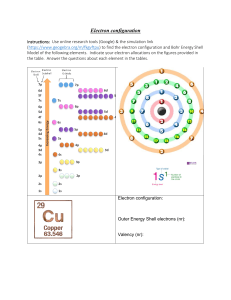
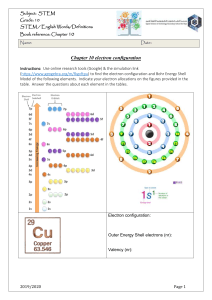
Biomentors Classes Online, Mumbai www.biomentors.online Chemistry Class Notes DAY-27 l.c om CHEMICAL BONDING: L1 Atom Always have tendency to attain stability by attaining the configuration of Nobel gas and lower its energy gm ai Valency: Combining power of an atom is called its valency. The outer most orbit of an atom is called valence shell and the number of Electron present in the valence shell is called valence electron 𝐶𝑙 → 2,8,7 𝑉𝑎𝑙𝑒𝑛𝑐𝑦 𝑆ℎ𝑒𝑙𝑙 Why Chemical bonds form? ni m 52 @ Chemical Bond: the attraction force which hold various constituents together in different chemical species is called chemical bond Kossel Lewis – concept (Octet rule) 𝑯𝒆𝟐 , 𝑵𝒆𝟐,𝟖 𝑨𝒓𝟐,𝟖,𝟖 ar Atom with incomplete Octet always have tendency to form chemical bond by gain of Electron, loss of 𝑒 − or sharing of electron is w After the formation of chemical bond the number of electron in the valence shell of central metal atom is 8 and its Co – valences are 4 𝐶𝐻4 𝐶6 → 2, 4 sr is ht Why chemical bond form The electron are denoted by dot or cross and the bond are denoted by straight line, it is known as Lewis – Structure . Exception of Lewis Octet rules: Molecule with incomplete Octet are the molecules in which number of electrons are less than 8 in the valence shell with incomplete octet. 𝐵𝐹3 = 𝐵5 → 2,3 , 1 1 @ gm 𝑁 → 1𝑠 2 2𝑠 2 2𝑝3 ai 𝑁𝑂2 = 𝑁7 → 2, 5 l.c om 𝐴𝑙𝐶𝑙3 = 𝐴𝑙13 → 2, 8, 3 𝐵𝑒𝐶𝑙2 = 𝐵𝑒4 → 2, 2 Molecule with Incomplete Octet (𝐵𝐹3 , 𝐴𝑙𝐶𝑙3 , 𝐵𝑒𝐶𝑙2 ) have tendency to gain electron pair, it is also known as Lewis acid or Electrophile Molecule with expansion Octet: these are those molecules in which no of electrons are more than 8 in the valency of control atom. 𝑃𝐶𝑙5 = 𝑃15 → 2, 8, 5 𝑆𝐹4 = 𝑆 → 2, 8, 6 𝑆𝐹6 = 𝑆 → 2, 8, 6 Sidgwick’s Rule for maximum Co – valency: If the number of 𝑒 − in the valency shall of control atom is more than 8 then Co – Valency are also possible more than 4 molecule with odd electron bond are those molecules in which unpaired electron’s are present in the valency shell of central atom. 𝑁𝑂 = 𝑁7 → 2, 5 sr is ht is w ar ni m 52 Molecule with odd electron bond 𝑁𝑂2 , 𝑁𝑂 are paramagnetic 𝑁𝑂2 is Brown colour gas 𝑁𝑂 = is colourless 2