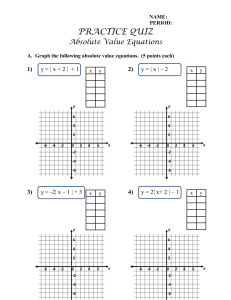
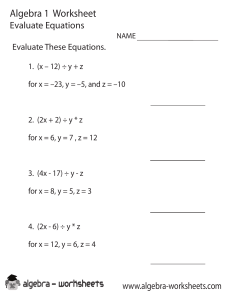Subido por: Libros de Ingeniería Química y más https://www.facebook.com/pages/InterfaseIQ/146073555478947?ref=bookmarks Si te gusta este libro y tienes la posibilidad, cómpralo para apoyar al autor. IntroduccIón a la termodInámIca en IngenIería químIca 00-SmithVanNess Preliminares.ind1 1 8/1/07 12:46:17 00-SmithVanNess Preliminares.ind2 2 8/1/07 12:46:17 IntroduccIón a la termodInámIca en IngenIería químIca SÉPtIma edIcIón J. m. Smith University of California, Davis H. c. Van ness m. m. abbott Rensselaer Polytechnic Institute reVISIón tÉcnIca misael Flores rojas Profesor de Termodinámica ESIQIE, Instituto Politécnico Nacional MÉXICO • AUCKLAND • BOGOTÁ • BUENOS AIRES • CARACAS • GUATEMALA LISBOA • LONDRES • MADRID • MILÁN • MONTREAL • NUEVA DELHI • NUEVA YORK SAN FRANCISCO • SAN JUAN • SAN LUIS • SANTIAGO SÃO PAULO • SIDNEY • SINGAPUR • TORONTO 00-SmithVanNess Preliminares.ind3 3 8/1/07 12:46:17 Director Higher Education: Miguel Ángel Toledo Castellanos Director editorial: Ricardo A. del Bosque Alayón Editor sponsor: Pablo E. Roig Vázquez Editora de desarrollo: Lorena Campa Rojas Supervisor de producción: Zeferino García García Traducción: Efrén Alatorre Miguel Esther Fernández Alvarado Emilio Sordo Zabay IntroduccIón a la termodInámIca en IngenIería químIca Séptima edición Prohibida la reproducción total o parcial de esta obra, por cualquier medio, sin la autorización escrita del editor. DERECHOS RESERVADOS © 2007, respecto a la séptima edición en español por McGRAW-HILL/INTERAMERICANA EDITORES, S.A. DE C.V. A Subsidiary of The McGraw-Hill Companies, Inc. Edificio Punta Santa Fe Prolongación Paseo de la Reforma 1015, Torre A Piso 17, Col. Desarrollo Santa Fe, Delegación Álvaro Obregón C.P. 01376, México, D. F. Miembro de la Cámara Nacional de la Industria Editorial Mexicana, Reg. Núm. 736 ISBN-13: 978-970-10-6147-3 ISBN-10: 970-10-6147-0 (ISBN edición anterior: 970-10-3647-6) Traducido de la séptima edición en inglés de la obra: INTRODUCTION TO CHEMICAL ENGINEERING THERMODYNAMICS, by J.M. Smith, H.C. Van Ness, M.M. Abbott. Copyright © 2005, 2001, 1996, 1987, 1975, 1959, 1949 by The McGraw-Hill Companies, Inc. All rights reserved. ISBN 10: 0-07-310445-0 ISBN 13: 978-0-07-310445-4 1234567890 09865432107 Impreso en México Printed in Mexico 00-SmithVanNess Preliminares.ind4 4 8/1/07 12:46:19 Contenido lista de símbolos Ix Prefacio xV 1 IntroduccIón 1.1 El panorama de la termodinámica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.2 Dimensiones y unidades . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.3 Medidas de cantidad o tamaño . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.4 Fuerza . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.5 Temperatura . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.6 Presión . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.7 Trabajo . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.8 Energía . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.9 Calor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1 1 2 2 3 5 6 8 10 15 2 la PrImera ley y otroS concePtoS báSIcoS 2.1 Experimentos de Joule . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.2 Energía interna . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.3 La primera ley de la termodinámica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.4 Balance de energía para sistemas cerrados . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.5 Estado termodinámico y funciones de estado . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.6 Equilibrio . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.7 Regla de las fases . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.8 El proceso reersible . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.9 Procesos con V y P constantes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.10 Entalpía . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.11 Capacidad calorífica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 2.12 Balances de masa y energía para sistemas abiertos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21 21 21 22 23 26 29 29 31 37 38 40 44 3 ProPIedadeS VolumÉtrIcaS de FluIdoS PuroS 3.1 Comportamiento PVT de sustancias puras . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3.2 Ecuaciones de estado iriales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3.3 El gas ideal . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 64 64 70 73 00-SmithVanNess Preliminares.ind5 5 8/1/07 12:46:19 i 3.4 3.5 3.6 3.7 Aplicación de las ecuaciones iriales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Ecuaciones cúbicas de estado . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Correlaciones generalizadas para gases . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Correlaciones generalizadas para líquidos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 87 90 99 109 4 eFectoS tÉrmIcoS 4.1 Efectos del calor sensible . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.2 Calores latentes de sustancias puras . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.3 Calor estándar de reacción . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.4 Calor estándar de formación . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.5 Calor estándar de combustión . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.6 Dependencia con la temperatura de ΔH ° . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4.7 Efectos térmicos de las reacciones industriales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 125 125 133 135 136 139 140 143 5 la Segunda ley de la termodInámIca 5.1 Presentación de la segunda ley . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.2 Máquinas térmicas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.3 Escalas de temperatura termodinámica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.4 Entropía . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.5 Cambios de entropía de un gas ideal . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.6 Planteamiento matemático de la segunda ley . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.7 Balance de entropía para sistemas abiertos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.8 Cálculo del trabajo ideal . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.9 Trabajo perdido . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.10 La tercera ley de la termodinámica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5.11 Entropía desde el punto de ista microscópico . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 159 160 161 163 167 170 173 176 181 185 188 188 6 ProPIedadeS termodInámIcaS de loS FluIdoS 6.1 Expresiones para la ealuación de una propiedad en fases homogéneas . . . . . . . . . . . . . . . . 6.2 Propiedades residuales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6.3 Las propiedades residuales a partir de ecuaciones de estado . . . . . . . . . . . . . . . . . . . . . . . . . 6.4 Sistemas de dos fases . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6.5 Diagramas termodinámicos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6.6 Tablas de propiedades termodinámicas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6.7 Correlaciones generalizadas para la ealuación de una propiedad para gases . . . . . . . . . . . 199 199 208 215 220 225 226 230 7 aPlIcacIoneS de la termodInámIca a loS ProceSoS de FluJo 7.1 Flujo en conductos de fluidos compresibles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7.2 Turbinas (expansores) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7.3 Procesos de compresión . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 254 255 268 273 8 generacIón de PotencIa a PartIr del calor 8.1 Planta de energía de apor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8.2 Motores de combustión interna . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8.3 Motores de reacción: motor de propulsión . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 290 291 302 310 9 reFrIgeracIón y lIcueFaccIón 9.1 El refrigerador de Carnot . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 317 317 00-SmithVanNess Preliminares.ind6 6 8/1/07 12:46:19 ii 9.2 9.3 9.4 9.5 9.6 Ciclo de compresión de apor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . La elección del refrigerante . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Refrigeración por absorción . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . La bomba de calor . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Proceso de licuefacción . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 318 321 323 326 327 10 equIlIbrIo VaPor/líquIdo: IntroduccIón 10.1 La naturaleza del equilibrio . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10.2 Regla de fase . Teorema de Duhem . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10.3 EVL: Comportamiento cualitatio . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10.4 Modelos simples para el equilibrio apor/líquido . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10.5 EVL mediante la ley de Raoult modificada . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10.6 EVL a partir de las correlaciones del alor K . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 338 338 339 341 347 358 363 11 termodInámIca de SolucIoneS: teoría 11.1 Relación de una propiedad fundamental . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.2 Potencial químico y equilibrio de fase . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.3 Propiedades parciales . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.4 Modelo de mezcla de gas ideal . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.5 Fugacidad y coeficiente de fugacidad: especies puras . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.6 Fugacidad y coeficiente de fugacidad: especies en solución . . . . . . . . . . . . . . . . . . . . . . . . . 11.7 Correlaciones generalizadas para el coeficiente de fugacidad . . . . . . . . . . . . . . . . . . . . . . . . 11.8 El modelo de solución ideal . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11.9 Propiedades de exceso . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 378 378 380 381 391 394 401 407 411 413 12 termodInámIca de SolucIoneS: aPlIcacIoneS 12.1 Propiedades de fase líquida a partir de la información de EVL . . . . . . . . . . . . . . . . . . . . . . . 12.2 Modelos para la energía de Gibbs de exceso . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12.3 Cambios en la propiedad de mezclado . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 12.4 Efectos térmicos de los procesos de mezclado . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 430 430 446 449 456 13 equIlIbrIo en reaccIón químIca 13.1 Coordenada de la reacción . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13.2 Aplicación de criterios de equilibrio a las reacciones químicas . . . . . . . . . . . . . . . . . . . . . . 13.3 Cambio en la energía de Gibbs estándar y la constante de equilibrio . . . . . . . . . . . . . . . . . . 13.4 Efecto de la temperatura en la constante de equilibrio . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13.5 Ealuación de las constantes de equilibrio . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13.6 Relación de las constantes de equilibrio con la composición . . . . . . . . . . . . . . . . . . . . . . . . 13.7 Conersiones de equilibrio para reacciones únicas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13.8 Regla de la fase y teorema de Duhem para sistemas con transformaciones químicas . . . . . 13.9 Equilibrios en reacciones múltiples . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 13.10 Celdas de combustible . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 483 484 488 489 492 496 498 502 514 518 529 14 temaS Sobre loS equIlIbrIoS de FaSe 14.1 Formulación gamma/phi del EVL . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14.2 EVL a partir de ecuaciones de estado cúbicas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14.3 Equilibrio y estabilidad . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 545 545 556 575 00-SmithVanNess Preliminares.ind7 7 8/1/07 12:46:20 viii 14.4 14.5 14.6 14.7 14.8 14.9 Equilibrio líquido/líquido (ELL) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Equilibrio vapor/líquido/líquido (EVLL) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Equilibrio sólido/líquido (ESL) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Equilibrio sólido/vapor (ESV) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Equilibrio de adsorción de gases en sólidos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Equilibrio osmótico y presión osmótica . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 581 590 597 602 606 621 15 Análisis termodinámico de procesos 15.1 Análisis termodinámico de procesos de flujo en estado estacionario . . . . . . . . . . . . . . . . . . 635 635 16 introducción A lA termodinámicA moleculAr 16.1 Teoría molecular de los fluidos . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16.2 Segundos coeficientes viriales a partir de las funciones potenciales. . . . . . . . . . . . . . . . . . . 16.3 Energía interna de gases ideales: punto de vista microscópico . . . . . . . . . . . . . . . . . . . . . . . 16.4 Propiedades termodinámicas y mecánica estadística. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16.5 Enlace de hidrógeno y complejo de transferencia de carga . . . . . . . . . . . . . . . . . . . . . . . . . . 16.6 Comportamiento de las propiedades de exceso . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16.7 Bases moleculares para el comportamiento de mezclas . . . . . . . . . . . . . . . . . . . . . . . . . . . . 16.8 EVL por simulación molecular . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 647 647 654 657 660 662 665 669 672 A Factores de conversión y valores de la constante de los gases 677 B propiedades de especies puras 679 c capacidades caloríficas y cambios de propiedad de formación 683 d programas representativos para computadora d.1 Funciones definidas . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . d.2 Solución de problemas de ejemplo con Mathcad® . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 688 688 691 e tablas de correlación generalizada de lee/Kesler 695 F tablas de vapor F.1 Interpolación . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 712 712 G diagramas termodinámicos 788 H método uniFAc 791 i método de newton 798 Índice de autores 803 Índice analítico 807 00-SmithVanNess Preliminares.ind8 8 10/1/07 14:02:01 Lista de símbolos A A A a a a a-i B B B̂ B′ B0, B1 Bij b bi C C Ĉ C′ C 0, C1 CP CV CP° ΔCP° CPH CPS CP°H Área Energía de Helmholtz molar o específica ≡ U − TS Parámetro, ecuaciones Empíricas, por ejemplo, en las ecuaciones (4 .4), (6 .76) y (12 .14) Aceleración Área molar de una fase adsorbida Parámetro en las ecuaciones de estado cúbicas Parámetros parciales en ecuaciones de estado cúbicas Segundo coeficiente irial, expansión en densidades Parámetro en ecuaciones empíricas, por ejemplo, en las ecuaciones (4 .4), (6 .76) y (12 .14) Segundo coeficiente irial reducido, definido por la ecuación (3 .62) Segundo coeficiente irial, expansión en presiones Funciones de la correlación generalizada del segundo coeficiente irial Interacción del segundo coeficiente irial Parámetro en ecuaciones de estado cúbicas Parámetro pracial en ecuaciones de estado cúbicas Tercer coeficiente irial, expansión en densidades Parámetro, en ecuaciones empíricas, por ejemplo, ecuaciones (4 .4), (6 .76) y (12 .14) Tercer coeficiente irial reducido, definido en la página 103 . Tercer coeficiente irial, expansión en presiones Funciones, correlación generalizada del tercer coeficiente irial Capacidad calorífica molar o específica, a presión constante Capacidad calorífica molar o específica, a olumen constante Capacidad calorífica en el estado estándar, a presión constante Cambio en la capacidad calorífica estándar de reacción Capacidad calorífica media para cálculos de entalpía Capacidad calorífica media para cálculos de entropía Capacidad calorífica estándar media para cálculos de entalpía ix 00-SmithVanNess Preliminares.ind9 9 8/1/07 12:46:21 x CP°S c D D D′ Ei EK EP F F F fi f i° fˆi G Gi° – Gi GE GR ΔG ΔG° ΔGf° g gc gi H Hi Hi° – Hi HE HR (HR)0, (HR)1 ΔH ∼ Δ H ΔH ° ΔH 0° ΔH °f h I Capacidad calorífica estándar media para cálculos de entropía Rapidez del sonido Cuarto coeficiente irial, expansión en densidades Parámetro en ecuaciones empíricas, por ejemplo, en las ecuaciones (4 .4) y (6 .77) Cuarto coeficiente irial, expansión en presiones Niel de energía Energía cinética Energía potencial graitacional Grados de libertad, regla de las fases Fuerza Constante de Faraday Fugacidad, especies puras i Fugacidad en el estado estándar Fugacidad de la especie i en solución Energía de Gibbs molar o específica ≡ H – TS Energía de Gibbs en el estado estándar de la especie i Energía de Gibbs parcial de la especie i en solución Energía de Gibbs de exceso ≡ G – G i d Energía de Gibbs residual ≡ G – G i g Cambio en la energía de Gibbs por mezclado Cambio en la energía de Gibbs estándar de reacción Cambio en la energía de Gibbs estándar de formación Aceleración local de la graedad Constante dimensional = 32 .1740(lbm)(ft)(lbr)–1(s)–2 Degeneración Entalpía molar o específica ≡ U + PV Constante de Henry de la especie i en solución Entalpía en el estado estándar de la especie pura i Entalpía parcial de la especie i en solución Entalpía de exceso ≡ H – H i d Entalpía residual ≡ H – H i g Funciones en la correlación de la entalpía residual generalizada Cambio de entalpía (“calor”) por mezclado; también calor latente de transición de fase Calor de solución Cambio en el entalpía estándar de reacción Calor estándar de reacción a la temperatura de referencia T0 Cambio en la entalpía estándar de formación Constante de Planck Representa una integral definida, por ejemplo en las ecuaciones (6 .65) 00-SmithVanNess Preliminares.ind10 10 8/1/07 12:46:22 xi I Kj Ki k L l li j m M M – Mi ME MR ΔM ΔM ° ΔM f° m ṁ N NA n ṅ ñ ni P P° Pc Pr P 0r , P 1r P0 pi Pisat Q Q· q q q q-i R 00-SmithVanNess Preliminares.ind11 11 Primer potencial de ionización Constante de equilibrio para la reacción química j Constante de equilibrio apor/líquido para la especie i ≡ yi / xi Constante de Boltzmann Fracción molar líquida del sistema Longitud Parámetro de interacción de una ecuación del estado, ecuación (14 .101) Número de Mach Masa molar (peso molecular) Valor moral o específico, de una propiedad termodinámica extensia Propiedad parcial de la especie i en solución Propiedad de exceso ≡ M – M i d Propiedad residual ≡ M – M i g Cambio en una propiedad por mezclado Cambio en una propiedad estándar de reacción Cambio en una propiedad estándar de formación Masa Rapidez de flujo de masa Número de especies químicas, regla de las fases Número de Aogadro Número de moles Rapidez de flujo de moles Moles de solente por mol de soluto Número de moles de la especie i Presión absoluta Presión en el estado estándar Presión crítica Presión reducida Funciones, correlación generalizada apor-presión Presión de referencia Presión parcial de la especie i Presión de apor de saturación de la especie i Calor Rapidez de transferencia de calor Rapidez de flujo olumétrico Parámetro en las ecuaciones de estado cúbicas Carga eléctrica Parámetro parcial en las ecuaciones de estado cúbicas Constante uniersal de los gases (tabla A .2) 8/1/07 12:46:22 xii r r r S – Si SE SR (SR)0, (SR)1 SG S·G ΔS ΔS ° ΔS °f T Tc Tn Tr T0 Tσ Tisat t t U U u V V – Vi Vc Vr VE VR ΔV W · W Wideal · Wideal Wperdido · Wperdido Relación de compresión Separación intermolecular Número de reacciones químicas independientes, regla de fases Entropía molar o específica Entropía parcial de la especie i en solución Entropía de exceso ≡ S – S i d Entropía residual ≡ S – S i g Funciones en la correlación de entropía residual generalizada Generación de entropía por cantidad unitaria de fluido Rapidez de generación de entropía Cambio de entropía por mezclado Cambio de entropía estándar de la reacción Cambio de entropía estándar de la formación Temperatura absoluta en kelin o rankine Temperatura crítica Temperatura normal de ebullición Temperatura reducida Temperatura de referencia Temperatura absoluta de los alrededores Temperatura de saturación de la especie i Temperatura, °C o (°F) Tiempo Energía interna molar o específica Función del par potencial intermolecular Velocidad Volumen molar o específico Fracción molar del sistema que es apor Volumen parcial de la especie i en solución Volumen crítico Volumen reducido Volumen de exceso ≡ V – V i d Volumen residual ≡ V – V i g Cambio de olumen por mezclado; también, cambio de olumen de la transición de fase Trabajo Rapidez de trabajo (potencia) Trabajo ideal Rapidez de trabajo ideal Trabajo perdido Rapidez de trabajo perdido 00-SmithVanNess Preliminares.ind12 12 8/1/07 12:46:23 xiii Ws · Ws xi xv yi Z Zc Z 0, Z 1 Z z z zi Trabajo de flecha para procesos de flujo Potencia de flecha para procesos de flujo Fracción molar de la especie i en general o en una fase líquida Calidad Fracción molar de la especie i en la fase apor Factor de compresibilidad ≡ PV / RT Factor de compresibilidad crítica ≡ PcVc / RTc Funciones en la correlación generalizada de factores de compresibilidad Funcion de partición Factor de compresibilidad de fase adsorbida definida por la ecuación (14 .108) Eleación por encima de un niel de referencia Fracción molar global o fracción molar en una fase sólida Superíndices E av R s sl t v ∞ Denota una propiedad termodinámica de exceso Denota una transición de fase de la fase adsorbida a apor Denota una propiedad termodinámica residual Denota fase sólida Denota transición de fase de sólido a líquido Denota un alor total de una propiedad termodinámica extensia Denota fase de apor Denota un alor a dilución infinita Letras Griegas α α α, β αβ β β l γ γl δ 00-SmithVanNess Preliminares.ind13 13 Función en las ecuaciones del estado cúbicas (tabla 3 .1, p .98) Polarizabilidad Como superíndices, identifican fases Como superíndices, denota una transición de fase de la fase α a la fase β Expansiidad del olumen Parámetro en las ecuaciones de estado cúbicas Constante de integración Relación de las capacidades caloríficas CP / CV Coeficiente de actiidad de la especie i en solución Exponente politrópico Constante en las ecuaciones de estado cúbicas 8/1/07 12:46:23 xi 0 ε η κ π µ µ µi νi ρ ρc ρr σ σ τ i φi φ̂i φ0, φ1 , ω Profundidad del pozo en la función de potencial intermolecular Permitiidad eléctrica en el acío Coordenada de reacción Eficiencia Comprensibilidad isotérmica Presión de dispersión en fase absorida Presión osmótica Número de fases, regla de las fases Coeficiente de Joule/Thompson Momento dipolar Potencial químico de la especie i Número estequiométrico de la especia i Densidad molar o específica ≡ 1/V Densidad crítica Densidad reducida Constante en las ecuaciones de estado cúbicas Diámetro de colisión molecular Relación de temperatura ≡ T / T0 [En la ecuación (6 .77), τ ≡ 1 – Tr ] Relación de coeficientes de fugacidad, definida por la ecuación (14 .2) Coeficiente de fugacidad de la especie pura i Coeficiente de fugacidad de la especie i en solución Funciones en la correlación generalizada de coeficientes de fugacidad constantes en las ecuaciones de estado cúbicas Factor acéntrico Notas c fs ° – · ˆ Δ Como subíndice, denota un olumen de control Como subíndice, denota corriente de flujo Como superíndice, denota el estado estándar La sobrebarra denota una propiedad parcial El sobrepunto denota una rapidez El circunflejo denota una propiedad en solucion Operador de diferencia 00-SmithVanNess Preliminares.ind14 14 8/1/07 12:46:24 Prefacio La termodinámica, uno de los temas centrales de la ciencia, está basada en leyes de aplicación universal. La justificación para presentar el tema desde el punto de vista de la Ingeniería Química es que tenemos la convicción de que es más efectivo pensar en el contexto de la disciplina que interesa y compromete al estudiante. Aunque es de naturaleza introductoria, el material de este texto no se considera tan simple. En realidad, no hay manera de hacerlo sencillo, ya que un estudiante que se inicia en el tema encontrará que tiene por delante una tarea demandante de descubrimiento. Se presentan conceptos, términos y símbolos nuevos con una frecuencia y rapidez desconcertantes, y en este punto resulta que la memoria tiene un papel significativo. Un reto aún más difícil es la necesidad de desarrollar la capacidad de razonamiento y aplicar los principios de la termodinámica en la solución de problemas prácticos. Por ello, mientras se mantiene el rigor característico del análisis termodinámico integral, hemos hecho esfuerzos para evitar la complejidad matemática innecesaria. Además, alentamos la comprensión al escribir frases simples y directas en voz activa y tiempo presente. Existen los elementos para ofrecer la motivación requerida, pero nuestro objetivo, como ha sido en todas las ediciones anteriores, también es proporcionar un método que pueda ser comprendido por cualquier estudiante dispuesto a ejercitarse con el esmero adecuado. Los primeros dos capítulos del libro presentan las definiciones básicas y un desarrollo de la primera ley. Los capítulos 3 y 4 tratan el comportamiento a través de la presión/volumen/temperatura de los fluidos y ciertos efectos térmicos, lo cual permite la aplicación inicial de la primera ley a problemas reales. La segunda ley y algunas de sus aplicaciones se consideran en el capítulo 5. Un tratamiento de las propiedades termodinámicas de los fluidos puros en el capítulo 6 permite la aplicación general de la primera y la segunda leyes, y proporciona un amplio tratamiento de los procesos con flujo en el capítulo 7. Los capítulos 8 y 9 abordan los procesos de producción de energía y de refrigeración. En el resto del libro, se hace referencia a mezclas fluidas y se tratan temas que corresponden exclusivamente a la termodinámica de la ingeniería química. Los capítulos 11 y 12 proporcionan una exposición detallada de la teoría y aplicación de la termodinámica de soluciones. El equilibrio de las reacciones químicas se cubre con detalle en el capítulo 13. El capítulo 14 se ocupa de temas como el equilibrio de fase, en donde se incluye un tratamiento extenso del equilibrio vapor/ líquido, así como del equilibrio osmótico y de adsorción. El capítulo 15 considera el análisis termodinámico de procesos reales, proporcionando una revisión de gran parte de los temas prácticos de interés para la termodinámica. xv 00-SmithVanNess Preliminares.ind15 15 10/1/07 14:03:43 xvi El material de estos capítulos es adecuado para un curso de un año a nivel licenciatura; no obstante, es útil en otro tipo de cursos siempre y cuando se utilice con discreción y de manera condicionada por el contenido de estos. Los primeros 13 capítulos incluyen material que es necesario en la formación del ingeniero químico. Por lo tanto, cuando se proporcione un curso de termodinámica de Ingeniería Química con duración de sólo un semestre, estos 13 capítulos representan el material suficiente. Las leyes y los principios de la termodinámica clásica no dependen de algún modelo en particular de la estructura de la materia; están libres de cualquier consideración a nivel molecular. Sin embargo, el comportamiento exhibido por la materia (gases, líquidos y sólidos) depende de su naturaleza en particular, por lo que en el capítulo 16 presentamos una introducción a la termodinámica molecular, a la cual se hace referencia de manera ocasional en capítulos anteriores. Este libro fue realizado de manera detallada a fin de convertirlo en una referencia útil para los cursos a nivel de posgrado y la práctica profesional. No obstante, debido a consideraciones de extensión, fue necesario hacer una selección prudente del material. De este modo, no incluimos ciertos temas dignos de atención, pero de naturaleza especializada. Éstos incluyen aplicaciones a polímeros, electrolitos y biomateriales. Estamos en deuda con muchas personas, estudiantes, profesores, revisores, quienes han contribuido de diversas maneras a la calidad de esta séptima edición, tanto en forma directa como indirecta, a través de preguntas y comentarios, elogios y críticas a lo largo de 55 años y seis ediciones de evolución. A todos ellos hacemos extensivo nuestro agradecimiento. J. M. Smith H. C. Van Ness M. M. Abbott Agradecemos en especial la valiosa contribución de los siguientes asesores técnicos para la presente edición en español: Alejandro J. Guzmán Gómez, Universidad Nacional Autónoma de México, FES Zaragoza Margarita Hernández Alvarado, Universidad Tecnológica de México, campus Sur Ma. del Carmen Doria Serrano, Universidad Iberoamericana, Ciudad de México Luis Neri Vitela, ITESM, campus Estado de México Rodolfo Gámez Aguilar, Instituto Tecnológico de Los Mochis Hidelberto Hernández Frías, Instituto Tecnológico de Los Mochis Fortunato Ramos Valenzuela, Instituto Tecnológico de Los Mochis Pedro Rochín Angulo, Instituto Tecnológico de Culiacán Guillermo Aguirre, Instituto Tecnológico de Mazatlán 00-SmithVanNess Preliminares.ind16 16 10/1/07 14:04:50 Capítulo 1 Introducción 1.1 EL PANORAMA DE LA TERMODINÁMICA La ciencia de la termodinámica nació en el siglo xix, cuando surgió la necesidad de describir el funcionamiento de las máquinas de vapor y establecer los límites de lo que éstas podían realizar. Así, como su nombre lo indica, la termodinámica es la potencia desarrollada por el calor, con aplicaciones obvias a las máquinas térmicas, de las cuales la máquina de vapor representa el primer ejemplo. De cualquier modo, los principios observados, válidos para las máquinas, se han generalizado sin dificultad y ahora se conocen como la primera y la segunda leyes de la termodinámica. Estas leyes no tienen demostración en sentido matemático; su validez estriba en la ausencia de experiencias contradictorias. Así, la termodinámica comparte con la mecánica y el electromagnetismo la base sobre la que se sustentan las leyes básicas, que no se deducen de algo más. Estas leyes conducen, mediante la deducción matemática, a un sistema de ecuaciones que encuentran aplicación en todas las ramas de la ciencia y de la ingeniería. El ingeniero químico hace frente de manera particular a una amplia variedad de problemas; por ejemplo, el cálculo de los requerimientos de calor y de trabajo para los procesos físicos y químicos, así como la determinación de las condiciones de equilibrio para las reacciones químicas y para la transferencia de especies químicas entre las fases. Las consideraciones termodinámicas no establecen la rapidez de los procesos químicos o físicos. La rapidez depende de las fuerzas impulsoras y de la resistencia; las fuerzas impulsoras son variables termodiná­ micas, pero las resistencias no. Ninguna formulación termodinámica, es decir, de características macroscópi­ cas, revela los mecanismos microscópicos (moleculares) de los procesos físicos o químicos. Por otra parte, el conocimiento del comportamiento microscópico de la materia puede ser útil en el cálculo de las propiedades termodinámicas. Los valores de las propiedades son esenciales para el uso práctico de la termodinámica. El ingeniero químico se ocupa de muchas especies químicas y a menudo no cuenta con datos experimentales, lo cual ha conducido al desarrollo de las “correlaciones generalizadas” que proporcionan estimaciones de la propiedad cuando se carece de información. La aplicación de la termodinámica a cualquier problema real comienza con la identificación de un agre­ gado particular de materia como punto central de atención. Este agregado de materia se llama sistema y su estado termodinámico está definido por algunas propiedades macroscópicas mensurables. Éstas dependen de las dimensiones fundamentales de la ciencia, de las cuales la longitud, el tiempo, la masa, la temperatura y la cantidad de sustancia son las de mayor interés. Se presenta un tratamiento elemental en el capítulo 6. 01­SmithVanNess.indd 1 8/1/07 12:47:11 1.2 CAPÍTULO 1. Introducción DIMENSIONES Y UNIDADES Las dimensiones fundamentales son primordiales, ya que son reconocidas por nuestras percepciones sen­ soriales y no son definibles en términos de algo más simple. Sin embargo, su empleo requiere la definición de escalas arbitrarias de medición, divididas en unidades de tamaño específico. Las unidades originales fue­ ron establecidas mediante un acuerdo internacional, y codificadas como el Sistema Internacional de Unidades (abreviado como SI, para el Système International). El segundo, la unidad del tiempo del SI, cuyo símbolo es s, corresponde a la duración de 9 9 63 770 ciclos de radiación asociada a una transición específica del átomo del cesio. El metro, cuyo símbolo es m, es la unidad fundamental de longitud que se define como la distancia que la luz viaja en el vacío durante /99 79 458 de un segundo. El kilogramo, con el símbolo kg, es la masa de un cilindro de platino/iridio que se conserva en la Oficina Internacional de Pesas y Medidas en Sèvres, Francia. La unidad de la temperatura es el kelvin, cuyo símbolo es K, y es igual a /73.6 de la temperatura termodinámica del punto triple del agua. En la sección .5 se hace un análisis detallado de la temperatura, que es la dimensión característica de la termodinámica. El mol, cuyo símbolo es mol, es la cantidad de sustancia representada por tantas entida­ des elementales (por ejemplo, moléculas) como el número de átomos que hay en 0.0 kg de carbono­. Esto es equivalente al “gramo mol” usado de manera común por los químicos. Los múltiplos y fracciones decimales de las unidades del SI se designan mediante prefijos. Los de uso más frecuente se enumeran en la tabla .. De esta manera, el centímetro está determinado como cm = 0 – m, y el kilogramo como kg = 0 3 g. Tabla 1.1: Prefijos para unidades del SI Múltiplo Prefijo 0−5 femto 0− 0−9 0−6 0−3 0− pico nano micro mili centi Símbolo Múltiplo Prefijo Símbolo f 0 hecto h p n µ m c 03 kilo mega giga tera peta k M G T P 06 09 0 05 Otros sistemas de unidades, como el sistema inglés de ingeniería, utilizan unidades que están relaciona­ das con las unidades del SI mediante factores fijos de conversión. Así, el pie se define como 0.3048 m, la libra masa (lbm) como 0.4535937 kg y la libra mol (lb mol) como 453.5937 mol. 1.3 MEDIDAS DE CANTIDAD O TAMAÑO Tres medidas de cantidad o tamaño son de uso común: • Masa, m • Número de moles, n • Volumen total, V t Para un sistema específico, estas medidas están en proporción directa una de otra. La masa, una medida primordial sin definición, se puede dividir entre la masa molar M, comúnmente llamada peso molecular, para 01­SmithVanNess.indd 2 8/1/07 12:47:11 1.4. 