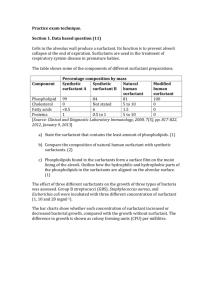
SPE 129892 Ping Zhao (SPE), A. J.Howes (SPE), Varadarajan Dwarakanath (SPE), Sophany Thach (SPE), Taimur Malik (SPE), Adam Jackson (SPE), Oya Karazincir (SPE), Curt Campbell, and Jeff Waite (SPE), Chevron Copyright 2010, Society of Petroleum Engineers This paper was prepared for presentation at the 2010 SPE Improved Oil Recovery Symposium held in Tulsa, Oklahoma, USA, 24–28 April 2010. This paper was selected for presentation by an SPE program committee following review of information contained in an abstract submitted by the author(s). Contents of the paper have not been reviewed by the Society of Petroleum Engineers and are subject to correction by the author(s). The material does not necessarily reflect any position of the Society of Petroleum Engineers, its officers, or members. Electronic reproduction, distribution, or storage of any part of this paper without the written consent of the Society of Petroleum Engineers is prohibited. Permission to reproduce in print is restricted to an abstract of not more than 300 words; illustrations may not be copied. The abstract must contain conspicuous acknowledgment of SPE copyright. Abstract The success of chemical flooding rests heavily on the ability to deliver high-quality and repeatable surfactant in the field. While high-quality surfactants can easily be produced at lab scale, high oil recovery can be achieved only if similar quality product is produced at field scale. Scale-up of chemical production from batch-scale laboratory processes to field-scale continuous production requires a comprehensive program to evaluate surfactants produced at the bench-, pilot-plant and commercial scale to establish surfactant performance. Given the difference between batch-scale and continuous processes, small differences in surfactant structure can result thereby inducing differences in phase behavior and consequently oil recovery. Rigorous laboratory work is therefore required to synthesize and characterize surfactant samples in order to understand the correlation between structures/composition and performance in oil recovery. Following laboratory synthesis, pilot-plant studies are used to investigate process variables in order to understand their viability range and their impact on the large-scale product. Other important concerns are: optimal operational conditions, process repeatability, supply chain management, logistics, quality assurance/quality control, feedstock availability and dedicated procurement team. We present a case study for a proposed light oil surfactant polymer flood where a rigorous path for scale-up of surfactants from lab to pilot and finally field scale was evaluated via phase behavior experiments and coreflood testing. We show that phase behavior results are well correlated with coreflood recovery. For EOR, they are superior performance criteria to traditional specifications of physical and compositional properties for quality control. For the two-surfactant system presented herein, 272 surfactant samples, 512 formulations and 20 corefloods were conducted. The results from this large effort were utilized to develop appropriate manufacturing and quality-control processes to ensure the delivery of high-performance surfactants for field application. Introduction Many early Chemical-EOR field trials failed to produce desired results and poor performance was attributed to the failure of chemical flooding technology. With hindsight, a contributing factor to poor performances was the failure to deliver to the field the same quality surfactant that was used in laboratory testing. It is difficult to estimate how many field trials failed for this reason as the problem was seldom discussed or even recognized in the literature despite its importance. Two field-test results hinted at the problem of under-performing surfactants. At Bell Creek, the petroleum-sulfonate blends used in the pilot contained far more polysulfonates than the products used in the laboratory design (Holm, 1982). Chromatographic separation (Salter, 1986) clearly occurred in the field with unfortunate consequences. Highly water-soluble and fast-moving polysulfonates broke through early causing severe emulsions in the produced fluids. The altered composition of the trailing micellar slug probably contributed to sub-optimal performance at Bell Creek. In one of the pilots in Indonesia (Bou-Mikael et al., 2000), post mortem analysis suggested that the synthetic sulfonate used in the field trial was not compositionally identical to the laboratory product though it was not clear how much this difference affected pilot performance. While many sub-surface uncertainties are uncontrollable, the quality of the commercial surfactants used in the field is within our control. A comprehensive evaluation program and QA/QC criteria are needed to ensure that field surfactant meets expected performance. To clearly understand changes in phase behavior due to