%.
FORM TP 2014150
TEST CODE O2II2O2O
MAY/JLINE 2014
CARIBBEAN EXAMINATIONS COUNCIL
CARIBBEAII ADVAI\CED PROF'ICIENCY EXAMINATION@
CHEMISTRY
't
I
I
I
Ill[IT 1 - Paper 02
2hoursilominutes
..1 tr, EASr
oo-
s Ec,
tofon'+A
636-2585
LTBRAR
c.r.s.ET
READ THE FOLLOWING INSTRUCTIONS CAREFTJLLY.
1.
This paper consists of sIX questions in Two sections. Answer ALL
questions.
2.
For SectionA, write your answers in the spaces provided in this booklet.
3.
For section B, write your answers in the spaces provided at the end of
each question in this booklet.
4.
ALL working MUST be shown.
5.
You may use a silent, non-programmable calculator to answer questions.
6.
A data booklet is provided.
DO NOT TTIRN THIS PAGE UNTIL YOU ARE TOLD TO DO SO.
Copyright @ 2012 Caribbean Examinations Council
All rights reserved.
02112020/C'APB2014
-2SECTION A
AnswerALL questions.
Write your answers in the spaces provided in this booklet.
MODULE 1
...
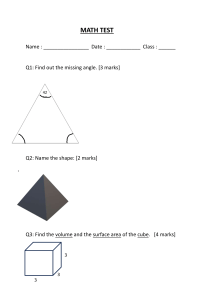
1.
(a)
_,
;. I
'i
i.,,.
FUNDAMENTALS IN crrEMIsrRY
Define-the'terms
. .'
(i)
mole
[2 marks]
(ii)
molar mass (include the units).
[2 marks]
(b)
A carbonate of a metal, M, has the formula M2CO3. The equation for the reaction of
with hydrochloric acid is given below.
MrCOt
MrCor(ag) + 2HCl(aq) + 2MCl(aq) + cor(e) + Hro(l)
6.125 gof M,CO, was used to prepare I dm3 of solution. 40 cm3 of the solution was
required to neutraiize exactly 23.6 cm3 of 0.150 mol dmr hydrochloric acid.
(i)
Calculate
a)
the number of moles of MrCOt
[2 marksl
GO ON TO THE NEXT PAGE
02tt2020/cAPE2014
-3-
b) the relative molecular mass of I\4rCOr.
[2 marksl
(ii)
J
I
Deduce the identity of M.
I
[2 marksl
(c)
Outline the experimental steps required to carry out the reaction described in (b) on
page2.
[5 marks]
Total 15 marks
GO ON TO THE NEXT PAGE
02112020/CAPE.20t4
-4MODULE 2
KINETICS AI\D EQUILIBRIA
2.
(a)
(i)
State the Bronsted-Lowry theory of acids and bases.
[1 markl
(ii)
Distinguish between a'strong acid'and a'weak acid'.
[2 marks]
(b)
25 cm3 of 0.1 mol dmr hydrochloric acid is titrated with 0.1 mol dmr of aqueous ammonia.
(D ln the space below, sketch the curve which is expected to illustrate the changes
that take Place during titration.
[2 marksl
GO ON TO THE NEXT PAGE
02rr2020/cAPE2014
---t
-5-
(iD
Suggest an indicator, glving a reason for your choice.
[2 marks]
(c)
A solution of a halic acid, HOX, readily decomposes at room temperature with oxygen
as the only gaseous product. The data in the table below applies to the decomposition of
aqueous HOX at room temperature.
Experiment
(i)
Initial Concentration of HOX
(mol dm-3)
Initial Rate
(mol dm-r 5-t1
I
1.6 x 10-3
0.t2
II
2.4 x l0-3
0.18
m
3.2 x l0-3
0.24
Deduce the order of the reaction.
A
o${
o
'1,:'J;; oron^'Yl
636-25A5
c. E. S. E. C.
[1 mark]
(iD Calculate the rate constant for the decomposition.
[2 marksl
02t12020/cAPE2014
GO ON TO THE NEXT PAGE
-6-
(d)
(D
Magnesium filings were added to the remaining solution at2 (c). What observation
is expected?
[l markl
(iD Outline an alternative experimental method to determine the rate of decomposition
of HOX at room temperature.
[4 marksl
Total 15 marks
GO ON TO THE NEXT PAGE
02r12020/cAPE2014
- nt-
MODULE 3
CHENIISTRY OF THE ELEMENTS
3.
(a)
Define the term 'transition element'.
I
[2 marksl
(b)
Cisplatin, [PtCNq)rClrJ, is a drug used in cancer treatrnent.
(i)
Deduce the oxidation number of the platinum atom.
[l markl
(iD Suggest the shape of the cisplatin molecule.
[1 markl
(c)
(D Complete the diagrams below by inserting,ffiolvs:to.-gho.w the electronic
configuration of EACH ion.
,,, r,,, ,'-' 4 i ,..
,{*t'" 4J.r:
'
$ :. *.. ;l r: i{
,1..
3d
a)
Fe2* : [Ar]
b)
Mnz* : [Ar]
-'4s
:l'l
tl
[2 marks]
(ii)
Use the information in (c) (i) b) to explain why Mn2* ions resist oxidation to Mn3*
ions.
[2 marksl
GO ON TO THE NEXT PAGE
02112020/CAPE2014
-8-
(d)
A sequence of reactions involving compounds of cobalt, with ions labelled A, B, C and
D, is shown below.
c
A
HrO
NH3
I
[Co(OH)o]](aq) + [Co(HrO)u]'*(aq) s
Cl-
[Co(NHr)u]'*(aq) <r [Co(Cl)J'-(aC)
Blue
(i)
List the colour of the following ions:
B:
C:
D:
[3 marks]
(iD Sketch the arrangement of bonds in the ion labelled C.
