2022
V-A346 Appendix IX A
Appendix IX
A. Determination of Sulfated Ash
Use Method I unless otherwise directed.
Method I
(No Ph. Eur. method)
Heat a platinum dish £0 redness for 10 minutes, allow to cool
in a desiccator and weigh. Unless otherwise specified in the
monograph, place 1 g of the substance being examined in the
dish, moisten with suf/urie acid, ignite gently, again moisten
with sulfuric acid and ignite at about 8000 • Cool, weigh again,
ignite for 15 minutes and repeat this procedure until two
successive weighings do not differ by more than 0.5 mg.
sodium carbonate solution and gradually heat the liquid until it
boils. Maintain the current of nitrogen or carbon dioxide, allow
the solution to boil for about J 0 minutes and cool the flask
by gradual immersion in water. Introduce, while momentarily
removing the stopper of the flask, a weighed quantity of
50 to 100 g of the substance being examined, heat gently and
boil for 45 minutes. Disconnect the absorption tubes before
turning off the current of nitrogen or carbon dioxide and titrate
the combined contents with O.IM sodium hydroxide VS.
Each mL OrO.1M sodium hydroxide VS is equivalent to
3.203 mg of sulfur dioxide.
Repeat the operation without the substance being examined.
The solution in the absorption tubes remains neutral.
Method II
(ph. Eur. merhod 2.5.29)
Method III
EQUIPMENT
(Ph. Eur. method 2.4.14)
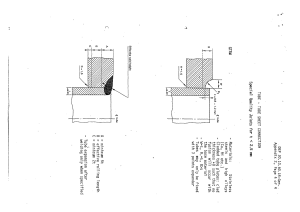
The apparatus as shown in Figure 2.5.29.-1 comprises:
- a ground-glass 3-neck round-bottomed flask (Al;
- a dropping funnel (B);
- a reflux condenser (G)j
- a receiving tube (D);
- a transfer tube (E);
- a gas pon.
Ignite a suitable crucible (for example, silica, platinum,
porcelain or quartz) at 600 ± 50°C for 30 min, allow to
cool in a desiccator over silica gel or other suitable desiccant
and weigh. Place the prescribed amount of the.substance to
be examined in the crucible and weigh. Moisten the
substance to be examined with a small amount of sulfuric
acid R (usually 1 mL) and heal gently at as Iowa
temperature as practicable until the sample is thoroughly
charred. After cooling, moisten the residue with a smaU
amount of sulfuri< acid R (usually 1 mL), heat gently until
white fumes are no longer evolved and ignite at 600 ± 50°C
until the residue is completely incinerated. Ensure that flames
are not produced at any time during the procedure. Allow
the crucible to cool in a desiccawr over silica gel or other
suitable desiccant, weigh it again and calculate the percentage
PROCEDURE
Method
Introduce 150 mL of warer R into lite flask (A) and
equilibrate the whole system by passing carbon dioxide R for
15 min at a rate of about 100 mIJmin.
E
of residue.
If the amount of the residue so obtained exceeds the
prescribed limit, repeat the moistening with sulfuric acid R
and ignition, as previously, for 30 min periods until
2 consecutive weighings do not differ by more than 0.5 mg
or until the percentage of residue complies with the
prescribed limb.
D
The amount of substance used for the test (usually 1-2 g) is
chosen so that at the prescribed limit the mass of the residue
(usually about 1 mg) can be measured with sufficient
accuracy.
B. Determination of Sulfur Dioxide
Method I
(No Ph. Eur. me/hod)
Apparatus A round-bottomed flask of 1000- 10 1500-mL
capacity is fitted with a water-cooled reflux condenser the
upper end of which is connected to two absorption tubes in
series. The flask is fitted with a gas inlet tube which reaches
nearly to the bottom of the flask. Each absorption tube
contains 10 mL of hydrogen peroxide solution (20 wi)
previously neutralised willt O.lM sodium hydroxide VS using
bromophenol bluesolution as indicator.
Method Place in lite flask 500 mL of warer and 20 mL of
hydrochloric acid. Pass through lite flask a steady current of
nitrogen or carbon dioxide that has been bubbled through di/uce
I This chapter has und~ phannacopoeiaJ hannonisation. S~ chapter
5.8 Phamuuqpoeial harmonisation.
Figure 2.5.29.-1.- Appararus for the determination of su/fur
dioxide rontenl
www.webofpharma.com
2022
Appendix IX C V-A347
To 10 mL of dilute hydrogen peroxide solution R, add 0.15 mL
of a I gIL solution of bromophenol blue R in ethanol
(20 per cent V/~ R. Add 0.1 M sodium hydroxide until a
violet-blue colour is obtained, without exceeding the
end-point. Pour the solution into the receiving tube (D) and
moum the tube on the apparatus as shown in Figure
2.5.29.-1.
