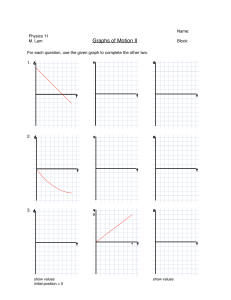
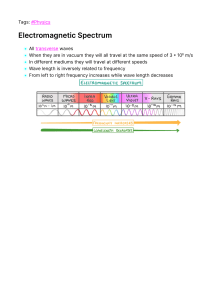
PHYSICS CXC REVISION SPEEDRUN A base (or fundamental) quantity is one that cannot be expressed as another quantity, e.g. length, time. A derived quantity is one that is a combination of base quantities. e.g. ‘speed’ is distance per unit time. BASE QUANTITIES • Length (L), unit = m • Mass (m), unit = kg • Time (t), unit = s • Current (I), unit = A • Temperature (T), unit = K • Amount of a substance (n), unit = mol • Luminous intensity (lv), unit = candela (cd) DERIVED QUANTITIES • Area (A), unit = m2 (m x m) • Volume (V), unit = m3 (m x m x m) • Pressure (P), unit = Pa (N/m2) • Force (F), unit = N (kg m/s2) • Charge (Q), unit = C (A x s) Prefixes • Micro (µ) = 10-6 (÷ 1,000,000) • Milli (m) = 10-3 (÷ 1000) • Centi (c) = 10-2 (÷ 100) • Kilo (k) = 103 (x 1000) • Mega(M) = 106 (x 1,000,000) Prefixes (examples) 5600ms to s = 5600 ÷ 1000 = 5.6s 300cm to m = 300 ÷ 100 = 3m 2.5kW to W = 2.5 x 1000 = 2500 W Scalar is a quantity that has magnitude but NO direction. e.g. distance, speed, area, volume, density Vector is a quantity that has BOTH magnitude and direction. e.g. displacement, velocity, acceleration, force, momentum Sum of parallel and anti-parallel vectors: Resolving non-parallel vectors Mass (unit = kg) The amount of matter contained in an object. It is a measure of an object’s inertia or resistance to change in motion. Weight (unit = N) The force exerted on a body’s mass by gravity. w = mg e.g. If an astronaut has a mass of 70kg on Earth, what is her weight on the Moon if its gravitational field strength is 1.6 N/kg? 70 x 1.6 = 112 N The centre of gravity of an object can be defined as the point at which the weight of a body acts. Wide base (low centre of gravity) = higher stability • Forces enable masses to overcome inertia, i.e. they are able to cause a change in an object’s acceleration, deceleration or direction (even shape and size, but NOT mass) • Forces are measured in Newtons (N) which can be derived as 1N = 1 kg m/s2. Type of Force GRAVITY WEIGHT FRICTION BUOYANCY / UPTHRUST ELECTROSTATIC Description MAGNETIC REACTION An attraction or repulsion caused by north and south poles. The force that always acts opposite to another, e.g. the forward push from swimming while pushing the water backward. TENSION CENTRIPETAL NUCLEAR An upward force exerted on a string or rope attached to a load. The pull towards a central point for an object moving in a circle. The attraction holding the nucleus of an atom together. Pulls objects towards the centre of the Earth. The effect of gravity on an object’s mass. The resistance an object experiences when rubbing a surface. The upward force exerted by a fluid. Attraction due to charged particles called electrons stored in an object. If a force is absent, there will be no change in motion or direction. In the car, two “resistant forces” are equal to the applied forward thrust of the car. This will give an overall resultant or net force of 0 Newtons. The car is at equilibrium and will keep moving at a constant velocity. ACCELERATION DUE TO GRAVITY In a vacuum, objects (whether it be a rock or paper) will accelerate and fall at the same rate. In air, the force of air resistance will affect falling speeds. GRAVITATIONAL CONSTANT (g) The acceleration due to gravity on Earth is 10 m/s2. This means that with each second under the force of only gravity, the velocity increases by 10 m/s. So, after 3s in freefall (with negligible air resistance), the final velocity would be 30 m/s. LEVERS are simple machines that have an input and an output. The input force is usually referred to as the EFFORT and the output as the LOAD. Levers have a point of rotation that these forces will turn about. This point of rotation is referred to as the PIVOT or FULCRUM. The Principle of Moments states that: THE SUM OF CLOCKWISE AND ANTICLOCKWISE MOMENTS ABOUT THE SAME PIVOT IS EQUAL AT EQUILIBRIUM. Calculating the effort, E: F1 x D1 (Effort) = F2 x D2 (Load) E x (0.40 + 0.80) = 600 x 0.80 1.20 E = 480 E = 480 / 0.12 = 400 N If C is the centre of gravity of the plank, calculate the weight of the plank. F1D1 + F2D2 = F3D3 (500 x 2.0) + (300 x 0.5) = F3 x 0.75 1000 + 150 = 0.75 F3 1150 = 0.75 F3 F3 F3 = 1150 / 0.75 = 1533.33 N Hooke’s Law states that: The extension of a spring is directly proportional to the force acting on it, given it is within its elastic limit. F = kx F = Force (N) x = extension (m) k = spring constant (N/m) The elastic limit of a spring is within the limit of proportionality between force and extension. If too much force is applied, the spring can experience permanent deformation. Hooke’s Law (worked example) A 200 N load is attached to a spring with an initial length of 100 cm. If its spring constant (k) is 5 N/cm, calculate the final length, L, of the spring. F = kx x=F÷k = 200 ÷ 5 = 40 cm L = 40 + 100 = 140cm Density is defined as mass per unit volume. The SI unit is