1.4.Force Force 1.4. Force 1.4. Force 1.4. Force 1.4.1.4. Force Force 1.4. 1.4. Force 1.4.Force Force 1.4. Fuerza weight, to yield weight, to to yield yieldnumber numberofof ofmoles: moles: weight, number moles: 333 333 333 3 3 to yield number of mmmoles: weight, totoyield yield number ofofmoles: moles: weight, toweight, yield number of moles: weight, to number of to number of weight, yield number moles: obtener weight, elweight, número de moles: toyield yield number ofmoles: moles: nnn== oror mm = m or m== =Mn Mn Mn M M m M m m mm mm = Mn =n= oror or m mMn ==m Mn =nnn= or oor m= =m Mn = Mn nnn= or Mn =M =m Mn Mofof Total the size isis quantity MM M= M Totalvolume, volume,representing representing the size ofaor system, isaam adefined defined quantitygiven givenasas asthe theproduct product Total volume, representing size aasystem, system, defined quantity given the product Mthe ofof lengths. It may be divided by the mass or number of moles of the system to yield ofthree three lengths. It may be divided by the mass or number of moles of the system to yield three lengths. It may be divided by the mass of moles of the system to yield Total volume, representing the size of aor is aquantity defined quantity given as the product Total volume, representing the size of system, isis defined quantity given asasproduct the product Total volume, representing the size of system, issystem, aesdefined defined quantity given as the product Total volume, representing the size of aaasystem, is aaadefined quantity given as the product Total volume, representing size of aaun system, is anumber given as the El volumen total, que representa el the tamaño de sistema, una cantidad definida como elproduct producto de specific or molar volume: Total volume, representing the size of system, defined quantity given the product Total volume, representing the size of a system, is a defined quantity given as the specific or molar volume: specific or molar volume: of three lengths. It may be divided by the mass or number of moles of the system to yield ofofthree three lengths. It may be divided by the mass or number of moles of the system to yield of three three lengths. It may be divided by the mass or number of moles of the system to yield of lengths. It may be divided by the mass or number of moles of the system to yield of lengths. It may be divided by the mass or number of moles of the system to yield tres longitudes. Se puede dividir entre la masa o el número de moles del sistema para obtener el volumen lengths. It may be divided by the or number of moles of the system to yield of threethree lengths. It may be divided by tthe massmass or number of moles of the system to yield specific or molar volume: specific orormolar molar volume: specific or molar volume: VV specific or volume: or molar volume: V tt molar ospecific específico: tt specific molar volume: specific or molar volume: = mV or V •••Specific volume: V ≡ t = mV mV or V = Specific volume: volume: V≡ ≡ V Specific V mmV tt V tor VVVtt m t VV t t t t t t = mV VmV • volume: Specific volume: ≡VV ≡ == mV or Vor = Specific volume: ≡ V ≡ or mV or o or Specific volume: • Volumen específico: t= VV= •••Specific volume: •••Specific mV VmV Specific volume: VVV≡ mV or or VV t= Specific volume: ≡Vm m m mV≡t m t mV Vt m tt •••Molar VV oror VV Molarvolume: volume: V≡≡ ≡ =nV nV or V t== Molar volume: nV t Vt tnn n t t V V VV t VV t t t t t t •volume: Molar volume: VV ≡ = nV VnV Molar volume: ≡≡V ≡ or == nV or VVort= Molar volume: ≡VVV≡ = nV or oor • Volumen molar: t= •••Molar volume: VV= •••Molar −1 −1 Molar volume: nV VnV Molar volume: V ≡ nV Vofspecific nor or of . .. Specific or molar density is defined as the reciprocal orormolar n n n n Specific or molar density is defined as the reciprocal specific molarvolume: volume:ρρρ≡≡ ≡VV V −1 Specific or molar density is defined as molar volume: n then reciprocal of specific or These quantities (V and ρ) are independent of the size of a system, and are examples These quantities (V and ρ) are independent of the size of a system, and are examples −1 −1ρ −1 −1 These quantities (V and ρ) are independent the size ofmolar a system, examples LaSpecific densidad molar oor específica sedefined define como el recíproco del volumen molar oand específico: Specific molar density isas as the reciprocal ofor specific or molar volume: ≡−1..V −1.. Specific orormolar molar density is defined asas the reciprocal ofof specific or molar volume: ≡ Specific or molar density is defined asdefined the reciprocal ofofspecific specific or molar volume: ρare ≡ρρVV Specific or density is as the reciprocal of specific or molar volume: ≡ VV ..and or molar density is defined the reciprocal of volume: ρpressure, ≡ −1 . Specific molar density is defined the reciprocal specific or molar volume: ρ ≡ V ofof intensive thermodynamic variables. They are functions of the temperature, . Specific or molar density is defined as the reciprocal of specific or molar volume: ρ ≡ V of intensive thermodynamic variables. They are functions of the temperature, pressure, and intensive thermodynamic variables. They areof functions of pressure, and Estas cantidades (V y ρ) sonand independientes del independent tamaño desize unofsize sistema son unexamples ejemplo de These quantities (Vare and ρ)independent are theathe size ofademás, a and system, and are examples These quantities (V and ρ) are independent of the size of ay, system, and are examples These quantities (V and ρ) are independent ofare the size of aof system, and are examples These quantities (V and ρ) are of the atemperature, system, and are These quantities (V ρ) independent the of system, are examples composition of a system, additional quantities that independent of system size. These quantities (V and ρ) are independent of the size of a system, and are examples These quantities (V and ρ) are independent of the size of a system, and are examples composition of a system, additional quantities that are independent of system size. composition of a system, additional quantities that are independent of system size. variables termodinámicas intensivas. Son funciones de la temperatura, de la presión y de la composición of intensive thermodynamic variables. They are functions of the temperature, pressure, ofofintensive intensive thermodynamic variables. They are functions ofoftemperature, the temperature, pressure, and and of intensive intensive thermodynamic variables. They are functions functions of the the temperature, pressure, andand thermodynamic variables. They the pressure, of thermodynamic variables. They are of pressure, and intensive thermodynamic variables. They arefunctions functions thetemperature, temperature, pressure, of of intensive thermodynamic variables. They areare functions of of the temperature, pressure, and and de un sistema, son cantidades adicionales e independientes del tamaño del sistema. composition of a system, additional quantities that are independent of system size. composition of system, additional quantities that are independent ofofsystem system size. composition of aaof system, additional quantities thatthat are independent of system system size.size. composition aaasystem, additional quantities are composition of additional quantities that of size. composition of system, additional quantities that areindependent independent system composition of asystem, system, additional quantities thatare areindependent independent of of system size.size. 1.4 1.4 1.4 FORCE FORCE 1.4 FORCE FUERZA 1.4 FORCE 1.4 FORCE 1.4 FORCE 1.4 FORCE 1.4 FORCE 1.4 FORCE 1.4 FORCE The TheSISIunit unitofofforce forceisisthe thenewton, newton,symbol symbolN,N,derived derivedfrom fromNewton’s Newton’ssecond secondlaw, law,which whichexex- The SI unit of force is the newton, symbol N, derived from Newton’s second law, which exLa unidadpresses del SI que corresponde a la fuerza es elmnewton, su símboloa; es N, yF seF= deduce de la newton segunda ley force FF product ofof thus The isis presses force Fasas asthe the product ofmass mass mand andacceleration acceleration a;from thus =ma. ma.second The newton is de presses force mass m and acceleration a; thus F = ma. The newton The SI unit ofthe force isnewton, the newton, symbol N,from derived Newton’s law, which exThe SI unit of force isFproduct the newton, symbol N, derived from Newton’s second law, which exThe SI unit of force is the newton, symbol N, derived from Newton’s second law, which exThe SI unit of force is the symbol N, derived from Newton’s second law, which exSI unit of force is the newton, symbol N, derived Newton’s second law, which ex−2 −2 Newton,The que expresa a la fuerza como el producto de la masa m y la aceleración a; es decir, F = ma. defined as the force which when applied to a mass of 1 kg produces an acceleration of 1 m s ; The SI unit of force is the newton, symbol N, derived from Newton’s second law, which exThe SI unit of force is the newton, symbol N, derived from Newton’s second law, which ex−2 definedpresses as the the force force which which when applied to mass of 11acceleration kg produces producesa; anthus acceleration of 11The m ssnewton ; El is defined as when applied to aa and mass of kg an acceleration of m ;is F as the product of mass m and F = ma. presses force F as the product of mass m and acceleration a; thus F = ma. The newton is presses force F as the product of mass m and acceleration a; thus F = ma. The newton is − presses force F as the product of mass m acceleration a; thus F = ma. The newton presses force F as the product of mass m and acceleration a; thus F = ma. The newton is −2 −2 newton sepresses define como fuerza que,product alunit aplicarse amuna de kg, de newton m is s −2 is ; de thus the newton isis athe derived 1and kg m . .. produce presses force F the of mass mmasa acceleration a; thus = ma. force Fla as product ofrepresenting mass and a; thus Funa=Faceleración ma. TheThe newton thus the newton is derived unit representing 1to kg msss−2 −2 thus the newton aaas derived unit representing 1acceleration kg m as the force which when a11mass of produces 1− kg. produces an acceleration of m;; s−2 ; defined asasforce the force which when applied to mass of kg produces an acceleration of m defined asdefined the force which when applied toapplied ato mass of kg produces an acceleration acceleration of 11of m defined as the force which when applied aaamass of 11is kg an acceleration 11ss1−2 m defined as the which when applied to arepresenta mass of kg produces an of m ;;ss1;−2 −2 −2 esta forma, el newton es una unidad deducida que kg m s defined the force which when applied to mass of 1 kg produces an acceleration of m s In the English engineering system of units, force treated as an additional independent defined as the force which when applied to a mass of 1 kg produces an acceleration of 1 m s In English engineering system of units, units, force is treated as an an additional additional independent independent ; −2 −2 −2 −2 In the English system of force treated thus the is aunit derived unit representing 1m kg m . as thus the newton is aengineering derived unit representing kg . .s−2como thus the newton isnewton derived unit representing kgfuerza m thus the newton is aunidades derived unit representing 11m kg m .force the newton is aawith derived representing 11la kg sspound ..ssis −2 −2 Enthus eldimension sistema inglés de de ingeniería, se trata una dimensión independiente thus the newton is a derived unit representing 1 kg m s dimension along length, time, and mass. The (lb ) is defined as thus the newton is a derived unit representing 1 kg m s . f along with length, time, and andsystem mass. of The pound force (lbff)) is isasdefined defined asthat thatforce force dimension along length, time, mass. The pound force (lb as that force InEnglish thewith English engineering units, force isas treated an additional independent InInEnglish the English engineering system of units, force is(lb treated asas an additional independent In accelerates the English engineering system of feet units, force is treated treated as an additional independent In the engineering system of units, force is treated as an additional independent In the engineering system of units, force is an additional independent adicional,which junto con la longitud, el tiempo y la masa. La libra fuerza ), se define como la fuerza que acele­ 1 pound mass 32.1740 per second per second. Newton’s law must here the English engineering system of units, force is treated an additional independent f In the English engineering system of units, force is treated as an additional independent whichdimension acceleratesalong 1 pound pound mass 32.1740 feet per second per second. Newton’s law must here which accelerates 1 mass 32.1740 feet per second per second. Newton’s law must with length, time, and mass. The pound (lb isas defined ashere that force dimension along with length, time, and mass. The pound force (lb )defined isisdefined defined asasthat that force dimension along with length, time, and mass. The pound force (lbffforce isff)defined as that force f )caso dimension along with length, time, and mass. The pound force is as force dimension along with length, time, and mass. The pound force (lb ))(lb is that force ra libra masa a 3.740 pies por segundo. La ley de Newton debe incluir en este una constante de include a dimensional proportionality constant for consistency with this definition: dimension along with length, time, and mass. The pound force (lb ) defined that force dimension along with length, time, and mass. The pound force (lb ) is defined as that force f includewhich dimensional proportionality constant for consistency with this definition: definition: include aaaccelerates dimensional proportionality constant for consistency with this accelerates 1ser pound mass 32.1740 feet perper second perfNewton’s second. Newton’s law here which pound mass 32.1740 feet per second per second. Newton’s law must here which accelerates pound mass 32.1740 feet per second per second. Newton’s law law must heremust which accelerates 11para mass 32.1740 feet per second per Newton’s must here which accelerates 111pound mass 32.1740 feet per second law here proporcionalidad dimensional consistente con esta definición: which accelerates 1pound pound mass 32.1740 feet per second persecond. second. Newton’s law must here which accelerates pound mass 32.1740 feet per second persecond. second. Newton’s lawmust must here a dimensional proportionality constant for consistency with this definition: include dimensional proportionality constant for consistency with this definition: include dimensional proportionality constant for consistency withwith thisthis definition: include aaadimensional proportionality constant for definition: include aainclude proportionality constant for with 11consistency include dimensional proportionality forconsistency consistency with this definition: include adimensional dimensional proportionality constant for consistency withthis thisdefinition: definition: Fconstant ma F== = 1ma ma F g c g 111gcc111 1 ma F= ma == ma =FFF= ma ma FFF= ma ma = gc g g g g 1 c c c c 1 2 −2 g32.1740(ft)(s) g)c )×× c 32.1740(ft)(s) −2 1(lb De donde,Whence, −2 mm f )f))== 1(lb = 1 ×× ×1(lb 1(lb Whence,22 1(lb Whence, m ) × 32.1740(ft)(s) f ggcgc1 1(lb 1 1 1 1 c11 2 −2 −2 −2 −2 22 22Whence, −2 =× 1(lb ) × 32.1740(ft)(s) ==× ××1(lb 1(lb ) )× ××m32.1740(ft)(s) 32.1740(ft)(s) Whence, 1(lb1(lb =ff))1(lb × 1(lb × Whence, f ) 1(lb )32.1740(ft)(s) Whence, 1(lb = ))× × 32.1740(ft)(s) Whence, 2 m m1(lb f))1(lb 2 −2 −2 m m f1(lb )= 32.1740(ft)(s) Whence, 1(lb =fgg32.1740(lb × 1(lb ) × 32.1740(ft)(s) Whence, −1 −2 g m m fc) = g −1 −2 c g c c and g )(ft)(lb ) (s) c mm)(ft)(lb f f))−1 (s) and =cg32.1740(lb 32.1740(lb )(ft)(lb (s)−2 y and ggcc = c gc m f −1 −1 −2(s)−2 −1 −2 −2 and force is equivalent )(ft)(lb and ==g32.1740(lb 32.1740(lb )(ft)(lb (s) and = 32.1740(lb )(ft)(lb c = 32.1740(lb f )−2 and gg=4.4482216 = ))−1 and ggcgc to = )(ft)(lb ))−1 c32.1740(lb m)(ft)(lb f(s) mN. −1 −2 The pound N. cg m f(s) m ffm The pound force is is equivalent equivalent to 4.4482216 and 32.1740(lb )−1(s) (s)−2 and c32.1740(lb m )(ft)(lb The pound force 4.4482216 N. c to m )(ft)(lb f ) f(s) La libra fuerza es equivalente a 4.4486 N. Because force and mass are different concepts, aaapound force and aaapound mass are Because force and mass are4.4482216 different concepts, pound force and poundson mass are Because and mass are different concepts, pound pound mass are Puesto quepound laforce fuerza yequivalent la son conceptos distintos, una libra fuerzaforce y unaand libra masa cantidades The pound force is equivalent to 4.4482216 N. The pound force isismasa equivalent to N. TheThe pound force isforce equivalent to 4.4482216 4.4482216 N. N. force is equivalent to 4.4482216 The pound is to N. The pound force equivalent to 4.4482216 N. different quantities, and their units do not cancel one another. When an equation contains both The pound force is equivalent to 4.4482216 N. different quantities, and their units do not cancel one another. When an equation contains both quantities, and their units dodifferent not one When an equation contains both diferentes,different y estas unidades no se cancelan entre sí. cancel Cuando una ecuación contiene ambas unidades, (lb Because force and mass are different concepts, aforce pound force amass pound mass are Because force and mass are different pound force and pound mass are f) y Because force and mass are different concepts, a pound pound force and pound mass are Because force and mass concepts, aaapound force and aaand mass Because force and mass are different concepts, aanother. and aaand pound are units, (lb )f))and (lb ), dimensional constant gconcepts, appear in the to force and mass are different concepts, pound force apound pound mass and mass areare different concepts, aalso pound force aequation pound mass areitare m cgcmust fBecause units,Because (lb andforce (lb ),the the dimensional constant must also appear inand theequation equation tomake make itareboth m units, (lb and (lb ), the dimensional constant g must also appear in the to make it (lb m ), la constante dimensional g también debe aparecer en la ecuación para hacerla dimensionalmente different quantities, and their units do not cancel one another. When an equation contains m c f different quantities, and their units do not cancel one another. When an equation contains both different quantities, and their units do not cancel one another. When an equation contains both c different quantities, their units do not another. When an contains both different quantities, and their units dodonot one another. When ananequation contains both dimensionally correct. different quantities, and their units docancel notcancel cancel one another. When anequation equation contains both different quantities, andand their units not cancel oneone another. When equation contains both dimensionally correct. dimensionally correct. correcta.units, units, (lb )(lb and (lb the dimensional constant gc must alsoin appear inequation the equation to make units, (lb ) and (lb ), the dimensional constant g must also appear in the equation to make it it units, (lb ) and (lb ), the dimensional constant g must also appear in the equation to make it m ),dimensional fm units, (lb ) and ), the constant g must also appear in the to make (lb ) and ), the dimensional constant g must also appear the equation to make it m c f m c f m c f c f units, the dimensional constant gc must appear in equation the equation to make units, (lbf )(lb and (lbm(lb ), the constant gc must alsoalso appear in the to make it itit m ), dimensional f ) and dimensionally correct. dimensionally correct. dimensionally correct. dimensionally correct. dimensionally correct. dimensionally correct. dimensionally correct. 2 Where 22Where Wherenon-SI non-SIunits units(e.g., (e.g.,English Englishunits) units)are areemployed, employed,parentheses parenthesesenclose enclosethe theabbreviations abbreviationsofof ofallall allunits. units. non-SI units (e.g., English units) are employed, parentheses enclose the abbreviations units. 22 22 2 Where units (e.g., English units) areunidades employed, parentheses enclose the abbreviations ofabreviaturas all units. de Where non-SI units (e.g., English units) are employed, employed, parentheses enclose the abbreviations abbreviations of alllas units. Where non-SI unitsnon-SI (e.g.,no English units) are employed, employed, parentheses enclose the abbreviations of all allof units. Where non-SI units (e.g., English are parentheses enclose the all units. non-SI units (e.g., English units) are parentheses enclose abbreviations of units. 2 Where 2 Where Donde seWhere empleen unidades sean del SIunits) (por inglesas), sethe utilizarán paréntesis con non-SI units English units) are employed, parentheses enclose the abbreviations ofunits. all units. non-SI unitsque (e.g.,(e.g., English units) are ejemplo, employed, parentheses enclose the abbreviations of all todas las unidades. 01­SmithVanNess.indd 3 8/1/07 12:47:16 4 CHAPTER 1. Introduction CHAPTER 1. Introduction 4 4 CAPÍTULO 1. Introducción Weight properly refers to the force of gravity on a body, and is correctly expressed in or in properly pounds force. ofamass areand often called “weights,” refersaUnfortunately, to force ofstandards gravitysobre on body, in Elnewtons peso Weight se refiere propiamente la the fuerza de gravedad un cuerpo y is se correctly expresa deexpressed manera and correcta en a balance toDesafortunadamente, compare masses is los called “weighing.” Thus, one must discern from they al uso newtons or fuerza. in pounds force. Unfortunately, standards ofmasa mass often called “weights,” and newtonsuse o enoflibras estándares de aare menudo se denominan “pesos” context whether force or mass is llama meant when “weight” incuando a one casual informal way. the use of apara balance to compare is“para called “weighing.” Thus, must discern from de una balanza comparar masas semasses le pesar”. Por isloused tanto, se or emplea la palabra “peso” de context whether force oresmass is meant whendentro “weight” is used in a casual informal una manera ocasional o informal preciso distinguir del contexto si se trata deoruna fuerza oway. de una masa. Example 1.1 Example 1.1 730 N in Houston, Texas, where the local acceleration of gravity Ejemplo 1.1 weighs An astronaut −2 is =pesa 9.792730 m What the donde astronaut’s mass on local the moon, where Angastronaut weighs 730 Nare in Houston, Texas, whereand thelaweight local acceleration Un astronauta Ns en.Houston, Texas, la aceleración de gravedad es deofggravity = 9.79 m s−2. −2 ? −2 g = 1.67 m s is g = 9.792 m s . What are the astronaut’s mass and weight on the moon, where − ¿Cuáles son la masa −2 y el peso del astronauta en la Luna, donde g = .67 m s ? g = 1.67 m s ? Solución 1.1 Solution 1.1 1.1 Con aSolution =With g, laaley Newton es: F is: = mg. donde, = de g, Newton’s law F =De mg. Whence, With a = g, Newton’s law is: F = mg. F 730 N Whence, 2 m= = 74.55 N m−1 s = F 730mNs−2 2 g 9.792 m= = 74.55 N m−1 s = −2 g 9.792 m s −, Como elBecause newton the N tiene unidades m skg newton N has de thekg units m s−2 , Because the newton N has the units kg m s−2 , m = 74.55 m= 74.55 kg kg m = 74.55 kg Esta masa delmass astronauta independiente de la ubicación, perobut el peso depende de on la aceleración This of the es astronaut is independent of location, weight depends the local delocal la gravedad. forma,isel pesoondel labut Luna es: acceleration of gravity. Thus theastronauta moon the en astronaut’s weight is: on the This mass of De the esta astronaut independent of location, weight depends local acceleration of gravity. Thus on the moon the astronaut’s − weight is: F(moon) 74.55kg kg ×× .67 1.67 m m ss−2 F(Luna) = = mg(moon) mg(Luna) == 74.55 F(moon) = mg(moon) = 74.55 kg × 1.67 m s−2 or F(moon) = 124.5 kg m s−2 = 124.5 N − o F(Luna) = 4.5 kg m s = 4.5 N or F(moon) = 124.5 kg m s−2 = 124.5 N Usesistema of the English engineering system of units requiresdeconversion of thedelas-peso del El uso del inglés de unidades de ingeniería requiere la conversión −2 . With 1 N equivalent to −of tronaut’s tovalores (lbf )engineering and values goftounits (ft)(s) English system requires conversion of the as- f) y m astronauta aUse (lbf)ofweight y the de los de gthe a (pie)(s ). Puesto que N equivale a 0.4809(lb 0.224809(lb m fto 3.28084(ft): tronaut’s weight ) and the values of g to (ft)(s)−2 . With 1 N equivalent to f ) andto1 (lb a 3.8084(pie): 0.224809(lbf ) and 1 m to 3.28084(ft): Weight of astronaut in Houston = 164.1(lbf ) El peso del astronauta en Houston = 64.(lb f) Weight of astronaut in Houston = 164.1(lbf ) g(Houston) = 32.13 and g(moon) = 5.48(ft)(s)−2 − g(Houston) = 3.3 g(Luna) = 5.48(pie)(s) −2 g(Houston) = 32.13 and y g(moon) = 5.48(ft)(s) Newton’s law then gives: then da: gives: Por esto,Newton’s la ley de law Newton 164.1(lbf ) × 32.1740(lbm )(ft)(lbf )−1 (s)−2 Fgc = m= )(ft)(lbf )−1 (s)−2 164.1(lbf ) × 32.1740(lb Fg m −2 gc 32.13(ft)(s) = m= −2 g 32.13(ft)(s) or m = 164.3(lb m) or m = 164.3(lbm ) Thus the astronaut’s mass in (lbmm) and weight in) (lbf ) in Houston are numerically o = 64.3(lb m almost theastronaut’s same, but mass on theinmoon not theincase: Thus the (lbm )this andisweight (lbf ) in Houston are numerically almost the same, but on the moon this is not the case: De esta forma, la masa del astronauta en (lbm) y (164.3)(5.48) su peso en (lbf) en Houston son casi numéricamg(moon) = 28.0(lbf ) = = no es el caso: mente iguales, pero enF(moon) la Luna éste (164.3)(5.48) mg(moon) gc 32.1740 = 28.0(lbf ) = F(moon) = gc 32.1740 mg.Luna/ .164:3/.5:48/ H 28:0(lbf ) F.Luna/ H H gc 32:1740 01­SmithVanNess.indd 4 8/1/07 12:47:19 5 1.5. Temperatura 1.5 TEMPERATURA La temperatura se mide por lo regular con termómetros de líquidos en capilares de vidrio, donde el fluido se expande cuando se calienta. De este modo, un tubo uniforme que está parcialmente lleno de mercurio, alcohol o algún otro fluido, indica el grado de “calentamiento” mediante la longitud de la columna del fluido. De cualquier modo, se asignan valores numéricos a los diversos grados de calentamiento por medio de una defi­ nición arbitraria. Para la escala Celsius,3 el punto de hielo (punto de congelación del agua saturada con aire a la presión atmosférica estándar) es cero, y el punto de vapor (punto de ebullición del agua pura a la presión atmosférica estándar) es 00. Es posible asignar a un termómetro una escala numérica sumergiéndolo en un baño de hielo y haciendo una marca correspondiente al cero en el nivel donde se encuentra el fluido, para después sumer­ girlo en agua hirviendo y hacer una marca para el valor de 00 en este nuevo nivel del fluido. La distancia entre las dos marcas se divide en 00 espacios equidistantes denominados grados. Para extender la escala del termómetro se marcan otros espacios de igual tamaño por debajo del cero y por encima del 00. Todos los termómetros, sin importar el fluido que contengan, proporcionan la misma lectura en cero y en 00 si se calibran con este método, pero generalmente en otros puntos no corresponden las lecturas porque los fluidos varían en sus características de expansión. De esta manera se requiere una elección arbitraria del fluido, y la escala de temperatura en el sistema SI cuya unidad es el kelvin y su símbolo es K, se apoya en el gas ideal como fluido termométrico. Puesto que la definición de la escala Kelvin depende de las propieda­ des de los gases, se hará un análisis detallado hasta el capítulo 3. Sin embargo, se observa que la escala abso­ luta depende del concepto de un límite inferior de la temperatura. Las temperaturas Kelvin se indican con el símbolo T, en tanto que las temperaturas Celsius se designan con el símbolo t, y se definen en relación a las temperaturas Kelvin mediante: t °C = T K − 73.5 La unidad de temperatura Celsius es el grado Celsius, °C, que en magnitud equivale al Kelvin.4 No obstante, las temperaturas en la escala Celsius son 73.5 grados menores que en la escala Kelvin. De esta forma el límite infe­ rior de la temperatura, conocido como el cero absoluto en la escala Kelvin, se presenta en −73.5 °C. En la práctica, la Escala Internacional de Temperatura de 1990 (ITS­90) se utiliza para la calibración de instrumentos científicos e industriales.5 La escala ITS­90 se define de modo que sus valores difieren de las temperaturas del gas ideal, pero que están en los límites de exactitud con los que actualmente se hacen las me­ diciones. Se basa en valores asignados de la temperatura para un número de estados de equilibrio de fase reproducibles de sustancias puras (puntos fijos) y en los instrumentos estándar calibrados a estas tempera­ turas. La interpolación entre las temperaturas de punto fijo se proporciona mediante fórmulas que establecen la relación entre las lecturas de los instrumentos estándar y los valores de la ITS­90. El termómetro de resis­ tencia de platino es un ejemplo de un instrumento estándar, que se utiliza para temperaturas que van desde −59.35 °C (punto triple del hidrógeno) hasta 96.78 °C (el punto de congelación de la plata). Además de las escalas Kelvin y Celsius existen otras dos que aún son utilizadas por los ingenieros de Estados Unidos: las escalas Rankine y Fahrenheit.6 La escala Rankine es una escala absoluta que se relaciona 3 Anders Celsius, astrónomo sueco (70­744). Adviértase que la palabra grado no se emplea en las temperaturas dadas en kelvins, y que la palabra kelvin entendida como una unidad no se escribe con mayúsculas. 5 El texto en lengua inglesa de la definición de ITS­90 está dado por H. Preston­Thomas, Metrologia, vol. 7, pp. 3­0, 990. 6 Gabriel Daniel Fahrenheit, físico alemán (686­736). 4 01­SmithVanNess.indd 5 8/1/07 12:47:19 6 CAPÍTULO 1. Introducción directamente con la escala Kelvin mediante: T(R) = .8 T K La escala Fahrenheit se relaciona con la de Rankine por una ecuación semejante a la relación entre las escalas Celsius y Kelvin: t(°F) = T(R) − 459.67 De este modo, el límite inferior de la temperatura en la escala Fahrenheit es −459.67(°F). La relación entre las escalas Celsius y Fahrenheit está dada por: t(°F) = .8 t °C + 3 Por consiguiente, el punto de congelación del agua es 3(°F) y el punto de ebullición normal del agua es (°F). El grado Celsius y el kelvin representan el mismo intervalo de temperatura, al igual que el grado Fah­ renheit y el Rankine. Las relaciones entre las cuatro escalas de temperatura se ilustran en la figura .. En ter­ modinámica, se da a entender una temperatura absoluta mediante una referencia informal de la temperatura. Celsius Kelvin Fahrenheit Rankine 100(C) 373.15 K 212(F) 671.67(R) Punto de vapor 0(C) 273.15 K 32(F) 491.67(R) Punto de hielo 273.15(C) 0K 459.67(F) 0(R) Cero absoluto Figura 1.1: Relaciones entre las escalas de temperatura. 