Without interrupting the stream of carbon dioxide, remove
the dropping funnel (B) and introduce into the flask (A)
25.0 g (m ) of the substance to he examined, rinsing with
100 mL of water R. Replace the dropping funnel, close the
tap and pour 80 mL of dilute hydrochloric acid R into the
funnel. Open the tap to allow the hydrochloric acid solution
to flow into me flask. Make sure that no sulfur dioxide
escapes by closing the tap before the last few millilitres of
hydrochloric acid solution drain OU[. Boil for 1 h.
Open the tap of the dropping funnel then stop the flow of
carbon dioxide . Transfer the contents of the receivingtube
(D) to a 200 mL conical flask, rinsing the rube with a little
water R. Heat on a water-bath for 15 min and allow to cool.
Add 0.1 mL of a I gIL solution of bromophenol blue R in
ethanol (20 per cent V/~ R and titrate with 0.1 M sodium
hydroxide until the colour changes from yellow to violetblue (V,). Carry out a blank titration (V,).
Results
Calculate me content of sulfur dioxide, in parts per million,
using the following expression:
n
32 030x(V,- V,) x m
VI
V2
volume of tirranr used in the titration, in millilitresj
volume of titrant used in the blank urraucn, in miUilitresi
II
molarityof the sodium hydroxidesolution used as titrant, in
m
moles per litrej
mass of the sample. in gruns.
C. Determination of Water
Use Method IA unless otherwise directed.
Method I Semi-micro Determination of Water
(Ph. Bur. manod 2.5.12)
The semi-micro determination of water is based upon the
quantitative reaction of water with sulfur dioxide and iodine
in a suitable anhydrous medium in the presence of a base
with sufficient buffering capacity.
APPARATUS
The apparatus consists of a titration vessel with:
- 2 identical platinum electrodes;
- tight inlets for introduction of solvent and titrant;
- an inlet for introduction of air via a desiccant;
- a sample inlet fitted with a stopper Of, for liquids, a
septum.
Inlet systems for introduction of dry nitrogen or for
aspiration of solvents may also be fitted.
The titration is carried out according to the instrument
supplier's instructions. Care is taken throughout the
determination to avoid exposure of reagents and solvents to
atmospheric moisture. The end-point is determined using 2
identical indicator electrodes connected to an electrical
source that maintains between the electrodes either a
constant current (2.2.65. Voitametl'k titration) or a constant
voltage (2.2.19. Amperomerric tilration). Where direct titration
is used (method A), addition of titrant causes either a
decrease in voltage where constant current is maintained or
an increase in current where constant voltage is maintained,
until the end-point is reached. Instruments with automatic
end-point detection are commonly used. Instrument
qualification is carried out according to established quality
system procedures, for example using a suitable certified
reference material (sodium aminosalicylau dihydraufor
C'/uipmen' qualifi<arion CRS may be used),
STANDARDISATION
To the titration vessel, add methanol R, dried if necessary, or
the solvent recommended by the supplier of the titrant.
Where applicable for the apparatus used) eliminate residual
water from the measurement cell or carry out a pre-titration.
Introduce a suitable amount of water in an appropriate form
(water R or a certified reference material) and carry out the
titration, stirring for the necessary time. The water equivalent
is not less than 80 per cent of that indicated by the supplier.
Standardise the titrant before the first use and at suitable
intervals thereafter.
Unless otherwise prescribed, use Method A.
METHOD A
Introduce into the titration vessel methanol R, or the solvent
indicated in the monograph or recommended by the supplier
of the titrant. Where applicable for the apparatus used,
eliminate residual water from the measurement ceU or carry
out a pre-titration. Introduce the substance to be examined
rapidly and cany out the titration, stirring for the necessary
extraction time.
METHODB
Introduce into the titration vessel methanol R, or the solvent
indicated in the monograph or recommended by the supplier
of the titrant. Where applicable for the apparatus used,
eliminate residual water from the measurement cell or carry
out a pre-titration. Introduce the substance to be examined
rapidly and in a suitable state of division. Add an accurately
measured volume of the titrant, sufficient to give an excess of
about 1 mL or the prescribed volume. Allow to stand
protected from light for 1 min or the prescribed time, with
stirring. Titrate the excess of reagent using methanol R or the
prescribed solvent, containing an accurately known quantity
of water.