kg/m3. Relative density is a given ratio of the density of a substance in reference to the density of the medium. e.g. Calculate the R.D. of oil in seawater, if oil has a density of 800 kg/m3 and seawater has a density of 1020 kg/m 3. R.D. = Density of oil / Density of seawater = 800 / 1020 = 0.78 (no unit) Archimedes’ Principle states that: The upward buoyant force that is exerted on an object immersed in a fluid is equal to the weight of fluid displaced. Floating occurs if an object’s buoyant force is greater than its weight, or if it is less dense than its medium. More surface area = more likely to float Sinking occurs if an object’s weight is greater than its buoyant force, or if it is denser than its medium. Distance is how much ground an object has covered despite direction. It is a scalar. Displacement is the overall change in position of an object in a straight line between its origin and destination. It is a vector. In the displacement-time graph, the gradient represents velocity. Gradient of upward slope: y2 – y1 = 25 - 0 x2 – x1 5– 0 = 5 m/s (velocity) Speed is distance per unit time. It is a scalar. Velocity is the displacement per unit time. It is a vector. Speed = distance / time Velocity = displacement / time Unit = m/s Acceleration is a change in velocity over time. The unit is m/s2. a = (v – u) t v = final velocity u = initial velocity Calculate a car’s acceleration after braking for 4 seconds if the velocity decreased from 24 m/s to 16 m/s. a = (16 – 24) = -8 4 4 = -2 m/s2 In the velocity-time graph, the gradient represents acceleration. In the velocity-time graph, the displacement travelled is represented as the area under the graph. The unit is metres (m). Newton’s 1st Law An object at rest remains at rest, or an object in motion remains in motion at a constant velocity, unless an unbalanced force acts upon it. Newton’s 2nd Law (example) A 2000 kg minibus increases its velocity from 8 m/s to 14 m/s in half of a minute. Calculate the force required to do this. a = (v – u) / t = (14 – 8) / 30s = 6 / 30 = 0.2 m/s2 F = ma = 2000 x 0.2 = 400N Newton’s 2nd Law The force on a body is directly proportional to its acceleration. (F = ma) Newton’s 3rd Law • Every action force has an equal and opposite reaction force. • If a Body A acts on Body B, then B exerts an equal and opposite force on A. Linear momentum (p) is the product of mass and velocity for an object travelling in a straight line. Impulse is the force that produces a change in momentum. Momentum = mass x velocity p = mv ▲p = F x t Unit = kg m/s or Ns The law of conservation of linear momentum states that: With no external forces, the total momentum before collision is equal to the total momentum after collision. If the wreckage moves as a combined mass after collision, calculate its net velocity and direction. Energy is simply defined as the capacity for doing work. The SI unit for energy is Joule (J). A Joule is defined as the work needed to move 1N by a 1m distance, so 1J = 1Nm. The law of conservation of energy states that Energy can neither be created nor destroyed. It can only be converted from one form to another. Some types of energy: - Chemical, e.g. batteries, fuel - Kinetic, e.g. moving car, falling water - Gravitational potential, e.g. boulder on cliff - Elastic potential, e.g. compressed spring - Thermal, e.g. fire - Electromagnetic, e.g. sunlight - Nuclear, e.g. fission reactors Work is energy used to produce some type of mechanical change. Measured in Joules (J). Power is the rate of energy conversion. Measured in Watts (W). A 500 N package is lifted 20 m above the ground. This takes 25s to occur. Calculate the work done and power. W = F x d = 500 x 20 = 10 000 J P =W/t = 10 000 / 25 = 400 W (or 400 J/s) EFFICIENCY refers to the percentage or ratio of useful output compared to total supplied input. KINETIC ENERGY (KE) is the energy possessed by an object in motion or during collision. GRAVITATIONAL POTENTIAL ENERGY (GPE) is the energy possessed by an object by virtue of its position or height. KE and GPE Conversions A coconut of mass 2 kg falls from a distance of 30m to 10m. Assuming negligible air resistance, calculate its velocity as it falls. (g = 10 N/kg) ▲GPE = mg▲h = 2 x 10 x (30 – 10) = 20 x 20 = 400 J 100% of GPE converts to KE KE = ½ mv2 v2 = KE / (½m) = 400 / 1 = 400 v = √400 = 20 m/s KE and GPE Conversions • • • • • 1 – Maximum GPE, minimum KE 2 – GPE converting to KE 3 – Minimum GPE, maximum KE 4 – KE converting to GPE 5 – Maximum GPE, minimum KE ALTERNATIVE ENERGY SOURCES - Solar panels. Converts sunlight to electric. - Hydroelectric dams. Converts KE from water to electric. - Geothermal. Uses KE from steam wells below surface. - Tidal. Uses natural ebb of ocean and waves. - Biofuels. Uses methane or ethanol from harvests. - Wind turbines. Uses KE from natural winds. - Nuclear. Uranium fission yields high amounts of energy. Reducing energy wastage – Switching from incandescent