compositional and molecular changes, surfactants must be analyzed and their performance in corefloods must be evaluated at all stages of their production, from laboratory synthesis to pilot-plant and finally commercial-plant manufacturing. The scale-up from batch synthesis in the Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Evaluation and Manufacturing Quality Control of Chemicals for Surfactant Flooding 2 SPE 129892 laboratory to a continuous process in the pilot plant is particularly fraught with difficulties. Pilot-plant conditions must be carefully evaluated to understand their impacts at each processing step (e.g. alkylation, and sulfonation for alkyl aryl sulfonates). The variation in the feedstock and its availability can further complicate issues, especially at the commercial scale. Finally, blending, transportation and storage of the final commercial product may impact surfactant performance as well. Background A multi-year effort was devoted to develop a surfactant formulation for a light oil chemical flood pilot. This intensive program includes surfactant and polymer screening, development and laboratory testing of high performance formulations for the field pilot, and scale up of surfactant manufacturing from bench scale to the pilot-plant- and commercial scale. From the surfactant screening program for the light oil pilot, a two-surfactant system (AAS, an alkyl aryl sulfonate and OS, an olefin sulfonate, both manufactured by Chevron Oronite) emerges as the most promising formulation for the proposed pilot (Zhao, 2008). The addition of a second surfactant (OS) offers significant advantages: 1) OS boosts the performance of AAS, the single surfactant used in the first pilot; 2) a two-surfactant system adds flexibility in optimizing its EOR performance with light oil crude; 3) a two-surfactant system allows small adjustments in the final commercial-scale blends to compensate for possible variations in commercial quality of either surfactant. Certain changes in the operating conditions at the pilot-plant or commercial-plant scale can have a large impact on the product conversion (activity), isomeric distribution, byproducts, etc. These changes can significantly impact surfactant phase behavior and coreflood performance as will be seen later. To define the optimal operating conditions in the plant, over two hundred surfactants (batch and plant samples) and over five hundred formulations were painstakingly tested in phasebehavior experiments and twenty corefloods. This intensive program, which includes several pilot-plant and commercial scale runs, was undertaken to ensure that commercial-scale surfactants can be made reproducibly and meet the specifications and performance of the benchmark samples used in laboratory process design. Materials and Methods Phase behavior procedures were similar to those described by Levitt and Jackson, 2006. In a borosilicate glass pipette, two ml of oil were mixed with 2 ml of surfactant solution in brine at different salinities. The pipettes were then sealed and placed in an oven at reservoir temperature and allowed to mix and equilibrate. The oil-microemulsion and microemulsion-water interfaces were recorded for use in calculating the solubilization ratios of oil and water. In all phase-behavior experiments, the total surfactant concentration was 2 wt% and the ratio of the two surfactants was 3 to 1. The co-solvent concentration was set at 3 wt%. The most promising formulations from the phase behavior experiments are further tested in core flood experiments and their performance measured against a target oil recovery. Briarhill sandstone was used as surrogate for reservoir rock as it had similar permeability and cation exchange capacity to the reservoir rock. Cores were assembled and potted in epoxy with taps to measure differential pressure internally and across the core. Cores were initially saturated with synthetic reservoir brine at reservoir. Following the brine flood the cores were flooded with crude oil at a relatively high pressure gradient until a water cut of 2% or less was observed. The cores were aged at reservoir temperature then water-flooded at a low pressure gradient (1 psi/ft) which translates to a flow rate of approximately 1-5 ft/day. The effluent fluid samples and pressure drops were observed until there was no oil in the effluent and the pressure drop across the core stabilized. Finally, the cores were flooded with the surfactant. The surfactant slugs used in all the corefloods contained an AMPS co-polymer produced by SNF. Surfactant Manufacturing and Scale-up Developing commercially viable multi-component surfactant systems for an EOR field trial is challenging and requires a logically staged process to meet the desired performance criteria. For the light oil project, the surfactant acceptance workflow used for each stage is illustrated in Figure 1. This paper will cover each of the stages: (1) surfactant development, (2) scale up of surfactant production, (3) formulation blending, and (4) on-site delivery and evaluation. Successful completion of these stages can lead to a valuable EOR field trial that generates the data necessary to evaluate the long-term viability of full field expansion. Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Physical properties and chemical composition are typically used in surfactant industry to ensure that product meets specifications. Unfortunately, the phase behavior and physical properties of surfactants are sensitive to often small compositional and isomeric variations (Salter, S.J., 1983), which are not easily quantified to the desired precision by routine analyses. Phase behavior and core-floods provide the ultimate assurance of surfactant quality and performance. In this work, we found that seemingly small, unsuspected changes in surfactant composition caused by variations in synthesis steps, plant operating conditions and feedstock can lead to significant changes in surfactant performance in Chemical EOR. A comprehensive program has been developed to ensure high and reproducible performance for the commercial surfactant. SPE 129892 3 In addition to selecting the appropriate surfactant(s) for a specific reservoir, EOR performance criteria must be clearly defined. The performance tests for this project were the commonly used techniques: phase behavior, corefloods, and reservoir simulations. The surfactant formulation performance screening criteria at Stage 1 (Figure 1) were: optimal salinity (S*), optimum solubilization ratio (SP), equilibration time into fluid middle phases, microemulsion viscosity and core flood oil recovery. With the desired surfactant chemistries and performance criteria defined, the next step is to prepare laboratory samples of the surfactants using varying conditions to determine the performance sensitivity to surfactants with varying molecular properties. This allows one to understand the relationship between surfactant structure and performance. Simultaneously the raw materials, intermediates, and surfactants were characterized using standard analytical techniques and in some cases new analytical methods. For choosing the raw materials used in surfactant synthesis, a procurement team is helpful to understand the availability and quality of raw materials in the market place. Typical process conditions that are varied include reaction temperatures, pressures, catalyst type and loading, raw material charge molar ratios, flow rates, and reactor type (e.g., fixed bed, batch or continuous). In these studies, it is important to select process variables and ranges that make sense from a commercial feasibility perspective even though this work is still at the laboratory synthesis stage. Working with commercially unreasonable conditions and/or raw materials can lead to unattainable scale-up of the surfactant synthesis from the laboratory to full plant scale manufacturing. The molecular properties of all raw materials, intermediates, and surfactants were probed using the following analytical techniques (Greay and Chan, 1990 and Varadaraj et. al. 1990): nuclear magnetic resonance spectroscopy, (Carbon and Proton NMR), infrared (IR) and mass spectroscopy (MS), gas chromatography (GC), supercritical fluid chromatography (SFC) and high performance liquid chromatography (HPLC). Molecular properties such as the chain length (Kadam, 2006) aromatic ring isomers (Hsieh and Shah, 1977), aromatic ring size (Zhigang et.al. 2006), and alkyl chain attachment (Borchardt, 1987) are known to affect interfacial surfactant properties such as surface tension, critical micelle concentration, surface adsorption, and cloud point. Other surfactant properties that are important include pH, metals content, water content, molecular weight, activity and physical properties such as viscosity, density, flashpoint, sediment, color, and appearance. For each laboratory surfactant sample the analytical, molecular and physical properties were recorded along with the corresponding performance. Included with the individual surfactant performance was performance testing results of combinations of the primary surfactant, co-surfactant, and co-solvent at various treat rates. As illustrated in Figure 1, Stage 1 is an iterative process in which alky aryl sulfonate and olefin sulfonate samples were prepared, their analytical, physical, and molecular properties measured and then their performance determined. The performance and analytical results are then evaluated and changes in molecular properties are considered for additional surfactant syntheses. This process may take several iterations until acceptable performance is achieved and a surfactant or formulation is deemed acceptable. Once a sufficient amount of performance and analytical data has been generated, the performance results along with the analytical data were statistically analyzed to derive structure performance information and identify and quantify the critical