SUITABIUTY
The accuracy of the detennination with the chosen titrant
must be verified for each combination of substance, titrant
and solvent to be examined. The following procedure, given
as an example, is suitable for samples containing 2.5-25 mg
of water.
The water content of the substance to be examined is
determined using the reagent/solvent system chosen.
Thereafter, in the same titration vessel, sequential known
amounts of water, corresponding to about 50-100 per cent of
the amount found in the substance to be examined, are
added in an appropriate fonn (at least 5 additions) and the
water content is determined after each addition. Calculate
the percentage recovery (r) after each addition using the
following expression:
r=IOO
WI
W2
w,
W,
amount of water added, in mllllgrams;
amount of water found, in milligrams.
www.webofpharma.com
2022
V-A348 Appendix IX C
Calculate the mean percentage recovery
0').
The reagem/solvent system is considered to be acceptable if' is
between 97.5 percentand 102.5 percen.
Calculate the regression line. The x-axis represents the
cumulative water added whereas the y-axis represents the
sum of the initial water content determined for the substance
(M) and the cumulative water determined after each
addition. Calculate the slope (b), the intercept with the y-axis
(a) and the intercept of the extrapolated calibration line with
the x-axis (d).
Calculate the percentage errors (el and
~
using the
following expressions:
e,
e,
Q
d
M
s
c
N
IU
a-M
~IOO-­
M
=
1001dl~M
they-axis intercept. in milligrams of water,
me x-axis intercept, in milligrams of water;
Willer content l)f Ihe subsrance, in rni.Ulgnuns of water.
The reagentfsolvent system is considered to be acceptable if:
- le,1 and le,l are not greater than 2.5 per cent;
-
b is between 0.975 and 1.025.
Method II Determination of Water by Distillation
(Ph. Eur. method 2.2.13)
The apparatus (see Figure 2.2.13.-1) consists ofa glass
flask (A) connected by a tube (D) to a cylindrical tube (B)
filled with a graduated receiving tube (E) and reflux
condenser (G). The receiving tube (E) is graduated in
0.1 mL. The source of heat is preferably an electric heater
with rheostat control or an oil bath. The upper portion of the
flask and the connecting tube may be insulated.
Method Clean me receiving tube and the condenser of the
apparatus, thoroughly rinse with water, and dry.
Introduce 200 mL of toluene R and about 2 mL of water R
into the dry flask. Distil for 2 h, then allow to cool for about
30 min and read the water volume to the nearest 0.05 mL.
Place in the flask a quantity of the substance, weighed with
an accuracy of 1 per cent, expected to give about 2 mL to
3 mL of water. H the substance has a pasty consistency,
weigh it in a boat of metal foil. Add a few pieces of porous
material and heal the flask gently for 15 min. When the
toluene hegins to boil, distil at the rate of about two drops
per second until most of the water has distilled over, men
increase the rate of distillation to about four drops per
second. When the water has all distilled over, rinse the inside
of the condenser tube with toluene R. Continue the
distiUation for 5 min, remove the heat, aUow the receiving
tube to cool to room temperature and dislodge any droplets
of water which adhere to the walls of the receiving tube.
When the water and toluene have completely separated, read
the volume of water and calculate the content present in the
substance as millilitres per kilogram, using the formula:
m
R.
R2
the mass in grams of the substance to be examined,
the number of millilitres of water obtained in the lint:
distiUauon.
the total number of millilitres of water obtained in the 2
distillations.
o
B
: s
•
, 3
E
....
'"
~
2
! Gml
'"'"
A
165
I
I
I
I
I
Figure 2.2.13.-1. - Apparatus for the detenninarion of water by
disu"Ualion
Dimensions in mi//imeeres
Method III Coulometrlc TItration
(ph. Eur. method 2.5.32)
PRINCIPLE
The coulometric titration of water is based upon the
quantitative reaction of water with sulfur dioxide and iodine
in an anhydrous medium in the presence of a base with
sufficient buffering capacity. In contrast to the volumetric
method described in general chapter 2.5.12. Water: semi-micro
determination, iodine is produced electrochemically in the
reaction cell by oxidation of iodide. The iodine produced at
www.webofpharma.com
Appendix IX D V-A349
2022
the anode reacts immediately with the water and the sulfur
dioxide contained in the reaction cell. The quantity of water
in the substance is directly proportional to the quantity of
electricity (in coulombs), corresponding to electric current (in
amperes) multiplied by time (in seconds), used for iodine
generation up until the titration end-point. When all of the
water in me reaction cell has been consumed, the end-point
is reached and thus an excess of iodine appears. 1 mole of
iodine corresponds to 1 mole of water, an amount of
electricity of 10.71 C corresponds to 1 mg of water.