to fluorescent lights – Carpooling and public transport – Turning off lights or A.C. unit when not in use – Use more energy-efficient appliances – Hybrid or electric vehicles Pressure is force acting per unit area. Pressure in a fluid increases with depth and density of the fluid. Calculate the total pressure at the base of a 20m deep container of oil of density 800 kg/m 3 if the atmospheric pressure is given as 101 kPa. P = density x g x h = 800 x 10 x 20 = 160,000 Pa Total pressure = 160,000 + 101,000 = 261,000 Pa Manometers are used to find pressures of fluids of interest. The difference in column height is used to calculate pressure. Pascal’s Law states: The pressure applied to a point in an incompressible fluid is evenly distributed to all points in that fluid. Wider area = Larger force The pressure exerted by the fluid is equal at all points. HEAT is a form of energy that is transferred from areas of higher temperature to lower temperature until the objects and their surroundings are at equilibrium, the same temperature. HEAT energy is converted from mechanical energy. The scientist, James Joule, proved this with the experiment set-up below. THE KINETIC THEORY OF MATTER indicates that molecules in a gas move freely and rapidly along straight lines. In a liquid and solid, intermolecular bonds form, which reduce movement in the medium. COMPARING SOLIDS, LIQUIDS, GASES Temperature represents the average kinetic energy per molecule in a substance. Its SI unit is Kelvin (K). To convert to Kelvin, add 273 to the Celsius value, e.g. 100oC = 100 + 273 = 373 K Lower fixed point is the temperature of pure melting ice at 1 atmosphere. Its value is 0oC or 273 K. Upper fixed point is the temperature of pure dry steam at 1 atmosphere. Its value is 100oC or 373 K. Absolute zero is the temperature at which there is heat or internal energy in the molecules. Its value is -273oC or 0 K. At this point, the pressure is zero. A thermometer has a physical property that changes steadily with temperature e.g. when mercury is heated, it expands proportionately and moves along the bore of the thermometer. A clinical thermometer has a thinner, larger bulb (faster conduction), a narrower bore (more sensitive readings) and a constriction (prevents liquid from immediately returning to bulb). A thermocouple uses difference in voltage between hot and cold junctions for rapid, accurate readings in temperature. CONDUCTION is the transfer of heat through the vibrations of molecules to adjacent molecules in a medium. CONVECTION is the transfer of heat through bulk movement of the medium itself. RADIATION is the transfer of heat through the propagation of electromagnetic waves. It can occur through a vacuum. Conductors are materials with free electrons (such as metals), which allow the efficient transfer of heat. Insulators do not have many free electrons and may have structural gaps or air spaces that do not efficiently transfer heat, such as cloth or polystyrene. Air is a POOR CONDUCTOR of heat. Vacuum flasks keep liquids at constant temperatures by limiting heat transfer. Good absorbers (and good emitters) of heat are usually: - Black in colour - Rough in texture - Small in area - Dull in nature Good reflectors of heat are usually: - White in colour - Smooth in texture - Large in area - Shiny in nature Specific Heat Capacity of a material is defined as the amount of heat energy required to change the temperature of 1kg of the material by 1 Kelvin. e.g. Water has a specific heat capacity of 4200 J/kg K. Specific heat capacity is a constant. If the temperature of 1kg of water is to be raised from 10oC to 11oC, it would require 4200 J. To raise it from 10oC to 12oC, it would require 8400 J. Example question: A 1.2 kW heater was used to heat a 2 kg mass of water for 5 minutes. The initial temperature of the water is 20oC. If the specific heat capacity of water is 4200 J/kg K, calculate the final temperature of the water. E = mc▲T P=E÷t ▲T = E ÷ (mc) E = P x t = 1200 W x (5 x 60s) = 360,000 ÷ (2 x 4200) = 360,000 J = 42.86oC T (final) = 20 + 42.86 = 62.86oC Heat capacity of a body is defined as the amount of heat energy required to change the temperature of the body by 1 Kelvin. e.g. Water has a specific heat capacity of 4200 J/kg K. And a large container of water has 10 kg. It would require 42000 J (4200 x 10) to change the raise the temperature of the 10 kg container of water by 1 Kelvin. Heat capacity of a body is defined as the amount of heat energy required to change the temperature of the body by 1 Kelvin. e.g. Water has a specific heat capacity of 4200 J/kg K. And a large container of water has 10 kg. It would require 42000 J (4200 x 10) to change the raise the temperature of the 10 kg container of water by 1 Kelvin. Testing for specific heat capacity The metal aluminum block is heated for 3 minutes with a 5A current, 10V supply. If the initial and final temperatures of the 2kg block are 30oC and 35oC