surfactant properties that correlate to acceptable performance of the multi-component formulation. Using the results from statistical analysis, additional laboratory samples were prepared using narrower process condition ranges to understand how small changes in the critical surfactant properties affect performance. In addition, surfactant syntheses were repeated to determine surfactant preparation repeatability. These results provided the basis for defining the acceptable ranges of surfactant properties critical to acceptable performance. Once a formulation, or set of formulations, has been identified, it is prudent to run storage stability studies to identify any possible surfactant degradation upon storage. For this project, we ran separate storage stability studies for each component of the formulation and the entire formulation itself. If any component or the whole formulation is not stable for at least several months, it may not be suitable for storage and shipment to a reservoir for EOR and alternative formulations may need to be developed. As the surfactant development progresses through Stage 1 and even into Stage 2, environmental compliance Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Stage 1: Surfactant Development The first step in the development of a surfactant system for any reservoir is to determine the surfactant, co-solvent, and, if necessary, co-surfactant chemistries necessary to achieve acceptable enhanced oil recovery performance. Historically, EOR surfactant chemistries have included various alky aryl sulfonates, olefin sulfonates, and alcohol ether sulfates. The selection of the appropriate chemistry depends on specific reservoir properties: temperature, pressure, oil properties, rock characteristics, reservoir brine composition, etc. For the light oil project the primary surfactant chosen was an alkyl aryl sulfonate (AAS). A co-surfactant olefin sulfonate (OS) was selected based on the use of olefin sulfonates with high wax crude oils by Zhao et. al. (2008). Common co-solvent chemistries include alcohols and glycol ethers and for the light oil project, the co-solvent chosen was based upon previous work for light oil reservoirs and high wax content crude oils by Dwarakanath et. al. (2008). 4 SPE 129892 issues such as EPA registration, pre-manufacturing notification (PMN), and, if necessary, toxicological testing must be addressed. It is of little value to develop a surfactant that cannot be used outside the laboratory. Listed in Table 1 are various samples of AAS synthesized in the laboratory under selected conditions to explore the impact of physical, compositional and molecular properties on performance. To compare their performance, various AAS samples were systematically tested in formulations containing the same benchmark OS. Similarly, all new OS lab- and pilot-plant samples were tested with the benchmark AAS. In all, 272 lab- and pilot-plant surfactant samples were tested in 512 different formulations. Figures 3 and 4 summarize the results of phase-behavior evaluations in which 91 AAS lab samples were tested with benchmark OS surfactant. Only 5 formulations met both the optimal salinity and solubilization ratio requirements. The physical, analytical and molecular properties of the AAS samples, especially those with good performances, are carefully correlated to synthesis conditions and feedstock. Often minor changes in certain molecular components of the AAS led to surprisingly large shifts in either S* or SP*, or both (Figure 4). This evaluation process led to the conclusion that the best route to boost the performance of the formulation was to modify the properties of the OS. Once a formulation, or set of formulations, is established, it is prudent to run storage stability studies to identify possible sample degradation. If any part of the formulation, or the whole formulation, is not stable for months at a time, it may not be suitable for storage and shipment to a field for EOR. Alternative formulations may need to be developed or the components in the formulation may need to be stored separately until the time of the field trial. Stage 2: Scale-up of Surfactant Production In our experience, meeting the performance criteria during scale-up of the surfactant manufacturing process from the laboratory to the plant is the most difficult stage of chemical surfactant development for EOR performance. Traditionally, the next step following laboratory preparations is to use pilot plant facilities to prepare materials. Pilot plants must be carefully chosen such that they provide process information that can be scaled to a full scale plant. For sulfonate chemistries, Roberts describes the optimization of linear alkyl benzene sulfonation using falling film sulfonation reactors (Roberts, 2003) and various sulfonation technologies for linear alkylbezene, primary alcohols, ethoxylated alcohols, and alpha olefins (Roberts, 1998). These articles explain the complexities of these