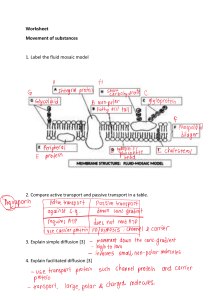
Chapter 5: Diffusion Chapter 5: Diffusion ISSUES TO ADDRESS... • What is diffusion? • By what atomic mechanisms does diffusion occur? • What are examples of diffusion in materials processing? • What equations do we use to solve diffusion problems? • How does the rate of diffusion depend on temperature? Chapter 5 - 1 Diffusion Diffusion - Mass transport by atomic motion • ______________________ - Gases & Liquids – random (___________) motion - Solids – ______ diffusion and _________ diffusion • Interdiffusion - diffusion of atoms of one material into another material • Self-diffusion – atomic migration in a pure metal Chapter 5 - 2 Diffusion • Atoms tend to _________ from regions of ____ concentration to regions of ____ concentration. Before diffusion After diffusion Figs. 5.1, Callister & Rethwisch 10e. Concentration Profiles Chapter 5 - 3 1 Chapter 5: Diffusion Diffusion • Self-diffusion: Migration of __________ in pure metals Locations of 4 labeled atoms before diffusion Locations of 4 labeled atoms after diffusion C C A D A D B B Chapter 5 - 4 Diffusion Mechanism I Vacancy Diffusion • ___________________ exchange positions • applies to host and ___________________ impurity atoms • diffusion rate depends on: -- number of ___________ -- activation _________ to exchange. increasing elapsed time Chapter 5 - 5 Diffusion Mechanism II ____________ Diffusion • Small, _____________ atoms move from one interstitial position to an adjacent one Fig. 5.2 (b), Callister & Rethwisch 10e. More rapid than ___________ diffusion Chapter 5 - 6 2 Chapter 5: Diffusion Processing Using Diffusion Case hardened region • Case _______________: - Example of ____________ diffusion - Outer surface selectively hardened by diffusing carbon atoms into surface - Presence of C atoms makes iron (steel) harder • Example: __________________ gear - Case hardening improves wear resistance of gear - Resulting residual compressive stresses improve resistance to fatigue failure Chapter-opening photograph, Chapter 5, Callister & Rethwisch 10e. (Courtesy of Surface Division, Midland-Ross.) Chapter 5 - 7 Processing Using Diffusion Diffusion in Semiconducting Devices • _______ – Diffusion of very small concentrations of atoms of an impurity (e.g., P) into the semiconductor silicon. • Process: 1. Deposit ___ rich layers on surface 2. __________ the sample to drive in P 3. Result is P doped ______________ regions silicon silicon Chapter 5 - 8 Rate of Diffusion • Diffusion is a time-dependent process. • Rate of Diffusion- expressed as diffusion flux, J mass of diffused species M ⎛ kg ⎞ J ≡ Flux ≡ = ⎜ ⎟ (area)(time) At ⎝ m2 -s ⎠ • Measured experimentally – Use thin sheet (______________) – cross-sectional area A – Impose _________________ gradient across sheet – Measure mass of diffusing species (M) that passes through the sheet over time period (t) J= M l dM = At A dt M= mass diffused J ∝ slope time Chapter 5 - 9 3 Chapter 5: Diffusion Steady-State Diffusion Rate of ___________ (or flux) independent