1.6 PRESIÓN La presión P ejercida por un fluido sobre una superficie se define como la fuerza normal ejercida por el fluido por unidad de área de la superficie. Si la fuerza se mide en N y el área en m, la unidad es el newton por metro cuadrado o N m−, llamado pascal y representado con el símbolo Pa, y es la unidad básica de la presión para el SI. En el sistema inglés de ingeniería una unidad común es la libra fuerza por pulgada cuadrada (psi, por sus siglas en inglés). El estándar primario para mediciones de presión es la balanza de peso muerto, en la cual una fuerza conocida se equilibra con una presión del fluido que actúa sobre un área conocida, donde P ≡ F/A. En la figu­ ra . se muestra un diseño sencillo. El pistón se ajusta cuidadosamente al cilindro dejando una separación 01­SmithVanNess.indd 6 8/1/07 12:47:20 1.6. Pressure 1.6. Presión 1.6. Pressure 7 7 7 muy pequeña. Se small. ponen Weights pesas en are la bandeja hasta presión del aceite, queoil, tiende a hacer el pistón se clearance placed on the que pan la until the pressure of the which tends que to make eleve, se equilibre precisamente por la fuerza de gravedad en el pistón y todo lo que éste soporta. the pistonsmall. rise, isWeights just balanced by on thethe force gravity on the piston andwhich all that it supports. clearance are placed panof until the pressure of the oil, tends to makeCon esta fuerza dada por la ley de Newton la presión del aceite es: Withpiston this force Newton’s the pressure of the the rise, given is justby balanced by law, the force of gravity onoil theis:piston and all that it supports. With this force given by Newton’s law, the pressure of the oil is: mg F = P= mg FA A = P= where m is the mass of the piston, pan, and weights; is la theaceleración local acceleration andy A es el A A donde m es la masa del pistón, de la bandeja y de las pesas; gges local deof la gravity; gravedad, A is the cross-sectional area of the piston. Gauges in common use, such as Bourdon gauges, área dewhere sección pistón. Los instrumentos degmedida de uso común, como el manómetro de m transversal is the mass del of the piston, pan, and weights; is the local acceleration of gravity; and are by comparison with gauges. Bourdon, secalibrated calibran por comparación con las balanzas de peso muerto.use, such as Bourdon gauges, A is the cross-sectional area of thedead-weight piston. Gauges in common are calibrated by comparison with dead-weight gauges. Pesa Bandeja Pistón Cilindro Aceite Weight Weight Pan Piston Pan Piston Cylinder Cylinder Oil Oil Figure 1.2:1.2: Balanza Figura Dead-weight gauge. Figure 1.2: muerto. de peso Dead-weight gauge. To pressure A la fuentesource To pressure de presión source Because a vertical column of a given fluid under the influence of gravity exerts a pressure at its base in direct proportion height, pressure also expressed as la theinfluencia equivalent Because a vertical columntode ofitsaun given fluid underisthe influence gravity exerts a pressure Puesto que una columna vertical fluido determinado que está of bajo deheight la gravedad, of a fluid column. This is the basis for the use of manometers for pressure measurement. at its base in direct proportion to its height, pressure is also expressed as the equivalent heightcomo la ejerce una presión en su base que va en proporción directa con su altura, la presión también se expresa Conversion height to force perbasis unitÉste areathe Newton’s lawpressure applied to the force of of a fluid column. This isdethefluido. for offrom manometers measurement. altura equivalente deofuna columna esfollows eluse fundamento para el for uso de manómetros en mediciones gravity acting on the mass of fluid in the column. The mass is given by: m = Ahρ, where A is Conversion of height to force per unit area follows from Newton’s law applied to the force of de presión. La conversión de la altura a fuerza por unidad de área surge a partir de la ley de Newton aplicada a la the cross-sectional area of the column, h is its height, and ρ is the fluid density. Therefore, gravity acting on the mass of fluid in the column. The mass is given by: m = Ahρ, where A is fuerza de la gravedad que actúa sobre la masa del fluido en la columna. La masa está dada por: m = Ahρ, donde the cross-sectional area of the column, its and is la thedensidad fluid density. Therefore, A es el área de sección transversal de la columna, es height, su altura y ρρ es del fluido. Por lo tanto, F h ishmg Ahρg P= = = = hρg FA mg Ahρg A A P= = = = hρg The pressure to which a fluid height A corresponds A Ais determined by the density of the fluid (which depends itsa identity and temperature) andisthe local acceleration of gravity. Thusfluid the The pressure to on which a fluid height corresponds determined by the del density of (que the La presión que corresponde una altura del fluido se determina por la densidad mismo depende de ◦ C in a standard gravitational (torr) is the pressure equivalent of 1 millimeter of mercury at 0 (whichy depends on itsyidentity and temperature) local acceleration ofes gravity. Thusequivalente the su identidad temperatura) de la aceleración local deand la the gravedad. Así, el (torr) la presión field, and ismercurio equal to a133.322 a standard gravitational (torr) is de the pressure equivalent 1 millimeter of mercury at 0◦yCesinigual de milímetro 0 °C enPa. unofcampo gravitacional estándar, a 33.3 Pa. Another unit of pressure is the standard atmosphere (atm), the approximate averagepor presfield, and isdeequal to 133.322 Pa. Otra unidad presión es la atmósfera estándar (atm), que es la presión promedio ejercida la atmós­ sure exerted by the earth’s atmosphere at sea level, defined as 101,325 Pa, 101.325 kPa, or El bar, Another unit pressure the standard atmosphere (atm), thekPa, approximate average MPa. presfera de la Tierra a nivel delofmar, y se is define como 0 35 Pa, 0.35 o bien, 0.035 5 Pa, is equal to 0.986923(atm). 0.101325 The bar, an0 SI5 unit asa 10 sure exerted the earth’s atmosphere at sea level, defined una unidad del SI,MPa. seby define como Pa ydefined es igual 0.98693 (atm).as 101,325 Pa, 101.325 kPa, or Most gauges readings which areque difference between the pressure of 0.101325 MPa. bar, an SIgive unit defined as 105 Pa, isthe equal to 0.986923(atm). La mayoría depressure losThe manómetros de presión dan lecturas representan la diferencia entre la presión de interest and the pressure of the surrounding atmosphere. These readings are known as gauge Most pressure gauges give readings which are the difference the pressure of interés y la presión atmosférica de los alrededores. Estas lecturas se conocenbetween como presiones manométricas pressures, be converted to absolute pressures by These addition of theEn barometric pressure. interest andand theacan pressure ofabsolutas the surrounding atmosphere. readings are as gauge y se pueden convertir presiones al sumarles la presión barométrica. losknown cálculos termodinámi­ Absolute pressures must be used in thermodynamic calculations. pressures, andpresiones can be converted cos se deben utilizar absolutas.to absolute pressures by addition of the barometric pressure. Absolute pressures must be used in thermodynamic calculations. 01­SmithVanNess.indd 7 8/1/07 12:47:23 CHAPTER 1. Introduction 8 8 CAPÍTULO 1. Introducción Example 1.2 Ejemplo 1.2 A dead-weight gauge with a 1-cm-diameter piston is used to measure pressures very Se utiliza una balanza de peso muerto con un pistón de 1 cm de diámetro para medir presiones con accurately. In a particular instance a mass of 6.14 kg (including piston and pan) mucha precisión. En un caso particular, una masa de 6.14 kg (incluyendo el pistón−2 y la bandeja) alcan­ brings it into balance. If the local acceleration of gravity is 9.82 m s , what is the za el equilibrio. Si la aceleración local de la gravedad es 9.82 m s−, ¿cuál es la presión manométrica gauge pressure being measured? If the barometric pressure is 748(torr), what is the medida? Si la presión barométrica es 748(torr), ¿cuál es la presión absoluta? absolute pressure? Solución 1.2 Solution 1.2 La fuerza ejercida por la gravedad sobre el pistón, la bandeja y las pesas es: The force exerted by gravity on the piston, pan, and weights is: F = mg = (6.4)(9.8) = 60.95 N F = mg = (6.14)(9.82) = 60.295 N Gauge pressure = Presión manométrica 60.295 F = 76.77 N cm−2 = A (1/4)(π )(1)2 The absoluteabsoluta pressurees: is therefore: Por lo tanto la presión P = 76.77 + (748)(0.013332) = 86.74 N cm−2 P = 76.77 + (748)(0.0333) = 86.74 N cm– P = 867.4 kPa P = 867.4 kPa or o Example 1.3 At 27◦ C the reading on a manometer filled with mercury is 60.5 cm. The local ac−2 celeration Ejemplo 1.3 of gravity is 9.784 m s . To what pressure does this height of mercury correspond? A 27 °C la lectura en un manómetro lleno de mercurio es de 60.5 cm. La aceleración local de la grave­ dad es 9.784 m s −. ¿A qué presión le corresponde esta altura del mercurio? Solution 1.3 Solución 1.3 Recall the equation in the preceding text, P = hρg. At 27◦ C the density of is 13.53 cm−3anterior, . Then, P = hρg. A 7 °C la densidad del mercurio es Recuerde mercury la ecuación del gtexto −3 3.53 g cm . Por esto, P = 60.5 cm × 13.53 g cm−3 × 9.784 m s−2 = 8,009 g m s−2 cm−2 or o 1.7 P = 60.5 cm × 3.53 g cm−3 × 9.784 m s− = 8 009 g m s− cm− P = 8.009 kg m s−2 cm−2 = 8.009 N cm−2 = 80.09 kPa = 0.8009 bar P = 8.009 kg m s− cm− = 8.009 N cm− = 80.09 kPa = 0.8009 bar 1.7 WORK TRABAJO Se realiza Work trabajo quewhenever una fuerza actúeacts a través de auna distancia. Por definición, la cantidad WW is siempre performed a force through distance. By definition, the quantity of de trabajo está dadaispor la ecuación: work given by the equation: d W = F dl (1.1) dW = F dl (.) 01­SmithVanNess.indd 8 8/1/07 12:47:24 1.7. Work 1.7. Work Work 1.7. 1.7. Trabajo 9 99 9 where F is the component of force acting along the line of the displacement dl. When intedonde F es la componente de la fuerza que acting actúa aalong lo largo la of línea de desplazamiento dl. Cuando se inte­ where the component of force along thede line of the displacement dl. When intewhere isis the component force the line displacement dl. integrated,FFthis equation yieldsof the workacting of a finite process. Bythe convention, work is When regarded as gra, esta ecuación proporciona el trabajo para un proceso finito. Por convención, el trabajo se considera grated, this equation yields the work of a finite process. By convention, work is regarded as grated, equation yields the work of asame finitedirection process.asBy work regarded as como positivethis when the displacement is in the theconvention, applied force and is negative when positivopositive cuandowhen el desplazamiento está en la misma dirección que la fuerza aplicada, y negativo cuando se positive when the displacement is in the same direction as the applied force and negative when the displacement they are in opposite directions. is in the same direction as the applied force and negative when encuentra en direcciones opuestas. they are in opposite directions. they areThe in opposite directions. work which accompanies a change in volume of a fluid is often encountered in Con frecuencia, enwhich termodinámica el trabajo se acompaña cambio en el volumen de un The work which accompanies change in volume volumede ofun fluid isisof often encountered inlíquido. The work accompanies change in of aa fluid often encountered in thermodynamics. A common exampleaais the compression or expansion a fluid in a cylinder Un ejemplo común es la compresión o expansión de un fluido en un cilindro, resultado del movimiento de un thermodynamics. A common example is the compression or expansion of a fluid in a cylinder thermodynamics. common example is the compression or expansion of aonfluid a cylinder resulting from theAmovement of a piston. The force exerted by the piston the in fluid is equal pistón. resulting La fuerzafrom ejercida por el pistón sobre el fluido es igual al producto del área del pistón y la presión del resulting from the movement of a piston. The force exerted by the piston on the fluid is equal movement of aand piston. The force exerted thedisplacement piston on theoffluid equalis to the product the of the piston area the pressure of the fluid.byThe the is piston fluido. to El desplazamiento del pistón es igual al cambio de volumen total del fluido dividido entre el área to the product of the piston area and the pressure of the fluid. The displacement of the piston is the product of the pistonchange area and of theby fluid. the piston is equal to the total volume ofthe thepressure fluid divided the The area displacement of the piston. ofEquation (1.1) del pistón. Por lo tanto, la ecuación (.)of sethe convierte en: by equal to the total volume change of the fluid divided by the area of the piston. Equation (1.1) equal to the total volume change fluid divided the area of the piston. Equation (1.1) therefore becomes: thereforebecomes: becomes: therefore Vt d W = −P A dVVt t W= =−P −PAAdd A ddW AA or, because A is constant, d W = −P d V tt (1.2) or,abecause because constant, W= =−P −PddVVt (1.2) (.) o, debido que A es or, AAconstante, isisconstant, ddW (1.2) � t � V2t Integrating, W = −� VV2t2 P d Vt tt (1.3) Integrando, Integrating, W= =− − V1t PPddVV (1.3) (.3) Integrating, W (1.3) t VV1t1 The“negativos” minus signseninestas theseecuaciones equationssearehacen madenecesarios necessarypor bylathe sign convention adopted for para el Los signos convención de signos adoptada The minus signs in these these equations arecylinder made necessary necessary by the the sign sign convention adopted for The minus signs in equations are made by convention adopted for work. When the piston moves into the so as to compress the fluid, the applied force trabajo. Cuando el pistón se mueve en el cilindro para comprimir el fluido, la fuerza aplicada y su desplaza­ work. When thepiston piston moves into the cylinderthe soas as tocompress compress thepositive. fluid,the the applied force the into cylinder so the fluid, applied and itsWhen displacement aremoves in the samethe direction; work is therefore The minus sign menos mientowork. se encuentran en la misma dirección; por lo tanto, el to trabajo es positivo. Se requiere el force signo and its displacement are in the same direction; the work is therefore positive. The minus sign and its displacement are in the same direction; the work is therefore positive. The minus sign is required because the volume change is negative. For an expansion process, the applied force porque el cambio de volumen es negativo. Para un proceso de expansión, la fuerza aplicada y su desplaza­ isrequired requiredbecause becausethe the volume change negative. For an expansion process, theisapplied appliedforce force volume change isisnegative. an process, the are in opposite directions. TheFor volume change in this case andel signo mientoisand estánitsendisplacement direcciones opuestas. El cambio de volumen en expansion este caso es positivo, y positive, se requiere and its displacement are in opposite directions. The volume change in this case is positive, and and its displacement are in opposite directions. The volume change in this case is positive, and the minus sign is required to make the work negative. menos para hacer negativo el trabajo. theminus minussign signisisrequired requiredto tomake makethe thework worknegative. negative. the P P2 PP22 2 22 P PP P P 0 P1 PP10 1 t 00 V t path. Figura 1.3: Diagrama que muestra trayectoria función de V t . Figure 1.3: Diagramlashowing a P de vs.PVen Figure1.3: 1.3:Diagram Diagramshowing showingaa PPvs. vs.VVt t path. path. Figure V2tt VV2t2 V t t Vt t 1 11 Vt V1tt VV1t1 VV 7 Equation (1.3) expresses the work done by a finite compression or expansion process. 777La figu­ Equation (1.3) expresses the work done by a finite compression or expansion process. LaFigure ecuación (.3) expresa el trabajo hecho por un proceso de compresión o expansión finitas. t Equation (1.3) expresses the work done by a finite compression or expansion process. 1.3 shows a path for compression of a gas from point 1 with initial volume t Vt1t at Figure 1.3Pshows shows aa path path forla compression of un gas P from point 1con 1 with with initialinicial volume ra .3 muestra una trayectoria compresión del punto volumen V VVa presión P Figure 1.3 for compression of aa gas from point initial volume atat pressure 2 para with volume Vt2tt at de pressure 1 to point 2 . This path relates the pressure at11any t 2 pressure P to point with volume V at pressure P . This path relates the pressure at any al punto con volumen V a presión P . Esta trayectoria relaciona la presión en cualquier punto del proceso 1 2 pressure P to point 2 with volume V at pressure P . This path relates the pressure at any 1 2 2 point of the process to the volume. The 2 work required is given by Eq. (1.3) and is proportional point ofarea the processthe tothe thevolume. volume. The work required isofgiven given by Eq. (1.3) and isproportional proportional con el volumen. El trabajo requerido está dado por la ecuación (.3) y esby proporcional al is área bajo curva de la point the process to The work required and to theof under curve of Fig. 1.3. The SI unitis work isEq. the(1.3) newton-meter or la joule, to the area under the curve of Fig. 1.3. The SI unit of work is the newton-meter or(ft joule, figura .3. La area unidad delEnglish trabajo delof SI Fig. es el1.3. newton­metro o often joule, cuyois es J. Enforce el sistema de tosymbol the the curve Thethe SIunit unit of work the foot-pound newton-meter or joule, J. Inunder the engineering system used issímbolo the lbfinglés ). symbol Inthe the English engineering system theunit unitoften oftenused used thefoot-pound foot-poundforce force(ft (ftlb lbff).). ingeniería la unidad pie­libra fuerza (pie lbf)system se emplea con mucha frecuencia. symbol J.J.In English engineering the isisthe 7 7 However, as explained in Sec. 2.8, it may be applied only in special circumstances. 77However, However,asasexplained explainedininSec. Sec.2.8, 2.8,ititmay maybe beapplied appliedonly onlyininspecial specialcircumstances. circumstances. Sin embargo, como se explica en la sección .8, sólo se aplica en circunstancias especiales. 01­SmithVanNess.indd 9 8/1/07 12:47:27 0 1.8 10 10 10 CAPÍTULO 1. Introducción CHAPTER 1. Introduction CHAPTER 1. 1. Introduction Introduction CHAPTER ENERGÍA 1.8 ENERGY 1.8 ENERGY ENERGY 1.8 El principio general de conservación de energía se estableció alrededor de 850. El germen de este principio, of conservation of energy established about 1850. The germ of this tal comoThe se general aplica aprinciple la mecánica, está implícito en los was trabajos de Galileo (564­64) y de Isaac The general principle of conservation conservation of energy energy was established about 1850. The The germ germ of this thisNewton The general principle of of was established about 1850. of principle as it applies to mechanics was implicit in the work of Galileo (1564–1642) and Isaac (64­76). De as hecho, es una consecuencia directa de in la the segunda ley de Newton sobre el movimiento, una principle it applies to mechanics was implicit work of Galileo (1564–1642) and Isaac asesitdefinido applies to mechanics was in yfrom the of Galileo (1564–1642) and Isaac Newton (1642–1726). Indeed, it follows Newton’s second law of motion once vez queprinciple el trabajo como producto deimplicit ladirectly fuerza el work desplazamiento. Newton (1642–1726). Indeed, it follows directly from Newton’s second law of motion once Newton (1642–1726). Indeed,ofit force follows directly from Newton’s second law of motion once work is defined as the product and displacement. work is is defined defined as as the the product product of of force force and and displacement. displacement. work Energía cinética Kinetic Energy Kinetic Energy Energy Kinetic a body mass m, acted upon by a force F, is a distance dl during a differential CuandoWhen un cuerpo deof masa el que actúa fuerza F,displaced se desplaza una distancia dl, durante un intervalo When body of massm,m, m,enacted acted upon byuna force F, is is displaced distance dl during during differential When aatiempo body of mass upon by aa force F, displaced aa distance dl aa differential interval of time dt, the work done is given by Eq. (1.1). In combination with Newton’s second diferencial de dt, el trabajo que se realiza está dado por la ecuación (.). De manera conjunta interval of of time time dt, dt, the the work work done done is is given given by by Eq. Eq. (1.1). (1.1). In In combination combination with with Newton’s Newton’s second second con la law this equation becomes: segundainterval ley de Newton, esta ecuación se convierte en: law this this equation equation becomes: becomes: law dW = ma dl W == = ma ma dl dl dW dl dd W ma By definition the acceleration is a ≡ du/dt, where u is the velocity of body. Thus, By definition the acceleration is a ≡ du/dt, where is the the velocity velocity of of the the body. body. Thus, Thus, By definition the acceleration is a ≡donde du/dt, where uu is Por definición, la aceleración es a ≡ du/dt, u es la velocidad del cuerpo.the De este modo, dl du dl du du dl = m dl d W = m du dl = m du d W = m dt dl = m d W = m dt dt dt du dt dt Because the definition of velocity is ≡ dl/dt, the expression for work becomes: Because the definition definition of velocity velocityesis isuuu u≡≡ ≡dl/dt, dl/dt, the expression for work becomes: becomes: Puesto que la definición de la velocidad la the expresión para for el trabajo es: Because the of dl/dt, expression work dW = mu du W == = mu mu du du dW dd W mu This equation may now be integrated for a finite change in velocity from u to u : This equation equation may nowintegrar be integrated integrated for finitefinito change invelocidad velocity from from to Esta ecuación ahora semay puede para unfor cambio en la de uuu111a to u:uu222:: This now be aa finite change in velocity � � � � u2 � � uu 22 � uu 221 � 22 � u 2 2 u 2 u du = m u 2 − u 1 W = m du = =m m 22 − − 21 W= =m m u uu du W u1 22 22 u 11 � � � 2� � 2 2 � mu mu mu 2 2 2 2 1 mu mu 2 2 mu 2 or W = − = � (1.4) 2 1 o 2 − mu or W= = mu =� � mu (1.4) (.4) 2 22 or W − 22 1 = (1.4) 2 2 2 2 1 2 in Eq. (1.4) is a kinetic energy, a term introduced by Lord Each of the quantities 1 mu 2 in Eq. (1.4) is a kinetic energy, a term introduced by Lord en Each of the the quantities 122 mu 2 in Cada una de las cantidades −¹ mu la Eq. ecuación es unaenergy, energíaa cinética, un término introducido mu (1.4) is(.4) a kinetic term introduced by Lord Each of quantities 8 2 ² Kelvin in 1856. Thus, by definition, 8 Kelvin inen1856. 1856. Thus, by definition, definition, por lordKelvin Kelvin8 8in 856.Thus, En estos términos, por definición, by 1 2 (1.5) E K ≡ 11 mu ≡ (1.5) (.5) mu22 E K (1.5) E K ≡ 22 mu 2 La ecuación (l.4) muestra que elthat trabajo hechodone sobreonuna cuerpo que se acelera aitpartir de una velocidad inicial u Equation (1.4) shows the work body in accelerating from an initial velocity Equation (1.4) shows that the work done on a body in accelerating accelerating itAfrom from an initial initial velocity hasta una velocidad final u es igual al cambio de la energía cinética del cuerpo. la inversa, si el cuerpo en movi­ Equation (1.4) shows that the work done on a body in it an velocity u 1 to final velocity u is equal to the change in kinetic energy of the body. Conversely, if to aaa final final por velocity u 22 is is equal to the theque change in kinetic kinetic energy ofpor theelbody. body. Conversely, ifcambio miento se desacelera la acción de una fuerza se opone, el trabajo hecho cuerpo es igual a su uau11moving to velocity u equal to change in energy of the Conversely, if 2 body is decelerated by the action of aa resisting force, the work done by the body is a moving moving body issistema decelerated by the the action action of resisting force, the work workendone done by, the the body is iscinética en energía cinética. En elis de unidades SI con laSIamasa en kg yunits la velocidad m kg s − la energía aequal body decelerated by of resisting force, the by body to its change in kinetic energy. In the system of with mass in and velocity − − equal to its change in kinetic energy. In the SI system of units with mass in kg and velocity E K tiene unidades de kgenergy min kinetic s E. Puesto que newton la of unidad kgiskg mthe s composite , velocity EK se mide en −1 ,its 2ess−2 equal to change energy. Inelthe SI m system units compuesta with mass in and in m has the units of kg the newton −1 , kinetic 2 s−2 .. Because in m m sss−1 kinetic energy EK has la theecuación units of of(.4), kg m m2ésta Because the newton is the the composite composite −2es K con newton­metros o joules. De acuerdo la unidad del trabajo. −2 in , kinetic energy E has the units kg s . Because the newton is K unit kg m is measured in newton-meters or joules. In accord with Eq. (1.4), this is the −2 ,, E unit kg kg m m sss−2 EK K is measured in newton-meters or joules. In accord with Eq. (1.4), this is the unit ,E K is measured in newton-meters or joules. In accord with Eq. (1.4), this is the unit of work. unit of of work. work. unit 8 Lord Kelvin 8 LordoKelvin, William Thomson (84­907) fue un físico que, junto con who, el físico alemán Rudolf Clausius (8­888), William Thomson (1824–1907), wasinglés an English physicist along with the German physicist 8 Lord Kelvin, or or William Thomson (1824–1907), was an English physicist who, along with the German physicist establecióRudolf los8 Lord fundamentos para la ciencia moderna de la termodinámica. Kelvin,(1822–1888), or William Thomson (1824–1907), English physicist who, along with the German physicist Clausius laid the foundations forwas the an modern science of thermodynamics. Rudolf Clausius (1822–1888), laid the foundations for the modern science of thermodynamics. Rudolf Clausius (1822–1888), laid the foundations for the modern science of thermodynamics. 01­SmithVanNess.indd 10 8/1/07 12:47:30 1.8. Energy Energy 1.8. Energía 1.8. 1.8. Energy 11 11 11 2 /g , where En el sistema de engineering ingeniería, lasystem, energíakinetic cinéticaenergy se expresa como −¹ mu gc tiene In the the inglés English is expressed expressed as 2112/g mu gc el valor c ,2donde c In English engineering − system, kinetic energy is as mu ² c , where gc 1 − 2 2/g −1 −2 3.740has y las unidades (lb )(pie)(lb ) (s) . De este modo, la unidad de la energía cinética en este siste­ In the English engineering system, kinetic energy is expressed as mu /g , where m and the funits (lbm )(ft)(lbf )−1 (s)−2 . Thus the unit of the value value 32.1740 32.1740 kinetic c energy gin 2 kinetic has the and the units (lbm )(ft)(lbf ) −1(s) −2. Thus the unit of energy inc ma es: has this system system is:32.1740 and the units (lbm )(ft)(lbf ) (s) . Thus the unit of kinetic energy in the value this is: −2 this system is: )(ft)22(s) (s)−2 mu22 (lbm)(ft) mu (lb m E = = = (ft lb ) 2 K 2 −1−2 −2 = (ft lbff) E K = mu (lb m )(ft)f )(s) 2gc = (lbm )(ft)(lb (s) −1 −2 2g E K = c = (lbm )(ft)(lbf ) −1(s) −2 = (ft lbf ) 2gc la inclusión (lbminclusion )(ft)(lb Dimensional consistency here requires the of Aquí, laDimensional consistenciaconsistency dimensionalhere requiere defg) cof . (s) requires the inclusion ggcc.. Dimensional consistency here requires the inclusion of gc . Potential Energy Energía potencial Potential Energy Potential Energy If aa body body of of mass mass m m is is raised raised from from an initial initial elevation elevation zz 1 to to aa final final elevation elevation zz 2,, an an upward upward If Si un cuerpo de masa m se mueve desde unaanaltura inicial z a una final z, se debe una fuerza 1 altura 2 ejercer force at least equal to the weight of the body must be exerted on it, and this force must move If a body of mass m is raised from an initial elevation z to a final elevation z , an upward 1 forcealatmenos least equal of the ybody bedebe exerted on it, aand this force move ascendente igual to al the pesoweight del cuerpo, esta must fuerza moverse lo largo de2lamust distancia z − z. through the distance distance − weight z 1.. Because Because the weight ofbethe the body is is the forcethis of gravity gravity on it, it, the force at the least equal tozz 2the of thethe body mustof exerted on it, force and force must move through weight of on the Puesto que el peso del cuerpo 2es−lazfuerza de la gravedad sobre el body mismo, the la fuerza mínima requerida está dada 1 minimum force required iszgiven given by Newton’s Newton’s law:of the body is the force of gravity on it, the through the distance z 2 −is the weight 1 . Because minimum force required by law: por la ley de Newton: minimum force required is given by Newton’s law: F= = ma = mg F F = ma ma = mg F = ma = mg where g is the local acceleration of gravity. The minimum minimum work work required required to to raise raise the the body body is is is the locallocal acceleration of gravity. The donde gwhere es product la gaceleración de lathe gravedad. El elevation: trabajo mínimo requerido para to elevar althe cuerpo es the of this force and change in where g is the local acceleration of gravity. The minimum work required raise body is el pro­ theesta product ofpor thiselforce anden thelachange in elevation: ducto de fuerza cambio elevación: the product of this force and the change in elevation: − z ) = mg(z mg(z 2 − − z1) W= = F(z 2 − W W =F(z F(z2 − zz11) = mg(z 2− zz1)) W = F(z 2 − z 1 ) = mg(z 2 − z 1 ) − mz mz 1gg = = �(mzg) �(mzg) (1.6) or W= = mz mz 2gg − (1.6) or W 2 1 o (1.6) (.6) or W = mz 2 g − mz 1 g = �(mzg) We see from Eq. (1.6) that work done on a body in raising it is equal to the change in the We see from Eq. (1.6)que thatelwork done on asobre body el incuerpo, raising it islevantarlo, equal to the change the En la ecuación (.6) vemos trabajo hecho al esits igual al in cambio quantity mzg. Conversely, if a body is lowered against a resisting force equal to weight, We see from Eq. (1.6) that work done on a body in raising it is equal to the change in the en la quantity mzg. Conversely, ifsiaelbody is lowered against a resisting force equalcontrario, to its weight, the cantidad mzg. De manera inversa, cuerpo desciende con una fuerza en sentido pero igual a su work done by the the body is is equal equal to the the change inagainst the quantity quantity mzg.force Eachequal of the thetoquantities quantities mzg quantity mzg. Conversely, if a body ischange loweredin a resisting its weight,mzg the done by body to the mzg. Each of 9 peso, elwork trabajo hecho por el cuerpo es igual al cambio en la cantidad mzg. Cada una de las cantidades in Eq.done (1.6)by is athe a potential potential energy. Thus, by definition, definition, work body isenergy. equal to the change in the quantity mzg. Each of the quantities mzg mzg 9 Thus, in Eq. (1.6) is by 9 De 9 en la ecuación (.6) es una energía potencial. esta forma, por definición, in Eq. (1.6) is a potential energy. Thus, by definition, ≡ mzg (1.7) E P (1.7) E P ≡ mzg ≡ mzg mzg (1.7) (.7) EEPP ≡ In the SI system of units with mass in kg, elevation in m, and the acceleration of gravity in In the system of units with mass in kg, elevation in m, and the acceleration of gravity in −2 ,SI 2 s−2 . This En el sistema de unidades SIunits con la the masa en in kg,kg, la m elevación en m, m, y lanewton-meter aceleración de lajoule, gravedad en potential energy has units of kg is the or the unit m s In the SI system of with mass elevation in and the acceleration of gravity in m s−, −2 2 −2 kg m 2 s −2. This is the newton-meter or joule, the unit m s −2, potential energy has the units of− la energía potencial tiene unidades de kg m of s kg . Éste newton­metro o joule, laorunidad del unit trabajo, de of work, in agreement agreement with Eq. (1.6). , potential energy hasEq. the units m ses .elThis is the newton-meter joule, the m swork, of in with (1.6). acuerdoofcon la ecuación (.6). In the the English engineering engineering system, potential potential energy energy is is expressed expressed as as mzg/g mzg/gc.. Thus Thus the the work, in agreement with Eq. (1.6). In English system, c En el of sistema inglés deengineering ingeniería, la energía potencial se expresa como mzg/g la unidad unit potential energy in this system is: In the English system, potential energy is expressed as mzg/g . Thus the de la c . Así, c unit of potential energy in this system is: energía unit potencial en este sistema es: of potential energy in this system is: −2 mzg (lbm)(ft)(ft)(s) )(ft)(ft)(s)−2 mzg (lb m E = = = (ft (ft lb lbf)) P −2−2 = −1 E P = mzg f m )(ft)(ft)(s) g = (lb(lb )(ft)(lb (s)−2 m)(ft)(lb f))−1 (s) E P = gcc = (lb = (ft lbf ) m f gc (lbm )(ft)(lbf )−1 (s)−2 Again, must be included for dimensional dimensional consistency. De nueva cuenta, debe be incluirse gc for para asegurar la consistency. consistencia dimensional. Again, ggcc must included Again, gc must be included for dimensional consistency. Energy Conservation Conservation Conservación de la energía Energy Energy Conservation In any any examination examination of of physical physical processes, processes, an an attempt attempt is is made made to to find find or or to to define define quantities quantities In En cualquier examen de procesos físicos of se the intenta encontrar o occur. definirOne cantidades que permanezcan which remain constant regardless changes which such quantity, early recog-constan­ In any examination of physical processes, an attempt is made to find or to define quantities remain constant regardless of the changes which occur. One such quantity, early recogtes, sinwhich importar los cambios que ocurran. Una de estas cantidades, reconocida al principio del desarrollo nized in the development development of mechanics, mechanics, is mass.which The great great utility of thequantity, law of of conservation conservation whichin remain constant regardless of the changes occur. One of such early recognized the of utility the law de la mecánica, es la masa. La gran utilidad deislamass. ley deThe conservación de masa sugiere que existen otros nized in the development of mechanics, is mass. The great utility of the law of conservation 9 9 This term was proposed in 1853 by the Scottish engineer William Rankine (1820–1872). 9 This termpropuesto was proposed in 1853 byingeniero the Scottish engineer William Rankine (1820–1872). Este término en 853 por el escocés William Rankine (80­87). 9 This fue term was proposed in 1853 by the Scottish engineer William Rankine (1820–1872). 