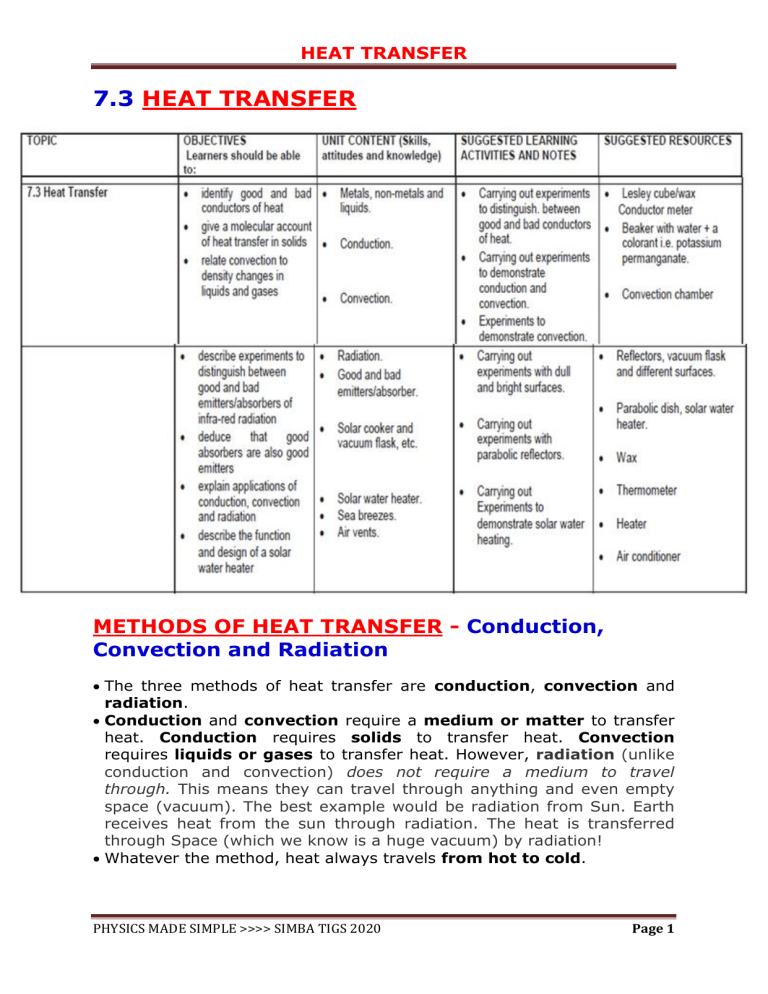
HEAT TRANSFER 7.3 HEAT TRANSFER METHODS OF HEAT TRANSFER - Conduction, Convection and Radiation • The three methods of heat transfer are conduction, convection and radiation. • Conduction and convection require a medium or matter to transfer heat. Conduction requires solids to transfer heat. Convection requires liquids or gases to transfer heat. However, radiation (unlike conduction and convection) does not require a medium to travel through. This means they can travel through anything and even empty space (vacuum). The best example would be radiation from Sun. Earth receives heat from the sun through radiation. The heat is transferred through Space (which we know is a huge vacuum) by radiation! • Whatever the method, heat always travels from hot to cold. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 1 HEAT TRANSFER 1. CONDUCTION • Definition: Conduction is the transfer of heat through a medium from one particle to another without any flow of the material medium. • Conduction is the main heat transfer process in solids, much less so in liquids and gases. It is a slow process compared to convection and radiation. • All metals are good conductors, and hard materials like diamond. • Examples of bad conductors are air, wood, water, stone, paper, cork, wool, fur, feathers, plastic, polystyrene, fibreglass, glass, fabrics and foam. • These bad conductors are called insulators. • Conduction cannot occur when there are no particles present, so a vacuum is a perfect insulator. Experiment to compare heat conduction by different materials - Pins are stuck at the end of metal rods using paraffin wax. - The opposite end of the rods are heated with the same burner flame. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 2 HEAT TRANSFER - The heat from the flame is conducted along the length of each rod. - Metals do not all conduct heat equally well. When the heat reaches the paraffin wax, the wax melts and eventually the pin falls. - The pin falls off the best conductor first. In order of heat conductivity they are: Copper, aluminium, brass, iron, Lead - Four precautions taken in this experiment to carry out a fair test are: 1) Rods of equal length should be used. 2) The rods should be of equal thickness. 3) The rods should be held in the same burner flame for the same length of time. 4) The rods should be supported on an insulating stand, e.g. brick, which does not conduct away the heat. Do not use a metallic stand, e.g. tripod stand, because it would conduct some of the heat away. • Alternatively, the rods can be heated using boiling water as shown in the following diagram. The results would be the same as explained above. Experiment to compare heat conduction by conductors and insulators - When heated by passing it through a flame several times, the paper on the brass side is scorched but that on the wood side is badly scorched. - This is because the brass, conducts heat away quickly preventing scorching of the paper on that side. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 3 HEAT TRANSFER Experiment: Water is a poor conductor of heat An experiment is carried out as shown in the diagram. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 4 HEAT TRANSFER The ice takes a long time to melt, even though the water at the top of the tube is boiling because water is a poor conductor of heat. Uses of conductors and insulators 1. If heat is required to travel quickly through a solid then a good conductor such as metal is used. Car engines, saucepans boilers and radiators are made of metals. 2. If heat is required not to travel quickly then an insulator (bad conductor) is used e.g. the handles of saucepans, kettles etc., are made of substances such as wood, rubber or plastic. 3. Materials that trap air are very good insulators, since air is a bad conductor. Wool, fur, feathers, fibreglass contain tiny pockets of trapped air which reduce conduction and increase insulation. 