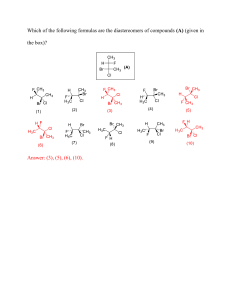
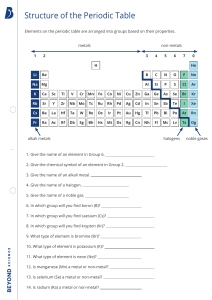
G8 Chemistry 1.4 Chemical names and Formulas Learning Objectives ZK Curriculum •Identify the meanings of chemical symbols理解 化学符号的意义 •Use the symbols of elements and write down the formulae of some substances利用元素 符号写出一些物质的化学式 English Curriculum •State the formulas of the elements and compounds named in the subject content 1.4.2 Naming compounds Types of compounds Type1: one metal + one non-metal Type2: one non-metal + one non-metal Type3: one metal + two or more non-metals 1.4.2 Naming compounds 1.4.2 Naming compounds Type1: one metal + one non-metal Writing Formulas from Names • 1st word = metal name • 2nd word = non-mental name with “ide” ending. Eg. NaCl ➔ sodium chloride NaBr ➔ sodium bromide NaF ➔ sodium fluoride 1.4.2 Naming compounds Type2: one non-metal + one non-metal Prefixes are used to show how many atoms are present in each chemical formula. Mono (or no prefix) – 1 Di – 2 Tri – 3 CO2 •No mono prefix is used on first element Carbon Dioxide •Di means 2 oxygens!! 1.4.2 Naming compounds Type2: one non-metal + one non-metal Mono (or no prefix) – 1 Di – 2 Tri – 3 CO ➔ carbon monoxide PCl3 ➔ phosphorus trichloride SiO2 ➔ silicon dioxide N2O ➔ dinitrogen monoxide 1.4.2 Naming compounds Type2: one non-metal + one non-metal Writing Names from Formulas silicon dioxide ➔ SiO2 sulfur trioxide ➔ SO3 1.4.2 Naming compounds Type3: one metal + two or more non-metals Writing Formulas from Names • 1st word = metal name • 2nd word = common polyatomic ions name Symbol polyatomic ions Name OH hydroxide SO4 sulphate/sulfate CO3 NO3 PO4 carbonate nitrate Phosphate E.g., Al2(SO4)3 ➔ aluminum sulphate CaCO3 ➔ calcium carbonate NaOH ➔ sodium hydroxide Zn(NO3)2 ➔ zinc nitrate SUMMARY