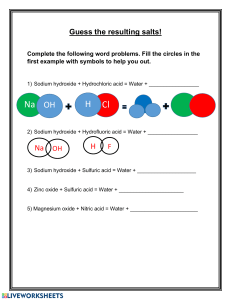
WORD EQUATIONS Write a word equation to sum up the following reactions. 1) Magnesium reacts with oxygen to form magnesium oxide. ................................................................................................................................ 2) Hydrochloric acid reacts with magnesium to form magnesium chloride and hydrogen. ................................................................................................................................ 3) Sodium hydroxide reacts with hydrochloric acid to form sodium chloride and water. ................................................................................................................................ 4) Nitric acid reacts with iron to form iron nitrate and hydrogen. ................................................................................................................................ 5) Copper carbonate reacts with hydrochloric acid to form copper chloride, water and carbon dioxide. ................................................................................................................................ 6) Calcium oxide reacts with nitric acid to form calcium nitrate and water. ................................................................................................................................ CHEMICAL EQUATIONS 1) Complete the following table. Name Formula Hydrochloric acid HCl Sodium chloride NaCl Sodium hydroxide NaOH Potassium hydroxide KOH Hydrogen fluoride Magnesium chloride HF MgCl2 Types of atoms How many of each type? 2) Write balanced chemical equations for the following reactions. a) Hydrogen gas reacts with oxygen gas to form water. ................................................................................................................................ b) Electricity is passed through water which reacts to form hydrogen gas and oxygen gas. ................................................................................................................................ c) Hydrochloric acid reacts with sodium hydroxide to form sodium chloride and water. ................................................................................................................................ d) Hydrochloric acid reacts with elemental magnesium metal to form magnesium chloride and hydrogen gas. ................................................................................................................................ e) Potassium metal reacts with water to form potassium hydroxide and hydrogen gas. ................................................................................................................................ MODELLING EQUATIONS Sketch a model for the following equations which you balanced above. 1) Hydrogen gas reacts with oxygen gas to form water. 2) Potassium metal reacts with water to form potassium hydroxide and hydrogen gas.