4. Air on its own is also used as an insulator. Layers of still air are used e.g. air in the cavity between walls and in double glazing. How conduction occurs in solids - Conduction in solids takes place by two processes which are molecular/atomic vibrations and free electron flow/diffusion. - In metals, conduction happens due to the combination of vibrations of molecules and free electron diffusion. While in non metals, the mechanism for conduction is only atomic or molecular vibrations. (a) Atomic/Molecular Vibrations for both metals and non-metals. When one end of the solid is heated, the molecules/atoms at the hotter end gain kinetic energy and vibrate faster about a fixed position. These molecules collide with their less energetic neighbouring molecules and transfer some of their energy to them which in turn gain kinetic energy. Heat is then passed from molecule/atom to molecule/atom until it reaches the colder end of the solid. The process will only stop when the temperature of the solid becomes uniform and constant. It is a rather slow process. (b) Free electron flow/diffusion for metals only. A metal consists of a ‘sea of free electrons’ surrounding atoms. When a metal is heated, the free electrons gain kinetic energy and move faster. These electrons can move freely in the spaces between molecules before colliding with other electrons or atoms and transfer some energy to them. Free electron diffusion is a very quick process, much faster than PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 5 HEAT TRANSFER molecular/atomic vibration. The combination of these two processes makes conduction in metals faster. Rate of heat transfer via conduction • Solid > liquid > gas • Reason: Solids are good conductors of heat because molecules/atoms in solids are more closely packed and hence collisions are more frequent. • Liquids and gases are poor conductors of heat because particles are far apart and hence collisions are less frequent. 5) CONVECTION • Definition: Convection is process of heat transfer by actual movement of the medium (gas or liquid) particles. • This occurs in fluids (liquids and gases), where atoms are able to move. • Convection is quicker than conduction and slower than radiation. The process of convection • When part of a gas or liquid is heated it expands, becomes less dense and rises. Cooler, denser gas or liquid above it sinks and takes its place. This in turn is heated and the process repeats itself. • This results in a movement of material in a circular path, called convection current. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 6 HEAT TRANSFER Experiment to demonstrate convection in a liquid Crystals of potassium permanganate are placed in the bottom of the water filled flask and heated. As the warmer water rises it drags a stream of dissolving potassium permanganate with it and we see a stream of purple rising. This stream then falls back down as the cooler liquid falls to replace the warm liquid. The purple stream follows the path of the convection current. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 7 HEAT TRANSFER Experiment to demonstrate convection in a gas The air above the candle rises as it is heated, becoming less dense than the cold air around it. Cooler air, which is denser than the warm air falls down the right hand chimney. The stream of smoke follows the convection current and smoke is pulled with the moving air through the box where it is heated and rises out of the left hand chimney. Examples of convection currents in practice 1. Heaters – keep a room warm throughout by setting up convection currents in the air in the room. 2. Air Conditioning – keeps rooms cool by setting up convection currents in the air, in the opposite sense to when the room is heated 3. Coastal breezes PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 8 HEAT TRANSFER Sea breeze (aka Daytime breezes) - During the day the temperature of the land increases faster than that of the sea because the specific heat capacity of the land is lower. The hot air above the land rises and is replaced by cooler air from over the sea. A convection current is set up which causes the breeze to come in from the sea. Land breeze (aka Night-time breezes) - At night the sea has more heat to lose and cools more slowly because of the higher specific heat capacity of the sea. The air above the sea is warmer than above the land and cool air from the land sets up a breeze blowing from the land to the sea. 4. A hot water system When the boiler is turned on, convection current is set up in pipe A from the top of the boiler to the top of the hot water tank. Cold water flows down pipe B from the bottom of the tank to the bottom of the boiler. This convection current allows all of the water to circulate and be heated. When hot water is drawn from the top of the tank, it is replaced by the cold water from the tank, along pipe C. Pipe E provides a pressure release PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 9 HEAT TRANSFER overflow in case the water boils. D shows the normal water level in the overflow pipe. 5. Fridges cool food by convection currents. 