01­SmithVanNess.indd 11 8/1/07 12:47:32 CHAPTER 1. Introduction 12 CAPÍTULO 1. Introducción of mass suggests that other conservation principles could be of comparable value. With reprincipios de conservación que podrían tener un valor comparable. Con respecto a la energía observamos que spect to energy we observe that Eqs. (1.4) and (1.6) both show that work done on a body is las ecuaciones (.4) y (.6) muestran que el trabajo realizado sobre un cuerpo es igual al cambio en una can­ equal to the change in a quantity which describes the condition of the body in relation to its tidad que describe la condición del cuerpo en relación con sus alrededores. En cada caso, el trabajo efectuado surroundings. In each case the work performed can be recovered by carrying out the reverse puede recuperarse realizando el proceso inverso y regresando el cuerpo a su condición inicial. Esta observa­ process and returning the body to its initial condition. This observation leads naturally to the ción conduce naturalmente a la idea de que, si el trabajo se efectúa sobre un cuerpo al acelerarlo o elevarlo, thought that, if the work done on a body in accelerating it or in elevating it can be subsequently éste se puede recuperar posteriormente; de esta manera, el cuerpo tiene la habilidad o capacidad de realizar recovered, then the body by virtue of its velocity or elevation contains the ability or capacity trabajo en virtud de su velocidad o elevación. Este concepto ha demostrado ser tan útil en la mecánica del to do the work. This concept proved so useful in rigid-body mechanics that the capacity of a cuerpo rígido, que a la capacidad que tiene un cuerpo para realizar trabajo se le dio el nombre de energía, body for doing work was given the name energy, a word derived from the Greek and meaning palabra derivada del griego, que significa “en trabajo”. Por lo tanto, se dice que el trabajo para acelerar un “in work.” Hence the work of accelerating a body produces a change in its kinetic energy: cuerpo produce un cambio en su energía cinética: � � mu 22 W = �E KK = � 2 y el trabajo realizado para on elevar un cuerpo produce un cambio en su energía potencial: and the work done a body in elevating it produces a change in its potential energy: W W= =E EPPP = = Δ(mzg) �(mzg) Si un cuerpo recibe cuandowhen se eleva, retiene energía hasta or queretains realizathis el trabajo If a body is energía given energy it is conserva elevated, othen the esa body conserves del cualenergy es capaz. Cuando un cuerpo se eleva después le permite libremente, en energía until it performs the work of ywhich it issecapable. Ancaer elevated body, gana allowed to fall cinéti­ ca lo que pierde en energía potencial, de manera que capacidad parasorealizar no for cambia. freely, gains in kinetic energy what it loses in su potential energy that itstrabajo capacity doingPara un cuerpo que libremente esto significa que: falling body this means that: workcae remains unchanged. For a freely ΔEKK + +�E E P P==00 �E K P o or mu 222 mu 212 2 − 1 + mz g − mz g = 0 22 11 2 2 La validez esta ecuación fue confirmada innumerables experimentos. De este modo, desarrollo del Thede validity of this equation has beenpor confirmed by countless experiments. Thus the el developconcepto de energía condujoof deenergy maneraled lógica al principio de conservación de energía para todosfor losall procesos ment of the concept logically to the the principle of energy conservation estrictamente amplia evidencia experimentalevidence que justifica esta generalización fue obtenida sin purely mecánicos. mechanicalLa processes. Ample experimental to justify this generalization was dificultad. readily obtained. Son posibles de energía mecánica, la gravitational energía cinética y de laenergy potencial Other otras formsformas of mechanical energy besidesademás kineticde and potential are gravi­ tacional.possible. La más evidente la energía potencial de configuración. Cuando se acomprime un resorte, el traba­ The mostes obvious is potential energy of configuration. When spring is compressed, jo es hecho una fuerza el resorte puede realizar tarde this cierto trabajo contra una workpor is done by anexterna. externalPuesto force. que Because the spring can latermás perform work against fuerza de resistencia, posee capacidad para efectuarlo. Ésta es lawork. energíaThis potencial de configuración. a resisting force, thelaspring possesses capacity for doing is potential energy of Exis­ te energía del mismo Energy tipo tanto en una estirada en una barra metal deformada en la región configuration. of the sameliga form exists como in a stretched rubberdeband or in a bar of metal elástica.deformed in the elastic region. La generalidad del principio de conservación de la energía en mecánica aumenta si consideramos al The generality of the principle of conservation of energy in mechanics is increased if we trabajo mismo como unaitself forma energía. Esto esThis claramente ya que los cambios tantoand en la ener­ look upon work as adeform of energy. is clearlyaceptable, permissible, because both kineticgía cinética como en la changes energía potencial iguales al trabajo hecho para producirlos potential-energy are equal son to the work done in producing them [Eqs. (1.4)[ecuaciones and (1.6)]. (l.4) y (.6)]. Sin embargo, el trabajo es energía enand tránsito y nunca se considera como en unwork cuerpo. However, work is energy in transit is never regarded as residing in residente a body. When is Cuan­ do se realiza trabajo no aparece simultáneamente trabajo en otra se convierte en otra forma de done and doesy not appear simultaneously as como work elsewhere, it is parte, converted into another form energía.of energy. Al agregado o conjunto sobre el que se concentra la atención seislecalled nombra todo lo demás se The body or assemblage on which attention is focused thesistema. system.AAll else is le llamacalled alrededores. Cuando se When realizawork trabajo, éste esit hecho sobre el sistema, the surroundings. is done, is donepor bylos thealrededores surroundings on the system, oorvicever­ sa; en tanto, la energía se transfiere de los alrededores al sistema, otodethe manera inversa. formaItdeisenergía vice versa, and energy is transferred from the surroundings system, or the La reverse. conocida como trabajo sólo existe durante esta transferencia. En contraste, las energías cinética y potencial 01­SmithVanNess.indd 12 8/1/07 12:47:34 1.8. Energy 13 only during this transfer that the form of energy known as work exists. In contrast, kinetic and 1.8. Energía 3 potential energy reside with the system. Their values, however, are measured with reference to the surroundings; i.e., kinetic energy depends velocity with respect to the surroundings, residen en el sistema. Sin embargo, sus valores se midenon con respecto a los alrededores; es decir, la energía and potential energy depends on elevation with respect to a datum level. Changes in kinetic cinética depende de la velocidad con respecto a los alrededores, y la energía potencial depende de la altura and potential energy do not depend on these reference conditions, provided they are fixed. con respecto a un nivel de referencia. Los cambios en las energías cinética y potencial, una vez fijados, no dependen de estas condiciones de referencia. Example 1.4 An elevator Ejemplo 1.4 with a mass of 2,500 kg rests at a level 10 m above the base of an elevator shaft. It is raised to 100 m above the base of the shaft, where the cable holding it Un elevador con The una masa de falls 2 500freely kg descansa en un a 10 m sobre la base del pozo de un ele­ breaks. elevator to the base ofnivel the shaft and strikes a strong spring. vador. Cuando se eleva a 100 m sobre la base del pozo, se rompe el cable que lo sostiene. El elevador The spring is designed to bring the elevator to rest and, by means of a catch arrangecae libremente hacia la base del pozo y golpea contra un fuerte resorte. El resorte está diseñado ment, to hold the elevator at the position of maximum spring compression. Assuming para poner althe elevador en reposo medio deand un diseño entire process to y, bepor frictionless, taking gde= retención, 9.8 m s−2 ,mantener calculate:al elevador en la po­ sición de máxima compresión del resorte. Si se supone que todo el proceso es sin fricción, y tomando − , calcule: The potential energy of the elevator in its initial position relative to the base of g = 9.8 m s(a) the shaft. a) La energía potencial del elevador en su posición inicial con respecto a la base del pozo. (b) The work done in raising the elevator. b) El trabajo hecho al subir el elevador. (c) The potentialdelenergy of the elevator in its highest position relative to the base c) La energía potencial elevador en su posición más alta con respecto a la base del pozo. of the shaft. d) La velocidad y la energía cinética del elevador justo antes de que golpee en el resorte. (d) The velocity and kinetic energy of the elevator just before it strikes the spring. e) La energía potencial del resorte comprimido. (e) The energy of the compressed f) La energía delpotential sistema que se compone del elevadorspring. y el resorte: 1) al inicio del proceso, 2) cuan­ do el(felevador alcanza su altura máxima, 3) justo antes de queand el elevador incida enstart el resorte, ) The energy of the system consisting of the elevator spring (1) at the 4) después de que el elevador ha llegado al reposo. of the process, (2) when the elevator reaches its maximum height, (3) just before the elevator strikes the spring, (4) after the elevator has come to rest. Solución 1.4 Suponga que el subíndice Solution 1.4 denota las condiciones iniciales; el subíndice , las condiciones cuan­ do el elevador está en su posición más alta, y el subíndice 3, las condiciones justo antes de que el subscript 1 designate the initial conditions; subscript 2, conditions when the elevadorLet llegue al resorte. elevator is at its highest position; and subscript 3, conditions just before the elevator strikes the spring. a) Por la ecuación (.7), E P = mz g = ( 500)(0)(9.8) = 45 000 J (a) By Eq. (1.7), E P1 = mz 1 g = (2,500)(10)(9.8) = 245,000 J � z2 � z2 By Eq. (.), (1.1), W = F dl = mg dl = mg(z 2 − z 1 ) b) Por la(b) ecuación z1 whence de donde (c) By Eq. (1.7), W = (2,500)(9.8)(100 − 10) = 2,205,000 J W = ( 500)(9.8)(00 − 0) = 05 000 J E P2 = mz 2 g = (2,500)(100)(9.8) = 2,450,000 J Note that c) Por la ecuación (.7), P2 − 1 . 500)(00)(9.8) = 450 000 J EWP2==Emz gE =P( Observe que 01­SmithVanNess.indd 13 z1 W = E P − E P . 8/1/07 12:47:35 4 14 14 14 14 CHAPTER CHAPTER 1.1. Introduction Introduction CAPÍTULO 1. Introducción CHAPTER 1. Introduction CHAPTER 1. Introduction CHAPTER 1. Introduction 14 d) Del principio de the conservación de energía mecánica se puede energy, escribir que lamay suma dethat los (d) (d) From From the principle principle of of conservation conservation of of mechanical mechanical energy, one onemay write write thatcam­ (d) From(d)the principle of conservation of mechanical energy, energy, one mayone write that From the principle of conservation of mechanical may write that bios de lasthe energías cinética y potencial durante los procesos desde lasthe condiciones a con3cones cero; the sum sum of of the the kinetickineticand and potential-energy potential-energy changes changes during during the process process from from the sum the of the kineticand potential-energy changes during the process from consum ofthe theprinciple kineticand potential-energy changes during theone process from From of conservation of mechanical energy, may writeconthat es decir,(d)ditions ditions 2 2 to to 3 3 is is zero; zero; that that is, is, ditions 2ditions to sum 3 is2zero; that is, that to the 3 is zero; the of kineticandis,potential-energy changes during the process from con++ �E ==00 E oror EE+K3 3E −−EEKK2 E ++EEP3P30−−EEP2P2==00 �E o ditions�E 2�E to 3KKis zero; that P2→3 Pis, 2→3 2→3 2→3 =�E 0P2→3 �E + �E =or0 orK 3 − EEKK23 K − EPK3 2− +2EPP2 3 = − E P2 = 0 K 2→3 + �E K 2→3P2→3 and and EPE2→3 are zero.Therefore, Therefore, However, EEP3 Sin embargo, E cero. Por �E = zero. 0consiguiente, or E K 3 − E K 2 + E P3 − E P2 = 0 �E KK P3PTherefore, K KyE 2+ 2son 3are 2→3 are zero. However, EHowever, However, K 2 andE KP 2 3and E P3 are zero. Therefore, == EEP=2P2= 2,450,000 EEK=K3Therefore, E P3 are However, E K 2 and E EEzero. E =450 000J JJ J K3 J2,450,000 =32,450,000 EP K3 = E PK23 = P2 = 2,450,000 =2E EKPK23 3= 2,450,000 J 2E (2)(2,450,000) (2)(2,450,000) 1 1 2 2 2E KE2K232E (2)(2,450,000) K3 ,3 , 2u3u3= ==(2)(2,450,000) = = With With 1 EEK2K 2 3= 1=2 2mu 2mu 3, 3= 3 mu u With Con EEK3 = = mu , u = = With E K3 mm 2,500 2,500 2 K 3 33 2 33 m3 m 2,500 2,500 1 2, 2 = 2E K 3 = (2)(2,450,000) = mu u With E K3 Whence, Whence, uu3m 44.27 44.27m ms−1 s−1 3 3 2 3== Whence,Whence, u3 = 44.27 s−1 u 3m= 44.27 m 2,500 s−1 − De donde, = 44.27 44.7 m m ss−1 Whence, uupotential = 33potential (e) (e) Because Because the the changes ininthe the energy energy ofofthe thespring spring and andthe thekinetic kineticenergy energy (e) Because the changes inchanges the potential energy of the spring the kinetic (e) Because the changes in the potential energy of theand spring and theenergy kinetic energy of of the the elevator elevator must must sum sum to to zero, zero, of elevator must sum toen zero, of the elevator must sum tothe zero, e) the Puesto los cambios la in energía potencial del resorte y en laand energía cinética del elevador (e)que Because the changes potential energy of the spring the kinetic energy deben ser (spring) (spring) + + �E �E (elevator) (elevator) = = 0 0 �E �E of cero, the elevator�E must(spring) sum to zero, PP �E (elevator) KK= 0 + =0 P �E P (spring) K + �E K (elevator) The Theinitial initialenergy potential potential energy energy ofofthe the+spring spring and the thefinal final kinetic energy energy ofofthe theeleele(elevador) (spring) �Eand (elevator) 0kinetic �E (resorte) Pspring K and The initial of the the final kinetic energy of the eleThepotential initial potential energy of theand spring the final = kinetic energy of the elevator vator are are zero; zero; therefore, therefore, the the final final potential potential energy energy of of the the spring spring must must equal equal the the vator arevator zero; theenergy finalthe potential energy of the final spring must equal aretherefore, zero; therefore, final potential energy of the spring mustthe equal the The initial potential of the spring and the kinetic energy of the elekinetic kinetic energy energy of of the the elevator elevator just just before before it it strikes strikes the the spring. spring. Thus Thus the the final final popoLa energía potencial inicial lastrikes energía cinética final del elevador son cero; por lo kinetic energy of the elevator justresorte before spring. Thus theThus final po-equal kinetic energy oftherefore, the del elevator justyitbefore it the strikes the spring. the final povator are zero; the potential energy of energía the spring must the justo tential tential energy energy ofofthe the spring spring isfinal is2,450,000 2,450,000 J.J.igual tanto, energy latential energía potencial final del resorte debe ser a la cinética del elevador tential of the spring is 2,450,000 J. energy of the spring is 2,450,000 energy elevator beforeJ. it strikes the Thusesthe 450 final000 po- J. Así,just la energía potencial finalspring. del resorte antes dekinetic que incida conofelthe resorte. tential energy of theand spring isspring 2,450,000 J. are (f (f ) ) If If the the elevator elevator and the the spring together together are taken taken as as the the system, system, the the initial initial energy energy (f ) If the(felevator and the spring are takenare astaken the system, the initial ) If the elevator and thetogether spring together as the system, theenergy initial energy ofofthe thethe system isisthe the potential potential energy energy ofofthe the elevator, elevator, orJ. or245,000 245,000 J.J.energy The The total total energy energy f) the Si elsystem elevador ysystem elpotential resorte juntos seof consideran como el sistema, la energía inicial del sistema es of is energy the elevator, or 245,000 The total of the system is the potential energy of the elevator, or 245,000 J. The total energy (f ) If the elevator and the spring together are taken as the system, the initial energy of of the the system system can can change change only only if if work work is is transferred transferred between between it it and and the the surroundsurroundla the energía potencial del elevador, o 45if 000 J. La energía total del sistema sólo cambia si se trans­ of system can change only if work is transferred between it and the surroundof the system can change only work is transferred between it and the surroundofings. the system the potential energy of the elevator, 245,000 J.by The total energysobre el ings. As Asélthe the elevator elevator isisraised, raised, work work isis done on onor the the system system by the surroundings surroundings fiere trabajo entre yis alrededores. Conforme sedone levanta el elevador sethe hace trabajo ings. Asings. the elevator islos raised, isifwork done on the system by the surroundings As the elevator is work raised, is done on the between system by the surroundings of the system can change only work is transferred it and the surroundin in the the amount amount of of 2,205,000 2,205,000 J. J. Thus Thus the the energy energy of of the the system system when when the the elevator elevator sistema por de of los2,205,000 alrededores en unathe cantidad de the 05when 000 the J.when De esta la energía in the amount of 2,205,000 J. isThus energy of theon system elevator in theacción amount J.the Thus energy ofthe system theforma, elevator ings. As the elevator raised, work is done system by the surroundings reaches reaches its its maximum maximum height height is is 245,000 245,000 + + 2,205,000 2,205,000 = = 2,450,000 2,450,000 J. J. Subsequent Subsequent del sistema cuando el height elevador alcanza su altura máxima es 45 000 + 05 000 = 450 000 J. reaches its maximum is 245,000 + 2,205,000 = 2,450,000 J. Subsequent reaches its maximum height is 245,000 + 2,205,000 = 2,450,000 J. Subsequent in changes the amount ofocurren 2,205,000 J. Thus the energy of the system when the elevator changes occur occur entirely entirely within within the the system, system, with with no no work work transfer transfer between between the the syssys- en­ Los cambios siguientes por completo dentro del sistema, sin transferencia trabajo changes occur entirely within the system, with no+with work transfer between the syschanges occur entirely within the245,000 system, no work transfer between thedesysreaches its maximum height is 2,205,000 = 2,450,000 J. Subsequent tem tem and and surroundings. surroundings. Hence Hence the the total total energy energy of of the the system system remains remains constant constant atat en tre eland sistema y los alrededores. Por lothe tanto, energía total del sistema permanece tem surroundings. Hence the total energy the system remains constant at theconstante tem and surroundings. Hence totallaof energy of the system remains constant at changes occur entirely within the system, with no work transfer between sys2,450,000 2,450,000 J. J. It It merely merely changes changes from from potential potential energy energy of of position position (elevation) (elevation) of of the the 450 000 sólo cambia changes de energía potencial deof posición (elevación) del elevador a energía 2,450,000 J.J.and ItÉsta merely from potential energy position (elevation) of the 2,450,000 J. It changes merely from potential energy position (elevation) of the tem surroundings. Hence total of theofsystem remains constant atofof elevator elevator to tokinetic energy energy ofofthe the the elevator elevator totoenergy potential potential energy energy ofofconfiguration configuration cinéticaelevator del elevador, ykinetic a energía potencial de laenergy configuración en el resorte. elevator to kinetic energy of the elevator to potential of configuration of to kinetic energy of the elevator to potential energy of configuration of 2,450,000 J. It merely changes from potential energy of position (elevation) of the the thespring. spring. the spring. the spring. elevator to kinetic energy thedeelevator to potential energy of configuration of Este ejemplo ilustra el uso de laofley conservación de la energía mecánica. Sin embargo, se the spring. This This example illustrates illustrates application of ofthe thelaw lawofofconservation conservation ofofexactos mechanical mechanical supone todo elexample proceso ocurre application sinapplication fricción; resultados obtenidos son sólo para This que example illustrates application of the law of conservation of mechanical This example illustrates oflos the law of conservation of mechanical energy. energy. However, However, the the entire entire processisisto assumed assumed totooccur occur without withoutthe friction; friction; the the procesos tan idealizados como éste. energy. However, the entire is process assumed occur friction; energy. theprocess entire process isofassumed occur without the This However, example illustrates application theidealized law to ofwithout conservation offriction; mechanical results results obtained obtained are are exact exact only only for for such such an an idealized process. process. results obtained are exactare only for suchprocess an such idealized process. results exact only for idealized energy.obtained However, the entire isanassumed toprocess. occur without friction; the results obtained are exact only for such an idealized process. During the the period period ofofdevelopment development of ofthe thelaw lawofofconservation conservation ofofmechanical mechanical energy, energy, heat heat During period development ofdethe of ofdemechanical energy, heat Durantethe elDuring periodo del desarrollo la law ley conservación la energía mecánica, en general During theofperiod of development of de theconservation law of conservation of mechanical energy, heat no se was was not not generally generally recognized recognized as as a a form form of of energy, energy, but but was was considered considered an an indestructible indestructible fluid fluid was notwas generally recognized as of a form of energy, but wasseof considered an indestructible fluid reconocía alnot calor como forma deasenergía; más bien, leconservation consideraba como fluido indestructible lla­ generally recognized a form of energy, but was considered an un indestructible fluid During the una period development of the law of mechanical energy, heat called called caloric. caloric. This This concept concept was was firmly firmly entrenched, entrenched, and andmuchos for formany many years years no nohizo connection connection was was called This concept was firmly entrenched, and for many years no connection was mado caloric. calórico. Este concepto estaba firmemente establecido, y por años no se conexión entre el called caloric. This concept was firmly entrenched, and for many years no connection was was not between generally recognized asfrom afrom form of energy, but was considered anofof indestructible made made between heat heat resulting resulting friction friction and andthe theestablished established forms forms energy. energy. The Thefluid law lawofof made heat resulting from friction and the established forms of energy. The law of calor between resultante de la fricción y las formas establecidas de la energía. Por lo tanto, la ley de la conservación made between heat resulting from friction and the established forms of energy. The law of called caloric. of This concept was firmly limited entrenched, and for many years no connection was conservation conservation ofenergy energy was was therefore therefore limitedininapplication application totofrictionless frictionless mechanical mechanical proproconservation of energy was therefore limited in application to frictionless mechanical prode la energía estaba limitada a was procesos mecánicos sininthe fricción. No to es forms necesaria talmechanical limitación; hoy conservation of energy therefore limited application frictionless promade between heat resulting from friction and established of energy. The law of en día, cesses. cesses. No No such such limitation limitation is is necessary; necessary; heat heat like like work work is is now now regarded regarded as as energy energy in in transit, transit, cesses. No such limitation is necessary; heat like work now as concepto energy transit, tanto alcesses. calor como aloftrabajo sewas lesisconsidera como energía en regarded tránsito, un que ganó aceptación en No such limitation necessary; heat like work is nowtoregarded as in energy in transit, conservation energy therefore limited inisapplication frictionless mechanical processes. No such limitation is necessary; heat like work is now regarded as energy in transit, 01­SmithVanNess.indd 14 8/1/07 12:47:38 Problemas 5 los años posteriores a 850, en gran parte por los experimentos clásicos de J. P. Joule. Estos experimentos se consideran de manera detallada en el capítulo , pero primero examinaremos algunas de las caracterís­ ticas del calor. 1.9 CALOR Sabemos por experiencia que un objeto caliente que se pone en contacto con otro objeto frío tiende a enfriar­ se, al mismo tiempo que este último se calienta. Una visión razonable es que algo se transfiere del objeto ca­ liente al frío, y a ese algo le llamamos calor Q.0 En estos términos se dice que el calor siempre fluye de una temperatura más alta a una más baja, lo cual nos lleva al concepto de temperatura como la fuerza impulsora para la transferencia de energía como calor. Con más precisión, la rapidez de transferencia de calor de un cuerpo a otro es proporcional a la diferencia de temperatura entre los dos cuerpos; cuando no existe diferen­ cia de temperatura, no hay transferencia neta de calor. En sentido termodinámico, el calor nunca se considera como algo que está almacenado dentro de un cuerpo. Al igual que el trabajo, solamente existe como energía en tránsito desde un cuerpo hacia otro; o, en términos termodinámicos, entre un sistema y sus alrededores. Cuando se agrega energía en forma de calor a un cuerpo, se almacena no como calor sino como energías ci­ nética y potencial de los átomos y de las moléculas que constituyen el sistema. A pesar de la naturaleza transitoria del calor, con frecuencia se le ve sólo desde el punto de vista de su efecto sobre el cuerpo del cual o al cual se transfiere. De hecho, alrededor de 930 las definiciones de unida­ des de calor estaban basadas en los cambios de temperatura de una masa de una unidad de agua. De esta forma, la caloría se definió por mucho tiempo como la cantidad de calor que, al transferirse a un gramo de agua, elevaba su temperatura un grado Celsius. Del mismo modo, la unidad térmica inglesa (o Btu, British termal unit) se definió como la cantidad de calor que, al transferirse a una libra masa de agua, elevaba su temperatura en un grado Fahrenheit. Aunque estas definiciones proporcionan una “sensación” de la magnitud de estas unidades de calor, dependen de experimentos realizados con agua y, por lo tanto, están sujetas a cam­ bios en la medida en que las mediciones se vuelven más precisas. En la actualidad se reconoce a la caloría y al (Btu) como unidades de energía, y se definen con respecto al joule, la unidad de energía del SI, que es igual a N m. Éste es el trabajo mecánico realizado por la fuerza de un newton que actúa a través de una distancia de un metro. Todas las demás unidades de energía se definen como múltiplos del joule. El pie­libra fuerza, por ejemplo, es equivalente a .355879 J, la caloría es igual a 4.840 J, mientras que el (Btu) es igual a 055.04 J. La unidad SI para la potencia es el watt, cuyo símbolo es W y se define como una relación de energía de un joule por segundo. La tabla A. en el apéndice A proporciona una lista amplia de factores de conversión para la energía, así como para otras unidades. PROBLEMAS 1.1. ¿Cuál es el valor de gc y cuáles son las unidades en un sistema donde el segundo, el pie y la libra masa están definidos como en la sección ., y el poundal es la unidad de fuerza requerida para que a (lbm) se le dé una aceleración de (pie)(s)−? 0 Un punto de vista igualmente razonable sería considerar que se transfiere “frío” desde el objeto frío hacia el objeto caliente. 01­SmithVanNess.indd 15 8/1/07 12:47:39 CHAPTER 1. CHAPTERCAPÍTULO 1. Introduction Introduction 1. Introducción 16 6 16 1.2. current is electrical dimension, ampere (A) 1.2. corriente eléctrica es fundamental la dimensiónSI eléctrica fundamental el SI, y su unidad el ampere (A). 1.2.LaElectric Electric current is the the fundamental SI electrical dimension,enwith with ampere (A) as asesunit. unit. Determine units for the following quantities, as combinations of fundamental SI units. Determine las unidades de las siguientes cantidades, como combinaciones de las unidades fundaDetermine units for the following quantities, as combinations of fundamental SI units. mentales del SI. (a) (a) Electric Electric power; power; (b) (b) Electric Electric charge; charge; (c) (c) Electric Electric potential potential difference; difference; a)(d) Potencia eléctrica; b) carga eléctrica; c) diferencia de potencial eléctrico; Electric resistance; (e) Electric capacitance. (d) Electric resistance; (e) Electric capacitance. d) resistencia eléctrica; e) capacitancia eléctrica. sat represented as of 1.3. saturation pressure P is often often as aa function function of temperature temperature 1.3.LaLiquid/vapor Liquid/vapor saturationde pressure P sat is 1.3. presión de saturación un líquido/vapor Psatrepresented con frecuencia se representa como una función de by an equation of the form: an equation ofmedio the form: laby temperatura por de una ecuación de la forma siguiente: bb sat sat /torr log10 P log /torr = = aa − − t/◦◦ C + c 10 P t/ C + c Here, parameters a, b, are substance-specific constants. Suppose required to Aquí, parámetros y cccson específicas de las sustancias. Suponga Here,los parameters a, a, b, band and are constantes substance-specific constants. Suppose it it is is requiredque to se requiere sat sat sat represent P by the equivalent equation: represent P by the equivalent equation: representar P mediante la ecuación equivalente: B B sat ln /kPa = =A A− − T /K + C ln P P sat /kPa T /K + C Muestre cómo relacionados en las dos ecuaciones. Show the parameters in two equations related. Show how how theestán parameters in the the los twoparámetros equations are are related. 1.4. what absolute the Fahrenheit temperature scales give 1.4. temperatura absoluta las do escalas Celsiusand y Fahrenheit el mismo valor ¿Cuál 1.4.¿AAt Atqué what absolute temperature temperature do the Celsius Celsius and Fahrenheitdan temperature scalesnumérico? give numerical esthe esesame valor? the same numerical value? value? What What is is the the value? value? 1.5. Pressures up 3,000 measured aa dead-weight gauge. piston diameter 1.5. presiones superiores 3 000 bar se with miden con una balanza peso muerto. El diámetro del 1.5.Las Pressures up to to 3,000 bar bara are are measured with dead-weight gauge.deThe The piston diameter is 4 mm. What is the approximate mass in kg of the weights required? pistón es deWhat 4 mm. ¿Cuál es la masamass aproximada de las pesas requeridas? is 4 mm. is the approximate in kg ofen thekgweights required? 1.6. Pressures up to measured with dead-weight gauge. The piston 1.6. presiones a 3 are 000(atm) se miden una balanza de peso El diámetro del 1.6.Las Pressures up superiores to 3,000(atm) 3,000(atm) are measured with aacon dead-weight gauge. The muerto. piston diamdiam) of the weights required? eter is 0.17(in). What is the approximate mass in (lb pistón es de 0.7 (pulg). ¿Cuál es la masa aproximada (lb ) de las pesas requeridas? m m the weights required? eter is 0.17(in). What is the approximate mass in (lbm ) of ◦ C (open to the atmosphere at one end) 1.7. reading on aa mercury 1.7. lectura de un de mercurioat 5◦°C (abierto la atmosphere atmósfera enatuno sus extremos) C (open to athe onede end) 1.7.LaThe The reading onmanómetro mercury manometer manometer ata 25 25 −2 . Atmospheric local is m pressure esis 56.38cm. cm.The La aceleración local deof gravedad es de 9.83 s−. La presión atmosférica es de isde56.38 56.38 cm. The local acceleration acceleration oflagravity gravity is 9.832 9.832 m ss−2m . Atmospheric pressure is the in being measured? The of 0.78 kPa.kPa. ¿CuálWhat es la is presión absolutapressure en kPa que debe medirse? La densidad del mercurio a 5 °C is 101.78 101.78 kPa. What is the absolute absolute pressure in kPa kPa being measured? The density density of ◦◦ C−3 −3 at esmercury de 3.534 g cm mercury at 25 25 C is is.13.534 13.534 g g cm cm−3 .. ◦◦ F) (open to the atmosphere at one end) is 1.8. reading on aa mercury at l.8. lectura en un de mercurio 70(°F) (abierto a laatmosphere atmósfera at enone unoend) de sus F) (open to the is extremos) 1.8.LaThe The reading on manómetro mercury manometer manometer at a70( 70( −2 −. La −2 .. Atmospheric pressure The acceleration of is es25.62(in). de 5.6 (pulg). La aceleración de la gravedad es de 3.43(pie)(s) presión atmosfé­ Atmospheric pressure 25.62(in). The local local accelerationlocal of gravity gravity is 32.243(ft)(s) 32.243(ft)(s) is Hg). What the absolute (psia) measured? density rica es de 9.86 (pulg de is Hg). es lapressure presiónin en (psia) que debeThe medirse? is 29.86(in 29.86(in Hg). What is the¿Cuál absolute pressure inabsoluta (psia) being being measured? The densityLa densidad ◦◦ F) is 13.543 g cm−3 −3 −3 of mercury at 70( . del 3.543 g cm. . ofmercurio mercury aat70(°F) 70( F)esis de 13.543 g cm 1.9. Liquids that boil at low are stored as their 1.9. líquidos a temperaturas relativamente bajas son almacenados como líquidos bajo 1.9.Los Liquids thatque boilhierven at relatively relatively low temperatures temperatures are often often stored as liquids liquids under under their vapor pressures, which at ambient temperature can be quite large. Thus, n-butane sus presiones de vapor, lasatcuales serían más grandes temperatura ambiente. Así, el n­butano al­ vapor pressures, which ambient temperature canabe quite large. Thus, n-butane stored aa liquid/vapor system is 2.581 bar temperature 300 K. macenado un sistema líquido/vapor está aof presión de aa.58 bar paraof de stored as as como liquid/vapor system is at at aa pressure pressure ofuna 2.581 bar for for temperature ofuna 300temperatura K. 33 ) of this kind is sometimes 3 done in spherical tanks. SugLarge-scale storage (>50 m 300 K. El almacenaje de grandes cantidades (>50 m ) de estainclase de sustancias en ocasiones kind is sometimes done spherical tanks. SugLarge-scale storage (>50 m ) of this two reasons why. segest realiza tanques esféricos. Sugiera dos posibles razones del por qué. gest twoen reasons why. 1.10. first accurate the of gases were made 1.10. primer preciso paraof las propiedades de los gases a alta fue creado en 1.10.ElThe The first instrumento accurate measurements measurements ofmedir the properties properties of high-pressure high-pressure gases werepresión made by by E. France 1869 and Before developing the Francia por E. H.in entre 869 y 893. Antes de desarrollar la balanza de peso muerto, Ama­ E. H. H. Amagat Amagat inAmagat, France between between 1869 and 1893. 