ND4et
""u"intt$:
[e.s.e.c.
[2 marks]
(e)
State the changes that would be observed when a solution of copper(Il) ions is treated with
concentrated hydrochloric acid followed by concentrated ammonia.
[2 marks]
Total 15 marks
GO ON TO THE NEXT PAGE
02r12020/cAPE2014
-9-
i
SECTION B
AnswerALL questions.
Write your answers in the spaces provided at the end of each question.
MODULE 1
FTJhIDAMENTALS IN CHEMISTRY
4.
(a)
Carbon combines with hydrogen covalently to form a gas, ethane.
(i)
State the electronic configuration of carbon in its ground state using sp notation.
[1 markl
-t
(iD Account for the number and type of orbitals around EACH carbon atom in a
molecule of ethane.
[3 marksl
(iiD Use the VSEPR theory to deduce the arrangement of the orbitals and the bond
angles around EACH carbon atom in a molecule of dichloroethane, CHTCICILCL
[3 marks]
(b)
(i)
State THREE assumptions of the kinetic theory as it pertains to ideal gases.
[3 marks]
(iD Under what conditions of temperature and pressure will a real gas deviate from
ideal gas behaviour?
(c)
[1 mark]
A gas with a mass of 0.299 g was collected in a 400 cm3 container at a pressure of
4.2 x I04 Pa and a temperature of 25 "C.
Calculate the relative molecular mass (M) of the gas.
[R: 8.31 J K-r mol-t]
[4 marksl
Total 15 marks
Write the answer to Question 4 here.
(a)
(i)
(iD
GO ON TO TI{E NEXT PAGE
02rr2020/cAPE20t4
- l0Write the answer to Question 4 here.
(a)
(iiD
(b) (D
GO ON TO THE NEXT PAGE
02rr2020lcAPE2014
- ll Write the answer to Question 4 here.
(b)
(iD
I
I
I
I
I
I
I
I
I
"t
(c)
I
I
GO ON TO THE NEXT PAGE
02tr2020/cAPE 2014
-12MODULE 2
KINETICS AI\D EQUILIBRIA
5.
(a)
o)
Define the terms
(D solubility
[2 marksl
(iD solubility product.
[2 marks]
At 25 oC, a saturated solution of lead(Il) azide, Pb(Nr)r, is prepared by dissolving
0.025 gin water to make 100.0 cm3 of solution.
(D Calculate the solubility, in mol dm-3, of the lead(Il) azide.
[2 marksl
(iD Lead(tr) azide dissociates in a saturated solution according to the following equation:
eb(Nr)r(s) .r
(c)
Pb2*(aq + 2Nr-(a9)
Write the expression for the solubility product of lead(Il) azide.
[2 marksl
(iii)
calculate the solubiliry product of lead(Il) azide.
[2 marksl
(D
Sketch a diagram showing the distribution of energies in a gas at a given
[2 marksl
temperature, T,.
(iD
On the same diagram as in 5 (c) (i), insert another curve showing the distribution
[1 mark I
of energies of the molecules at a higher temperature, Tr.
(iiD Using your sketch, explain the difference in the rates of reaction at temperatures
T, and Tr.
[2 marksl
Total 15 marks
Write the answer to Question 5 here.
(a) (D
GO ON TO THE NEXT PAGE
02tt2020/cAPE2014
.-l
-13Write the answer to Question 5 here.
(a)
(ii)
(b)
(i)
i
)
I
I
I
I
I
ll
,l
I
I
i',l
rJ
l.
I
t't.
(ii)
""*^:iB#oNotet
c, E. s. E. c.
,J
l.
,-t
II
(iiD
I
'l
I
I
J
I
j
I
I
:
GO ON TO THE NEXT PAGE
02112020/C.APE20t4
:
-14Write the answer to Question 5 here.
(c) (D-(iD
(iii)
GO ON TO THE NEXT PAGE
021L2020/CAPE2014
j
-15MODULE 3
CHEMISTRY OF THE ELEMENTS
-j
I
J
I
6.
I
I
(a)
(b)
t
-t
Define the term 'polarization'.
(i)
State the trend in ionic size of the elements from Na to
Al.
[1 markl
(iD Explain the following observation. (Include relevant equations to support your
I
answer.)
I
The pH of an aqueous solution of sodium chloride is 7, while the
corresponding value for aluminium chloride is 3.
[7 marksl
I
I
I
'l
[2 marks]
(c)
I
I
t
When a stream of chlorine gas is passed through an aqueous solution of a metal halide,
A, of molecular mass 212.5, a reddish-brown solution, B, is formed. Shaking B with
tetrachloromethane in a separatory funnel produces C, a purple organic layer, and D, a
colourless aqueous layer.
(D
State the name of the halide ion inA.
[1 markl
(ii)
Write the formula equation for the reaction ofA with chlorine.
[2 marksl
(iiD Describe an additional simple test to confirm the identity of the halide ion inA.
[2 marksl
Total 15 marks
Write the answer to Question 6 here.
I
(a)
I
J
J
J
l
l
GO ON TO THE NEXT PAGE
02112020/CAPE 2014
- 16Write the answer to Question 6 here.
(b)
(i)
(ii)
GO ON TO THE NEXT PAGE
021r2020/cAPE 2014
-r7Write the answer to Question 6 here.
(c)
(i)
(ii)
(iiD
F eAlr_ll3o"o.
i.I
I
I
636'2585
I
I
J
c. E. S. E.C.
I
I
i
J
I
I
I
I
j
END OF TEST
l
:
i
I
IF YOU FINISH BEFORE TIME IS CALLED, CHECK YOUR WORK ON THIS TEST.
02tt2020/cAPE 2014
I
j