respectively, calculate its specific heat capacity. E = IVt = 5 x 10 x (3 x 60) = 9000 J E = mcΔT c = E ÷ (mΔT) = 9000 ÷ (2 x (35 – 30)) = 9000 ÷ 10 = 900 J/kg oC Method of Mixtures A 50g block is placed in 200g of water. The block was heated to 100oC. The temperature of the block dropped to 30oC and the water rose from 30oC to 35oC. Using 4.2J/g K as the specific heat capacity of water, calculate the specific heat capacity of the block, in J/g K. Heat gained by water = Heat lost by metal mcΔT (water) = mcΔT (substance) mcΔT (water) = mcΔT (block) 200 x 4.2 x 5 = 50 x c x (100 – 30) 4200 = 3500 c c = 4200 ÷ 3500 = 1.2 J/g K Specific latent heat of fusion is the energy required to change 1 kg of a solid to a liquid without a temperature change. Specific latent heat of vapourization is the energy required to change 1 kg of a liquid to a gas without a temperature change. The unit for each is J/kg. When latent heat is being released or absorbed, there is no temperature change. State of matter changes as molecular bonds are broken or reformed. EXAMPLE QUESTION A student heats 200g of ice at 0oC until it turns to steam at 100oC. How much energy was needed to do this? • • • [specific heat capacity of water = 4200 J/(kg K)] E = mLf = 0.2 x (3.36 x 105) = 67200 J E = mcΔT = 0.2 x 4200 x 100 = 84000 J [specific latent heat of fusion of ice = 3.36 x 105 J/kg] [specific latent heat of vapourization of water = 2.25 x 106 J/kg] E = mLv = 0.2 x (2.25 x 106) = 450 000 J E (total) = mLf + mcΔT + mLv = 601 200 J Air pressure is the result of collisions of gas molecules with the walls of the container the gas is held within. The higher the frequency of collisions with the walls, the higher the pressure. The number of gas molecules in the container will also increase the frequency of collisions and increase the pressure. Gas molecules in high temperature have more kinetic energy and thus, move faster, have more momentum and collide more often with the walls. Temperature and pressure have a directly proportional relationship. There are THREE GAS LAWS: 1. Boyle’s Law 2. Charles’ Law 3. Pressure Law And a combination of the three: The General (or Combined) Gas Law BOYLE’S LAW: For a fixed mass of gas at constant temperature, pressure and volume are inversely proportional. Basically: Low volume = high pressure High volume = low pressure BOYLE’S LAW (example) A syringe at a fixed temperature has a gas at an initial volume of 50ml at 4 kPa. At what volume would the pressure be 10 kPa? 50 x 4 = 10 x V2 200 = 10 V2 V2 = 200 / 10 V2 = 20ml CHARLES’ LAW: For a fixed mass of gas at constant pressure, volume and temperature are directly proportional. Basically: High temperature = high volume Low temperature = low volume CHARLES’ LAW (example) When filled with 24m3 of helium at a temperature of 32oC, a hot air balloon is able to float. What is the volume of the balloon at 75oC? PRESSURE LAW: For a fixed mass of gas at constant volume, pressure and temperature are directly proportional. Basically: High temperature = high pressure Low temperature = low pressure PRESSURE LAW (example) A car tyre is pumped to a pressure of 2 x 105 Nm-2 when the temperature is 23oC. Later in the day, the temp. rises to 34oC. Calculate the new pressure in the tyre if the volume was constant. COMBINED GAS LAW: In this scenario, no factor except the mass of the gas is constant. You can always start with this law for any gas law problem. Just eliminate the quantity that is constant, e.g. eliminate T1 and T2 for Boyle’s Law. COMBINED GAS LAW (example) An ideal gas has a volume of 150 cm3 at a temperature of 300 K and a pressure equivalent to 76 cm Hg. What will its volume be if the temperature goes up to 320 K and the pressure drops to 70 cm Hg? What is a WAVE? A wave is a propagation of energy from one location to another. ANATOMY OF A WAVE: The wavelength (λ) is the distance between any two successive points in phase. The amplitude (A) is the height of the wave, or the maximum displacement of the oscillation. ANATOMY OF A WAVE: The period (T) is the time taken for one complete oscillation. In the graph, it is 0.6 seconds. The frequency (f) is number of oscillations passing a point per second. f=1÷T = 1 ÷ 0.6 = 1.67 Hz or 1.67 waves per second PERIOD (worked example) The maximum audible range of a human is 20,000 Hz. Calculate the period of the sound wave. T=1÷f = 1 ÷ 20 000 = 0.00005 seconds ANATOMY OF A WAVE: High frequency waves have more oscillations per second. High amplitude waves are ‘taller’, have greater displacements. VELOCITY OF A WAVE (example): If a sound wave has a frequency of 15 Hz and a wavelength of 22 m, calculate its velocity. v=fλ = 15 x 22 = 330 m/s TRANSVERSE WAVES Are waves with crests and troughs; and the displacement of particles is perpendicular to its propagation. Examples are: light, radio waves, gamma rays, X-rays. LONGITUDINAL WAVES Are waves with compressions and rarefactions; and the displacement of