Moisture is eliminated from the reaction cell by pre-titration,
i.e. the electrolyte reagent is titrated to dryness before starting
the sample analysis. Individual determinations can be carried
out successively in the same reagent solution) under the
following conditions:
each component of the test mixture is compatible with
me other components;
- no other reactions take place;
- the volume and the water capacity of the electrolyte
reagent are sufficient.
Coulornetric titration is intended for the quantitative
determination of small quantities of water (from 10-llg),
however a working range of 100 J.tg to 10 mg of water is
recommended for reproducibility reasons.
Accuracy and precision of the method are predominantly
governed by the sample preparation and the extent to which
atmospheric moisture is excluded from the system. Control
of the system must be monitored by measuring the amount
of baseline drift.
-
APPARATUS
The apparatus consists of a reaction cell, electrodes and a
magnetic stirrer. The reaction cell consists of a large anode
companment and a smaller cathode compartment.
Depending on the design of the electrode, both
compartments can be separated by a diaphragm. Each
compartment contains a platinum electrode. liquid or
solubilised samples are introduced through a septum, using a
syringe. Alternatively, an evaporation technique may be used
in which me sample is heated in an oven and the water is
evaporated and carried into the cell by means of a stream of
dry inert gas. The introduction of solid samples into the cell
should in general be avoided. However, if it has to be done it
is effected through a sealable port; appropriate precautions
must be taken to avoid the introduction of moisture from air,
such as working in a glove box in an atmosphere of dry inert
gas. The analytical procedure is controlled by a suitable
electronic device, which also displays the results.
Instrument qualification is carried out according to
established quality system procedures, for example using a
suitable certified reference material. Sodium aminosalicylau
dihydrate for equipmen, qualification CRS may be used when
proceeding by direct or liquid sample introduction, whereas
amoxicilJin lrihydrate for performance verifica.wn CRS may be
used with the evaporation technique.
METIIOD
Fill the compartments of the reaction cell with e/e<lrolyte
reagent for the micro determination 0/ WtUer' R according to the
manufacturer's instructions and perform the coulometric pretitration to a stable end-point. Introduce the prescribed
quantity of the substance to be examined into the reaction
cell and titrate again to a stable end-point, stirring for at least
30 s, unless otherwise indicated in the monograph. If an oven
is used, me prescribed quantity of sample is introduced into
the oven and heated. After evaporation of the water from the
sample into the reaction cell, the titration is started.
Alternatively, the evaporated moisture is immediately titrated
while heating the sample in the oven to avoid loss of
evaporated water already collected in the reagent solution
during prolonged heating. Read the value from the
instrument's output and calculate if necessary me percentage
or quantity of water that is present in the substance. When
appropriate to the type of sample and the sample
preparation, perform a blank titration.
VERIFICATION OF ACCURACY
At appropriate intervals, such as at least at the beginning and
the end of a series of sample titrations, introduce a defined
quantity of water, in the same order of magnitude as the
quantity of water in the sample, using a suitable certified
reference material and perform the coulometric titration.
The recovery is within the range of 97.5 per cent to
102.5 per cent for an addition of 1000 ~g of H 2 0 and within
the range of 90.0 per cent to 110.0 per cent for the addition
of 100 ~g of H 2 0 .
D. Determination of Loss on Drying
(Ph. Eur. method2.2.32)
PRINCIPLE
Loss on drying is the loss of mass after drying under
specified conditions, calculated as a percentage (mlm).
Drying to constant mass means that 2 consecutive weighings
do not differ by more than 0.5 mg, the 2n d weighing
following an additional period of at least 30 min of drying
under the conditions prescribed for the substance to be
examined.
EQUIPMENT
The equipment typically consists of:
- weighing bottles that are made of suitable inert material
and can easily be dried to constant mass; their diameter is
large enough so that the layer of the substance to be
examined does not exceed about 5 mm;
- an analytical balance by which it is possible to determine
a change in mass of 0.1 mg;
- depending on the procedure to be applied, a desiccator) a
vacuum cabinet, a vacuum oven or an ordinary laboratory
oven; in any case, the temperature of ovens is adjustable
to the specified temperature ± 2 °Cj vacuum ovens in
which the pressure can at least be reduced to about 2 kPa
are suitable; ovens are qualified according to established
quality system procedures, for example by using a suitable
certified reference material (sodium aminosalicylate
dihydrate for equipment qualificalion CRS may be used).