chemistries and outline guidelines to be successful in scaling up sulfonation processes. For the light oil project, statistically designed pilot plant studies were used to determine the impact of larger scale processing conditions on the alkyl aryl sulfonate and olefin sulfonate surfactant molecular properties followed by performance testing. Not uncommonly, pilot plant produced surfactants had different analytical properties compared to the laboratory prepared materials which required additional pilot plant studies and further performance testing such that this stage is also an iterative process as in Stage 1 and exemplifies why Stage 2 is the most difficult stage of an EOR project. A thorough discussion of the results at Stage 2 is required to determine how best to proceed. If a decline in performance is unacceptable, one may need to re-examine the entire pilot plant process. For example, there may be a need to examine pilot plant data to determine if any operational upsets occurred, identify any key differences in raw materials used, and examine more closely samples from the pilot plant. Some off-line processing of material may be required as well. Ultimately, additional pilot plant runs may be required to generate acceptable performing material with altered process conditions. Once acceptable pilot plant material is generated, additional storage stability studies were conducted to determine if there is any degradation in performance upon storage under nominal storage conditions. Next, document and clearly define all process conditions and product specifications with tolerance ranges. The conditions and specifications are then used in a plant trial in a commercial facility. For this project, small plant trials in commercial facilities were initially conducted to gain experience with process operations and determine the range of surfactant properties that can be generated. Like the initial lab work, samples were analyzed and performance tested using phase behavior. Figure 5 shows the poor performance of the initial pilot sample. These results were used to enhance the performance of future plant trial materials. Figure 6 show a comparison of the initial pilot sample, the improved pilot sample and the benchmark sample. All of these samples were also core flooded. Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Phase-Behavior Performance— The formulations developed for this project (AAS + OS + Co-Solvent) must meet or exceed two main performance criteria: 1) The optimal salinity (S*) should be between 0.7-1.1 wt% of Na2CO3. 2) The optimum solubilization parameter (SP*) should be greater than 8. These criteria applied to all laboratory (batch) samples, pilot-plant and commercial-plant samples used in phase-behavior experiments. Figure 2 shows the phase behavior of AAS and OS surfactant samples used as benchmark: 1) Optimal salinity, S* = 0.75 wt%. 2) Optimum solubilization parameter, SP= 9. SPE 129892 5 Coreflood Performance—All corefloods used the following sequence of slugs: • • • 0.15 PV surfactant slug at optimal salinity 0.10 PV of polymer chase (with 1.5 wt% co-solvent) More than 1.0 PV of polymer chase in synthetic reservoir brine. Once an acceptable pilot plant material was generated for the pilot, storage stability studies were conducted to monitor any long term performance degradation. The next task was to document and clearly define all process conditions and product specifications with tolerance ranges. The conditions and specifications are then used in a plant trial in a commercial facility. Small plant trials in commercial facilities were initially conducted to gain experience with process operations and determine the range of surfactant properties that can be generated. Like the pilot plant work, samples were analyzed and performance tested. The results obtained were used to enhance the performance of future plant trial materials. Finally, storage stability studies were conducted on the plant trial materials to monitor any long term product degradation. Figure 8 shows the phase behavior of AAS and OS surfactant 2009 Plant Trial samples which were stored at 40°C for 115 days. The S* is about 1.10 wt% of Na2CO3 and the optimum solubilization parameter (SP) is greater than 8. Both of these results meet the requirement for acceptable behavior. Stage 3: Formulation Blending Stage 3 of the surfactant development flow sheet involves blending the primary surfactant, co-surfactant, and co-solvent to produce the EOR formulation. Before shipment of the final blend to the reservoir, surfactant samples are blended in the laboratory and performance tested. During the performance the treat rates of each surfactant can be varied to determine the optimal blend. As illustrated in Stage 3 of Figure 1, this process is iterative and may require discussions if the final formulation performance is less than satisfactory. Once a final formulation is agreed upon, a large scale blend is conducted and the