particles is parallel to its propagation. An example is sound. SOUND WAVES They are longitudinal and mechanical in nature. Mechanical means that they require a medium to propagate. It cannot propagate through a vacuum. The denser the medium, the higher the velocity. Sound travels fastest in a solid and slowest in a gas. PITCH AND LOUDNESS Pitch correlates with frequency. Loudness correlates with amplitude. The human audible range Is 20 Hz – 20 kHz. A sound that exceeds this is called ultrasound. ECHOES An echo is a reflection of sound. It can be used in SONAR to detect sea depths. An ultrasound signal is sent via a transducer, reflects off a dense surface and is picked back up by a detector. The speed is calculated by: ECHOES (example problem) In an experiment, Ravi stands 60m away from a wall and claps two blocks together 20 times. Chantal records the time for the 20 echoes as 7.2 seconds. Calculate the speed of sound from this data. Time for 1 echo = 7.2 ÷ 20 = 0.36s Speed = 2d ÷ t = (60 x 2) ÷ 0.36 = 333.33 m/s ELECTROMAGNETIC SPECTRUM From lowest frequency to highest frequency (or longest wavelength to shortest) ACRONYM RICH MEN IN VEGAS USE XPENSIVE GADGETS: RADIO – broadcasting, RADAR MICROWAVES – cellular communications INFRA-RED – heat emission, night vision VISIBLE LIGHT – optical fibres, human eye vision ULTRAVIOLET – sterilization of equipment, tanning beds X-RAYS – radiographs of bones (due to electron bombardment) GAMMA RAYS – killing of cancer cells, radioactive decay DIFFRACTION Occurs when a wave passes through a narrow aperture (opening) and thus spreads out over a large area as it continues to progress. All waves can undergo diffraction. INTERFERENCE Occurs when two waves superpose with each other to form a resultant wave that might either raise or lower the amplitude. Constructive interference means that the waves are exactly superposed. Destructive interference means that the waves are out of phase by ½ of a wavelength. THEORIES OF LIGHT Newton – “Light is a stream of particles or corpuscles.” Huygens – “Light is a transverse wave.” Young – “Light is a wave that can undergo interference.” Einstein – “Light can behave as both a wave and a particle.” (Quantum Theory) YOUNG’S DOUBLE SLIT EXPERIMENT Where the waves are in phase (CONSTRUCTIVE interference), the crests and troughs are aligned and result in bright fringes (called MAXIMAS). Where the waves are out of phase (DESTRUCTIVE interference), they result in dark fringes (called MINIMAS). Troughs of one wave ‘cancel’ out the crests of another. REFLECTION OF LIGHT Occurs when an incident light ray bounces off a surface. A normal is an imaginary line perpendicular (or 90o) to a boundary or surface. All relevant angles are measured from the normal to the ray. REFLECTED IMAGES are: 1. The same height as the object 2. The same distance from the mirror as the object 3. Laterally inverted (flipped sideways) 4. Virtual (cannot be projected) LAWS OF REFLECTION 1. The incident ray, reflected ray and normal all lie on the same plane. 2. The angle of incidence is equal to the angle of reflection. REFRACTION OF LIGHT In the glass block, the emergent ray is parallel to the incident ray. Lateral displacement is the perpendicular distance the ray shifted from its initial path. The denser the medium, the greater the lateral displacement. If a wave moves into a denser medium, its: - Wavelength decreases - Speed decreases - Frequency remains unchanged. DISPERSION OF LIGHT Occurs when light comprised of various wavelengths passes through a prism. The light splits into different colours. Red has the longest wavelength and is refracted the least. Violet has the shortest wavelength and is refracted the most. REFRACTIVE INDEX The refractive index (n) is the ratio of the sines of the angles of incidence and refraction of a ray passing from one medium to another. REFRACTIVE INDEX (worked examples) Calculate the refractive index from air to glass. REFRACTIVE INDEX (worked examples) Calculate the angle of refraction if n = 1.50. LAWS OF REFRACTION 1. The incident ray, refracted ray and normal all lie on the same plane. 2. The refractive index is the ratio of the sines of the angles of incidence and refraction for a wave moving from one medium to the next. (also called Snell’s Law) CRITICAL ANGLE Is the angle of incidence that produces an angle of refraction equal to 90o. It can be calculated with: n = sin 90 / sin c = 1 / sin 42 = 1 / 0.67 = 1.49 TOTAL INTERNAL REFLECTION If the angle of incidence surpasses the critical angle, the ray is reflected instead of refracted. This is total internal reflection. TOTAL INTERNAL REFLECTION Optical fibres, periscopes and road reflectors all utilise the idea of total internal reflection. CONVEX LENSES These lenses allow light rays to converge upon a singular point at the focal point. Examples include microscopes and magnifying glasses. CONCAVE LENSES These lenses allow light rays to diverge. Examples