Equipment using other means of drying such as microwaves)
halogen lamps, infrared lamps or mixed technologies may be
used provided they are demonstrated to be fit for purpose.
PROCEDURE
It is recommended to perform the test in an environment
that has minimal impact on sample measurement
(e.g. humidity).
Weigh an empty weighing bottle that has been previously
dried under the conditions prescribed for me substance to be
examined for at least 30 min, then weigh the weighing bottle
filled with the prescribed quantity of substance to be
examined. Dry to constant mass or for the prescribed time.
Where the drying temperature is indicated by a single value
rather than a range, drying is carried out at the prescribed
www.webofpharma.com
V-A3S0 Appendix IX E
2022
temperature ± 2°C. Use one of the following procedures,
unless otherwise prescribed in the monograph.
-
a If-tube (U,) containing 30 g of reaystallised iodine
pentoxide R in granules, previously dried at 200°C and
-
In a desiccator: the drying is carried out over about 100 g
of molecular sieve R at atmospheric pressure and at room
temperature.
- In vacuo: the drying is carried out over about 100 g of
molecular sieve-R-at-a pressure not exceeding 2.5 kPa, at
room temperature or at the temperature prescribed in the
monograph.
- In an oven at a specified temperature: the drying is
carried out at annospheric pressure in an oven at the
temperature prescribed in the monograph.
After drying in an oven, allow the weighing bottle and the
sample to cool to room temperature in a desiccator and
weigh the weighing bottle containing the dried sample.
The mass of the sample is the difference between the mass of
the filled weighing bottle and the mass of the dried empty
weighing bottle.
The loss on drying is the difference in the mass of the sample
before and after drying, expressed as a percentage) mlm being
implicit.
kept at a temperature of 120 °C (1) during the test; the
iodine pentoxide is packed in the robe in 1 ern columns
separated by 1 em columns of glass wool to give an
effective length of 5 cm;
- a reaction tube (F2 ) containing 2.0 mL of poeassium iodide
solution Rand 0.15 mL of starch solutUm R.
Method Flush the apparatus with 5.0 L of argon R and, if
necessary, discharge the blue colour in the iodide solution by
adding the smallest necessary quantity of freshly prepared
0.002 M sodium thiosulfate. Continue flushing until not more
than 0.045 mL of 0.002 M sodium thiosul/a,. is required after
passage of 5.0 L of argon R. Pass the gas to be examined
from the cylinder through the apparatus, using the prescribed
volume and the flow rate. Flush the last traces of liberated
iodine into the reaction tube by passing through the
apparatus 1.0 L of argon R. Titrate the liberated iodine with
0.002 M sodium thiosulfate. Carry out a blank test, using the
prescribed volume of argon R. The difference between the
volumes of 0.002 M sodium thiosulfate used in the titrations is
not greater than the prescribed limit.
METHODD
Gases absorb light at one or more specific wavelengths. This
property is widely used to aUow highly selective measurement
of their concentrations.
E. Limit Test for Carbon Monoxide in
Medicinal Gases
Description and principle of measurement
The concentration of carbon monoxide in other gases can be
determined using an infrared analyser.
The infrared analyser generally consists of a light source
emitting broadband infrared radiation, an optical device, a
sample cell and a detector. The optical device may be
positioned either before or after the sample cell; it consists of
one or several optical filters, through which the broadband
radiation is passed. The optical device in this case is selected
for carbon monoxide. The measurement light beam passes
through the sample cell and may also pasa througb a
reference cell if the analyser integrates such a feature (some
use an electronic system instead of a reference cell).
(ph. Bur. method 2.5.25)
METHOD I
Apparatus The apparatus (Figure 2.5.25.-1) consists of
the foUowing parts connected in series:
- • Ll-tube (U,) containing anhydrous ri&a gel R
impregnated with chromium trWxide Rj
-
a wash bottle (F,) containing 100 mL of a 400 gIL
solution of potassium hydroxide R;
a Uctube (U2 ) containing pellets of poUlSSium hydroxide R;
a U-tube (U.) containing diphosphoTUS pemoxide R
dispersed on previously granulated, fused pumice;
U1
F1
U2
U3
U4
F2
o
o
II
II
II
~
11-'1·-
-= I 1-.