3 RADIATION • Definition: Radiation is a method of heat transfer whereby heat is transmitted from one body to another in the form of e.m waves/infra-red waves without the aid of a medium. • Radiation is a process of heat transfer in which heat directly passes from one body to other body without affecting the medium. • Radiation is the method of heat transfer in gases and in a vacuum. It is the quickest of the 3 methods. • It involves the movement of energy from one place to another in the form of infra-red waves. - Radiation is the continual emission of infra-red (IR) waves from the surface of all bodies, transmitted without a medium. - All objects and surfaces absorb or emit radiant heat or IR radiation. The hotter the object, the greater the amount of radiant heat emitted. Factors that affected the rate at which radiant heat is emitted are 1. Colour and texture of the surface • Dull black surfaces are better absorbers and emitters of radiant heat than shiny white surfaces. • Shiny surfaces are also good reflectors of radiant heat. 2. Surface area • The object with larger surface area will absorb and emit radiant heat at a faster rate. 3. Surface temperature PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 10 HEAT TRANSFER • The object with higher surface temperature will absorb and emit radiant heat at a faster rate. Note: • • • Radiation does not require a medium to transfer heat energy and can take place in a vacuum. In explaining heat energy transfer involving radiation, we can only use the term “infra-red radiation” or “radiant heat”. A good absorber of radiant heat is also a good emitter of radiant heat. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 11 HEAT TRANSFER Experiment: Good or bad absorbers of radiated heat Heat cannot reach the tin by conduction because air is an insulator. Also hot air rises, so no convected heat reaches the metal plates. Radiated heat from the burner is absorbed by the two metal plates. This heat is then conducted through the metal, melting the wax and the cork falls, falling first from the dull (matt) black surface before the coin from the shiny surface. This shows that the dull (matt) black surface absorbs radiated heat better than the shiny surface. Matt (dull) black is the opposite of gloss black. Gloss black is shiny black. Matt (dull) black is a better absorber and emitter of radiant heat than gloss (shiny) black. Conclusion: For surfaces at the same temperature, dark, dull (matt) or black surfaces are better absorbers of heat than bright, shiny (glossy) or white surfaces. A poor absorber is called a good reflector, so shiny/glossy/white surfaces are better reflectors than dull black surfaces. Applications: Matt (dull) black is the opposite of gloss black. Gloss black is shiny black. Matt (dull) black is a better absorber and emitter of radiant heat than gloss (shiny) black. Gloss (shiny) black actually reflects away most of the heat and absorbs very little. This is why some black cars are actually PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 12 HEAT TRANSFER painted with gloss black so that they will not absorb much radiant heat from the sun so keeping the inside cool. Most cars and houses are pointed with bright/shiny/white colours for the same reason. TRY THIS QUESTION Explain each of the following in terms of thermal energy (heat) transfer : (a) People in very hot climates wear white clothing in order to stay cool. (b) Clean snow does not melt quickly in bright sunshine, whereas dirty snow does. (c) After finishing the London marathon, athletes are wrapped in aluminium-coated plastic sheets. (d) Some casserole dishes which are used to cook food in ovens are black, but the outside of an electric kettle is shiny. Experiment 1: Good or bad emitters of radiant heat. Initially the copper plate is heated. One side is shiny and the other is dull black. If you place your hands at equal distances either side of the hot sheet, then the one opposite the dull black side feels hotter than the hand opposite the shiny side, showing that the dull black side emitted more heat than the shiny side. Conclusion: Dull, black surfaces are better emitters of heat than bright, shiny or white surfaces. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 13 HEAT TRANSFER Experiment 2: Good or bad emitters of radiant heat. • One can is wrapped with a shiny aluminium foil and another is wrapped with a dull (matt) black paper. • The two cans are then filled with hot water. • The temperature readings are monitored. • The temperature of the can with the dull black surface is found to decrease more rapidly than that of the shiny tube. Conclusion For surfaces at the same temperature, dark, matt black surfaces are better emitters of thermal radiation than bright, shiny surfaces. Examples of radiation 1. All hot objects give off radiant heat as infra–red radiation. 2. Heat travels from the sun through a vacuum of outer space by radiation. 3. Cooling fins on the heat exchangers on the back of refrigerators are painted black to help heat loss by radiation. 4. Tea pots and kettles are shiny so they lose less heat by radiation. 5. Fire protective suits are shiny to reflect heat away from the fireman’s body. People suffering from exposure / hypothermia are wrapped in shiny aluminium foil to reflect heat back to the body and keep them warm. Vacuum (Thermos) flask A vacuum flask is designed to keep a liquid hot or cold by minimizing heat loss or gain in four possible ways, namely • Conduction, PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 14 HEAT TRANSFER • • • Convection, Radiation and Evaporation. However, heat loss by radiation is harder to stop as it can occur in a vacuum. 1. Vacuum between glass walls reduce heat loss by conduction and convection. 2. Silvered glass walls minimize heat loss through radiation by reflecting the radiant heat back to the liquid across the vacuum. 3. Plastic stopper is a poor conductor and, therefore, minimise heat loss by conduction. Convection and evaporation can only occur when it is removed during use. 4. Felt pad/foam support traps air which is a poor conductor of heat. 5. Trapped air above the liquid minimizes heat loss by conduction as air is a poor conductor of heat. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 15 HEAT TRANSFER Solar water heater • The solar water heater uses radiant energy from the sun to heat water. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 16 HEAT TRANSFER Operation of the solar water heater 1. Cold water is piped to… 2. Reservoir/storage tank then to…. 3. Solar heater on the roof where it is heated before being piped to… 4. Insulated hot water storage tank then flows to… 5. User taps. Reasons for the design of a solar water heater 1. GLASS COVER-transparent so allows sunlight to pass through. The glass plate and narrow air space also prevent heat loss by convection by trapping heat like a greenhouse. 2. THIN COPPER TUBES- are good conductors which readily pass heat to water. They have thin walls so they do not keep much of the heat themselves. 3. BLACKENED COPPER PLATE-acts as good absorber of radiation/heat. 4. EXPANDED POLYSTYRENE- separates the heating panel from the roof. A good insulator which prevents heat loss through the roof by conduction. 5. PLACED ON ROOF TOP-to maximise the capture of heat from the sun. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 17 HEAT TRANSFER 6. The hot water storage tank is insulated to prevent heat loss to surroundings. This keeps the water hot for a long time. SOLAR COOKER • Solar cookers use heat from the sun as their source of energy. • Solar cookers are constructed from shiny foil-covered parabolic (curved) surfaces. • To use a solar cooker, a black pot containing the food is placed at the focal point of the solar cooker. • The black pot absorbs the heat most efficiently because black surfaces are good absorbers of heat. • The shiny surface of the cooker reflects the sun’s rays onto the pot. The pot heats up cooking the food. PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 18 HEAT TRANSFER PICTURE: Solar cooker PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 19 HEAT TRANSFER Advantages of solar cookers: 1. 2. 3. 4. Eco friendly. Use a renewable energy source. Used in rural areas. Retains all the nutrients in food due to slow cooking. Disadvantages of solar cookers: 1. 2. 3. 4. Cannot be used at night. Cannot be used on cloudy days. Solar radiations are not uniform over the earth’s surface. Maximum temperature is 100oC therefore cannot exceed 140oC required for frying. TRY THESE QUESTIONS PHYSICS MADE SIMPLE >>>> SIMBA TIGS 2020 Page 20