include flashlights and peepholes. REAL AND VIRTUAL IMAGES Real images can be projected. They occur when the object distance is greater than the focal length. Virtual images cannot be projected. They occur when the object distance is less than the focal length. Pinhole cameras create real, inverted images. LENS DIAGRAMS LENS DIAGRAMS THE LENS FORMULA THE LENS FORMULA (worked example) An object is placed 24cm away from a convex lens of 8cm focal length. Calculate the image distance, v. ELECTROSTATICS When a silk cloth is rubbed with a glass rod: - The rod loses electrons and becomes +ve. - The cloth gains electrons and becomes –ve. Positive charges don’t move because they are bound to the nucleus of the atom. INDUCTION Objects can also be charged by placing them next to each other and using a charged object within proximity. This method is called charging by induction. Examples of technology that use electrostatic forces: • 1. Photocopiers • 2. Electrostatic painting • 3. Electroplating CHARGE Charges are comprised of coulombs (C). Think of a coulomb as a bundle of electrons that can either be stored or transmitted. Worked example: The makers of a cellphone have upgraded its battery capacity from 4320C to 9000C. If a charger delivers a current of 0.6A, how much more time will it take to charge the new battery than the old? ΔQ = 9000 – 4320 = 4680 C Δt = ΔQ ÷ I = 4680 ÷ 0.6 = 7800 s (or 130 mins) ELECTRIC FIELDS An electric field is defined as a region around a charged particle or object within which a force would be exerted on other charged particles or objects. Electric fields flow out from positive and into negative. So flow occurs from positive to negative. Voltage The amount of energy contained per unit of charge. 1 Volt = 1 Joule/Coulomb (J/C) Also called potential difference and emf (electromotive force). Think of it as the ‘force’ that pushes charges across a conductor. Current The number of unit charges that pass a point per second. 1 Ampere = 1 Coulomb/second (C/s) If you have a 2A charger for your phone, it will deliver 2 coulombs per second to the battery. Current flows ‘conventionally’ from positive to negative. RESISTANCE The opposition to current flow in a circuit. 1 Ω= 1 Volt/Ampere (V/A) Wires of thin diameter tend to restrict flow of current and so have greater resistance. Longer wires also have greater resistance due to heat losses. Ohm’s Law The current through a conductor is directly proportional to voltage and inversely proportional to resistance. Ohm’s Law (worked example) A lamp is marked 12V, 3A. Calculate the lamp’s: (i) Resistance (ii) Energy if left on for a minute (i) V = IR R=V/I = 12 / 3 =4Ω (ii) P = VI = 12 x 3 = 36 W E =Pxt = 36 x 60s = 2160 J OR E = VIt = 12 x 3 x 60 = 2160 J Ammeters and Voltmeters Ammeters measure current. They are connected in series due to their low resistance. Voltmeters measure voltage. They are connected in parallel due to their high resistance. Direct (d.c.) and alternating current (a.c.) d.c. is unidirectional and has a mostly fixed value, e.g. AA batteries. a.c. is bidirectional and has values that fluctuate between positive and negative, e.g. transformers, power lines Rectification An a.c. can be converted to a d.c. with the use of a semiconductor diode or a rectifier. This produces half-waves in a voltage-time graph. If the diode is reversed, no current will flow. Primary and Secondary Cells Voltage-Current (VI) Graphs The graph shapes for: 1. Ohmic conductor 2. Filament lamp 3. Electrolyte 4. Semi-conductor diode Series Circuits There is only one current path. If either A or B is removed, no current will flow. The voltage in the cell is ‘shared’ between A and B. Higher resistances require more voltage. Current is equal in all components in series. The total resistance, Rs, is the sum of all resistances. Rs = R1 + R2 … Series Circuits (example) Calculate the following: (i) Total resistance (ii) Current (iii) Voltage through A (i) Rs = R1 + R2 = 5 + 3 = 8Ω (ii) I = V / R = 6 / 8 = 0.75 A (iii) V (A) = IR = 0.75 x 5 = 3.75 V Parallel Circuits There are multiple paths. If either A or B is removed, current can still flow. The voltage in each path is the same as the voltage of the cell. The current in each path is determined by the path’s resistance. Higher resistances have lower currents. Parallel Circuit Resistance Resistance in a parallel circuit is calculated with: Parallel Circuit (Current) Total current can be calculated by finding the currents through each path: Path A → I = V/R = 6/5 = 1.2 A Path B → I = V/R = 6/3 = 2.0 A Total = 1.2 + 2.0 = 3.2 A OR I = V/R = 6/1.875 = 3.2 A (using total resistance) Combined Series and Parallel Circuit In the circuit above, R2 and R3 are in series on path B. R1 will be parallel to the other two. A1 measures the total current in the circuit (as it is connected to the power source). A2 measures the current in Path A. Combined Series and Parallel Circuit Reading on A2: I=V/R = 12 / 2 = 6A Reading on A1: = Current in Path A + Current in Path B = 6A + 2A = 8A Total current of Path B: R = R2 + R3 =2+4=6 I =V/R = 12 / 6 = 2A Fuses and Wiring Fuses are made of metals with low melting points. Fuse gets heated up → Fuse breaks → Circuit is broken Fuses and switches are always connected to live wires. Neutral wires complete the circuit. Ground or earth wires deposit extra electrons into the ground. Electrical Hazards 1. Damp wires and broken insulation can result in electrocution or electrical fires. 