100 m
::1 j_-
_1,--I,,1--,
_.1-
Figure 2.5.25.-1. - Apparatus for the determination of carbon monoxide
Dimensions in millimetres
www.webofpharma.com
Appendix IX G V-A3S1
2022
When carbon monoxide is present in the sample cell,
absorption of energy in the measurement light beam will
occur according to the Beer-Lambert law and this produces a
change in the detector signal. This measurement signal is
compared to a reference signal to generate an output related
to the concentration of carbon monoxide. The generated
signal is linearised in order to obtain the carbon monoxide
concentration. To prevent the entry of particles into the
sensors, which could cause stray-light phenomena, the
apparatus is fitted with a suitable filter.
Required technical specifications
When used for a limit test, the carbon monoxide infrared
analysermeets me following technical specifications:
- lim;' 0/detection: (generaUy defined as a signal-to-noise
me
ratio of 2) maximum 20 per cent of
maximum
admissible concentration;
- repeatability: maximum relative standard deviation of
10 per cent of the maximum admissible concentration,
determined on 6 measurements;
- linean'ty: maximum 10 per cent of the maximum
admissible concentration.
The technical specifications must be met in the presence of
the other gas impurities in the sample.
F. Determination of Carbon Dioxide in
Medicinal Gases
broadband radiation is passed. The optical device in this case
is selected for carbon dioxide. The measurement light beam
passes through the sampJe cell and may also pass through a
reference cell if the analyser integrates such a feature (some
use an electronic system instead of a reference ceJl).
When carbon dioxide is present in the sample cell,
absorption of energy in the measurement light beam will
occur according to the Beer-Lambert law and this produces a
change in the detector signal. This measurement signal is
compared to a reference signal to generate an output related
to the concentration of carbon dioxide, The generated signal
is linearised in order to obtain the carbon dioxide
concentration. To prevent the entry of particles into the
sensors, which could cause stray-light phenomena, the
apparatus is fitted with a suitable filter.
Required technical specifications
When used for a Jimit test, the infrared analyser meets the
following technical specifications:
- limit of detection: (generally defined as a signal-to-noise
ratio of 2) maximum 20 per cent of the maximum
admissible concentration;
- repeatability: maximum relative standard deviation of
] 0 per cent of the maximum admissible concentration,
determined on 6 measurements;
- linean·ty: maximum J 0 per cent of the maximum
admissibJe concentration.
The technical specifications must be met in the presence of
the other gas impurities in the sample.
(Ph. Eur. method 1.5.24)
Gases absorb light at one or more specific wavelengths, This
property is widely used to allow highly selective measurement
of their concentrations,
Descrlpdon and principle of measurement
The concentration of carbon dioxide in other gases can be
determined using an infrared analyser.
The infrared analyser generally consists of a light source
emitting broadband infrared radiation, an optical device, a
sample cell and a detector. The optical device may be
positioned either before or after the sample cell and it
consists of one or several optical filters, through which the
G. Determination of Nitrogen Monoxide
and Nitrogen Dioxide in Medicinal Gases
(ph. Eur. method 1.5.16)
Nitrogen monoxide and nitrogen dioxide in gases are
determined using a chemiluminescence analyser
(Figure 2.5.26.-1).
The apparatus consists of the following:
- a device for filtering, checking and controlling the flow of
the gas to be examined,
Sample now control
Converter
NO
N02~
Reaction chamber
Opllcal filler
IRefrigeraled chamber
~-
-0-
Filter 10eliminate
ozone
Photomultiplier lube
Ozone generator
~---10
system
. Controls
- NO - (NO+NO,) cycle
NO
(NO+NO,)
NO,
Figure 2.5.26.-J. - Chemiluminescence analyser
www.webofpharma.com
V-A352 Appendix IX H
-
-
-
a converter that reduces nitrogen dioxide (0 nitrogen
monoxide) [Q determine the combined content of nitrogen
monoxide and nitrogen dioxide. The efficiency of the
convener has to be verified prior to use,
a controlled-flow-rate ozone generator; the ozone is
produced by high-voltage electric discharges across two
electrodes; the ozone generator is supplied with pure
oxygen or with dehydrated ambient air and the
concentration of ozone obtained must greatly exceed the
maximum content of any detectable nitrogen oxides,
a chamber in which nitrogen monoxide and ozone can
react,
-
a system for detecting light radiation emitted at a
wavelength of 1.2 JIm, consisting of a selective optical
filter and a photomultiplier tube.