1893. Before developing the dead-weight dead-weight gauge, he worked aa mine shaft, and mercury measurements gat trabajó el pozoin una mina y utilizó un aamanómetro de mercuriofor medir presiones mayo­ gauge, heen worked inde mine shaft, and used used mercury manometer manometer forpara measurements more bar. Estimate the of resof 400 bar.to Determine altura manómetro requerido. ofapressure pressure to more than thanla400 400 bar.del Estimate the height height of manometer manometer required. required. 01­SmithVanNess.indd 16 8/1/07 12:47:41 Problems Problemas 17 7 1.11. An instrument to measure the acceleration of gravity on Mars is constructed of a spring 1.11. Un instrumento para mediralamass aceleración de la Marte está the construido con un resorte de from which is suspended of 0.40 kg. Atgravedad a place onenearth where local accelerdonde se suspende una masa de 0.40 kg. En un lugar de la Tierra donde la aceleración −2 ation of gravity is 9.81 m s , the spring extends 1.08 cm. When the instrument pack- local de la −, el resorte se extiende .08 cm. Cuando el aparato desciende en Marte, la radio gravedad es 9.8 m s age is landed on Mars, it radios the information that the spring is extended 0.40 cm. informa resorte acceleration se extiende 0.40 cm. ¿Cuál es la aceleración de la gravedad marciana? What isque theelMartian of gravity? 1.12. variación deof la fluid presión de un with fluidoheight con laisaltura está by descrita por la ecuación diferencial: 1.12.LaThe variation pressure described the differential equation: dP = −ρg dz Here,ρ ρes is and yggisesthe local acceleration anun ideal Aquí, la specific densidaddensity específica la aceleración local deofla gravity. gravedad.For Para gas ideal, ρ = gas, ρ = MP/RT , where M is molar mass and R is the universal gas constant. M P / RT, donde M es la masa molar y R es la constante universal de los gases. Modele la atmós­ Modeling thecolumna atmosphere as an isothermal column of°C ideal gasdeatcalcular 10◦ C, estimate fera como una isotérmica de un gas ideal a 0 a fin la presiónthe ambiental en ambientdonde pressure Denver, z =al 1(mile) to sea level. ForMair, Denver, z −1 = in(milla) conwhere respecto nivel delrelative mar. Para el aire, tome = take 9 g mol−; los ; values of R are given in App. A. M = 29 g mol valores de R se dan en el apéndice A. 1.13. A group of engineers has landed on the moon, and they wish to determine the mass 1.13. Un ingenieros ha aterrizado la Luna y desea masaatdea algunas ofgrupo some de rocks. They have a spring en scale calibrated to determinar read poundslamass locationrocas. Tiene una escala de resorte calibrada para leer libras masa en una posición donde la aceleración de la −2 where the acceleration of gravity is 32.186(ft)(s) . One of the moon rocks gives a −). Una de las rocas lunares da una lectura de 8.76 en esta escala. ¿Cuál gravedad es 3.86(pie)(s reading of 18.76 on this scale. What is its mass? What is its weight on the moon? − esTake la masa? ¿Cuál=es el peso en −2la g(moon) 5.32(ft)(s) . Luna? Tome g(Luna) = 5.3(pie)(s ). 1.14. luz externa de seguridad de 70 wattsonseaverage, utiliza en 0 A horas día.costs Un foco nuevo 1.14.Una A 70-watt outdoor security light burns, 10 promedio hours a day. newalbulb cuesta $5.00, y su tiempo de vida es de casi 000 horas. Si la electricidad cuesta $0.0 por kW­hora, $5.00, and the lifetime is about 1,000 hours. If electricity costs $0.10 per kW-hour, ¿cuál es el costo anual de “seguridad” por luz? what is the yearly price of “security,” per light? 1.15. confinado en un cilindro de .5by (pie) de diámetro por medio un pistón, sobre el 1.15.Un Agas gasseis encuentra confined in a 1.25(ft)-diameter cylinder a piston, on which rests adeweight. cual descansa una pesa. La masa del pistón y la pesa en conjunto es de 50(lb ). La aceleración local de m The mass of the piston and weight together is 250(lbm ). The local acceleration of − y la presión atmosférica es de 30.(pulg de Hg). −2 lagravity gravedad es de 3.69(pie)(s) is 32.169(ft)(s) , and atmospheric pressure is 30.12(in Hg). Whatesislathe forceen in(lb (lbf))ejercida exerted on thegas gaspor by la theatmósfera, atmosphere, the piston, and the a) (a)¿Cuál fuerza en el el pistón y la pesa? Suponga que f weight, assuming no friction between the piston and cylinder? no existe fricción entre el pistón y el cilindro. Whatesislathe pressure in (psia)? b) (b)¿Cuál presión del of gasthe engas (psia)? (c) If the gas in the cylinder is heated, it expands, pushing the piston and weight c) Si el gas en el cilindro se calienta, se expande y empuja el pistón y la pesa hacia arriba. Si el upward. If the piston and weight are raised 1.7(ft), what is the work done by(pie the lb )? ¿Cuál pistón y la pesa se levantan .7(pie), ¿cuál es el trabajo realizado por el gas en f )? What is the change in potential energy of the piston and weight? gas in (ft lb f es el cambio en la energía potencial del pistón y la pesa? 1.16. A gas is confined in a 0.47-m-diameter cylinder by a piston, on which rests a weight. 1.16. Un gas se encuentra confinado en un cilindro de 0.47 m de diámetro por medio de un pistón, sobre The mass of the piston and weight together is 150 kg. The local acceleration of gravity el cual descansa una pesa. La masa del pistón y la pesa en conjunto es de 50 kg. La aceleración is 9.813 m s−2 , and atmospheric pressure is 101.57 kPa. local de la gravedad es de 9.83 m s−, y la presión atmosférica es de 0.57 kPa. (a) What is the force in newtons exerted on the gas by the atmosphere, the piston, and the weight, assuming no friction between piston cylinder? el pistón y la pesa? Supon­ a) ¿Cuál es la fuerza en newtons ejercida sobrethe el gas porand la atmósfera, (b)gaWhat is the pressure of the gas in kPa? que no hay fricción entre el pistón y el cilindro. If the in the del cylinder heated, it expands, pushing the piston and weight b) (c)¿Cuál es gas la presión gas eniskPa? thecilindro piston and weight are 0.83 what el is pistón the work done the arriba. Si c) Siupward. el gas enIf el se calienta, se raised expande y m, empuja y la pesabyhacia in kJ? is the change in potential of therealizado piston and weight? el gas pistón y laWhat pesa se levantan 0.83 m, ¿cuál energy es el trabajo por el gas en kJ? ¿Cuál es el cambio en la energía potencial del pistón y la pesa? 01­SmithVanNess.indd 17 8/1/07 12:47:43 8 CAPÍTULO 1. Introducción 1.17. Verifique si el joule es la unidad en el sistema internacional para las energías cinética y potencial. 1.18. Un automóvil que tiene una masa de 50 kg se encuentra viajando a 40 m s−. ¿Cuál es la energía cinética en kJ? ¿Cuánto trabajo debe efectuarse para detenerlo? 1.19. Las turbinas de una planta hidroeléctrica son alimentadas por una caída de agua de 50 m de altura. Al suponer una eficiencia de conversión de la energía potencial en eléctrica de 9%, y 8% de pérdi­ da de potencia resultante en la transmisión, ¿cuál es la rapidez de flujo de masa de agua requerida para encender un foco de 00 watts? 1.20. A continuación se encuentra una lista de factores de conversión aproximados, que se emplean para cálculos “rápidos”. Ninguno de ellos es exacto, pero en la mayoría hay un margen de error de ± 0%. Haga uso de la tabla A. (apéndice A) para establecer los factores de conversión exactos. • (atm) ≈ bar • (Btu) ≈ kJ • (hp) ≈ 0.75 kW • (pulgada) ≈ .5 cm • (lbm) ≈ 0.5 kg • (milla) ≈ .6 km • (cuarto) ≈ litro • (yarda) ≈ m Agregue sus propios términos a la lista. La idea es mantener los factores de conversión en una forma simple que sea fácil de recordar. 1.21. Considere la siguiente propuesta para un calendario decimal. La unidad fundamental es el año de­ cimal (año), igual al número convencional de segundos (SI) requeridos para que la Tierra complete una órbita alrededor del Sol. Otras unidades son definidas en la tabla siguiente. Desarrolle, hasta donde sea posible, factores para convertir unidades del calendario decimal a unidades del calendario convencional. Analice los puntos a favor y en contra de esta propuesta. 01­SmithVanNess.indd 18 Unidades del calendario decimal Símbolo Definición Segundo Seg 0–6 Año Minuto Hora Día Semana Mes Min H Día Sem Mes 0–5 Año 0–4 Año 0–3 Año 0– Año 0– Año Año Año 8/1/07 12:47:43 Problemas 9 19 Problems Problems 19 1.22. Los costos de la energía varían en gran medida con la fuente de energía: el carbón cuesta $5.00/to­ 1.22. Energy coststiene vary un greatly with energyde source: coal @y$25.00/ton, gasoline @ a pump nelada, la gasolina precio creciente $.00/galón la electricidad vale $0.000/kW­hora. 1.22. Energy costs vary greatly with energy source: coal @ $25.00/ton, gasoline @ a pump price of es $2.00/gal, and electricity Conventional is to put En la práctica conveniente expresarlos@en$0.1000/kWhr. una base común en $ GJ−. practice [Un gigajoule equivale price of $2.00/gal, and electricity @ $0.1000/kWhr. Conventional practice is to put these on a common basis by expressing them insuponga $−1GJ−1 . valores [A gigajoule is approximately aproximadamente a 0 6 (Btu).] Para este propósito de calentamiento totales (sin these on a common basis by expressing them in $ GJ −3 . [A gigajoule is approximately 106 (Btu).] purpose, assumeygross ofgasolina. 29 MJ kg−1 for coal and corrección), de 9For MJthis kg− para el carbón de 37heating GJ m values para la 106 (Btu).] For this purpose, assume gross heating values of 29 MJ kg−1 for coal and −3 37 GJ m for gasoline. 37 GJ m−3 for gasoline. − a) Clasifique en orden las tres fuentes de energía con respecto a los costos de energía −1 . en $ GJ . (a) Rank order the three energy sources with respect to energy cost in $ GJ b) (a)Explique la notable discrepancia que hay enrespect los resultados numéricos inciso a). Discuta las Rank order the three energy sources with to energy cost in $ del GJ−1 . (b) Explain the largededisparity in the numerical results of Part (a). Discuss the advanventajas y desventajas las tres fuentes de energía. (b) Explain the large disparity in the numerical results of Part (a). Discuss the advantages and disadvantages of the three energy sources. tages and disadvantages of the three energy sources. 1.23. Los costos del equipo de una fábrica de especies químicas rara vez varían en proporción al tamaño. 1.23. Chemical-plant equipment costs vary in proportion to size. In the simplest case, el caso más sencillo, el costo varíararely con S conforme a la 1.23.EnChemical-plant equipment costsCrarely varyelintamaño proportion to size. In theecuación simplestalométrica: case, cost C varies with size S according to the allometric equation cost C varies with size S according to the allometric equation C = αSβ β C = αS C = αS β La magnitud del exponente β por lo regular se encuentra entre 0 y . Para una amplia variedad de The size β is typically between 0 and 1. For a wide varity of equipment types equipos su exponent valorexponent es de aproximadamente The size β is typically between0.6. 0 and 1. For a wide varity of equipment types it is approximately 0.6. it is approximately 0.6. a) Para 0 < β < muestre que el costo por tamaño unitario disminuye con el incremento de tama­ (a) For 0 < β < 1, show that cost per unit size decreases with increasing size. (”Econ(a)ñoFor 0 < β < de 1, show that cost per unit size decreases with increasing size. (”Econ(“economía escala”). omy of scale.”) omy of scale.”) b) Considere el caso de un tanque de almacenamiento esférico. El tamaño se mide regularmente (b) Consider the case of a spherical storage tank. The size is commonly measured by (b)mediante Consider case ofinterno a tspherical storage tank. commonly measured by el the volumen V it. Demuestre que The β = size /3.is¿De qué parámetros o propiedades se internal volume V . Show that β = 2/3. On what parameters or properties would t . Show i internal thatα?β = 2/3. On what parameters or properties would espera quevolume dependaVila cantidad you expect quantity α to depend? you expect quantity α to depend? 1.24. Un laboratorio elabora un informe con la siguiente información de presión de vapor (P sat) para una 1.24. A laboratory reports the following vapor-pressuresat(P sat ) data for a particular organic 1.24.especie A laboratory the following vapor-pressure (P ) data for a particular organic orgánicareports particular: chemical: chemical: t/◦ C P sat /kPa t/◦ C P sat /kPa t/◦ C P sat /kPa t/◦ C P sat /kPa −18.5 3.18 32.7 41.9 −18.5 3.18 32.7 41.9 −9.5 5.48 44.4 66.6 −9.5 5.48 44.4 66.6 0.2 9.45 52.1 89.5 0.2 9.45 52.1 89.5 11.8 16.9 63.3 129. 11.8 16.9 63.3 129. 23.1 28.2 75.5 187. 23.1 28.2 75.5 187. Correlate the data by fitting them to the Antoine equation: Correlate the data de by la fitting them to the Antoinelosequation: Haga la correlación información ajustando datos a la ecuación de Antoine: B ln P sat /kPa = A − B ln P sat /kPa = A − T /K + C T /K + C That is, find numerical values of parameters A, B, and C by an appropriate regression is,encuentre find numerical valuesnuméricos of parameters B, and C by EsThat decir, los valores de losA,parámetros A, an B yappropriate C medianteregression un procedimiento de procedure. Discuss the comparison of correlated with experimental values. What is procedure. DiscussTambién the comparison correlated with experimental What is regresión apropiado. compareoflos valores correlacionados convalues. los experimentales. ¿Cuál the predicted normal boiling point of this chemical? predicted normal boiling point of this chemical? esthe el punto de ebullición normal pronosticado para esta especie química? −1 . Between 1.25. (a) In summer 1970, the pump price of gasoline was about $0.35(gal) −1 . Between −. Entre 1.25. 1970, theelpump gasolineera was $0.35(gal) 1.25. a) (a)EnInelsummer verano de 970, precioprice de laofgasolina de about $0.35(gal) 970 y 000, la tasa 1970 and 2000, the average rate of inflation was about 5% per year. What might 1970 and the average rate of inflation was 5%¿Cuál per year. promedio de2000, inflación fue de aproximadamente 5% about por año. es el What preciomight esperado para el be the expected pump price in summer 2000? What conclusion might one reach be thedelexpected pump in summerse2000? one reach verano año 000? ¿A price qué conclusión puedeWhat llegarconclusion a partir de might estos cálculos? from this calculation? from this calculation? 01­SmithVanNess.indd 19 8/1/07 12:47:45 0 CAPÍTULO 1. Introducción CHAPTER 1. Introduction 20 b) Un doctor en ingeniería inició su carrera en 970, con un salario de $6 000(año)−, se retiró en − año 000engineer con un salario $80career 000(año) tanto le mantenerse −1 , retiredcon este sa­ (b)el A Ph.D. startingdehis in 1970. ¿Qué at a salary ofconvendrá $16,000(yr) lario dada una tasa de inflación de 5% anual? −1 in 2000 at a salary of $80,000(yr) . How well did his salary keep up with an c) Los incrementos matrículas inflation rate ofen 5%lasper year? de las principales universidades privadas de Estados Unidos presentan una tasa de inflación de aproximadamente 3% anual. Haga uso de esta observación (c) Tuition increases at major private universities in the United States have led inflapara sugerir estrategias de pago para la futura matrícula de un niño en una universidad privada. tion rates by about 3% per year. Use this observation to suggest strategies for Suponga la ausencia de ayuda económica, una tasa de inflación anual de 5% y una matrícula ac­ paying the future tuition for a child at a private university. Assume no financial tual de $5 000(año)−. aid, an annual inflation rate of 5% per year, and a current tuition of $25,000(yr)−1 . Recall the la compound interest formula: Recuérdese fórmula de interés compuesto: C(t2 ) = (1 + i)t2 −t1 C(t1 ) whereC C can simbolizar be cost, salary, etc., t1 andetc., t2 indicate times, and itiempos, is a ratey (inflation, donde puede costos, salarios, t y t representan la i es una tasa (infla­ interest, etc.)etc.) expressed as acomo decimal. ción, interés, expresada un número decimal. 01­SmithVanNess.indd 20 8/1/07 12:47:46 Capítulo 2 La primera ley y otros conceptos básicos 2.1 EXPERIMENTOS DE JOULE El concepto moderno de calor se desarrolló después de experimentos decisivos llevados a cabo por James P. Joule1 (1818-1889), en el sótano de su casa cerca de Manchester, Inglaterra, durante la década de 1840. Los experimentos de Joule se caracterizaron por ser bastante simples en los aspectos esenciales, pero tomó precauciones minuciosas para asegurar su exactitud. En su más famosa serie de mediciones colocó en un recipiente aislado cantidades conocidas de agua, aceite y mercurio, y después mezcló el fluido con un agitador giratorio. La cantidad de trabajo hecha sobre el fluido con el agitador fue medida con precisión y se anotó cuidadosamente el cambio de temperatura del fluido. Joule encontró que para cada fluido se requiere una cantidad fija de trabajo por unidad de masa, por cada grado de aumento en la temperatura causado por el agitador, y que la temperatura original del fluido se puede restituir por la transferencia de calor a través del simple contacto con un objeto más frío. De esta manera, Joule demostró finalmente que existe una relación cuantitativa entre el trabajo y el calor y que, por lo tanto, el calor es una forma de energía. 2.2 ENERGÍA INTERNA En experimentos como los realizados por Joule, la energía que se agrega a un fluido en forma de trabajo se transfiere posteriormente desde el fluido como calor. ¿Dónde se encuentra esta energía entre su adición al fluido y la transferencia desde el mismo? Pensando de modo lógico se diría que está contenida en el fluido, pero en otra forma llamada energía interna. La energía interna de una sustancia no incluye la energía que ésta puede poseer como resultado de su posición macroscópica o su movimiento. Más bien, se refiere a la energía de las moléculas internas de la sustancia. Debido a su movimiento incesante, todas las moléculas poseen energía cinética de traslación y, exceptuando a las moléculas monoatómicas, también poseen energía cinética de rotación y vibración interna. La 1 Estos experimentos y su influencia en el desarrollo de la termodinámica fueron descritos por H. J. Steffens, James Prescott Joule and the Concept of Energy, Neale Watson Academic Publications, Inc., Nueva York, 1979. 21 02-SmithVanNess.indd 21 8/1/07 12:48:40 22 CAPÍTULO 2. La primera ley y otros conceptos básicos adición de calor a una sustancia aumenta su actividad molecular provocando un aumento en su energía interna. El trabajo hecho sobre la sustancia puede tener el mismo efecto, como lo demostró Joule. La energía interna de una sustancia también incluye la energía potencial que resulta de las fuerzas intermoleculares (véase la sección 16.1). En una escala submolecular la energía se asocia con los electrones y los núcleos de los átomos, así como con la energía de enlace que resulta de las fuerzas que mantienen unidos a los átomos como moléculas. Esta forma de energía se llama interna para distinguirla de las energías cinética y potencial, las cuales se asocian con la sustancia debido a su movimiento o posición macroscópica y se consideran formas externas de energía. La energía interna no tiene una definición termodinámica concisa. Es un concepto básico en termodinámica. No se puede medir de manera directa ni existen medidores de energía interna. Como resultado, los valores absolutos son desconocidos; sin embargo, esto no es una desventaja en el análisis termodinámico, ya que sólo se requieren cambios en la energía interna. 2.3 LA PRIMERA LEY DE LA TERMODINÁMICA El reconocimiento del calor y la energía interna como formas de energía hace posible generalizar la ley de la conservación de energía mecánica (sección 1.8) que incluye el calor y la energía interna además del trabajo y las energías externa, potencial y cinética. De hecho, la generalización se puede extender a otras formas, tales como las energías superficial, eléctrica y magnética. La abrumadora evidencia de la validez de esta generalización ha permitido elevar su categoría al nivel de una ley de la naturaleza, conocida como la primera ley de la termodinámica. Un enunciado formal es: Aunque la energía adopta muchas formas, la cantidad total de energía es constante, y cuando la energía desaparece de una forma, aparecerá simultáneamente en otras formas. Al aplicar la primera ley a un proceso dado, la esfera de influencia del proceso se divide en dos partes: el sistema y sus alrededores. La región en la que ocurre el proceso está separada y se conoce como sistema; todo aquello con lo que el sistema interactúa son los alrededores. Un sistema puede ser de cualquier tamaño, y sus fronteras pueden ser reales o imaginarias, rígidas o flexibles. Con frecuencia, un sistema se compone de una sola sustancia; en otros casos puede ser complejo. En cualquier situación, las ecuaciones termodinámicas se escriben con referencia a un sistema bien definido. Enfocamos nuestra atención al proceso de interés particular, así como al equipo y material directamente involucrados en el proceso. De cualquier modo, la primera ley se aplica al sistema y a sus alrededores, y no sólo al sistema. Para cualquier proceso, la primera ley requiere: Δ(Energía del sistema) + Δ (Energía de los alrededores) = 0 (2.1) donde el operador diferencia “Δ” indica cambios finitos en las cantidades encerradas entre paréntesis. El sistema puede cambiar en su energía interna, en su energía potencial o cinética, así como en las energías potencial o cinética de sus partes finitas. En el contexto de la termodinámica, el calor y el trabajo representan energía en tránsito a través de las fronteras que separan al sistema de sus alrededores, que nunca se encuentra almacenada o contenida en el sistema. Por otra parte, la energía potencial, cinética e interna reside y se almacena en la materia. En la práctica, la ecuación (2.1) adopta formas especiales que son adecuadas para aplicaciones prácticas específicas. El desarrollo de estas formas y sus posteriores aplicaciones son el tema del resto de este capítulo. 02-SmithVanNess.indd 22 8/1/07 12:48:40 2.4. Balance de energía para sistemas cerrados 2.4 23 BALANCE DE ENERGÍA PARA SISTEMAS CERRADOS Si la frontera de un sistema impide la transferencia de materia entre éste y sus alrededores, se dice que es un sistema cerrado, y su masa es necesariamente constante. El desarrollo de los conceptos básicos en termodinámica se facilita con un examen cuidadoso de los sistemas cerrados, y por esta razón serán tratados con todo detalle en las secciones siguientes de este capítulo. En la práctica industrial son más importantes los procesos en los que la materia cruza las fronteras del sistema como corrientes que entran y salen del equipo de proceso. A estos sistemas se les llama abiertos, y se abordarán en este capítulo una vez presentado el material necesario. Dado que en un sistema cerrado no entran ni salen corrientes, no se transporta energía asociada con la materia a través de la frontera que divide el sistema de sus alrededores. En ese caso todo intercambio de energía entre un sistema cerrado y sus alrededores aparecerá como calor y trabajo, y el cambio de energía total de los alrededores equivaldrá a la energía neta transferida hacia o desde ellos como calor o trabajo. Por lo tanto el segundo término de la ecuación (2.1) se puede sustituir por Δ(Energía de los alrededores) = ± Q ± W El calor Q y el trabajo W siempre se refieren al sistema, y la elección de los signos empleados con estas cantidades depende de la dirección de transferencia de energía con respecto al sistema, las cuales se consideran positivas. La convención moderna de los signos hace que los valores numéricos de ambas cantidades sean positivos para la transferencia al interior del sistema desde los alrededores. Las cantidades correspondientes tomadas con referencia a los alrededores, Qalrededores y Walrededores, tienen el signo opuesto, por ejemplo, Qalrededores = –Q y Walrededores = –W. Con esto entendemos: Δ(Energía de los alrededores) = Qalrededores + Walrededores = –Q –W (2.2) La ecuación (2.1) ahora será:2 Δ(Energía del sistema) = Q + W (2.3) Esta ecuación significa que el cambio en la energía total de un sistema cerrado es igual a la energía neta transferida como calor y trabajo hacia el sistema. Los sistemas cerrados a menudo se someten a procesos durante los cuales cambia únicamente la energía interna del sistema. Para tales procesos, la ecuación (2.2) se reduce a: ΔU t = Q + W (2.3) donde Ut es la energía interna total del sistema. La ecuación (2.3) se aplica a los procesos que involucran cambios finitos en la energía interna del sistema. Para cambios diferenciales: dU t = dQ + dW (2.4) 2 La convención del signo utilizado aquí es recomendada por la Unión Internacional de Química Pura y Aplicada. De cualquier modo, la elección original del signo para el trabajo y el que se usa en las primeras cuatro ediciones de este texto es la opuesta, y el lado derecho de la ecuación (2.2) se escribía entonces como Q – W. 02-SmithVanNess.indd 23 8/1/07 12:48:41 24 CAPÍTULO 2. La primera ley y otros conceptos básicos En las ecuaciones (2.3) y (2.4) los símbolos Q, W y U t están relacionados con el sistema entero, que puede ser de cualquier tamaño y debe estar definido con claridad. Todos los términos requieren expresiones en las mismas unidades de energía. En el sistema SI la unidad es el joule. Otras unidades en uso son la caloría, el (pie lbf) y la (Btu). El volumen total V t y la energía interna total U t dependen de la cantidad de material en un sistema, y se denominan propiedades extensivas. En contraste, la temperatura y la presión, las principales coordenadas termodinámicas para fluidos homogéneos puros, son independientes de la cantidad de materia, y se conocen como propiedades intensivas. Para un sistema homogéneo, un medio alternativo para expresar las propiedades extensivas, como V t y U t, es: V t = mV o V t = nV y U t = mU o U t = nU donde los símbolos simples V y U representan el volumen y la energía interna de una cantidad unitaria de materia, ya sea una unidad de masa o un mol. Éstas se denominan respectivamente propiedades específicas o molares y son intensivas, independientes de la cantidad de materia que en realidad está presente. Aunque V t y U t para un sistema homogéneo de tamaño arbitrario son propiedades extensivas, el volumen específico y molar V (o densidad) y la energía interna específica y molar U son intensivas. Observe que las coordenadas intensivas T y P carecen de contrapartes extensivas. Para un sistema cerrado de n moles, las ecuaciones (2.3) y (2.4) se escriben ahora como: Δ(nU) = n ΔU = Q + W (2.5) d(nU) = n dU = dQ + dW (2.6) En esta forma, dichas ecuaciones muestran de manera explícita la cantidad de sustancia contenida en el sistema. Las ecuaciones termodinámicas con frecuencia se escriben para una unidad representativa de la cantidad de materia, ya sea unidad de masa o mol. Así para n = 1, las ecuaciones (2.5) y (2.6) serán: ΔU = Q + W y dU = dQ + dW La base para Q y W se da a entender siempre por la masa o número de moles asociado con el lado izquierdo de la ecuación de energía. La ecuación (2.6) es la fuente fundamental de todas las relaciones de una propiedad que une a la energía interna con las cantidades mensurables. No representa una definición de la energía interna, ya que ésta no existe. Tampoco conduce a los valores absolutos para la energía interna. Lo que proporciona es la manera de calcular los cambios en esta propiedad; sin ésta, la primera ley de la termodinámica no se podría formular. De hecho, esa ley requiere de una afirmación previa de la existencia de la energía interna, la naturaleza esencial que se resume en el siguiente axioma: Existe una forma de energía, conocida como energía interna U, que es una propiedad intrínseca del sistema y que se relaciona mediante una función con las coordenadas mensurables que caracterizan a dicho sistema. Para sistemas cerrados, que no están en movimiento, los cambios en esta propiedad se dan por las ecuaciones (2.5) y (2.6). 02-SmithVanNess.indd 24 8/1/07 12:48:42 25 2.4. Balance de energía para sistemas cerrados Ejemplo 2.1 Fluye agua en una cascada de 100 m de altura. Considérese a 1 kg de agua como el sistema y suponga que no hay un intercambio de energía con los alrededores. a) ¿Cuál es la energía potencial del agua en la parte superior de la cascada con respecto a la base de la misma? b) ¿Cuál es la energía cinética del agua justo antes de llegar al fondo? c) Después de que el kg de agua entra al río que corre bajo la cascada, ¿qué cambio ocurre en su estado? Solución 2.1 El kg de agua no intercambia energía con los alrededores. Por lo tanto, para cada parte del proceso la ecuación (2.1) se reduce a: Δ(Energía del sistema) = ΔU + ΔE K + ΔE P = 0 a) De la ecuación (1.7), con g igual a su valor estándar, E P = mzg = 1 kg × 100 m × 9.8066 m s –2 kg m 2 = 980.66 = 980.66 N m = 980.66 J s2 b) Durante la caída libre del agua no existe un mecanismo para convertir energía potencial o cinética en energía interna. Así, ΔU debe ser cero: ΔE K + ΔE P = E K 2 – E K 1 + E P 2 – E P 1 = 0 Como una excelente aproximación, sea EK1 = EP2 = 0. Entonces, E K 2 = E P 1 = 980.66 J c) Conforme el kg de agua choca con el fondo y se mezcla con el agua que forma parte del río, la turbulencia resultante tiene el efecto de convertir la energía cinética en energía interna. Durante este proceso, ΔEP es esencialmente cero, y la ecuación (2.1) será: ΔU + ΔE K = 0 o ΔU = E K 2 – E K 3 No obstante, suponiendo que la velocidad del río es baja, podemos despreciar a EK3. De este modo, ΔU = E K 2 = 980.66 J El resultado global del proceso es la conversión de la energía potencial del agua en energía interna de la misma. Este cambio en la energía interna se manifiesta por un aumento de la temperatura del agua. Ya que se requiere una cantidad de energía de 4 184 J kg–1 para aumentar en 1 °C la temperatura del agua, el incremento en la temperatura es de 980.66/4 184 = 0.234 °C, suponiendo que no hay transferencia de calor con los alrededores. 02-SmithVanNess.indd 25 8/1/07 12:48:42 CHAPTER CHAPTER2. TheFirst FirstLaw Lawand andOther OtherBasic BasicConcepts Concepts CHAPTER 2.2.The The First Law and Other Basic Concepts 26 26 26 26 CAPÍTULO 2. La primera ley y otros conceptos básicos 2.5 2.5 THERMODYNAMIC THERMODYNAMICSTATE STATEAND ANDSTATE STATEFUNCTIONS FUNCTIONS 2.5 THERMODYNAMIC STATE AND STATE FUNCTIONS 2.5 ESTADO TERMODINÁMICO Y FUNCIONES DE ESTADO The Thenotation notationof Eqs.(2.3) (2.3)through through(2.6) (2.6)suggests suggeststhat thatthe theinternal-energy internal-energyterms termson onthe theleft left The notation ofofEqs. Eqs. (2.3) through (2.6) suggests that the internal-energy terms on the left are are different different in in kind kind from from the the quantities quantities on on the the right. right. Those Those on on the the left left reflect reflect changes changes in in the are different in kind from the quantities on the right. Those on the left reflect changes in the La notación de las ecuaciones (2.3) a la (2.6) sugiere que los términos de energía interna en el ladothe izquierdo thermodynamic state state of thesystem system as reflected byits thermodynamic properties, properties, amongwhich which thermodynamic state ofofthe the system asasreflected reflected by itsitsthermodynamic thermodynamic properties, among which son dethermodynamic una clase diferente de los de la derecha. Los by términos de la izquierda reflejan among cambios en el estado are aretemperature, temperature, pressure, pressure, anddensity. density. For Formediante aaahomogeneous homogeneous pure puresubstance substance we weknow knowfrom fromla temare temperature, pressure, and density. For homogeneous pure substance we know from termodinámico del sistema. Dichoand estado se refleja sus propiedades termodinámicas como experience experience that that fixing fixing two two of of these these properties properties also also fixes fixes all all the the others, others, and and thus thus determines determines experience that fixing two of these properties also fixes all the others, and thus determines its peratura, la presión y la densidad. Se sabe por experiencia que para una sustancia pura homogénea,its alitsfijar dos thermodynamic thermodynamic state. state. For For example, example, nitrogen nitrogen gas gas at at a a temperature temperature of of 300 300 K K and and a a pressure pressure thermodynamic state. For example, nitrogen gas at a temperature of 300 K and a pressure de sus propiedades termodinámicas, automáticamente se establecen las demás y así se determina su estado 55 5kPa of 10 kPa (1 bar) bar)has hasel aaanitrógeno fixed fixedspecific specific volume volume and andaaafixed fixed molar molar internal energy. energy.105 kPa ofof10 10 kPa (1(1 bar) has fixed specific volume orordensity density and fixed molar internal energy. termodinámico. Por ejemplo, en forma de or gas adensity una temperatura de 300 internal K y una presión Indeed, Indeed, it it has has a a complete complete set set of of intensive intensive thermodynamic thermodynamic properties. properties. If If this this gas gas is is heated heated or Indeed, it has a complete set of intensive thermodynamic properties. If this gas is heated ororconjun(1 bar) tiene volumen específico o densidad fijos y una energía interna molar fija. En efecto, tiene un cooled, cooled, compressed compressed or or expanded, expanded, and and then then returned returned to to its its initial initial temperature temperature and and pressure, pressure, its cooled, compressed or expanded, and then returned to its initial temperature and pressure, its to completo de propiedades termodinámicas intensivas. Si este gas se calienta o enfría, se comprimeits o expanintensiveproperties properties are are restored to their initialvalues. values. They They do do not notdepend dependon onthe thepast pastrecuperarán history history intensive properties are restored tototheir their initial values. They do not depend on the past history de, y aintensive continuación regresa a restored su temperatura yinitial presión iniciales, sus propiedades intensivas sus the thesubstance substance nor noron onthe themeans means by bywhich which reachesaaanterior agiven givenstate. state. They Theydepend depend onlymedios on on por ofof the substance nor on the means by which ititreaches reaches given state. They depend only on valoresof iniciales. Tales propiedades no dependen de itla historia de la sustancia ni deonly los present present conditions, conditions, however however reached. reached. Such Such quantities are areknown known as state functions. functions. When When two two present conditions, however reached. Such quantities are known asasstate state functions. When two los cuales alcanzan un estado determinado, sólo dequantities las condiciones actuales sin importar cómo éstas se hayan 33 3the of of them them are are held held at at fixed fixed values values for for a a homogeneous homogeneous pure pure substance, substance, the thermodynamic thermodynamic state state of them are held at fixed values for a homogeneous pure substance, the thermodynamic state alcanzado. Dichas cantidades son conocidas como funciones de estado. Cuando dos de ellas se mantienen en 3 se determina the thesubstance substance is fullydetermined. determined. This Thismeans means that thataaastate state function, function,such such as specific internal internal de la ofof the substance isisfully fully determined. This means that state function, such asasspecific specific internal valoresof fijos para una sustancia pura homogénea, totalmente el estado termodinámico energy, energy, is is a a property property that that always always has has a a value; value; it it may may therefore therefore be be expressed expressed mathematically mathematically as energy, is a property that always has a value; it may therefore be expressed mathematically as sustancia. Esto significa que una función de estado, tal como la energía interna específica, es una as propiedad a a function function of of such such coordinates coordinates as as temperature temperature and and pressure, pressure, or or temperature temperature and and density, density, and and its its a function of such coordinates as temperature and pressure, or temperature and density, and its que siempre tiene un valor; por lo tanto, se puede expresar matemáticamente como una función de coordenavalues values may may be identified withpoints points on onaaagraph. graph.y densidad, mientras que sus valores se pueden idenvalues may bebeidentified identified with points on graph. das tales como la temperatura ywith presión, o temperatura On On the the other other hand, hand, the the terms terms on on the the right rightsides sidesof Eqs.