2. Electrocution can also occur if the earth wire is improperly connected, causing electrons to build up in the frame of an appliance. Magnetism - Temporary magnets (e.g. iron) can be easily magnetized and are usually found in electromagnets. - Permanent magnets (e.g. steel) retain their magnetism for a long time and are usually found in compass needles and metal detectors. Magnetic Induction An unmagnetized magnetic material can be magnetized if it is placed in the proximity of a magnet or an electric field. Magnetic fields run perpendicular to electric fields, and are part of the same force. One cannot exist without the other. When the circuit is connected, electrons will flow from the battery to the iron nail, temporarily magnetizing it. Fleming’s Left Hand Rule Two of the above quantities can ‘create’ the third once they are perpendicular to each other: Example: If a wire is moved (thrusted) perpendicular to a magnetic field, a current can be induced in that wire. If a current-carrying wire is perpendicular to a magnetic field, the wire will be thrusted in a certain direction. Fleming’s Left Hand Rule (1st example) What direction is the wire being thrusted? Fleming’s Left Hand Rule (1st example) Fleming’s Left Hand Rule (2nd example) If the wire is being thrusted out of the page, is the current left or right? Fleming’s Left Hand Rule (2nd example) If the wire is being thrusted out of the page, is the current left or right? Right Hand Grip Rule The (x) means the current is going into the page. The dot (.) means the current is coming out of the page. Right Hand Grip Rule Fleming’s Left Hand Rule in a Coil The thrusts on AB and CD will create a turning force or moment. This allows the coil to spin in a motor. No thrust will occur on BC because the magnetic field is parallel, not perpendicular, to the current there. d.c. motors The motor converts electrical to mechanical energy. The motor spins due to forces created by the current being perpendicular to the magnetic field. d.c. motor (split ring) The split ring (commutator) breaks the circuit every halfturn. This prevents the coil from reversing direction, allowing it to spin continuously. d.c. motors To generate more turning force: 1. Add more turns in the coil 2. Increase the current 3. Use stronger magnets a.c. generators The generator converts mechanical to electrical energy. An outside energy source (e.g. wind) spins the external rotator, which turns the wire loop in the magnetic field. This generates current. a.c. generators To generate more current: 1. Add more turns in the coil 2. Rotate the coil faster 3. Use stronger magnets Electromagnetic Induction The voltage induced in a coil is proportional to the rate of magnetic force across it. – Faraday’s Law Electromagnetic Induction No current or voltage is induced if the magnet is not moving relative to the coil. The galvanometer needle points up at zero. TRANSFORMERS Transformers can either increase or decrease voltage, by decreasing or increasing current. Step-up transformers increase voltage by having more secondary turns than primary. Step-down transformers decrease voltage by having less secondary turns. TRANSFORMERS Transformers need a.c. power sources to allow for a constantly changing magnetic field in the primary turns. This electromagnetizes the core and induces a current in the secondary turns. TRANSFORMERS Transformers need a.c. power sources to allow for a constantly changing magnetic field in the primary turns. This electromagnetizes the core and induces a current in the secondary turns. TRANSFORMERS FORMULAS TRANSFORMERS (worked example) Electrical power produced by Powergen in Trinidad is stepped up from 11,000V at 8000A to 110,000V for transmission to Tobago. (i) If the number of turns in the secondary coil is 900, calculate the number of turns in the primary coil for an ideal transformer. (ii) Calculate the transmission current for the ideal transformer in (a). TRANSFORMER LOSSES Transformers that have no power losses are called IDEAL. The following factors result in power losses, and how to limit them: 1. HEAT LOSSES – Use thicker, lower-resistance wires 2. EDDY CURRENTS – Laminate the core. 3. HYSTERESIS (magnetization delay) – Use a perm-alloy core. LOGIC GATES Logic gates are electronic operators that send signals based on satisfied conditions (1 – true) or unsatisfied (0 – false). There are 5 types you should know, for now: 1. NOT