H. Determination of Oxygen in Medicinal
Gases
(ph. Bur. method 2.5.27)
Oxygen in gases is determined using a paramagnetic analyser.
The principle of the method is based on the high
paramagnetic sensitivity of the oxygen molecule. Oxygen
exerts a strong interaction on magnetic fields, which is
measured electronically, amplified and converted to a reading
of oxygen concentration. The measurement of oxygen
concentration is dependent upon the pressure and
temperature and, if the analyser is not automatically
compensated for variations in temperature and pressure, it
must be calibrated immediately prior to use. As the
paramagnetic effect of oxygen is linear, the instrument must
have a suitable range with a readability ofO.t per cent or
better,
Calibration of the instrument Make the setting in the
following manner:
- set the zero by passing nitrogen RJ through the instrument
until a constant reading is obtained;
- set the scale 10 100 per cent by passing oxygen R through
the instrument at the same flow rate as for nitrogen RJ
until a constant reading is obtained.
Assay Pass the gas to he examined through the instrument
at a constant flow rate until a constant reading is obtained.
Record the concentration of oxygen in the gas to be
examined.
.
J. Determination of Water in Medicinal
Gases
(ph. Bur. method 2.5.28)
Water in gases is determined using an electrolytic
hygrometer, described below.
The measuring cell consists of a thin film of diphosphorus
pentoxide, between 2 coiled platinum wires which act as
electrodes. The water vapour in the gas to be examined is
absorbed by the diphosphorus pentoxide, which is
transformed to phosphoric acid, an electrical conductor.
A continuous voltage applied across the electrodes produces
electrolysis of the water and the regeneration of the
diphosphorus pentoxide. The resulting electric current, which
is proportional to the water content in the gas to be
2022
examined, is measured. This system is self-calibrating since it
obeys Faraday's law.
Take a sample of the gas to be examined. Allow the gas to
stabilise at room temperature. Purge the cell continuously
until a stable reading is obtained. Measure the water content
in the gas to be examined, making sure that the temperature
is constant throughout the device used to introduce the gas
into the apparatus.
The electrolytic hygrometer achieves accurate sample flows
by using a mass flow controller to deliver a constant
volumetric flow rate to ensure that the water content is
determined accurately. The calibration of the mass flow
controller is normally performed using nitrogen. When using
gases other than nitrogen for calibration, consult the
manufacturer's instructions for the appropriate conversion
factors and ensure that the correct cell is used for the type of
gas to be examined.
K. Gas Detector Tubes
(Ph. Bur. method 2.1.6)
Gas detector tubes are cylindrical) sealed tubes consisting of
an inert transparent material and are constructed to allow the
passage of gas. They contain reagents adsorbed onto inert
substrates that are suitable for the visualisation of the
substance to be detected and, if necessary, they also contain
preliminary layers and/or adsorbent filters to eliminate
substances that interfere with the substance to be detected.
The layer of indicator contains either a single reagent for the
detection of a given impurity or several reagents for the
detection of several substances (monolayer tube or multilayer
tube).
The test is carried out by passing the required volume of the
gas to be examined through the indicator tube. The length of
the coloured layer or the intensity of a colour change on a
graduated scale gives an indication of the impurities present.
The calibration of the detector tubes is verified according to
the manufacturer's instructions.
Operating conditions Examine according to the
manufacturer's instructions or proceed as follows.
The gas supply is connected to a suitable pressure regulator
and needle valve. Connect the flexible tubing fitted with a
Y -piece to the valve and adjust the flow of gas to be
examined to purge the tubing in order to obtain an
appropriate flow (Figure 2.1.6.-1). Prepare the indicator tube
and fit to the metering pump, following the manufacturer's
instructions. Connect the open end of the indicator tube to
the short leg of the tubing and operate the pump by the
appropriate number of strokes to pass a suitable volume of
gas to be examined through the tube. Read the value
corresponding to the length of the coloured layer or the
intensity of the colour on the graduated scale. If a negative
result is achieved, indicator tubes can be verified with a
calibration gas containing the appropriate impurity.
In view of the wide variety of available compressor oils, it is
necessary to verify the reactivity of the oil detector tubes for
the oil used. Information on the reactivity for various oils is
given in the leaflet supplied with the tube. If the oil used is
not cited in the leaflet, the tube manufacturer must verify the
reactivity and if necessary provide a tube specific for this oil.