of time Flux (J) proportional to ___________ gradient: J ∝ dC dx C = concentration x = diffusion direction C1 C1 ____________ of diffusion C C2 x1 if linear x C2 J = −D x2 dC ΔC C2 − C1 ≅ = dx Δx x 2 − x1 dC dx D = diffusion coefficient Chapter 5 - 10 Diffusion Example Chemical Protective Clothing (CPC) • ________________ is a common ingredient of paint removers. Besides being an irritant, it also may be absorbed through skin. When using this paint remover, protective gloves should be worn. • Lets investigate whether butyl rubber gloves (_____ cm thick) commonly found in the _______ can be used as protective gloves. • Note: The maximum allowable flux for a 150 lb person is less than _______________________ • Compute the diffusion flux of methylene chloride through the gloves. Chapter 5 - 11 CPC Example (cont.) • Solution – diffusion flux of methylene chloride assume __________ conc. gradient glove C1 paint remover 2 tb = 6D skin C2 x1 x2 J = − (110 x 10-8 cm2 /s) J = −D C − C1 dC ≅ −D 2 dx x 2 − x1 Data: D = ____________ C1 = _____________ C2 = 0.02 g/cm3 x2 – x1 = 0.04 cm (0.02 g/cm3 − 0.44 g/cm3 ) g = 1.16 x 10-5 (0.04 cm) cm2 -s Note: this is more than 30 times the allowable flux. Unsafe to use these gloves for paint removal. Chapter 5 - 12 4 Chapter 5: Diffusion Influence of Temperature on Diffusion • Diffusion coefficient increases with increasing _____ D = Do exp - Qd RT D = __________ coefficient [m2/s] Do = pre-exponential [m2/s] Qd = __________ energy [J/mol] R = gas constant [____________] T = absolute temperature [K] Chapter 5 - 13 Influence of Temperature on Diffusion (cont.) D transform data ln D Temp = T 1/T ⎛ Q ⎞ D = D0 exp ⎜ − d ⎟ ⎝ RT ⎠ take natural log of both sides lnD = lnD0 − Qd RT Chapter 5 - 14 Influence of Temperature on Diffusion (cont.) 300 600 1000 10-8 1500 D has _________________ dependence on T T(°C) C in D (m2/s) C in α-Fe C in γ-Fe Al 1.0 Dinterstitial >> Dsubstitutional in 0.5 α-F e Al -Fe 10-20 Fe α- γ in Fe n Fe Fe i γ- 10-14 C in 1.5 Al in Al Fe in α-Fe Fe in γ-Fe 1000 K/T Adapted from Fig. 5.6, Callister & Rethwisch 10e. (Data for Fig. 5.7 taken from E.A. Brandes and G.B. Brook (Ed.) Smithells Metals Reference Book, 7th ed., Butterworth-Heinemann, Oxford, 1992.) Chapter 5 - 15 5 Chapter 5: Diffusion VMSE: Screenshot of Diffusion Plots Chapter 5 - 16 Influence of Temperature on Diffusion (cont.) Derive an equation relating the ________ coefficients at two temperature T1 and T2 using the ________ derived on slide 14. Q "1% Q "1% lnD2 = lnD0 − d $$ '' and lnD1 = lnD0 − d $$ '' R # T2 & R # T1 & ______________ equation at T1 from equation at T2 lnD2 − lnD1 = ln Q ⎛ 1 1⎞ D2 = − d ⎜⎜ − ⎟⎟ D1 R ⎝ T2 T1 ⎠ Take the ___________ of each side to get the final equation ⎡ Q ⎛ 1 1 ⎞⎤ D2 = D1 exp ⎢− d ⎜⎜ − ⎟⎟⎥ ⎢⎣ R ⎝ T2 T1 ⎠⎥⎦ Chapter 5 - 17 Example Diffusion Problem At 300°C the diffusion coefficient and _______________ for Cu in Si are D1(300°C) = 7.8 x 10-11 m2/s Qd = 41.5 kJ/mol Compute the _____________________ D2 at 350°C? ⎡ Q ⎛ 1 1 ⎞⎤ D2 = D1 exp ⎢− d ⎜⎜ − ⎟⎟⎥ ⎢⎣ R ⎝ T2 T1 ⎠⎥⎦ T1 = 273 + 300 = 573 K T2 = 273 + 350 = 623 K ⎡ −41,500 J/mol ⎛ 1 1 ⎞⎤ D2 = (7.8 x 10−11 m2 /s) exp ⎢ − ⎜ ⎟⎥ 8.314 J/mol-K 623 K 573 K ⎠⎦ ⎝ ⎣ D2 = _____________ Chapter 5 - 18 6 Chapter 5: Diffusion Non-steady State Diffusion • The ___________ of diffusing species is a function of both time and position C = C(x,t) • For non-steady state diffusion, we seek solutions to ____________________ ____________ Law ∂C ∂2C =D 2 ∂t ∂x This form of the equation assumes D is independent of concentration Chapter 5 - 19 Non-steady State Diffusion • Consider the diffusion of copper into a bar of aluminum Surface conc., Cs of Cu atoms bar pre-existing conc., Co of copper atoms Cs Fig. 5.4, Callister & Rethwisch 10e. Boundary/Initial Conditions at t = 0, C = Co for 0 ≤ x ≤ ∞ at t > 0, C = CS for x = 0 (constant surface conc.) C = Co for x = ∞ Chapter 5 - 20 Non-steady State Diffusion (cont.) ( ) C x,t − Co Cs − Co ⎛ x ⎞ = 1− erf ⎜ ⎟ ⎝ 2 Dt ⎠ C(x,t) = Conc. at point x at time t erf(z) = error ___________ z and erf(z) values are given in Table _______ Fig. 5.5, Callister & Rethwisch 10e. Chapter 5 - 21 7 Chapter 5: Diffusion Non-steady State Diffusion Example Problem An FCC iron-carbon alloy initially containing ______ % C is carburized at an elevated temperature and in an atmosphere in which the surface carbon concentration is maintained at 1.0 wt%. If, after _____, the concentration of carbon is 0.35 wt% at a position ________ below the surface, determine the temperature at which the treatment was carried out. Chapter 5 - 22 Example Problem (cont.): Solution: use Eqn. 5.5 ⎛ x ⎞ C(x,t) − Co = 1− erf ⎜ ⎟ Cs − Co ⎝ 2 Dt ⎠ Data for problem tabulated as follows: – t = 49.5 h x = ________ – Cx = 0.35 wt% Cs = 1.0 wt% – Co = __________ ⎛ x ⎞ C(x,t) − Co 0.35 − 0.20 = = 1− erf ⎜ ⎟ = 1− erf(z) Cs − Co 1.0 − 0.20 ⎝ 2 Dt ⎠ erf(z) = ______ Chapter 5 - 23 Example Problem (cont.): We must now determine from Table 5.1 the value of z for which the error function is 0.8125. An interpolation is necessary as follows z erf(z) 0.90 z 0.95 0.7970 0.8125 0.8209 Now solve for D z − 0.90 0.8125 − 0.7970 = 0.95 − 0.90 0.8209 − 0.7970 z = 0.93 z= x 2 Dt D= x2 4z 2t ⎛ x2 ⎞ (4 x 10−3 m)2 1h ∴D = ⎜⎜ 2 ⎟⎟ = = 2.6 x 10−11 m2 /s 2 ⎝ 4z t ⎠ (4)(0.93) (49.5 h) 3600 s Chapter 5 - 24 8 Chapter 5: Diffusion Example Problem (cont.): • To solve for the temperature at which D has the above value, we use a rearranged form of Equation 5.9a } T= Qd R(lnDo − lnD) From Table 5.2, for diffusion of C in FCC Fe Do = 2.3 x 10-5 m2/s Qd = 148,000 J/mol T= 148,000 J/mol (8.314 J/mol-K)[ln (2.3 x 10−5 m2 /s) − ln (2.6 x 10−11 m2 /s)] T = 1300 K = _______ Chapter 5 - 25 Summary • Solid-state diffusion is mass transport within solid materials by stepwise atomic motion • Two diffusion mechanisms - Vacancy diffusion - Interstitial diffusion dC dx • Fick’s First Law of Diffusion J = −D • Fick’s Second Law of Diffusion - non-steady state diffusion ∂C ∂2C =D 2 ∂t ∂x • Diffusion coefficient - Effect of temperature ⎛ Q ⎞ D = D0 exp ⎜⎜ − d ⎟⎟ ⎝ RT ⎠ Chapter 5 - 26 9