(2.3) (2.3)through through(2.6), (2.6),representing representing On the other hand, the terms on the right sides ofofEqs. Eqs. (2.3) through (2.6), representing tificar como puntos en una gráfica. heat heat and and work work quantities, quantities, are are not not properties; properties; they they account account for for the the energy energy changes changes that thatoccur occur heat and work quantities, are not properties; they account for the energy changes that occur Por otra parte, los términos en el lado derecho de las ecuaciones (2.3) a la (2.6) representan cantidades in in the the surroundings. surroundings. They They depend depend on on the the nature nature of of the the process, process, and and may may be be associated associated with with in the surroundings. They depend on the nature of the process, and may be associated with de calor y trabajo, que no son propiedades; consideran los cambios de energía que ocurren en los alrededores. areas areas rather than thanpoints points onaaagraph, graph, as suggested by byFig. Fig.1.3. 1.3.Although Although time isnot not aaathermothermoareas rather than points on graph, asassuggested suggested by Fig. 1.3. Although time is not thermoDependen derather la naturaleza del on proceso y pueden estar asociados con áreas antestime queis con puntos en la gráfica dynamic dynamic coordinate, coordinate, the the passage passage of of time time is is inevitable inevitable whenever whenever heat heat is is transferred transferred or or work workis is tiemdynamic coordinate, the passage of time is inevitable whenever heat is transferred or work isdel como lo sugiere la figura 1.3. Aunque el tiempo no es una coordenada termodinámica, el transcurso accomplished. accomplished. accomplished. po es inevitable cada vez que se transfiere calor o se realiza un trabajo. The Thedifferential differential of state function represents represents an infinitesimal infinitesimal change change in its value. value. InteInteThe differential ofofaaastate state function represents anan infinitesimal change ininsu itsits value. InteLa diferencial de una función defunction estado representa un cambio infinitesimal en valor. La integración gration gration of of such such a a differential differential results results in in a a finite finite difference difference between between two two of of its its values, values, e.g.: e.g.: gration of such a differential results in a finite difference between two of its values, e.g.: de esta diferencial da como resultado una diferencia finita entre dos de sus valores, por ejemplo: ���PP2P2 ���VV2V2 22 22 dddPPP= − − P P = = �P �P and and dddVVV= �V y ==PP2P − P = �P and ==VV2V2 2− −−VV1V1 1= ==�V �V 2 1 1 2 1 PP11P11 VV11V11 The Thedifferentials differentials of heatand andno work work arenot notchanges, changes, but butare areinfinitesimal infinitesimal amounts. amounts. When When inteinte- estas Las diferenciales de calor trabajo sonare cambios sino cantidades infinitesimales. Cuando se integran, The differentials ofofyheat heat and work are not changes, but are infinitesimal amounts. When integrated, grated, these these differentials differentials give give not notfinite finite changes,but but finite finiteamounts. amounts.Thus, Thus, diferenciales no presentan cambios finitos, sinochanges, cantidades finitas. Así, grated, these differentials give not finite changes, but finite amounts. Thus, ��� ��� dddQ and and ddW dWW= y QQ= ==Q QQ and ==W WW For For aaaclosed closed system system undergoing undergoing the thesame same change change state by byseveral severalde processes, processes, experiexperiPara un sistema cerrado sometido al mismo cambio de in estado por medio diferentes procesos, los For closed system undergoing the same change ininstate state by several processes, experiment ment shows shows that that the the amounts amounts of of heat heat and and work work required required differ differ for for different different processes, processes, but but that experimentos muestran queamounts las cantidades calor y trabajo difieren para los distintos procesos, ment shows that the of heatde and work requiredrequeridas differ for different processes, but that that the the sum sum Q Q + + W W is is the the same same for for all all processes. processes. This This is is the the basis basis for for identification identification of of internal internal enen- de la pero que suma QW +W es la misma para todo proceso. es el fundamento para of la internal identificación thelasum Q+ is the same for all processes. This isÉste the basis for identification entt is t isgiven t by ergy as state statefunction. function. The The same sameEl value value of �U given Eq. Eq. (2.3) (2.3) regardless regardless of the process, process, energíaergy interna función de estado. mismo valor ΔUby está dado por la ecuación (2.3) independienergy asasaaacomo state function. The same value ofof�U �U isde given by Eq. (2.3) regardless ofofthe the process, provided provided only only that that the the change change in in the the system system is is between between the the same same initial initial and and final final states. states. only that the change in the system is between theentre samelos initial and final states. tementeprovided del proceso, siempre que el cambio en el sistema ocurra mismos estados inicial y final. 33For 3 For systems systems more more complex complex than than aa simple asimple simple homogeneous homogeneous pure pure substance, substance, the the number number of state state functions functions that that must must be For systems more complex than homogeneous pure substance, the number ofof state functions that must bebe arbitrarily arbitrarilyspecified specifiedin orderto definethe thestate stateof thesystem systemmay maybe differentfrom fromtwo. two.The Themethod methodof determining arbitrarily specified ininorder order totodefine define the state ofofthe the system may bebedifferent different from two. The method ofofdetermining determining this this number isisis the the subject subject of Sec. Sec. 2.7.simple sustancia pura homogénea, el número de funciones de estado que deben especifithis number the subject ofof Sec. 2.7. Para losnumber sistemas más complejos que2.7. una carse en forma arbitraria con la finalidad de definir el estado del sistema puede ser distinto de dos. El método para determinar este número es el tema de la sección 2.7. 3 02-SmithVanNess.indd 26 8/1/07 12:48:44 2.5. Thermodynamic State and State Functions 27 2.5. Estado termodinámico y funciones de estado Example 2.2 27 A gas is confined in a cylinder by a piston. The initial pressure of the gas is 7 bar, Ejemplo and the2.2 volume is 0.10 m3 . The piston is held in place by latches in the cylinder wall. apparatus is placed total vacuum. What is the change the Un gasThe estáwhole confinado en un cilindro porinuna pistón. La presión inicial del energy gas es de 7 bar, of y el volumen 3 apparatus if the restraining latches are removed so that the gas suddenly expands es 0.10 m . El pistón se mantiene en su lugar mediante seguros en las paredes del cilindro.toTodo el its initialenvolume, piston striking latches at the end the process? aparatodouble se encuentra un vacíothe total. ¿Cuál es el other cambio en la energía del of aparato si se eliminan los seguros restrictivos, permitiendo así que el gas se expanda de manera repentina al doble de su volumen original, y al final 2.2 del proceso el pistón choca con otros seguros? Solution SoluciónBecause 2.2 the question concerns the entire apparatus, the system is taken as the gas, piston, and cylinder. No work is done during the process, because no force Dado que la pregunta quemoves, ver conand todo aparato se tomaráthrough como sistema el gas,surel pistón y external to thetiene system noelheat is transferred the vacuum el cilindro. Durante el proceso no se realiza trabajo, ya que no se aplica una fuerza externa al rounding the apparatus. Hence Q and W are zero, and the total energy of the sistema,system ni se transfiere calor del vacío hacia los alrededores del aparato. Por lo tanto, Q does not change. Without further information we can say nothing abouty W son cero y lathe energía total del cambia. Sin mayor es posible nada más distribution ofsistema energy no among the parts of theinformación system. Thisnomay well bedecir differacerca de la distribución de la energía entre las partes del sistema. Esto puede ser diferente de la ent than the initial distribution. distribución inicial. Example 2.3 If the process Ejemplo 2.3 described in Ex. 2.2 is repeated, not in a vacuum but in air at atmo- spheric pressure of 101.3 kPa, what is the energy change of the apparatus? Assume Si se repite el proceso en elbetween ejemplo 2.2, en el vacío el aire a unaair presión atmosféthe rate of heatdescrito exchange the no apparatus andsino theensurrounding is slow rica de compared 101.3 kPa,with ¿cuál esrate el cambio enthe la energía aparato? Suponga que la rapidez de intercamthe at which processdel occurs. bio de calor entre el aparato y el aire de los alrededores es lenta en comparación con la rapidez con la que ocurre el proceso. Solution 2.3 SoluciónThe 2.3system is chosen as before, but here work is done by the system in pushing the atmosphere. It is evaluated product of the force ofsistema atmospheric Se eligeback el mismo sistema que antes, pero ahoraaselthe trabajo es realizado por el al empujar el pressure on the back side of the piston F = P A and the displacement of the por la atm pistón de regreso a la atmósfera. Este trabajo se evalúa como el producto de la fuerza ejercida t /A. Here, A is the area of the piston and �V t is the volume �l =en�V presión piston atmosférica el lado opuesto del pistón F = Patm A y el desplazamiento del mismo, Δ l = gas.del This is work the system on thedelsurroundings, is a realizaΔV t / A. change Aquí, A of es the el área pistón y ΔV tdone es el by cambio de volumen gas. Esto es eland trabajo negative quantity; thus, do por el sistema sobre los alrededores, y es una cantidad negativa; en estos términos, kN W = −F �l = −Patm �V t = −(101.3)(0.2 − 0.1) kPa m3 = −10.13 2 m3 m W = −10.13 kN m = −10.13 kJ W = –10.13 kN m = –10.13 kJ Heat transfer between the system and surroundings is also possible in this case, En este but casothe también esisposible transferencia de calor entre el has sistema y susand alrededores, problem workedlafor the instant after the process occurred before pero el problema está planeado para elhas instante después que elThus proceso ocurrido y antes appreciable heat transfer had time to takedeplace. Q is ha assumed to be zero de que se tengainelEq. tiempo (2.2),suficiente giving para que se presente una transferencia de calor apreciable. Así, suponiendo que Q es cero en la ecuación (2.2), obtenemos �(Energy of the system) = Q + W = 0 − 10.13 = −10.13 kJ Δ(Energía del sistema) = Q + W = 0 – 10.13 = –10.13 kJ The total energy of the system has decreased by an amount equal to the work done on the surroundings. La energía total del sistema ha disminuido en una cantidad igual al trabajo realizado sobre los alrededores. o 02-SmithVanNess.indd 27 or 8/1/07 12:48:46 28 CAPÍTULO 2. La primera ley y otros conceptos básicos Ejemplo 2.4 En la figura 2.1, cuando se lleva un sistema del estado a al b a lo largo de la trayectoria acb, fluyen 100 J de calor hacia el sistema y éste realiza 40 J de trabajo. a) ¿Cuánto calor fluye hacia el sistema a lo largo de la trayectoria aeb, si el trabajo hecho por el sistema es de 20 J? b) El sistema regresa de b a a siguiendo la trayectoria bda. Si el trabajo hecho sobre el sistema es de 30 J, ¿el sistema absorbe o libera calor? ¿Cuánto? b c d P Figura 2.1: Diagrama para el ejemplo 2.4. e a V Solución 2.4 Supóngase que el sistema se modifica sólo en su energía interna y que se puede aplicar la ecuación (2.3). Para la trayectoria acb, y desde luego para cualquier trayectoria que conduzca de a hacia b, t = Q ΔUba acb + Wacb = 100 – 40 = 60 J a) Para la trayectoria aeb, t = 60 = Q ΔUba aeb + Waeb = Q aeb – 20 de donde Q aeb = 80 J b) Para la trayectoria bda, t = – ΔU t = – 60 = Q ΔUba bda + W bda = Q bda + 30 ba y Q bda = –60 – 30 = –90 J En consecuencia, se transfiere calor desde el sistema hacia los alrededores. 02-SmithVanNess.indd 28 8/1/07 12:48:47 29 2.6. Equilibrio 2.6 EQUILIBRIO Equilibrio es una palabra que denota una condición estática, es decir, la ausencia de un cambio. En termodinámica significa no sólo la ausencia de un cambio sino de cualquier tendencia hacia el cambio en una escala macroscópica. De esta manera existe un sistema en equilibrio bajo la condición de que no puede ocurrir en él ningún cambio de estado. Puesto que cualquier tendencia hacia el cambio es causada por una fuerza impulsora de uno u otro tipo, la ausencia de estas tendencias también indica la carencia de cualquier fuerza impulsora. Por tanto, en un sistema en equilibrio todas las fuerzas están en un balance exacto. Si en realidad ocurre un cambio en un sistema que no está en equilibrio, éste depende tanto de la resistencia como de la fuerza impulsora. Muchos sistemas experimentan cambios no mensurables aun bajo la influencia de grandes fuerzas impulsoras, porque su resistencia al cambio es muy grande. Las diferentes clases de fuerzas impulsoras tienden a producir distintos tipos de cambios. Por ejemplo, las fuerzas mecánicas no equilibradas como la presión en un pistón llegan a ocasionar transferencia de energía en forma de trabajo; las diferencias de temperatura suelen producir flujo de calor; los gradientes en el potencial químico tienden a originar que las sustancias sean transferidas de una fase a otra. En equilibrio, tales fuerzas están balanceadas. En muchas aplicaciones de la termodinámica no interesan las reacciones químicas. Por ejemplo, una mezcla de hidrógeno y oxígeno en condiciones ordinarias no está en equilibrio químico, debido a la gran fuerza impulsora para la formación de agua. Sin embargo, si la reacción química no se ha iniciado, el sistema puede estar en equilibrio térmico y mecánico a largo plazo, y sólo se analizarán los procesos puramente físicos sin considerar las posibles reacciones químicas. 2.7 REGLA DE LAS FASES Como se ha indicado con anterioridad, el estado de un fluido homogéneo puro se determina cada vez que se dan valores definidos a dos propiedades termodinámicas intensivas. En contraste, cuando dos fases están en equilibrio, el estado del sistema se establece al especificar sólo una propiedad. Por ejemplo, una mezcla de vapor y agua líquida en equilibrio a 101.33 kPa sólo puede existir a 100 °C. Es imposible cambiar la temperatura sin modificar también la presión, si se quiere que el vapor y el agua líquida continúen existiendo en equilibrio. Para sistemas de fases múltiples en equilibrio, el número de variables independientes que deben fijarse en forma arbitraria a fin de establecer su estado intensivo se proporciona por la célebre regla de las fases de J. Willard Gibbs,4 quien la dedujo por medio de un razonamiento teórico en 1875. Aquí se presenta sin comprobación, en la forma que se aplica para sistemas que no tienen reacciones químicas:5 F=2–p +N (2.7) donde p es el número de fases, N es el número de especies químicas y F representa los grados de libertad del sistema. 4 Josiah Willard Gibbs (1839-1903), físico matemático estadounidense. La justificación de la regla de las fases para un sistema que no contiene reacciones químicas está dada en la sección 10.2, mientras que la regla de las fases para sistemas con reacciones químicas se considera en la sección 13.8. 5 02-SmithVanNess.indd 29 8/1/07 12:48:48 30 CAPÍTULO 2. La primera ley y otros conceptos básicos El estado intensivo de un sistema en equilibrio se establece cuando su temperatura, presión y composiciones de todas las fases se encuentran fijas. Debido a eso, éstas son variables de la regla de las fases, pero no todas son independientes. La regla de las fases proporciona el número de variables de este conjunto, las cuales deben especificarse de manera arbitraria para fijar todas las demás variables restantes de la regla de las fases, y con ello el estado intensivo del sistema. Una fase es una región homogénea de materia. Un gas o mezcla de gases, un líquido o una solución líquida y un sólido cristalino son ejemplos de fases. Una fase no necesita ser continua; son ejemplos de fases discontinuas un gas disperso como burbujas en un líquido, un líquido disperso como gotas en otro líquido con el cual no es miscible, y cristales de un sólido dispersos ya sea en un gas o en un líquido. En cada caso, una fase dispersa está distribuida en todas las partes de una fase continua. Siempre ocurre un cambio repentino de las propiedades en las fronteras entre las fases. Pueden coexistir varias fases, pero deben estar en equilibrio para aplicar la regla de las fases. Un ejemplo de un sistema de tres fases en equilibrio es una solución salina acuosa que está saturada en su punto de ebullición con un exceso de cristales de sal. Las tres fases (π = 3) son la sal cristalina, la solución acuosa saturada y el vapor generado en el punto de ebullición. Las dos especies químicas (N = 2) son agua y sal. Para este sistema, F = 1. Las variables de la regla de las fases son propiedades intensivas, las cuales son independientes de la extensión del sistema y de las fases individuales. De este modo, la regla de las fases proporciona la misma información tanto para sistemas grandes como para sistemas pequeños y para cantidades diferentes de fases presentes. Además, sólo las composiciones de fases individuales son variables de las reglas de las fases. Las composiciones totales o globales no son variables de la regla de las fases cuando ésta presenta más de una fase. El número mínimo de grados de libertad para cualquier sistema es cero. Cuando F = 0 el sistema es invariante; la ecuación (2.7) se transforma en p = 2 + N. Este valor de p es el número máximo de fases que pueden coexistir en equilibrio para un sistema que contiene N especies químicas. Cuando N = 1, este número es 3, característico de un punto triple (véase la sección 3.1). Por ejemplo, el punto triple del agua, donde el líquido, el vapor y la forma común de hielo existen juntos en equilibrio, se presenta a 0.01 °C y 0.0061 bar. Cualquier cambio de estas condiciones provoca la desaparición de al menos una de estas fases. Ejemplo 2.5 ¿Cuántos grados de libertad tienen cada uno de los siguientes sistemas? a) Agua líquida en equilibrio con su vapor. b) Agua líquida en equilibrio con una mezcla de vapor de agua y nitrógeno. c) Una solución líquida de alcohol en agua en equilibrio con su vapor. Solución 2.5 a) El sistema contiene una sola especie química que existe en dos fases (una líquida y otra gaseosa). Así, F = 2 – π+ N = 2 – 2 + 1 = 1 02-SmithVanNess.indd 30 8/1/07 12:48:48 31 2.8. El proceso reversible Este resultado está de acuerdo con el hecho de que para una presión de agua dada sólo se tiene un punto de ebullición. Es posible especificar la temperatura o la presión, no ambas, para un sistema que contiene agua en equilibrio con su vapor. b) En este caso están presentes dos especies químicas. Hay otra vez dos fases. Ahora, F = 2 – π+ N = 2 – 2 + 2 = 2 La adición de un gas inerte a un sistema de agua en equilibrio con su vapor cambia las características del sistema. Ahora la temperatura y la presión pueden variar en forma independiente, pero una vez que se fijan el sistema descrito puede existir en equilibrio sólo para una composición particular de la fase vapor. (Si se considera que la solubilidad del nitrógeno en agua es despreciable, la fase líquida es agua pura.) c) Aquí N = 2, y π = 2, y F = 2 – π+ N = 2 – 2 + 2 = 2 Las variables de la regla de las fases son la temperatura, la presión y las composiciones de la fase. Las variables de la composición son las fracciones de masa o de moles de las especies en una fase, y deben sumar la unidad para cada fase. De esa forma, si se fija la fracción molar de agua en la fase líquida, la fracción molar de alcohol se fija automáticamente. Estas dos composiciones no se especifican en forma arbitraria. 2.8 EL PROCESO REVERSIBLE El desarrollo de la termodinámica se facilita por la introducción de una clase especial de proceso en sistemas cerrados que se caracteriza como reversible: Un proceso es reversible cuando la dirección puede ser invertida en cualquier punto por un cambio infinitesimal en las condiciones externas. Expansión reversible de un gas La naturaleza de los procesos reversibles se ilustra con el ejemplo de una expansión simple de gas en una combinación pistón/cilindro. El aparato que se muestra en la figura 2.2 se supone que está en un espacio vacío. Se elige como sistema el gas retenido en el interior del cilindro; todo lo demás son los alrededores. Los procesos de expansión ocurren cuando se retira masa del pistón. Con la finalidad de simplificarlo, se supone que el pistón se desliza dentro del cilindro sin presentar fricción, y que ni el pistón ni el cilindro absorben o transmiten calor. Además, ya que la densidad del gas en el cilindro es baja y que la masa del gas es pequeña, se ignoran los efectos de la gravedad sobre el contenido del cilindro. Esto significa que los gradientes de presión inducidos por la gravedad en el gas son muy pequeños en comparación con su presión, y que los cambios en la energía potencial del gas son despreciables en comparación con los cambios de la energía potencial del montaje del pistón. 02-SmithVanNess.indd 31 8/1/07 12:48:48 32 CAPÍTULO 2. La primera ley y otros conceptos básicos m Figura 2.2: Expansión de un gas. Δl El pistón de la figura 2.2 confina el gas a una presión suficiente para equilibrar el peso del pistón y todo lo que éste soporta. Tal condición de equilibrio para el sistema no debe tender al cambio. Se debe eliminar masa del pistón para que se eleve. Primero suponga que una masa m se desliza repentinamente del pistón a una repisa (al mismo nivel). El pistón se acelera hacia arriba y alcanza su velocidad máxima en el punto en que la fuerza ascendente sobre el pistón se equilibra con su peso. A continuación el momentum del pistón lo lleva a su nivel máximo donde invierte la dirección. Si el pistón se mantuviera en esta posición de elevación máxima, su energía potencial aumentaría a un valor muy cercano al trabajo realizado por el gas durante el recorrido inicial. Sin embargo, cuando no existe restricción, el pistón oscila con la disminución de la amplitud, hasta alcanzar finalmente una nueva posición de equilibrio a un nivel arriba de su posición inicial. Las oscilaciones del pistón se amortiguan, ya que la naturaleza viscosa del gas de manera gradual convierte el movimiento ordenado global de las moléculas en un movimiento molecular caótico. Este proceso disipativo transforma parte del trabajo hecho inicialmente por el gas para elevar el pistón, de nuevo en energía interna del gas. Una vez que se inicia el proceso, ningún cambio infinitesimal en las condiciones externas puede invertir su dirección; el proceso es irreversible. Todos los procesos que se llevan a cabo en un tiempo finito con sustancias reales se acompañan en cierto grado por efectos disipativos de una u otra clase y, por lo tanto, todos son irreversibles. No obstante, se pueden imaginar procesos que están libres de efectos disipativos. Para el proceso de expansión de la figura 2.2, tales efectos tienen su origen en el retiro repentino de una masa finita del pistón. El desequilibrio resultante de fuerzas que actúan en el pistón causa su aceleración y conduce a su subsecuente oscilación. El retiro repentino de pequeñas cantidades de masa reduce pero no elimina este efecto disipativo. Incluso el retiro de una masa infinitesimal lleva a que el pistón oscile con una amplitud infinitesimal y, por lo tanto, a un efecto disipativo. Sin embargo, es posible imaginar un proceso en donde se retiran pequeñas cantidades de masa una tras otra con una rapidez tal que la elevación del pistón es continua, con pequeñas oscilaciones sólo al final del proceso. 02-SmithVanNess.indd 32 8/1/07 12:48:49 2.8. El proceso reversible 33 El caso límite del retiro de una sucesión de masas infinitesimales del pistón se aproxima cuando las masas m de la figura 2.2 son sustituidas por una pila de polvo, soplando en una línea de corriente muy fina desde el pistón. Durante este proceso, el pistón se eleva con una rapidez uniforme pero muy lenta, y el polvo se almacena siempre a niveles más altos. El sistema nunca se desplaza más que de manera diferencial, ya sea desde su equilibrio interno o del equilibrio con sus alrededores. Si se detiene el traslado de polvo desde el pistón y se invierte la dirección de la transferencia del polvo, el proceso invierte su dirección y continúa a la inversa a lo largo de su trayectoria original. Al final, tanto el sistema como sus alrededores regresan virtualmente a sus condiciones iniciales. El proceso original se aproxima a la reversibilidad. Sin la suposición de un pistón sin fricción no podemos suponer un proceso reversible. Si el pistón se pega debido a la fricción, se debe eliminar una masa finita antes de que se libere el pistón. De esta forma, no se mantiene la condición de equilibrio que es necesaria para la reversibilidad. Por otro lado, la fricción entre las dos partes deslizantes es un mecanismo para la disipación de la energía mecánica en energía interna. Este análisis se ha centrado en un simple proceso de sistema cerrado, la expansión de un gas en un cilindro. El proceso opuesto, la compresión de un gas en un cilindro, se describe de la misma manera. De cualquier modo, hay muchos procesos que son manejados por el desequilibrio de fuerzas distintas de las mecánicas. Por ejemplo, ocurre flujo de calor cuando existe una diferencia de temperatura, la electricidad fluye bajo la influencia de una fuerza electromotriz, y las reacciones químicas suceden porque existe un potencial químico. En general, un proceso es reversible cuando la fuerza impulsora neta sólo es de tamaño diferencial. Así, el calor se transfiere en forma reversible cuando fluye desde un objeto finito a temperatura T hacia otro objeto a la temperatura T – dT. Reacción química reversible El concepto de una reacción química reversible se ilustra con la descomposición del carbonato de calcio que, al ser calentado, forma óxido de calcio y dióxido de carbono gaseoso. En equilibrio, para una temperatura dada el sistema ejerce una presión de descomposición específica de CO2. Cuando la presión cae por abajo de este valor, se descompone el CaCO3. Supongamos que a un cilindro se le adapta un pistón sin fricción que contiene CaCO3, CaO y CO2 en equilibrio. Se le sumerge en un baño a temperatura constante, como se muestra en la figura 2.3, y el equilibrio térmico asegura la igualdad de la temperatura del sistema con la del baño. La temperatura se ajusta a un valor tal que la presión de descomposición es suficiente para equilibrar el peso sobre el pistón, una condición de equilibrio mecánico. La reacción química se mantiene balanceada (en equilibrio) por la presión del CO2. Cualquier cambio de las condiciones, incluso de manera ligera, altera el equilibrio y provoca que la reacción avance en una u otra dirección. Si el peso se aumenta en forma diferencial, la presión del CO2 se incrementa de manera diferencial, y el CO2 se combina con el CaO para formar CaCO3, permitiendo que la pesa baje lentamente. El calor generado por esta reacción incrementa la temperatura en el cilindro, y el calor fluye hacia el baño. La disminución diferencial del peso sobre el pistón provoca una cadena opuesta de eventos. Se obtienen los mismos resultados si la temperatura del baño aumenta o disminuye. Si la temperatura del baño aumenta en forma diferencial, fluye calor hacia el interior del cilindro y se descompone el carbonato de calcio. El CO2 generado ocasiona que la presión aumente en forma diferencial, lo cual origina que se eleven el pistón y el peso. Esto continúa hasta que se descompone totalmente el CaCO3. El proceso es reversible, ya que el sistema nunca se desplaza del equilibrio más que de forma diferencial, y sólo una disminución diferencial de la temperatura del baño hace que el sistema regrese a su estado inicial. 02-SmithVanNess.indd 33 8/1/07 12:48:50 34 34 CAPÍTULO LaLaw primera ley y otros CHAPTER 2. The 2. First and Other Basicconceptos Concepts básicos w Figura 2.3: Reversibilidad de una reacción química. T T CO2 w CO2 Figure 2.3: Reversibility of a chemical reaction. CaCO3 �CaCO CaO 3 CaO Termostato Thermostat Some chemical reactions can be carried out in an electrolytic cell, and in this case they Algunas químicas seapplied realizanpotential en una celda electrolítica, y en esteconsists caso pueden mantenerse en may bereacciones held in balance by an difference. If such a cell of two elecequilibrio al aplicar una diferencia de potencial. Si esta celda se compone de dos electrodos, uno de zinc y otro trodes, one of zinc and the other of platinum, immersed in an aqueous solution of hydrochloric de platino, enthat unaoccurs solución acid,sumergidos the reaction is: acuosa de ácido clorhídrico, la reacción que ocurre es: Zn + 2HCl � H2 + ZnCl2 cell is heldbajo under fixed conditions temperature and pressure, and the electrodes are con- exterLa celdaThe se mantiene condiciones fijas deof temperatura y presión, y los electrodos están conectados externally to a potentiometer. If the electromotive force byequilibra the cell isenexactly namentenected a un potenciómetro. Si la fuerza electromotriz producida porproduced la celda se forma exacta balanced by the potential of thelapotentiometer, the reaction is held inEs equilibrium. por la diferencia de potencial del difference potenciómetro, reacción se mantiene en equilibrio. posible hacer que The proceda reaction en may be made to proceed in the forward by a slight decrease in the opla reacción sentido directo disminuyendo un pocodirection la diferencia de potencial opuesta, y se puede potential difference, and it may be by a corresponding in la thecelda. potential revertir posing mediante un aumento correspondiente enreversed la diferencia de potencial deincrease la fem de difference above the emf of the cell. Resumen de lasRemarks observaciones en los procesos Summary on Reversible Processesreversibles A reversible process: Un proceso reversible: Is frictionless. • Es sin• fricción. • se Is sale neverdel more than differentially removed from equilibrium. • Nunca equilibrio más que de una manera diferencial. • Recorre una sucesión de estados equilibrio.states. • Traverses a succession ofde equilibrium • Está controlado fuerzaswhose cuyo desequilibrio tiene magnitud diferencial. • Is drivenpor by forces imbalance is differential in magnitude. • Se puede invertir en cualquier mediante un cambio diferencial en conditions. las condiciones externas. • Can be reversed at any punto point by a differential change in external • Cuando se invierte, vuelve a trazar su trayectoria y restaura el estado inicial del sistema y de sus alre• When reversed, retraces its forward path, and restores the initial state of system and dedores. surroundings. Una ecuación deducida en lainsección 1.7gives proporciona el of trabajo de compresión o expansión de un gas An equation derived Sec. 1.7 the work compression or expansion of a gas que es causado porthe el differential desplazamiento diferencial un pistón un cilindro: caused by displacement of de a piston in a en cylinder: ddW W = Vt = –−P P ddV 02-SmithVanNess.indd 34 (1.2) (1.2) 8/1/07 12:48:52 2.8. 