gate 2. AND gate 3. OR gate 4. NAND gate 5. NOR gate NOT GATE These invert the signal from input to output. AND GATE The only positive (1) output is when both inputs are 1. Can be used in username and password systems. OR GATE A positive (1) output is obtained when there is at least one positive (1) input. Can be used for burglar alarm systems. NAND and NOR GATES SOLVING LOGIC GATE PROBLEMS SOLVING LOGIC GATE PROBLEMS SOLVING LOGIC GATE PROBLEMS SOLVING LOGIC GATE PROBLEMS PARTICLES IN THE ATOM PARTICLES IN THE ATOM Nucleons (protons and neutrons) are held together in the nucleus by strong nuclear forces. Electrons orbit the nucleus in shells. This is the Bohr model. PARTICLES IN THE ATOM ATOMIC NUMBER The number of protons in the atom. In this case, the atomic number is 6. MASS NUMBER The number of protons and neutrons in the atom. In this case, the mass number is 12. RUTHERFORD GOLD FOIL EXPERIMENT A source fires a stream of +ve alpha particles through a thin slice of gold foil. A detector strip illuminates in the spots where the particles collide. Only the particles that hit or come close to a gold nucleus are deflected. Most don’t. Scientists who did experiment: GEIGER, MARSDEN, RUTHERFORD. RUTHERFORD GOLD FOIL EXPERIMENT ISOTOPES Isotopes are atoms of the same element, with the same atomic number but different mass number. They have the same number of protons but different number of neutrons. USES OF RADIOISOTOPES RADIOACTIVE DECAY Radioactive decay is spontaneous and random. It occurs when a nucleus gets far from a 1 proton : 1 neutron ratio. Unstable atoms that undergo decay lose mass over time due to release of alpha, beta and gamma radiation. TYPES OF RADIATION Alpha particles are helium nuclei (2p, 2n). Beta particles are fastmoving electrons. Gamma rays are highfrequency e.m. waves. TYPES OF RADIATION TYPES OF RADIATION Alpha is stopped by paper. Beta is stopped by aluminium. Gamma is stopped by lead. CLOUD CHAMBERS Cloud chambers are sealed dishes filled with alcohol or vapours that show the trails of radiation emissions. Alpha has thick trails. Beta has thinner, jagged trails. Gamma has scattered spots. RADIATION IN MAGNETIC FIELDS Use Fleming’s Left Hand Rule. Current (beta particles) to the right. They are deflected more because they weigh less. Alpha goes the opposite direction. Gamma is undeflected. RADIATION IN ELECTRIC FIELDS Positive alpha particles deflect towards negative plate. Negative beta particles deflect towards positive plate. Gamma rays are neutral, so remain undeflected. RADIATION SAFETY After Marie Curie (who discovered radium) succumbed to radiation-related disease, safety precautions were introduced. 1. Store radioactive materials in lead containers. 2. Wear safety gloves or use forceps when handling 3. Stay behind protective screens when necessary 4. Wear Hazmat suits in irradiated environments ALPHA DECAY An alpha particle (helium nucleus) is lost. The atomic number decreases by 2 and the mass number decreases by 4. BETA DECAY A beta particle (fastmoving electron) is lost. The atomic number increases by 1 and the mass number remains unchanged. WRITING DECAY EQUATIONS HALF-LIFE Half-life is the time taken for a radioactive substance to decay by half. For e.g. Iodine-131 has a half-life of 8 days. If there were 1000 atoms at first, there would be 500 atoms after 8 days, and 250 atoms after a further 8 days. HALF-LIFE (1st worked example) An 800mg sample of radon decays over a period of 20 days until only 25mg remains. What is the halflife of radon, in days? 800 → 400 → 200 → 100 → 50 → 25 Number of half-lives = 5 5 half-lives = 20 days 1 half-life = 20 days / 5 = 4 days HALF-LIFE (2nd worked example) If iodine-131 has a half-life of 8 days, how many days must pass before it reaches 1/16th its mass? 1→ ½ → ¼ → 1/8 → 1/16 Number of half-lives = 4 Time elapsed = 4 x 8 days = 32 days HALF-LIFE GRAPH NUCLEAR ENERGY NUCLEAR ENERGY Pros: 1. It is more efficient than fossil fuels and yields much more useable electrical energy. 2. It produces no greenhouse gases, does not contribute to climate change. Cons: 1. There is a risk of nuclear meltdown, e.g. Chernobyl 2. Radioactive waste is difficult to dispose of. CALCULATING NUCLEAR ENERGY The formula for nuclear energy is given as: CALCULATING NUCLEAR ENERGY (example) Calculate the energy released in the reaction above. CALCULATING NUCLEAR ENERGY (example) First, add the masses on the left: 2.014 + 3.016 = 5.03 u CALCULATING NUCLEAR ENERGY (example) Then, add the masses on the right: 4.003 + 1.009 = 5.012 u CALCULATING NUCLEAR ENERGY (example) Subtract the difference: ▲m = 5.03 - 5.012 = 0.018 u CALCULATING NUCLEAR ENERGY (example) Convert the ‘u’ to ‘kg’: ▲m = 5.03 - 5.012 = 0.018 u x (1.66 x 10-27 kg) = 2.988 x 10-29 kg CALCULATING NUCLEAR ENERGY (example) c = speed of light (3 x 108 m/s) Use Einstein’s formula to calculate E: ▲E = ▲mc2 = 2.988 x 10-29 kg x (9 x 1016) = 2.6892 x 10-12 J c2 = 9 x 1016 m/s