material is ready for delivery to the reservoir. Stage 4: Delivery and On-Site Evaluation The final step of the surfactant development flow sheet is to transport the surfactant formulation from a blending facility to the reservoir. It is imperative that one work with a supply chain and logistics team to ensure product integrity. A partial list of issues includes: mode of transport, type of containers, cross contamination, reliability of shipping companies, best shipping routes, environmental concerns, customs issues, and ultimately the capability of the reservoir facilities to receive, handle, sample, and weigh material. For this pilot, we have minimized many of these issues by shipping the blended formulation in sealed ISO containers and selected a reliable and safe delivery route to avoid any timing disruptions. Once on-site there will be some analysis and performance testing of each ISO container prior to injection to assure product integrity. As illustrated in Stage 4 of Figure 1, depending on these test results a decision may need to be made about whether or not to accept certain ISO containers or the entire shipment. These decisions are critical as they ultimately impact the quality of the EOR field trial results. Full Field Expansion If the light oil pilot is successful, manufacturing sufficient materials for a full field expansion is non-trivial. For the current formulation, there are insufficient raw materials currently available in the global market place to produce the amount of surfactant required for full field implementation. Dedicated commercial facilities would likely need to be built to supply the current formulation to the reservoir. However, modifications to surfactant chemistry, treat rates, and new and improved EOR technologies may make full field expansion more attainable. Research in this area is currently in progress. Conclusions Too often in the past, chemical EOR was deployed in field trials based entirely on the laboratory performance of surfactants often synthesized in small batches in the laboratory. Even today, some field trials still use surfactants whose “full commerciality” remains to be demonstrated. This work showed that: • Small compositional or molecular changes that are difficult to quantify via usual chemical or instrumental analyses can often lead to large performance differences. Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 The pressure drop in the experiments was kept below 1 psi/ft at a rate of 1 ft/day. Figure 7 shows oil recovery for three corefloods corresponding to the benchmark formulation, the formulation using initial pilot-plant samples of AAS and OS and the final formulation using improved pilot-plant samples of these surfactants. Coreflood results are also summarized in Table 2. As expected, the improved formulation with good phase behavior also performed better in coreflood. These results indicate excellent correlation between phase behavior results and coreflood recovery. They also further illustrate the importance of phase behavior testing and establishment of such benchmarks for surfactant performance. 6 SPE 129892 • • • • • • • Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 • Phase-behavior performance (minimally S* and SP*) and corefloods are the most sensitive and thus the most reliable criteria for surfactant performance in the field. A two-surfactant formulation gives the flexibility to optimize the performance with adjustment for one of the products in the blend. This flexibility is very useful at the commercial scale where sub-par performance of individual component can be expected. To ensure delivery of high-performance and repeatable surfactant product in the field, a comprehensive, timeconsuming but necessary program for commercial surfactant manufacturing must be developed One must establish surfactant performance criteria which are germane to field performance; traditional physical and chemical specifications alone are not sufficient to ensure expected performance. Important synthesis variables at the small-batch scale in the lab must be understood and correlated to physical analytical and molecular properties of the final product. Pilot plants must be used at key manufacturing steps of surfactants and a viable range of process variables should be explored to understand their impact on surfactant properties and performance. Blending, transportation, logistics for field delivery and storage must be carefully evaluated and a detailed QA/QC plan developed ahead of time to ensure that the field surfactant-blend performs as well as the small-scale product used in design. Full-field expansion requires raw-material quantities that may even exceed the global market capacity. The availability of feedstock required must be evaluated before field trial. SPE 129892 7 # Samples S* SP* Oil Recovery Soc Table2. Coreflood Summary Benchmark 2009 0.7 1.0 9 8 96% 92% 1.5%±2% 3.3%±2% Comments Lab samples Lab samples Large-scale lab samples Field