Arsine detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for the gold salt or other appropriate indicator.
www.webofpharma.com
2022
Appendix IX M V-A353
The minimum value indicated is 0.25 ppm or less, with a
relative standard deviation of at most 20 per cent.
Carbon dioxide detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for hydrazine and crystal violet indicators.
The minimum value indicated is 100 ppm with a relative
standard deviation of at most 15 per cent.
Carbon monoxide detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for di-iodine pentoxide, selenium dioxide and
fuming sulfuric acid indicators. The minimum value
indicated is 5 ppm or less, with a relative standard deviation
of at most 15 per cent.
Hydrogen sulfide detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for an appropriate lead salt indicator. The minimum
value indicated is 0.2 ppm or less, with a relative standard
deviation of at most 10 per cent.
Nitrogen monoxide and nitrogen dioxide detector tube
Sealed glass tube containing adsorbent filters and suitable
support. for an oxidelng layer (Cr(Vl) salt) and the
diphenylbenzidine indicator. The minimum value indicated is
0.5 pp~ with a relative standard deviation of at most
15 per-cent.
Oil detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for the sulfuric acid indicator. The minimum value
indicated is 0.1 mglm3 with a relative standard deviation of at
most 30 per cent.
Phosphine detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for the gold salt or other appropriate indicator.
The minimum value indicated is 0.2 ppm or less, with a
relative standard deviation of at most 20 per cent.
Sulfur dioxide detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for the iodine and starch indicator. The minimum
value indicated is 0.5 ppm with a relative standard deviation
of at most 15 per cent.
Water vapour detector tube
Sealed glass tube containing adsorbent filters and suitable
supports for the magnesium perchlorate indicator.
The minimum value indicated is 67 ppm or less, with a
relative standard deviation of at most 20 per cent.
2
3
4
}---{>(1--G"<I------,------7
5
6
I. Gas supply
2. Pressure regulator
3. Needle valve
4. V-pioce
5. Indicator tube
6. Indicator rube pump
7. End open to atmosphere
Figure 2.1.6.-1. - Apparatus fqr gas detector tubes
L. Determination of Nitrous Oxide in
Gases
(ph. Eur. method 2.5.35)
Gases absorb light at one or more specific wavelengths. This
property is widely used to allow highly selective measurement
of their concentrations.
Description and principle of measurement
The concentration of nitrous oxide in other gases can be
determined using an infrared analyser.
The infrared analyser generally consists of a light source
emitting broadband infrared radiation) an optical device, a
sample cell and a detector. The optical device may be
positioned either before' or after the sample cell and it
consists of one or several optical filters, through which the
broadband radiation is passed. The optical device in this case
is selected for nitrous oxide. The measurement light beam
passes through the sample cell and may also pass through a
reference cell if the analyser integrates such a feature (some
use an electronic system instead of a reference cell).
When nitrous oxide is present in the sample cell, absorption
of energy in the measurement light beam will occur
according to the Beer-Lambert law and this produces a
change in the detector signal. This measurement signal is
compared to a reference signal to generate an output related
to the concentration of nitrous oxide. The generated signal is
linearised in order to obtain the nitrous oxide concentration.
To prevent the entry of particles into the sensors) which
could cause stray-light phenomena) the apparatus is fitted
with a suitable filter.
M. Water-Solid Interactions:
Determination of Sorption.Deso~tion
Isotherms and of Water Activity
(Ph. Eur. method 2.9.39)
INTRODUCTION
Pharmaceutical solids as raw materials or as constituents of
dosage forms most often come in contact with water during
processing and storage. This may occur (a) during
,
crystallisation, Iyophilisation, wet granulation) or spray
drying; and (b) because of exposure upon handling and
storage to an atmosphere containing water vapour or
exposure to other materials in a dosage fonn that contain
water capable of distributing it to other ingredients. Some
properties known to be altered by the association of solids
with water include rates of chemical degradation in the
"solid-state", crystal growth and dissolution, dispersibility
and wetting) powder flow, lubricity, powder compactibility,
compact hardness and microbial contamination.
Although precautions can be taken when water is perceived
to be a problem, i.e. eliminating all moisture, reducing
contact with the atmosphere) or controlling the relative
humidity of the atmosphere, such precautions generally add
expense to the process with no guarantee that during the life
of the product further problems associated with moisture will
be avoided. It is also important to recognise that there are
many situations where a certain level of water in a solid is
required for proper performance) e.g. powder compaction.
I 17Iis chapter has undergone ph~a1 harmtmisation. Su chapter
5.8. Pharmacopoeial hannonisotion.
www.webofpharma.com