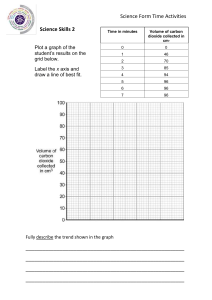
NAME DATE CLASS ANALYZING DATA Intensive and Extensive Properties Scientists have identified over 100 elements. Each element has its own unique set of physical and chemical properties. However, differences may also exist between samples of the same element. For example, a sample of iron may have a mass of 1 milligram, 1 gram, or 1 kilogram. The shape of the sample may be a cube, an irregular shape, or many tiny filings. The table shows the observed properties of several samples of three elements: hydrogen, sodium, and iron. Properties of Element Samples Sample Element 1 hydrogen 2 State Mass Volume Density Appearance Reactivity gas 8.2 mg 100 cm3 0.083 colorless reacts explosively with oxygen hydrogen gas 4.1 mg 50 cm3 0.02 colorless reacts explosively with oxygen 3 hydrogen gas 1.2 mg 15 cm3 0.08 colorless reacts explosively with oxygen 4 sodium solid 9.7 g 10 cm3 0.97 silvery-white cube reacts vigorously at surface with water and oxygen 5 sodium solid 2.4 g 2.5 cm3 0.96 silvery-white pieces reacts vigorously with water and oxygen 6 sodium solid 5.2 g 5.4 cm3 0.96 silvery-white flakes reacts quickly and vigorously with water and oxygen 7 iron solid 7.9 g 1.0 cm3 shiny, grayish flakes surface rusts when exposed to oxygen 8 iron solid 2.6 g 0.33 cm3 shiny, surface rusts grayish pellet when exposed to oxygen 9 iron solid 94.8 g 12 cm3 shiny, surface rusts grayish block when exposed to oxygen Copyright © Savvas Learning Company LLC. All Rights Reserved. Savvas is not responsible for any modifications made by end users to the content posted in its original format. NAME DATE CLASS 1. SEP Calculate Density is equal to mass divided by volume. Calculate the density for each of the nine samples, and record the values in the table. Be sure to include the appropriate unit for each value. 2. CCC Patterns What pattern do you observe in the data? 3. SEP Analyze Data The properties of matter can be classified into two groups. Extensive properties vary with the amount of matter in a sample. Intensive properties remain the same, or are independent of the amount of matter present. Analyze the data in the table to identify examples of extensive and intensive properties. Explain your classifications. 4. SEP Engage in Argument A classmate claims that state of matter is an extensive property because it can vary with temperature. For example, hydrogen changes into a liquid at very low temperatures and high pressure, while iron can melt into a liquid at very high temperatures. Do you agree with the classmate? Write an argument to support your position. Apply Concepts Two chemists each plan an investigation in which they will combine hydrogen gas with chlorine gas to observe the chemical reactivity. The plans differ in that one will use 25 milligrams each of hydrogen and chlorine, and the other will use 1000 times this amount, i.e., 25 grams each of hydrogen and chlorine. Can the investigations reach similar conclusions about the reactivity of hydrogen with chlorine? Explain. Copyright © Savvas Learning Company LLC. All Rights Reserved. Savvas is not responsible for any modifications made by end users to the content posted in its original format. NAME DATE CLASS Copyright © Savvas Learning Company LLC. All Rights Reserved. Savvas is not responsible for any modifications made by end users to the content posted in its original format.