2.8. The The Reversible Reversible Process Process 2.8. El proceso reversible 35 35 35 The The work work done done on on the the system system is is given given by by this this equation equation only only when when certain certain characteristics characteristics of of El trabajo hecho sobre el sistema se da por esta ecuación sólo cuando se efectúan ciertas del the reversible process are realized. The first requirement is that the system more the reversible process are realized. The first requirement is that the system be be no no características more than than procesoinfinitesimally reversible. El primer requisito es que el sistema se desplace sólo en forma infinitesimal de un estado displaced from a state of internal equilibrium, characterized by uniformity of infinitesimally displaced from a state of internal equilibrium, characterized by uniformity of de equilibrio internoand caracterizado porsystem la uniformidad de lahas temperatura y la presión. El sistemaincludsiempre tiene temperature pressure. then an set temperature and pressure. The The system then always always has an identifiable identifiable set of of properties, properties, includun conjunto identificable de propiedades, incluyendo la presión P. El segundo requisito es que el sistema no ing pressure P. The second requirement is that the system be no more than infinitesimally ing pressure P. The second requirement is that the system be no more than infinitesimally se desplace más que en forma infinitesimal del equilibrio mecánico con sus alrededores. En este caso, la predisplaced from mechanical equilibrium with its surroundings. In this event, the internal displaced from mechanical equilibrium with its surroundings. In this event, the internal prespressión interna P nunca está más que un poco fuera de equilibrio con la fuerza externa, y se puede hacer sure P is never more than minutely out of balance with the external force, and we may make sure P is never more than minutely out of balance with the external force, and we may make la sustituciónthe F= PA, que transforma la ecuación (1.1) la (1.2). Los(1.2). procesos para los se reúnen substitution F transforms Eq. into Processes for which these the substitution F = = P PA A that that transforms Eq.en(1.1) (1.1) into Eq. Eq. (1.2). Processes forque which these estos requisitos se conocen como mecánicamente reversibles y se puede integrar la ecuación (1.2): requirements are met are said to be mechanically reversible, and Eq. (1.2) may be integrated: requirements are met are said to be mechanically reversible, and Eq. (1.2) may be integrated: �� V tt V22 t (1.3) W = − P dd V Vt (1.3) W =− t P (1.3) V V11t The reversible process is ideal in itit produces the possible result. It aa The reversible reversible es process in that thatde produces the best best possible result.posible. It represents represents El proceso ideal is enideal el sentido que produce el mejor resultado Representa un limit to the performance of actual processes, but is never fully realized. An initial calculation limit to the performance of actual processes, but is never fully realized. An initial calculation límite en el desempeño de los procesos reales, ya que nunca se puede realizar por completo. Con frecuencia of work is made for aa reversible process, because the such is often often made reversible the choice choice is islabetween between such aa calculation calculation se hace of unwork cálculo inicial del for trabajo para unprocess, procesobecause reversible, porque opción estriba entre realizar o no and no calculation at all. The reversible work as the limiting value may be combined with an and no calculation at all. The reversible work as the limiting value may be combined withapropiadas an estos cálculos. El trabajo reversible con el valor limitante puede combinarse con las eficiencias appropriate efficiency to yield aa reasonable approximation to the work of an actual process. appropriate efficiency to yield reasonable approximation to the work of an actual process. para producir aproximaciones razonables del trabajo de los procesos reales. Example 2.6 Ejemplo 2.6 piston/cylinder A A horizontal horizontal piston/cylinder arrangement arrangement is is placed placed in in aa constant-temperature constant-temperature bath. bath. The slides cylinder friction, and external holds itit pistón The piston pistonhorizontal slides in in the the cylinder with with negligible negligible friction, anda an an external force force holds El Una combinación de pistón/cilindro se coloca en un baño temperatura constante. 33. The in place against an initial gas pressure of 14 bar. The initial gas volume is 0.03 m in place against an initial gas pressure of 14 bar. The initial gas volume is 0.03 m . The se desliza en el cilindro con una fricción insignificante, y una fuerza externa lo mantiene en su lugar en force on is and gas expands external forceinicial on the the piston is reduced reduced gradually, and the the gas del expands isothermally contra external de la presión delpiston gas que es de 14gradually, bar. El volumen inicial gas esisothermally 0.03 m3. La fuerza as its volume doubles. If the volume of the gas is related to its pressure so as its volume doubles. If the volume of the gas is related to its pressure so that that the theal doble externa sobre el pistón se reduce de manera gradual y el gas se expande de forma isotérmica tt is constant, what is the work done by the gas in moving the external product PV product PV is constant, what is the work done by the gas in moving the external de su volumen. Si el volumen del gas está relacionado con su presión, de modo que el producto PV t es force? force? constante, ¿cuál es el trabajo hecho por el gas al cambiar la fuerza externa? How much would be the were suddenly How much work would be done done the external external force were repentinamente suddenly reduced reduced to ¿Cuánto trabajo sework hubiera realizado si laififfuerza externaforce se reduce a la to mitad de half its initial value instead of being gradually reduced? half its initial value instead of being gradually reduced? su valor inicial en vez de hacer la reducción de manera gradual? Solution 2.6 Solución 2.6 El proceso como se en un principio, es mecánicamente y se puede The process, out as is reversible, and Therealizado, process, carried carried outdescribió as first first described, described, is mechanically mechanically reversible,reversible and Eq. Eq. (1.3) (1.3) t = k, entonces P =tt k / V t y ttPV aplicar la ecuación (1.3). Si = k, then P = k/V , and is applicable. If P V is applicable. If P V = k, then P = k/V , and W W= =− − With With Con and and y 02-SmithVanNess.indd 35 �� t V V22t t V V11t d V tt P P dV = = −k −k �� t V V22t t V V11t t t dd V V22t V t = −k ln V = −k ln t V V V tt V1t 1 t t 3 3 V V V11t = = 0.03 0.03 m m3 V22t = = 0.06 0.06 m m3 t 3 t V 1 = 0.03 m V 2 = 0.06 m 3 t t 5 t t kk = =P PV V = =P P11V V11 = = (14 (14 × × 10 105)(0.03) )(0.03) = = 42,000 42,000 JJ t = (14 × 10 5 )(0.03) = 42 000 J k = PV t =WP 1=V −42,000 1 W = −42,000 ln ln 22 = = −29,112 −29,112 JJ W = –42 000 ln 2 = –29 112 J 8/1/07 12:48:53 36 CHAPTER 2. The First Law and Other Basic Concepts 36 CAPÍTULO 2. La primera ley y otros conceptos básicos La presiónThe finalfinal es pressure is k 42,000 = 700,000 Pa P2 = t = o or 7 bar7 bar 0.06 V2 En el segundo después haber reducido a la mitad el gas In thecaso, second case,de reduction of the initial forcelabyfuerza half isinicial, followed by experimenta sudden una súbitaexpansion expansiónofcontra unaagainst fuerzaaconstante que equivale a una de of 7 bar. Con el the gas constant force equivalent to apresión pressure 7 bar. tiempo, la Eventually, transferencia de transfer calor regresa al the sistema a una condición de equilibrio idéntica al estaheat returns system to the same final equilibrium state as t es el mismo que antes, pero el do final alcanzado en el proceso reversible. Det is esta themanera, same asΔV before, but the work accomin the reversible process. Thus �V trabajo realizado estágiven dado by porEq. la ecuación (1.3).the Enwork su lugar, trabajothe realizado contra de plishedno is not (1.3). Rather, doneelagainst externalenforce la fuerza externa es igual a la presión externa equivalente el cambio de volumen: equals the equivalent external pressure times thepor volume change: 5)(0.06 5 W ×× 1010 – 0.03) = –21 000 J J W ==–(7 −(7 )(0.06 − 0.03) = −21,000 Thisesprocess is clearly irreversible, and compared with thereversible reversibleseprocess is tiene Este proceso claramente irreversible, y comparado con el proceso dice que said de: to have an efficiency of: una eficiencia 0.721 2121,000/29,112 000/29 112 = = 0.721 o or 72.1% 72.1% Example 2.7 Ejemplo 2.7 The piston/cylinder arrangement shown in Fig. 2.4 contains nitrogen gas trapped be- La combinación que se muestra en laThe figura 2.4 contiene gas nitrógeno atrapado debajo low thepistón/cilindro piston at a pressure of 7 bar. piston is held in place by latches. The del pistónspace a una above presiónthe de piston 7 bar. El pistón se mantiene en su lugar mediante unos seguros. El espacio is evacuated. A pan is attached to the piston rod and a mass sobre el pistón es kg evacuado. Se une un plato vástago delpiston pistón,rod, y una masa de 45 kg have se sujeta m of 45 is fastened to the pan. alThe piston, and panmtogether a al plato. El mass pistón,ofel23 vástago y el plato juntos tienen una masa de 23 kg. Los seguros que sostienen kg. The latches holding the piston are released, allowing the piston to mm Masa Mass Plato Pan Evacuated Espacio space evacuado Figura 2.4: Diagrama para el ejemplo 2.7.2.7. Figure 2.4: Diagram for Ex. 0.5m m 0.5 Cylinder Cilindro Piston Pistón Latch Seguro Gas under Gas bajo pressure presión rise rapidly until it strikes the top of the cylinder. The distance moved by the piston is −2 . Discuss the energy changes that 0.5liberan m. Thepermitiendo local acceleration gravity 9.8 m shasta al pistón se que ésteofsuba conisrapidez golpear la parte superior del cilindro. of thisde process. El pistón occur recorrebecause una distancia 0.5 m. La aceleración local de la gravedad es 9.8 m s–2. Analice los cambios de energía que ocurren debido a este proceso. 02-SmithVanNess.indd 36 8/1/07 12:48:56 37 2.9. Procesos con V y P constantes Solución 2.7 Este ejemplo sirve para ilustrar algunas de las dificultades encontradas al analizar los procesos irreversibles donde no hay flujo. Considere el gas sólo como sistema. De acuerdo con la definición básica, el trabajo hecho por el gas en los alrededores es igual a ∫ P ′ dV ′, donde P ′ es la presión ejercida por el gas sobre la cara del pistón. Ya que la expansión es muy rápida, existen gradientes de la presión en el gas, y no es posible evaluar P ′ ni la integral. Sin embargo, al regresar a la ecuación (2.1) se evita el cálculo del trabajo. El cambio en la energía total del sistema (el t gas) es igual al cambio en la energía interna, ΔU sistema . Para Q = 0, los cambios de energía en los alrededores consisten en los cambios en la energía potencial del pistón, del vástago, del plato y de la masa m, así como de los cambios en la energía interna del pistón, del vástago y del cilindro. Por lo tanto, la ecuación (2.1) se escribe como: t t + (ΔU alrededores + ΔE P alrededores) = 0 ΔU sistema donde Por lo tanto ΔE P alrededores = (45 + 23)(9.8)(0.5) = 333.2 N m t t ΔU sistema + ΔU alrededores = –333.2 N m = –333.2 J Los valores para ΔU tsistema y ΔU talrededores no pueden ser determinados. 2.9 PROCESOS CON V y P CONSTANTES Para n moles de un fluido homogéneo contenido en un sistema cerrado el balance de energía es: d(nU) = dQ + dW (2.6) donde Q y W representan siempre el calor y el trabajo total para cualquier valor de n. El trabajo de un proceso mecánicamente reversible en un sistema cerrado está dado por la ecuación (1.2), que se escribe a continuación: dW = –P d(nV) estas dos ecuaciones se combinan: d(nU) = dQ – P d(nV) (2.8) Este balance de energía es general para n moles de fluido homogéneo en un sistema cerrado que experimenta un proceso mecánicamente reversible. Proceso a volumen constante Si el proceso ocurre a un volumen total constante, el trabajo es cero. Por otra parte, para sistemas cerrados el último término de la ecuación (2.8) también es cero, porque tanto n como V son constantes. De este modo, dQ = d(nU)(V constante) Integrando se obtiene: Q = n ΔU (V constante) (2.9) (2.10) De esta manera, el calor transferido en un proceso mecánicamente reversible para un sistema cerrado a volumen constante, es igual al cambio en la energía interna del sistema. 02-SmithVanNess.indd 37 8/1/07 12:48:56 38 CAPÍTULO 2. La primera ley y otros conceptos básicos Proceso a presión constante Resolviendo para dQ, de la ecuación (2.8) se obtiene que: dQ = d(nU) + P d(nV) Para un cambio de estado a presión constante: dQ = d(nU) + d(n PV) = d[n(U + PV)] La aparición del grupo U + PV, tanto aquí como en otras aplicaciones, sugiere la definición conveniente de una nueva propiedad termodinámica. De esta forma, la definición matemática (y única) de la entalpía6 es: H ≡ U + PV (2.11) donde H, U y V son valores molares o por unidad de masa. La ecuación anterior ahora se puede escribir como: La integración produce: dQ = d(nH) (P constante) (2.12) Q = n ΔH (P constante) (2.13) De este modo, el calor transferido en un proceso mecánicamente reversible de un sistema cerrado a presión constante, es igual al cambio en la entalpía del sistema. Al comparar las ecuaciones (2.12) y (2.13) con las ecuaciones (2.9) y (2.10) se muestra que en los procesos a presión constante la entalpía desempeña un papel semejante a la energía interna en los procesos a volumen constante. 2.10 ENTALPÍA La utilidad de la entalpía se sugiere por las ecuaciones (2.12) y (2.13). También aparece en los balances energéticos para los procesos con flujo en relación con los intercambiadores de calor, evaporadores, columnas de destilación, bombas, compresores, turbinas, máquinas, etc., para el cálculo del calor y del trabajo. La tabulación de los valores de Q y de W para la combinación infinita de los posibles procesos es imposible. Sin embargo, las funciones de estado intensivas, tales como el volumen, la energía interna y la entalpía específicos, son propiedades intrínsecas de la materia. Una vez que son determinadas para una sustancia particular, sus valores para fases líquida y vapor se pueden tabular como funciones de T y P para su uso futuro en el cálculo de Q y de W para cualquier proceso que involucre dicha sustancia. La determinación de los valores numéricos para estas funciones de estado, así como su correlación y uso se tratarán en los últimos capítulos. Todos los términos de la ecuación (2.11) deben expresarse en las mismas unidades. El producto PV tiene unidades de energía por mol o por unidad de masa, al igual que U; por lo tanto, H también tiene unidades de energía por mol o por unidad de masa. En el sistema SI la unidad básica de la presión es el pascal o el 6 Una palabra propuesta por el físico holandés H. Kamerlingh Onnes, quien fue el primero en licuar el helio en 1908. Descubrió la superconductividad en 1911 y ganó el premio Nobel de Física en 1913. (Véase: Comunications from the Physical Laboratory of the University of Leiden, núm. 109, p. 3, pie de página 2, 1909.) 02-SmithVanNess.indd 38 8/1/07 12:48:56 39 2.10. Entalpía N m–2 y, para el volumen molar, m3 mol–1. El producto PV tiene por consiguiente las unidades N m mol–1 o J mol–1. En el sistema inglés de ingeniería una unidad común para el producto PV es el (pie lbf)(lbm)–1, que surge cuando la presión se encuentra en (lbf)(pie)–2 y con el volumen en (pie)3(lbm)–1. Este resultado generalmente se convierte a (Btu)(lbm)–1 mediante la división entre 778.16 y haciendo uso de la ecuación (2.11), porque la unidad inglesa de ingeniería común para U y H es el (Btu)(lbm)–1. Dado que U, P y V son funciones de estado, la H definida por la ecuación (2.11) también es una función de estado. Como U y V, H es una propiedad intensiva del sistema. La forma diferencial de la ecuación (2.11) es: dH = dU + d(PV) (2.14) Esta ecuación se aplica siempre que ocurre un cambio diferencial en el sistema. Al efectuar la integración se convierte en una ecuación para un cambio finito en el sistema: ΔH = ΔU + Δ(PV) (2.15) Las ecuaciones (2.11), (2.14) y (2.15) se aplican a la unidad de masa de una sustancia o a un mol. Ejemplo 2.8 Calcule ΔU y ΔH para 1 kg de agua cuando se evapora a la temperatura constante de 100 °C y a una presión constante de 101.33 kPa. Los volúmenes específicos del agua líquida y de su vapor en estas condiciones son 0.00104 y 1.673 m3 kg–1. Para este cambio se agrega al agua una cantidad de calor de 2 256.9 kJ. Solución 2.8 Se toma 1 kg de agua como el sistema, ya que es lo que nos interesa. Imaginemos que se encuentra contenido en los cilindros mediante un pistón sin fricción que ejerce una presión constante de 101.33 kPa. A medida que se agrega calor, el agua se evapora y se expande desde su volumen inicial hasta su volumen final. La ecuación (2.13) escrita para el sistema de 1 kg se tiene a continuación: ΔH = Q = 2 256.9 kJ Por la ecuación (2.15), ΔU = ΔH – Δ(PV) = ΔH – P ΔV Evaluando el término final: P ΔV = 101.33 kPa × (1.673 – 0.001) m3 = 169.4 kPa m3 = 169.4 kN m–2 m3 = 169.4 kJ Entonces 02-SmithVanNess.indd 39 ΔU = 2 256.9 – 169.4 = 2 087.5 kJ 8/1/07 12:48:57 40 2.11 40 40 40 CAPÍTULO 2. La primera ley y otros conceptos básicos CHAPTER 2. The First Law and Other Basic Concepts CHAPTER CHAPTER 2. 2. The The First First Law Law and and Other Other Basic Basic Concepts Concepts CAPACIDAD CALORÍFICA 2.11 HEAT HEATCAPACITY CAPACITY 2.11 2.11 CAPACITY El punto de vistaHEAT moderno con respecto al calor como energía en tránsito fue precedido por la idea de que un cuerpo tiene una capacidad para el calor. Entre más pequeño fuera el cambio de temperatura en un cuerpo The modern view of heat as energy in transit was preceded by the idea that a body has a The modern view energy in was preceded the that provocado la transferencia deas una cantidad dada de calor, mayorby su capacidad. En has realidad, una The por modern view of of heat heat as energy in transit transit was preceded bysería the idea idea that aa body body has aa capacity for heat. The smaller the temperature change in a body caused by the transfer of a capacity for The smaller the in caused by transfer of capacidad calorífica puede definirse C ≡ dQ/dT. change La dificultad con esta expresión que hace capacity for heat. heat. The smallercomo the temperature temperature change in aa body body caused by the thees transfer of aque a C, al given quantity of heat, the greater its capacity. Indeed, a heat capacity might be defined as given quantity of the greater Indeed, aa heat capacity be defined as igual que Q, sean cantidades del proceso que de una estado. obstante, given quantity of heat, heat,más thedependientes greater its its capacity. capacity. Indeed, heatfunción capacitydemight might beNo defined as sugieC ≡ d Q/dT . The difficulty with this is that it makes C, like Q, a process-dependent quantity CC ≡ .. The difficulty with this isis that itit makes like aa process-dependent re la definición de dos cantidades este nombre hecho son funciones dequantity estado relacio≡ ddQ/dT Q/dT The difficultycon with this thatanticuado, makes C, C,que likedeQ, Q, process-dependent quantity rather than a state function. However, it does suggest the definition of two quantities with this rather than function. However, does nadas sin ambigüedad con otras funciones deititestado. rather than aa state state function. However, does suggest suggest the the definition definition of of two two quantities quantities with with this this outmoded name that are in fact state functions, unambiguously related to other state functions. outmoded name that are in fact state functions, unambiguously related to other state functions. outmoded name that are in fact state functions, unambiguously related to other state functions. Capacidad caloríficaataConstant volumen constante Heat Capacity Constant Volume Heat Heat Capacity Capacity at at Constant Volume Volume The constant-volume heat capacity of a substance is defined as: La capacidad calorífica a volumen constante una sustancia se define The heat of is defined as: The constant-volume constant-volume heat capacity capacity of ade a substance substance defined as: como: � is � �� ∂U�� ∂U (2.16) (2.16) C ≡ ∂U (2.16) CCVVV≡ ≡ ∂T V (2.16) ∂∂TT VV Esta definición se acomoda a ambas capacidades, tanto la capacidad calorífica como a la capacidad This definition accommodates both the molar heata capacity and the specificmolar heat capacity (usuThis definition accommodates both the heat and specific heat capacity (usuThis definition accommodates bothcalor themolar molar heatcapacity capacity andthe theen specific heatde capacity (usucalorífica específica (usualmente llamada específico), dependiendo todo caso que U sea la energía ally called specific heat), depending on whether U is the molar or specific internal energy. called specific heat), on U isis the or internal energy. ally called specific Aunque heat), depending depending on whether whether Ureferencia the molar molar or specific specific internal energy. de una internaally molar o específica. esta definición no hace a cualquier proceso, se relaciona Although this definition makes no reference to any process, it relates in an especially simple this makes no to process, relates in especially Although this definition definition makes no reference reference to any any process,deititun relates in an an especially simple simple maneraAlthough especialmente simple con un proceso volumen constante sistema cerrado, el que la ecuaway to a constant-volume process in aaclosed system, for which Eq. (2.16) may bepara written: way process way to to aescribe a constant-volume constant-volume process in in aa closed closed system, system, for for which which Eq. Eq. (2.16) (2.16) may may be be written: written: ción (2.16) se como: dU = C V dT (const V ) (2.17) dU CCVVdT dT (const (2.17) dU =C (const VV)) (2.17) (2.17) dU = = V dT (V constante) � �� T T2 Integration yields: �U = T22 C V dT (const V ) (2.18) Integration �U (const VV)) (2.18) Integrando: (V constante) Integration yields: yields: �U = = T1 CCVV dT dT (const (2.18) (2.18) TT11 La combinación dewith este Eq. resultado ecuación (2.10) para unconstant-volume proceso mecánicamente a voluThis result (2.10) con for alamechanically reversible, process777reversible, gives: This result with Eq. (2.10) for aa mechanically reversible, constant-volume process gives: This result with Eq. (2.10) for mechanically reversible, constant-volume process gives: 7 men constante, nos da: � �� T T2 Q = n �U = n T22 C V dT (const V ) (2.19) Q VV)) (2.19) constante) Q= = nn �U �U = = nn T1 CCVV dT dT (V(const (const (2.19) (2.19) TT11 If the volume varieselduring the pero process but returns the end ofvalor the process Si el volumen varía durante proceso, al final de éste at regresa a su inicial, to noits esinitial correcto que IfIf the the volume volume varies varies during during the the process process but but returns returns at at the the end end of of the the process process to to its its initial initial = V1 and en las value, the process cannot rightly be called one of constant volume, even though V = V y ΔV = 0. De cualquier modo, los el proceso se llame volumen constante, aunque V 2 cambios 2 1 value, = VV11 and and value, the the process process cannot cannot rightly rightly be be called called one one of of constant constant volume, volume, even even though though VV22 = �Vde = 0. However, changes indestate functions y, arepor independent of path, and may therefore funciones estado son independientes la trayectoria lo tanto, pueden calcularse mediante ecuacio�V �V = = 0. 0. However, However, changes changes in in state state functions functions are are independent independent of of path, path, and and may may therefore therefore be calculated by equations for a truly constant-volume process withinicial the same initial and final (2.18) nes para un proceso a volumen constante real con las mismas condiciones y final. La ecuación � be process be calculated calculated by by equations equations for for aa truly truly constant-volume constant-volume process with with the the same same initial initial and and final final �� C V dT conditions. (2.18) , because , C V , and T Por are otra all state da entonces ΔU = ∫ CEquation U,then CV gives ygives T son�U todas=funciones de estado oU, propiedades. parte, Q y V dT, porque conditions. (2.18) conditions. Equation Equation (2.18) then then gives �U �U = = CCVV dT dT,, because because U U , CCVV,, and and TT are are all all state state functions. On the other hand, Q and W depend on path, and Eq. para (2.19) isy aWvalid expression W dependen de la trayectoria, y la ecuación (2.19) es una expresión válida Q, en general es cero sólo functions. functions. On On the the other other hand, hand, Q Q and and W W depend depend on on path, path, and and Eq. Eq. (2.19) (2.19) isis aa valid valid expression expression for Q, and W is inconstante. general zero, only for a constant-volume process. This is the de reason for Q y W. para unfor proceso a volumen Ésta es la razón para la distinción entre las funciones estado, for Q, Q, and and W W isis in in general general zero, zero, only only for for aa constant-volume constant-volume process. process. This This isis the the reason reason for for the distinction functions and Q and W . del Theproceso principle thatconcepto state functions are y útil. El principio de que las between funcionesstate de estado son independientes es un importante the the distinction distinction between between state state functions functions and and Q Q and and W W.. The The principle principle that that state state functions functions are are independent of the process is an important and useful concept. independent of process isis an and independent of the the los process an important important and useful useful concept. concept. Para el cálculo de cambios en las propiedades, un proceso real se puede sustituir por For the calculation of property changes, an actual process may be otro cualquiera que consiga el mismo cambio en el estado. For For the the calculation calculation of of property property changes, changes, an an actual actual process process may may be be replaced by any other process which accomplishes the same change replaced by any other process which accomplishes the same change replaced by any other process which accomplishes the same change De este modo,in puede seleccionarse un proceso alternativo, por ejemplo, debido a su simplicidad. state. in in state. state. Such an alternative process may be selected, for example, because of its simplicity. Such Such an an alternative alternative process process may may be be selected, selected, for for example, example, because because of of its its simplicity. simplicity. 7 Estas restricciones sirven para excluir el trabajo de agitación, que es intrínsecamente irreversible. 7 These restrictions serve to rule out work of stirring, which is inherently irreversible. 77These Theserestrictions restrictionsserve serveto torule ruleout outwork workof ofstirring, stirring,which whichisisinherently inherentlyirreversible. irreversible. 02-SmithVanNess.indd 40 8/1/07 12:48:59 2.11. Heat Capacity 2.11. Capacidad 2.11. Heat calorífica Capacity 2.11. Heat Capacity 41 41 41 41 HeatCapacity Capacity ataConstant Constant Pressure Capacidad calorífica presión constante Heat at Pressure Heat Capacity at Constant Pressure The constant-pressure heat capacity se is defined as: La capacidad calorífica a presión define como: The constant-pressure heat constante capacity is defined as: The constant-pressure heat capacity is defined � as: � � ∂ H� (2.20) (2.20) C P ≡� ∂ H � (2.20) C P ≡ ∂ ∂HT P ∂ T P (2.20) CP ≡ ∂caloríficas T P De nuevo, la definición se acomoda a las capacidades molar y específica, dependiendo Again, the definition accommodates both molar and specific heat capacities, depending de on si H es Again, theodefinition accommodates both molarseand specific heat capacities, depending simple on la entalpía molar específica. Esta capacidad calorífica relaciona de una manera especialmente para whether Hdefinition is the molar or specific enthalpy. Thisand heatspecific capacityheat relates in an especially simple Again, the both molar capacities, depending onestá bien whether H is the molaraccommodates or specific enthalpy. This heat capacity relates in an especially simple un proceso a presión constante en un sistema cerrado, para el que la ecuación (2.20) de igual manera way to aHconstant-pressure, closed-system process, for capacity which Eq. (2.20) isan equally well written: is the molar or specific enthalpy. This heat especially simple way to a constant-pressure, closed-system process, for which Eq.relates (2.20) in is equally well written: escrita:whether way to a constant-pressure, closed-system process, for which Eq. (2.20) is equally well written: d H ==CC PdTdT (P constante) (const P) (2.21) ddH H = CPP dT (const P) (2.21) (2.21) d H =�C P dT (const P) (2.21) � T2T2 whence �H = C dT (const P) (2.22) de donde constante) whence �H = � TT12 C PPdT (P(const P) (2.22) (2.22) whence �H = T1 C P dT (const P) (2.22) T1 process, this result may be combined with Eq. (2.13): For a mechanically reversible, constant-P a mechanically reversible, constant-P process, this result be combined with Eq. (2.13): Para unFor proceso mecánicamente reversible a presión constante, estemay resultado puede combinarse con la ecuaFor a mechanically reversible, constant-P process, ción (2.13): � T2 this result may be combined with Eq. (2.13): � T Q = n �H = n 2 C dT (const P) (2.23) Q = n �H = n � TT12 C PPdT (const P) (2.23) T P) (2.23) (2.23) Q = n �H = n 1 C P dT (P(const constante) T1 Because H , C , and T are state functions, Eq. (2.22) applies to any process for which P = P Because H , C PP, and T are state functions, Eq. (2.22) applies to any process for which P2 2= P1 1 whether is Tactually carried out at constant pressure. However, only for thePmechanDado que H, CPor y,not Tnot funciones deout estado, la ecuación (2.22) seprocess aplica afor cualquier proceso , it and are state functions, (2.22) applies toHowever, any which Because Hor C Pson 2 = P1 para el whether it istodas actually carried at Eq. constant pressure. only for the mechanically reversible, constant-pressure process can heat and work be calculated by the equations cual P2whether = P , se esté o no realizando realmente a presión constante. Sin embargo, sólo para un proceso mecáor not it constant-pressure is actually carried out at constant only by forthe theequations mechan� ically1 reversible, can heatpressure. and workHowever, be calculated � C P dT , and process Q = n �H , Q = n W = −Pn �V . nicamente reversible a presión constante se puede calcular el calor y el trabajo por medio de las ecuaciones ically can�V heat Q = nreversible, �H , Q =constant-pressure n � C P dT , and Wprocess = −Pn . and work be calculated by the equations Q = n ΔH, Q = n∫C dT y W = –Pn ΔV. Q = n �H , PQ = n C P dT , and W = −Pn �V . Example2.9 2.9 Example Ejemplo 2.9 Example 2.9 Air at 1 bar and 298.15 K (25◦ ◦ C) is compressed to 5 bar and 298.15 K by two different Air at 1 bar and 298.15 K (25 C) is compressed to 5 bar and 298.15 K by two different mechanically reversible processes: Se comprime a1 bar y 298.15 (25is°C), hasta 5 bar 298.15 mediante Air at 1 aire bar and 298.15 Kprocesses: (25K◦ C) compressed to 5y bar andK298.15 K bydos twoprocesos differentmecánimechanically reversible camente reversibles diferentes: mechanically reversible processes: (a) Cooling at constant pressure followed by heating at constant volume. (a) Cooling at constant pressure followed by heating at constant volume. (a) Cooling at constant pressure followed by heating at constant volume. (b) Heating at constant volumeseguido followeddebyuncooling at constant pressure. b) Calentamiento constante enfriamiento a presión constante. (b) Heatingaatvolumen constant volume followed by cooling at constant pressure. (b) Heating at constant volume followed by cooling at constant pressure. CalculeCalculate los requerimientos de calor de trabajo, yand ΔU y�U ΔHand del aire unaeach de las trayectothe heat and work yrequirements �H para of thecada air for path. Calculate the heat and workcaloríficas requirements and �U se and �H ofque the son air for each path. de la rias. Las siguientes capacidades para el aire supone independientes The following heatand capacities for air may be assumed independent of temperature: Calculate the heat work requirements �U and �H of theofair for each path. The following capacities for air may beand assumed independent temperature: temperatura: The following heat capacities for air may be assumed independent of temperature: −1 K−1 and C = 29.10 J mol−1 C = 20.78 −1 –1KK –1 C PPC=P 29.10 J mol C VV= 20.78 CV = 20.78 and y = 29.10 J mol and C P = 29.10 J mol−1 K−1 C V = 20.78 Assume also for air that P V /T is a constant, regardless of the changes it undergoes. Assume also for airelthat V /T is aesconstant, regardless of the changes it undergoes. Supóngase también para aire P que PV/T una constante, sin considerar los cambios que experimen3 mol −1 At 298.15 K and 1 bar the molar of air is 0.02479 m 3 mol −1 . Assume forelair thatthe P Vmolar /T isdel avolume constant, the it undergoes. 