Involves Breaking Ionic
Interactions
• Strong electrostatic interactions between the
solvated ions and water molecules lowers the
energy of the system
• Entropy increases as ordered crystal lattice is
dissolved
The Hydrophobic Effect
Water molecules adjacent to a hydrophobic molecule suffer restrictions in
orientation as they form hydrogen bonds with other water molecules
Chandler Nature 2005, Vol 437, p 640
The Hydrophobic Effect
Orientation of Water Around
a Nonpolar Solute
Caging
The Hydrophobic Effect
• A nonpolar solute "organizes" water
• The Hydrogen-bond network of water reorganizes
to accommodate the nonpolar solute
• This is an increase in "order" of water
• This is a decrease in ENTROPY
The Hydrophobic Effect
• Refers to the association or interaction of nonpolar
molecules or components of molecules in the
aqueous solution
• Is one of the main factors behind:
– protein folding
– protein-protein association
– formation of lipid micelles
– binding of steroid hormones to their receptors
• Does not arise because of some attractive direct
force between two nonpolar molecules
Hydrophobic Effect Favors Ligand Binding
• Binding sites in enzymes and receptors are often
hydrophobic.
• Such sites can bind hydrophobic substrates and
ligands, such as steroid hormones, which displace
water and increase entropy of the system.
• Many drugs are designed to take advantage of the
hydrophobic effect.
Amphiphilic Molecules
amphi both + pathos passion
also called "amphipathic"
• Refers to molecules that contain both polar
and nonpolar groups
• Equivalently - to molecules that are
attracted to both polar and nonpolar
environments
• Good examples - lipids
Water Surrounding Nonpolar Solutes Has
Lower Entropy
High entropy
Low
Entropy
Low entropy is thermodynamically unfavorable, thus hydrophobic
solutes have low solubility.
Origin of the Hydrophobic Effect
• Consider amphipathic lipids in
water.
• Lipid molecules disperse in the
solution; nonpolar tails of lipid
molecules are surrounded by
ordered water molecules.
• Entropy of the system
decreases.
• The system is now in an
unfavorable state.
Origin of the Hydrophobic Effect
• Nonpolar portions of the amphipathic molecule
aggregate so that fewer water molecules are ordered
and entropy increases.
• All nonpolar groups are sequestered from water, and the
released water molecules increase the entropy further.
• Only polar “head groups” are exposed.
Origin of the Hydrophobic Effect
• With high enough concentration of amphipathic
molecules, complete aggregation into micelles is
possible.
 0
0