batch Lab batches 2008 1.3 5.5 71% 10.6%±2% Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 15 27 5 1 91 Table1. Summary of AAS Laboratory Syntheses Analytical criteria met Phase Behavior Criteria Met 1 3 5 4 1 0 15 SPE 129892 8 Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Figure 1: Surfactant Acceptance Workflow 9 SPE 129892 Figure 3: Comparison of Optimal Salinity for Benchmark and 91AAS Laboratory samples Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Figure 2: Phase Behavior of Benchmark Sample: S* ~ 0.75 wt%; SP* > 8 10 SPE 129892 Figure 5: Example of Initial Pilot-Plant Sample with Unacceptable S* (>> 0.75%) and SP* (< 8) Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Figure 4: Comparison of Solubilization Ratio for Benchmark and 91 CS 2000 Laboratory samples SPE 129892 Figure 7: Comparison of Cumulative Oil Recovery Initial, Improved Pilot-Plant Samples vs. Benchmark Samples Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Figure 6: Comparison of Phase Behavior of Initial, Improved Pilot-Plants Samples with Benchmark Samples, 11 SPE 129892 12 Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Figure 8: Storage Stability of 2009 Plant Trial Sample SPE 129892 13 References Bou-Mikael, S., F. Asmadi, D. Marwoto, and C. Cease. “Light oil Surfactant Field Trial Tests Two Newly Designed Surfactants with High EOR Potential.” SPE 62488, presented at the SPE Asia Pacific Oil and Gas Conference in Brisbane, Australia, October 2000. Dwarakanath, V., T. Chaturvedi, A.C. Jackson, T. Malik, A. Siregar, and P. Zhao. “Using Co-Solvents to Provide Gradients and Improve Oil Recovery during Chemical Flooding in a Light Oil Reservoir.” SPE 113965 presented at the 2008 SPE/DOE Improved Oil Recovery Symposium in Tulsa, Oklahoma, April 2008. Greay, R.A., and A.F. Chan. “Sulfonation Studies of Monoisomeric Di- and Trialkylbenzenes.” Journal of the American Oil Chemists’ Society. 67 (1990): 132 – 141 (1990) Holm, L.W. Design, Performance, and Evaluation of the Uniflood Micellar-Polymer Process—Bell Creek Field. SPE 11196 presented at the 57th Annual Fall Technical Conference and Exposition of the Society of Petroleum Engineers of AIME, held in New Orleans, LA. Sept. 26-29. 1982 Hsieh, W.C., and D.O. Shah. “The Use of High Resolution NMR Spectroscopy for Characterizing Petroleum Sulfonates.” SPE 6600, presented at the 1977 SPE-AIME International Symposium on Oilfield and Geothermal Chemistry in La Jolla, California, June 1977. Jackson, A.C. “Experimental Study of the Benefits of Sodium Carbonate on Surfactants for Enhanced Oil Recovery.” M.S. Thesis, University of Texas, December 2006. Kadam, M.M., M.R. Sawant. “Interfacial and Thermodynamic Properties of Anionic-Nonionic Mixed Surfactant System: Influence of Hydrophobic Chain Length of the Nonionic Surfactant.” Journal of Dispersion Science and Technology. 27 (2006): 861 – 868. Levitt, D.B., Jackson, A.C., Heinson, C., Britton, L.N., Malik, T., Dwarakanath, V., Pope, G.A.: “Identification and Evaluation of High Performance EOR Surfactants,” SPE 100089, presented at the 2006 SPE/DOE Symposium on Improved Oil Recovery held in Tulsa, Oklahoma, April 2006. Roberts, D.W. “Optimization of the Linear Alkyl Benzene Sulfonation Process for Surfactant Manufacture.” Organic Process Research and Development. 7 (2003): 172 – 184. Roberts, D.W. “Sulfonation Technology for Anionic Surfactant Manufacture.” Organic Process Research and Development. 2 (1998): 194 – 202. Salter, S.J. Optimizing Surfactant Molecular Weight Distribution. 1. Sulfonate Phase Behavior and Physical Properties. Paper SPE 12036, presented at the Annual Technical Conference and Exhibition held at San Francisco, CA. October 5-8. 1983. Salter, S.J. Criteria for Surfactant Selection in Micellar Flooding. Paper SPE 14106, presented at International Meeting on Petroleum Engineering held in Beijing, China. March17-20. 1986 Varadaraj, R., P. Valint, J. Bock, S. Zushma, and N. Brons. “Synthesis and Fundamental Interfacial Properties of Two Isomeric Alkyl Xylene sulfonate Surfactants.” Langmuir. 7 (1990): 658 – 664. Zhigang, X., B. Chenguang, T. Yuehong, Q. Weihong, L. Zongshi, and C. Lubo. “Effect of Aromatic Ring in the Alkyl Chain on Surface Properties of Aryl Alkyl Sulfonate Surfactants.” Tenside Surfactants Detergents. 43 (2006): 142 – 145. Zhao, P., A.C. Jackson, C. Britton, D.H. Kim, L.N. Britton, D.B. Levitt, and G.A. Pope. “Development of High-Performance Surfactants for Difficult Oils.” SPE 113432 presented at the 2008 SPE/DOE Improved Oil Recovery Symposium in Tulsa, Oklahoma, April 2008. Downloaded from http://onepetro.org/SPEIOR/proceedings-pdf/10IOR/All-10IOR/SPE-129892-MS/1770310/spe-129892-ms.pdf/1 by Bandung Inst. of Tech., Ivan Kurnia on 09 January 2022 Borchardt, J.K. “Foaming Agents for EOR: Correlation of Surfactant Performance Properties With Chemical Structure.” SPE 16279 presented at SPE International Symposium on Oilfield Chemistry in San Antonio, Texas, February 1987.