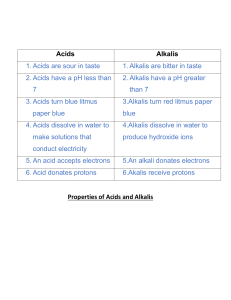
Grade 11 Chemistry Acids, Bases & Salts Quick Revision Worksheet Name:_______________________ 1) In terms of proton transfer, an acid is a ____________ _____________ and a base a _____________ __________________ 2) An alkali is a __________________ ____________ [2] [1] 3) Common bases are: ____________ ______________ ____________ __________________ ____________ __________________ ________________ [4] 4) Alkalis are most commonly the ____________________ of group ___ and ___ metals [2] 5) Acids release _____ in solution and alkalis release ______ in solution [2] 6) Acids have a pH _____________ 7 and alkalis have a pH ______________ 7 [2] 7) A neutralisation reaction follows the common equation of: ____________ + ____________ ____________ + ______________ [1] 8) Litmus is/turns ____________ in acid and ____________ in alkali Methyl orange indicator is/turns ____________ in acid and ____________ in alkali Phenolphthalein is/turns ___________________ in acid and ____________ in alkali [6] 9) ________________ indicator shows the exact strength/pH value of an acid/alkali solution [1] 10) pH is a measure of the ______________________________________________________ [1] 11) The ________________ the pH, the ________________ the H+ concentration and the _______________ the OH- concentration [3] 12) Strong acids have a pH range of ________ and weak acids a pH range of ________ Strong alkalis have a pH range of ________ and weak alkalis a pH range of ________ [4] 13) Acid + metal ____________ + ______________ Acid + metal oxide ____________ + ______________ Acid + metal hydroxide ____________ + ______________ Acid + metal carbonate ____________ + ______________ + _____________________ [4] 14) The salt formed in a neutralisation reaction is named according to the ____________ from the base and the ______________ from the acid [2] 15) The strength of an acid or alkali depends on its _________________________________________ and therefore, strong acids/alkalis dissociate (ionise) ________________________ in solution while weak acids/alkalis dissociate ______________________________ in solution [3] 16) The dissociation of weak acids or alkalis are ____________________ reactions with the equilibrium lying to the ___________ [2] 17) Concentration is the amount/number of _______________ in ______________ 18) A concentrated solution of either an acid or an alkali [2] contains a contains a ________________________________________________ per 1 dm3 of solution A dilute solution of either an acid or an alkali ________________________________________________ per 1 dm3 of solution [4] [Total: 46]