3m –1changes At 298.15 and 1volumen bar volume of air is 0.02479 ta. A 298.15 K yalso 1Kbar molar aire es deregardless 0.02479 mof mol . . At 298.15 K and 1 bar the molar volume of air is 0.02479 m3 mol−1 . a) Enfriamiento a presión constante seguido de un calentamiento a volumen constante. 02-SmithVanNess.indd 41 8/1/07 12:49:02 42 42 42 42 42 CHAPTER 2. CHAPTER 2. CHAPTER 2. CHAPTER 2. The First Law and Other Basic Concepts The First First Law Law and and Other Other Basic Basic Concepts Concepts The The First Law and Other Basic Concepts 42 CHAPTER 2. The 2. First and Other Basicconceptos Conceptsbásicos CAPÍTULO LaLaw primera ley y otros Solution 2.9 2.9 Solution Solution 2.9 Solución Solution 2.9 In2.9 each case take the system as 1 mol of air contained in an imaginary pisIn each case take the system as 1 mol of air contained in an imaginary pisIn each case arrangement. take the system as 1 mol of aircontenido contained in anare imaginary pisEn cadaIn caso considere al sistema como deprocesses aireair enin una combinación imaginaria ton/cylinder Because the considered mechanically each case take the system as1 1mol mol of contained anare imaginary pisSolution 2.9arrangement. ton/cylinder Because the processes considered mechanically ton/cylinder arrangement. Because processes considered are mechanically reversible, Debido the piston is estos imagined to the move in the cylinder without friction. The suponde pistón/cilindro. a que procesos seprocesses consideran mecánicamente reversibles, ton/cylinder arrangement. Because considered are mechanically reversible, the piston is imagined to the move in the cylinder without friction. The reversible, the piston issystem imagined to moveofinEl the cylinder without friction. The In each case take theel as 1fricción. mol air contained in es: an imaginary pisfinal volume is: ga que el pistón se mueve en cilindro sin volumen final reversible, theis:piston is imagined to move in the cylinder without friction. The final volume final volume is: ton/cylinder arrangement. Because the processes considered are mechanically � � final volume is: � 1� P1 � 1in �the reversible, the piston is Vimagined to move cylinder without friction. The P 1 = 0.02479 = 0.004958 m33 V2 = 1 P1 = 0.02479 � 1 � = 0.004958 m = V V 2 1 P 5 2 P 1 final volume is: V2 = V1 P12 = 0.02479 5 = 0.004958 m33 V2 = V1 P2 = 0.02479 � 5 � = 0.004958 m 51 constant pressure of 1 bar until the 2 is cooled at the (a) During the first step thePPair 1 is cooled at the 3 of 1 bar until the (a) During the etapa, firstVstep the air constant pressure = V = 0.02479 = 0.004958 3 a) Durante la primera el aire se enfría a presión constante deof1mthe barof hasta que sethe alcanza el 2 1 (a) During the first step the air is cooled at the constant pressure 1 at barthe until final volume offirst 0.004958 mPair isisreached. The temperature air end of 32 is 5 (a) During theof step them cooled at the constant pressure ofair 1 at barthe until final volume 0.004958 reached. The temperature of the endthe of 3 3 is reached.del . La temperatura aire al final de esta etapa de enfriamiento es: volumenfinal final de 0.004958 m volume of 0.004958 m The temperature of the air at the end of this cooling step is: final volumestep of 0.004958 m3 is reached. The temperature of the air at the end of this cooling is: this coolingthe step is: step the air is cooled�at the constant (a) During first � pressure of 1 bar until the this cooling step is: � � 3 is reached. V 0.004958 2 final volume of 0.004958 m The temperature of the � 0.004958 � = 59.63 V2 = 298.15� T� = T V K air at the end of � = 59.63 K 2 = 298.15 0.004958 T �� = T11V V 0.02479 this cooling step is: T = T V211 = 298.15 0.004958 0.02479 = 59.63 K T � = T11 V 1 = 298.15 � 0.02479 � = 59.63 K V 0.02479 Whence, V12 0.004958 De donde, Whence, T � = T1 = 298.15 = 59.63 K Whence, V 0.02479 Whence, Q = �H = C P1�T = (29.10)(59.63 − 298.15) = −6,941 J Q = �H = C P �T = (29.10)(59.63 − 298.15) = −6,941 J = (29.10)(59.63 − 298.15) = −6,941 J Q = C P �T Whence,�U = �H �H = −C �(P V)= �H − P �V − (29.10)(59.63 298.15) = −6,941 J Q P �T �U = = �H �H = − �(P V) = �H − P �V �U = �H − �(P V ) = �H − P �V 5 = �H −6,941 − (1V× 10�H )(0.004958 −− 0.02479) = −4,958 �U − )= − P �V− 5(29.10)(59.63 = 298.15)= =−4,958 −6,941JJJ Q= = �(P C = �H −6,941 − (1�T × 10 )(0.004958 0.02479) = −6,941 −P(1 × 1055 )(0.004958 − 0.02479) = −4,958 J = −6,941 − (1V×) 10 )(0.004958 − 0.02479) = −4,958 J �Uthe =second �H −step �(P = �H P �V During the volume is−held constant at V while the air is heated During the second step the volume is held constant at V22 while the air is heated 5 the air is heated During the second step the volume is held constant at V while to its final state. By Eq. (2.19), −6,941 − (1 × 10se)(0.004958 − 0.02479) −4,958 the airJ que is heated During the=second step the volume is held constant at Va22 while Durante lafinal segunda etapa, el(2.19), volumen mantiene constante V=2 mientras se calienta a to its state. By Eq. to its final state. By Eq. (2.19), to final. its final By Eq. (2.19), su estado Porstate. la ecuación (2.19), �T = (20.78)(298.15 − 59.63) = 4,958 J is heated �U = Q = C V the the Jair During�U the second is held constant at V2 while = (20.78)(298.15 − 59.63) = 4,958 = Q =step C �Tvolume �U =ByQEq. = C VV �T = (20.78)(298.15 − 59.63) = 4,958 J to its final �U state. �T = (20.78)(298.15 − 59.63) = 4,958 J = Q = C(2.19), V The complete process represents the sum of its steps. Hence, The complete process represents the sum of its steps. Hence, The complete process represents the sum of its steps. Hence,= 4,958 J �T = − �U = Q = C V El proceso la suma de sus lo 59.63) tanto, Thecompleto completerepresenta process represents the(20.78)(298.15 sumetapas. of its Por steps. Hence, Q = −6,941 + 4,958 = −1,983 J Q = −6,941 + 4,958 = −1,983 J Q = −6,941 + 4,958 = −1,983 J The complete process represents the sum of its –6 941 + 44,958 958 ==steps. –1 983Hence, JJ QQ==−6,941 + −1,983 and �U = −4,958 + 4,958 = 0 and �U = −4,958 + 4,958 = 0 and �U = −4,958 + 4,958 =0 J Q �U = −6,941 + 4,958 = −1,983 and −4,958 + 4,958 = 0 = Q + W , and therefore, Because the first law applies to=the entire process, �U Because the first law appliesΔU to the entire process, �U = Q + W , and therefore, y = –4 958 + 4 958 = 0 = Because the first law applies to the entire process, �U Q + W , and therefore, Because the first law applies to = the−4,958 entire process, �U = Q + W , and therefore, and �U + 4,958 = 0 0 = −1,983 + W whence W = 1,983 J 0 = −1,983 + Wa todo el whence W =W, 1,983 Debido a que la primera se aplica proceso, ΔU = W Q += y porJ lo tanto, 0 =ley −1,983 + to W the entire whence 1,983 Because the first applies process, �UW= = Q 1,983 + W , JJand therefore, 0 =law −1,983 +W whence Equation (2.15), �H = �U + �(P V ), also applies to the entire process. But Equation (2.15), �H = �U + �(P V ), also appliesWto=the entire process. But 1and 983 J process. 0 = –1�H 983P= +VW Equation �U Vdonde ), also applies to0, the entire But T , and(2.15), therefore, =+ P �(P V de . whence Hence �(P V ) =to T1 = 0 = −1,983 +�U =the 1,983 J process. But 1V 1W Equation �HP = V ), also applies entire T22, and (2.15), therefore, =+ P22�(P V22. Hence �(P V) W = 0, and T1 = 1 1 T1 = T2 , and therefore, P1 V1 = P2 V2 . Hence �(P V ) = 0, and and therefore, P1 V+1 Δ(PV), = P2 V2también . Hence se �(P V ) = 0, andel proceso. Pero T = T , y T1 = T2 , (2.15), La ecuación ΔH �H = ΔU todo 2 �H =V �U = 0aplica Equation (2.15), = �U �H + �(P ), also appliesa to the entire process. But1 = �U = 0 por lo tanto, = Ptherefore, = �H 0, yV2= �U =�(P 0 V ) = 0, and 2V2. Así, Δ(PV) P1 V1 = P . Hence T1 =PT12V, 1and 2 �H = �U = 0 (b) Two different steps are used in this case to reach the same final state of the air. (b) Two different steps are used inΔH this=case to reach the same final state of the air. ΔU == 0reach (b) Two different steps are used in case to the same final statevalue of theuntil air. In the first step the air is heated at�H athis constant volume equal to final its initial =case �Uto 0 the (b) Two different used at in reach same statevalue of theuntil air. In the first step thesteps air isare heated athis constant volume equal to its initial In the first step the air is heated at a constant volume equal to its initial value until the final pressure ofair5 is barheated is reached. The air volume temperature attothe end of this step is: In first step theof at a constant equalat itsend initial value until thethe final pressure 5 bar is reached. The para air temperature the of this step is: b) En este emplean dos etapas diferentes alcanzar mismo estado final the final pressure of 5 bar isused reached. air at thefinal end of this step is:aire. En (b)caso Twose different steps are in thisThe case totemperature reach theelsame state of thedel air. � � the etapa final pressure 5 bar is areached. The air at the end ofinicial, this step is: que se � � 5temperature la primera airethe seofcalienta igual suitsvalor Pun volumen In the firstelstep air a298.15 constant equalatoK initial value hasta until � is heated � = 1,490.75 �constante P22 at 5volume = T = T � 1 � � P 5 alcanza the la presión final deof5T5bar. temperatura del aire al final de esta etapa es: 2 = 298.15 = 1,490.75 K = La Tis1 reached. � bar P 1 final pressure The air temperature at the end of this step is: = 298.15 51 = 1,490.75 K T =T P P211 = 298.15 � 1 � = 1,490.75 K T � = T11 P 1 PP1 15 2 = 1,490.75 K = 298.15 T � = T1 P1 1 02-SmithVanNess.indd 42 8/1/07 12:49:07 2.11. Capacity 2.11. Capacidad 2.11. Heat Heatcalorífica Capacity 43 43 43 Para esta etapa volumen es constante, y and For this step the isis constant, For thisel step the volume volume constant, and Q Q= = �U �U = = CCVVV �T �T = = (20.78)(1,490.75 (20.78)(1,490.75 − − 298.15) 298.15) = = 24,788 24,788 JJ In at its final En la segunda etapa elstep airethe se air enfría a una presión = 5to bar estado final: In the the second second step the air isis cooled cooled at PP = = 5P 5 bar bar to itspara finalsustate: state: Q Q= = �H �H = = CCPPP �T �T = = (29.10)(298.15 (29.10)(298.15 − − 1,490.75) 1,490.75) = = −34,703 −34,703 JJ �U �U = = �H �H − − �(P �(PVV)) = = �H �H − − PP �V �V 55 = = −34,703 −34,703 − − (5 (5 × × 10 105)(0.004958 )(0.004958 − − 0.02479) 0.02479) = = −24,788 −24,788 JJ two steps Para lasFor dosthe etapas For the twocombinadas, steps combined, combined, as y como and antes and as before before Q Q= = 24,788 24,788 − − 34,703 34,703 = = −9,915 −9,915 JJ �U �U = = 24,788 24,788 − − 24,788 24,788 = = 00 W W= = �U �U − −Q Q= = 00 − − (−9,915) (−9,915) = = 9,915 9,915 JJ �H ΔH= = �U ΔU �H = �U == =000 The changes and calculated for in Los cambios en las propiedades y �H ΔH son calculados para change el cambio dado en el estaThe property property changes �U �UΔU and �Hque calculated for the the given given change in state state are are the same for both paths. On the other hand the answers to parts (a) and (b) do son iguales para trayectorias. otrahand parte, respuestas a los(a) incisos a) show yshow b) demuesthe same forambas both paths. On thePor other thelas answers to parts and (b) W the tran quethat Q yQ dependen de laon trayectoria. that QWand and W depend depend on the path. path. Example 2.10 Example 2.10 Ejemplo 2.10 Calculate the internal-energy and enthalpy changes that occur when air is changed Calculate Calculate the the internal-energy internal-energy and and enthalpy enthalpy changes changes that that occur occur when when air air is is changed changed ◦◦◦F)laand 333(lb de un Calculefrom los cambios ocurren en energía interna y en its la cuandois aire cambia an state of 10(atm), where molar from an initial initialque state of 40( 40( F) and 10(atm), where its entalpía molar volume volume isel36.49(ft) 36.49(ft) (lb −1 ◦ 3 –1 ◦◦F)su −1 estado mole) inicial−1 de 40(°F) y 10(atm), donde volumen molar es 36.49(pie) (lbPPmol) ,isaconstant un estado final ,, to aa final state 1(atm). Assume for VV/T mole) to final state of of 140( 140( F) and and 1(atm). Assume for air air that that /T is constant −1 ◦◦◦F)−1 −1 −1((PV/T −1.es de 140(°F) y 1C (atm). Suponga para el aire que mole) and V = 55 and and CCPPP== 7(Btu)(lb 7(Btu)(lb mole) F)−1 . constante y que CV = 5 y CP = 7 (Btu) and that that C VV= –1 –1 (lb mol) (°F) . Solution 2.10 Solution Solución 2.10 2.10 Because property changes independent of process that brings them about, Because property changes are are of the the themlos about, Ya que los cambios en la propiedad sonindependent independientes delprocess procesothat quebrings los causa, cálculos se calculations may be based on aa two-step, mechanically reversible process in which calculations may be based on two-step, mechanically reversible process in pueden basar en un proceso de dos etapas y mecánicamente reversible, en el cual 1(lbwhich mol) de aire 1(lb of (a) at constant volume to the and (b) 1(lb mole) mole) of air air isis (a) cooled cooled to calentado the final final pressure, pressure, (b) para es: a) enfriado a volumen constante paraatlaconstant presión volume final, y b) a presión and constante heated at constant pressure to the final temperature. The absolute temperatures heated at constant pressure to the final temperature. The absolute temperatures la temperatura final. Las temperaturas absolutas están dadas en la escala Rankine: here here are are on on the the Rankine Rankine scale: scale: T1 = 40 + 459.67 = 499.67(R) T2 = 140 + 459.67 = 599.67(R) TT111 = TT222 = = 40 40 + + 459.67 459.67 = = 499.67(R) 499.67(R) = 140 140 + + 459.67 459.67 = = 599.67(R) 599.67(R) Dado que PV = kT, la relación T/P ratio es constanteispara la etapa a). Por lo tanto, la temperatura interBecause Because PPVV = = kT kT,, the the ratio TT/P /P is constant constant for for step step (a). (a). The The intermediate intermediate media entre los dos pasos es:the two steps is therefore: temperature between temperature between the two steps is therefore: = (499.67)(1/10) ==49.97(R) TTT��� ′= = (499.67)(1/10) (499.67)(1/10) = 49.97(R) 49.97(R) y los cambios temperatura para lasfor dos son: are: and temperature changes the two and the thede temperature changes for theetapas two steps steps are: �T 49.97 499.67 −449.70(R) ΔT = 49.97 –449.70(R) aaaa= �T = 49.97–− −499.67 499.67== = −449.70(R) �T = 599.67 − 49.97 = 549.70(R) b �T 599.67–−49.97 49.97==549.70(R) 549.70(R) ΔTbbb = = 599.67 02-SmithVanNess.indd 43 8/1/07 12:49:10 44 CHAPTER CHAPTER2. 2. The TheFirst FirstLaw Lawand andOther OtherBasic BasicConcepts Concepts CHAPTER 2. The First Law and Other Basic Concepts 44 44 44 CAPÍTULO 2. La primera ley y otros conceptos básicos For Forstep step(a), (a),by byEqs. Eqs.(2.18) (2.18)and and(2.15), (2.15), For Eqs. (2.18) and Para la etapa a),step por(a), las by ecuaciones (2.18) y(2.15), (2.15) �U = CC �T �T = = (5)(−449.70) (5)(−449.70) = = −2,248.5(Btu) −2,248.5(Btu) �Uaa = �U a = CVVV �Taaa = (5)(−449.70) = −2,248.5(Btu) �H �Haa = = �U �U + +VV �P �P �H a = �Uaaa + V �Paaa = −2,248.5 + = −2,248.5 + (36.49)(1− −10)(2.7195) 10)(2.7195) = = −3,141.6(Btu) −3,141.6(Btu) = −2,248.5 + (36.49)(1 (36.49)(1 − 10)(2.7195) = −3,141.6(Btu) 3(atm)(ft) which The factor 2.7195 the VVV product from which is an an energy energy The factorconvierte 2.7195 converts converts the PPPPV product from (atm)(ft) El factor 2.7195 el producto de (atm)(pie) , el cual333,,,es una is unidad de energía a which is an energy The factor 2.7195 converts the product from (atm)(ft) into (Btu). unit, into (Btu). (Btu). unit, unit, into (Btu). the volume the air Forstep step (b), thefinal finalfinal volume of the airis: is: Para la For etapa b),(b), el volumen delof aire es: For step (b), the final volume of the air is: � � � �� � � � �10 PPP11TTT22 10� �599.67 599.67� 333 3 10 599.67 1 2 (pies) VVV22 = = = = VV = 36.49 36.49 = 437.93(ft) 437.93(ft) = 437.93(ft) 2 = V111PP22TT11 = 36.49 11 499.67 499.67 P2 T1 1 499.67 Por las By ecuaciones (2.22) (2.15), and ByEqs. Eqs.(2.22) (2.22) andy(2.15), (2.15), By Eqs. (2.22) and (2.15), �H = CC �T �T = = (7)(549.70) (7)(549.70) = = 3,847.9(Btu) 3,847.9(Btu) �Hbb = �H b = CPPP �Tbbb = (7)(549.70) = 3,847.9(Btu) �U = �H − P �V �U = �H − P �V �Ubb = �Hbb − P �Vbb b b b = = 3,847.9 3,847.9− −(1)(437.93 (1)(437.93− −36.49)(2.7195) 36.49)(2.7195) = = 2,756.2(Btu) 2,756.2(Btu) = 3,847.9 − (1)(437.93 − 36.49)(2.7195) = 2,756.2(Btu) Para lasFor dosthe etapas juntas, two For the twosteps stepstogether, together, For the two steps together, ΔU= = −2,248.5 –2 248.5 ++ 756.2 == 507.7 (Btu) �U 507.7(Btu) �U = −2,248.5 +22,756.2 2,756.2 = 507.7(Btu) �U = −2,248.5 + 2,756.2 = 507.7(Btu) ΔH = –3 141.6 + 3 847.9 = 706.3 (Btu) �H �H = = −3,141.6 −3,141.6+ +3,847.9 3,847.9 = = 706.3(Btu) 706.3(Btu) �H = −3,141.6 + 3,847.9 = 706.3(Btu) 2.12 BALANCES DE MASA Y ENERGÍA PARA SISTEMAS ABIERTOS 2.12 2.12 MASS MASS AND AND ENERGY ENERGY BALANCES BALANCES FOR FOR OPEN OPEN SYSTEMS SYSTEMS 2.12 MASS AND ENERGY BALANCES FOR OPEN SYSTEMS Aunque en las secciones anteriores nos hemos concentrado en sistemas cerrados, los conceptos mencionados Although focus preceding sections closed systems, the preAlthough the focus of of the the preceding sections has has been on on closed systems, the concepts pretienen un uso másthe extenso. Las leyes de conservación de been masa y de energía se aplican aconcepts todos los procesos, Although the focus of the preceding sections has been on closed systems, the concepts presented find far more extensive application. The laws of mass and energy conservation apply to sented find far more extensive application. The laws of mass and energy conservation apply to espeya seansented sistemas o cerrados. hecho, el sistema incluye el sistema cerrado como findabiertos far more extensiveDe application. The lawsabierto of mass and energy conservation applycaso to all processes, to open as well as to closed systems. Indeed, the open system includes the closed all processes, to open as well as to closed systems. Indeed, the open system includes the closed cial. Por tanto, el to resto deaseste se dedica al tratamiento sistemas al closed desarrollo de allloprocesses, open wellcapítulo as to closed systems. Indeed, the de open system abiertos includes ythe system case. The system asaaaspecial specialmás case. Theremainder remainderof ofthis thischapter chapteris istherefore thereforedevoted devotedto tothe thetreatment treatmentof of ecuaciones de as aplicación extensa. system as special case. The remainder of this chapter is therefore devoted to the treatment of open systems and thus to the development of equations of wide applicability. open systems and thus to the development of equations of wide applicability. open systems and thus to the development of equations of wide applicability. Medidas de flujo Measures of Measures of Flow Flow Measures of Flow Open characterized by flowing there four common measures of Opensystems systemsare arecaracterizan characterized bycorrientes flowingstreams; streams; thereare are fourcuatro common measures offlow: flow: Los sistemas abiertos se por que circulan; existen medidas de flujo comunes: Open systems are characterized by flowing streams; there are four common measures of flow: ... ... •••Mass flowrate, m Molar nnn qqq ••• Velocity, uuu Mass flowrate, m ṁ ••••Rapidez Molarflowrate, flowrate, Volumetric flowrate, Velocity, • Rapidez de flujo de masa, de flujo molar, ṅ••• •Volumetric Rapidez deflowrate, flujo volumétrico, q • Velocidad, u Mass flowrate, m Molar flowrate, Volumetric flowrate, Velocity, The are Themeasures measures offlow flow areinterrelated: interrelated: Las medidas de flujo of están correlacionadas: The measures of flow are interrelated: ... ... · ṁ =MnMn m = m =M m = M nn y and and and qqqq== uAuuuAAA = = donde where M es laM molar. En Importantly, forma importante, la rapidez de flujo relate de masa y molar se relaciona con la is mass. mass molar to where Mmasa ismolar molar mass. Importantly, massand and molarflowrates flowrates relate tovelocity: velocity: where M is molar mass. Importantly, mass and molar flowrates relate to velocity: velocidad: 02-SmithVanNess.indd 44 8/1/07 12:49:13 4545 2.12. and Balances forforOpen Systems 2.12.Mass Mass andEnergy Energy Balances Open Systems 2.12. Balances de masa y energía para sistemas abiertos 45 2.12. Mass and Energy Balances for Open Systems 45 2.12. 45 45 2.12. Mass Massand andEnergy EnergyBalances Balancesfor forOpen OpenSystems Systems . . . . mm Aρ (2.24a) n .n= u Aρ (2.24b) .ṁ===uuAr u Aρ (2.24a) = uAρ Aρ (2.24b) (2.24a) = uuAr (2.24b) a b m.. = u Aρ (2.24a) n.ṅ. = (2.24b) mm = (2.24a) (2.24b) = uuAρ Aρ (2.24a) nn = = uuAρ Aρ (2.24b) The area forforflow A is isthethecross-sectional area and ρ is isspecific orormolar area flow cross-sectional areaofde ofa aun aconduit, conduit, and specific molar area flow isis the cross-sectional area of conduit, and specific orasmolar El área The AThe para el for flujo es elAAárea de sección transversal conducto, yuρrρisis esused la densidad específica o density. Although velocity a vector quantity, its scalar magnitude here thethe The area for flow A is the cross-sectional area of a conduit, and ρ is specific or molar density. Although velocity is a vector quantity, its scalar magnitude u used here as The area for flow A is the cross-sectional area of a conduit, and ρ is specific or molar . . density.speed velocity is a vector quantity, its scalar magnitude u is used here as the molar.average Aunque laAlthough velocidad es una cantidad vectorial, su magnitud escalar u se utiliza en este caso como la . . ofofa astream direction normal toto A.A.Flowrates m,m, q here represent .uun,isis .n,and density. Although isisthe athe quantity, its scalar magnitude used average speed speed streaminen in direction normal Flowrates andflujo represent density. Although velocity a vector vector quantity, itsrespecto scalar magnitude asṁ,the the average of corriente a velocity stream in the direction normal to A. different Flowrates m, and qqhere represent rapidezmeasures promedio de una dirección normal con a A. Lainrapidez de deas ṅ, y q re.. n, .. used of quantity per unit of time. Velocity u is quite nature, as it does not average speed of a stream in the direction normal to A. Flowrates m, n, and q represent measures of quantity per unit of time. Velocity u is quite different in nature, as it does not average speed of a stream in the direction normal to A. Flowrates m, n, and q represent measures of quantity per unit of time. Velocity u is quite different in nature, as it does not presenta medidas de la cantidad por unidad de tiempo. La velocidad u es de una naturaleza muy diferente suggest thethe magnitude ofofflow. Nevertheless, it itis isan important design parameter. measures quantity unit of time. Velocity quite in nature, as suggest magnitude flow. Nevertheless, important design parameter. measures of quantity per per unit of time. Velocity uanisis quite different different in nature, as itit does does not not theof of Nevertheless, it es is uan important design parameter. porque suggest no sugiere lamagnitude magnitud delflow. flujo. Sin embargo, un importante parámetro de diseño. suggest suggestthe themagnitude magnitudeof offlow. flow. Nevertheless, Nevertheless,ititisisan animportant importantdesign designparameter. parameter. Example 2.11 Example 2.11 Example 2.11 . . Ejemplo 2.11 −1 in a pipe with inside diameter Example 2.11 Liquid n-hexane flows atata arate ofofmm −1 in a pipe with inside diameter Example 2.11 . ==0.75 Liquid n-hexane flows rate 0.75kg kgsss−1 . . Liquid n-hexane flows at a rate of m = 0.75 kg in a pipe with inside diameter . · =would .and DLiquid are n,.n, u?u? What these be the same mm −1quantities El n-hexano circula aq,una de 0.75 kg ss–1 una tubería con diámetro interno de ..if if n-hexane flows arelación rate of mm. m= 0.75 in aa pipe with inside diameter D== =55líquido 5cm. cm.What What are q,at and What would these quantities befor for the same Liquid n-hexane flows at aand rateu? of = would 0.75 kg kg s−1en in−3 pipe with inside diameter D cm. What are q, n, What these quantities be for the same m · . .. if m· si . = 2 cm? Assume for liquid n-hexane that ρ = 659 kg m . −3 D = 5D cm. ¿Cuáles son los valores de q, n y u? ¿Cuáles serían estas cantidades para la misma DD = = cm. What q, n, u? would quantities be =2525cm? cm? Assume for liquid n-hexane that =these 659kg kg m−3 . cm. Assume What are are q,liquid n, and and u? What What would these quantities be for for the the same same mm ifif D for n-hexane that ρρ = 659 –3 m−3 . D = 2 cm? que parafor elliquid n-hexano líquidothat r =ρρ659 kg DD = 22 cm? n-hexane = =Suponga cm? Assume Assume for liquid n-hexane that = 659 659mkg kg.m m−3.. Solution 2.11 . .−1 Solution 2.11 Solution 2.11 q q==mρ We have and −1 . −1 We have mρ and We have q = mρ and · r –1 ..= −1 Tenemos que q m y −1 We qq = −1 and Wehave have = mρ mρ and Solution 2.11 Solución 2.11 2.11 Solution . . . . −1 n .n= −1 .MM−1 =mm n= M −1 · Mm.–1 . n· =n.n.m= = mmM M −1 0.75 kgkgs s−1 −1 3 −1 0.75 whence q q== 0.75 kg −3 −1 s−1==0.00114 whence 0.00114mm3s3 s−1 −1 kgkg mm whence q =659 −3 = 0.00114 m3 s−1 0.75 kg ss−3 0.75 kg 659 3 de donde whence qq = = whence = 659 kg m−3 = 0.00114 0.00114m m ss−1 −3 −1 659 kg m −1 3 659 kg m )(10 )) −1 −1 . . (0.75 −1 )(103g3 ggkg kg−1 (0.75kg kgs ss−1 n .n= −1 )(10 kg )==8.703 kg 8.703mol mols ss−1 = (0.75 −1 3−1 −1 −1 3 −1 86.177 g mol = 8.703 mol n.. = −1 )(10 g kg ) (0.75 kg s g kg ) (0.7586.177 kg s )(10 g mol −1 −1 = nn = 86.177 g mol−1 = 8.703 8.703mol molss−1 = . . 86.177 ggmol Given m,m, quantities are independent velocity, 86.177 mol−1 ofofD.D. The . these Given these quantities are independent The velocity,however, however,dedeGivenonm, these quantities are independent D.a circular The velocity, however, −1 , where,offor ..diameter pends through u = q A cross-section, A Ade== −1 Given m, these quantities are independent of D. The velocity, however, de· pends on diameter through u = q A , where, for a circular cross-section, −1 Given these son quantities D.a circular Thela velocity, however, deDada m(π/4)D ,pends estas cantidades independientes de, D. Noofobstante, velocidad dependeAdel on diameter uare= independent q A−1 where, for cross-section, = diáme2m, . 2For DD==5through pends uu = qqAA−1,transversal aa circular .diameter For 5cm, cm, una (π/4)D –1,D pendsuon on diameter = , where, where, for forcircular, circular cross-section, == 5 cm, tro mediante =22 .qA donde para sección A =cross-section, (p/4)D2. ParaAAD= For = through 5through cm, (π/4)D (π/4)D �2� For DD = = 55cm, cm,π π� � (π/4)D 2.. For 2 −2 2 0.00196 m 2 A A== π 5� × 1010−2 mm�2= −2 × =0.00196 0.00196m m2 ��55× ��22 = A =4ππ 10−2 m 4 2 −2 4 AA = 5 × 10 m = 0.00196 m = 5 × 10 m = 0.00196 m2 4 3 −1 4 0.00114 −1 −1 0.00114mm m3s3 ss−1 whence u u== 0.00114 ==0.582 −1 de donde whence 0.582mm ms ss−1 2 3 −1 3 −1 = 0.00196 m whence u = 0.00114 0.582 2 s m s 0.00114 m 0.00196 m −1 2 whence uu = whence for D = 2 cm, = 0.00196 m22 = = 0.582 0.582m mss−1 Similarly, 0.00196 m 0.00196 m Similarly, for D = 2 cm, De manera similar,for para D= 2 cm, Similarly, D= 2 cm, Similarly, Similarly,for for DD = = 22cm, cm, 0.00114 22 0.00114= 3.63 m s−1−1 AA ===0.000314 mmm and u u== 0.00114 0.000314 yand 2 A 0.000314 = 3.63 m s−1 2 and u =0.000314 A = 0.000314 m2 0.00114 0.00114 = 3.63 m s−1 0.000314 and uu = AA = = 0.000314 0.000314m m2 and = 0.000314 = = 3.63 3.63m mss−1 0.000314 0.000314 Balance de masa para sistemas abiertos Mass MassBalance Balancefor forOpen OpenSystems Systems Mass Balance for Open Systems La región del espacio que es identificada para el análisis de systems sistemas abiertos se llama volumen de control; se Mass Balance for Open Systems Mass Balance for Open Systems The Theregion regionofofspace spaceidentified identifiedforforanalysis analysisofofopen open systemsis iscalled calleda acontrol controlvolume; volume;it itis is Theseparada region of identified for ofsurface. open systems is called control volume; it is encuentra despace sussurroundings alrededores mediante una superficie de control. El afluido dentro del volumen de separated from its bybyanalysis a acontrol The fluid within thethecontrol volume The of space identified for analysis open is aa control volume; itit is separated from its surroundings surroundings control surface. The fluid within control volume The region oftermodinámico space identified for analysis ofsurface. open systems systems is called called control volume; is from its byel aque control fluid within the control volume control separated es elregion sistema para se of escriben losThe balances de masa y de energía. El volumen separated separated from from its its surroundings surroundings by by aa control control surface. surface. The The fluid fluid within within the the control control volume volume 02-SmithVanNess.indd 45 8/1/07 12:49:20 46 46 46 46 46 46 CHAPTER 2. 2. TheThe First Law andand Other Basic Concepts CHAPTER First Other Basic Concepts CHAPTER 2. The First LawLaw and Other Basic Concepts CHAPTER 2. The First Law and Other Basic Concepts CAPÍTULO LaLaw primera ley y otros CHAPTER 2. The 2. First and Other Basicconceptos Conceptsbásicos ṁ1 ṁ1 ṁ1 ṁ1 ṁ1 ṁ 3 ṁ 3 ṁ 3 ṁ 3 ṁ1 ṁ 3 ṁ 3 Control volume Control volume Control volume dmcvdm /dt /dt Control volume cv dmVolumen /dt de control dmcv /dt cvvolume Control dmcv /dt dmcv /dt Figure 2.5: Schematic representation of a Figure Schematic representation Figure 2.5:2.5: Schematic representation of aof a control volume. Figure 2.5: Schematic representation a control volume. Figura 2.5: Representación esquemática de unofvolumen control volume. control Figure volume. 2.5: Schematic representation of a de control. control volume. ṁ 2 ṁ 2 ṁ 2 ṁ 2 ṁ 2 ṁ 2 Control surface Control surface Control surface Control surface Superficie de control Control surface is the thermodynamic system for which mass and energy balances are written. The control is the thermodynamic system which mass energy balances written. control is the thermodynamic system for for which mass andand energy balances are are written. TheThe control volume shown schematically in Fig. 2.5 is separated from its surroundings by an extensible is the thermodynamic system for which mass and energy balances are written. The controlpor una de control que se muestra en forma esquemática en la figura 2.5 está separado de sus alrededores volume shown schematically in Fig. 2.5 is separated from its surroundings by an extensible . . from . . its surroundings volume shown schematically in Fig. 2.5 is separated by an extensible · ym · están control surface. Two streams with flow rates m m are directed into the control volume shown schematically in 2.5 is rates separated from itsshown surroundings by anThe extensible .and . flujo is the thermodynamic system forFig. which mass energy balances are written. superficie de control extensiva. Dos corrientes con relaciones de m dirigidas hacia el volu1 and 1 2 directed control surface. streams with shown control 1 and 2 are . rates control surface. TwoTwo streams with flowflow m m. 22mare shown directed intointo the the control . 1 mand · . and one stream with flow rate m is directed out. Because mass is conserved, the rate control surface. Two streams with flow rates m and m are shown directed into the control . volume shown schematically in Fig. 2.5 is separated from its surroundings by an extensible men de volume, control, y una corriente con relación de flujo m está dirigida hacia afuera. Dado que la masa se con1 2 volume, stream with is .3directed Because mass is conserved, volume, andand oneone stream with flowflow raterate m. 33m is3directed out. Because mass is conserved, the the raterate . out. change of mass within the control volume, dm /dt, equals the net rate of flow of mass volume, and one stream with flow rate m is directed out. Because mass is conserved, the rate control surface. Two streams with flow rates m and m are shown directed into the control serva, laof rapidez de cambio de la masa dentro del volumen de control, dm / dt, es igual a la rapidez neta cv cv /dt, 3 1 dm 2 cvthe of change of mass within control equals of flow of mass de of change of mass within the the control volume, dm equals the net net raterate of flow of mass . volume, cv /dt, the control volume. The convention is that flow is positive when directed into the control of change of mass within the control volume, dm /dt, equals the net rate of flow of mass volume, and one stream with flow rate m is directed out. Because mass is conserved, the rate está flujo deinto masa en el interior del volumen de control. La convención es que el flujo es positivo cuando 3 is that flow control volume. convention is positive when directed control intointo the the control volume. TheThe convention is that flowcvis positive when directed intointo the the control and negative when directed out. The mass balance is expressed mathematically by: into the control volume. The convention is that flow is positive when directed into the control ofhacia change of mass within the control volume, dm /dt, equals the net rate of flow of mass dirigidovolume el volumen de control, y negativo cuando se dirige hacia afuera. El balance de masa se expresa cv balance is expressed mathematically by: volume negative when directed mass volume andand negative when directed out.out. TheThe mass balance is expressed mathematically by: volume negative when directed out. The mass balance is expressed mathematically by: into the and control volume. The convention is that flow is positive when directed into the control en forma matemática por: dm cvThe volume and negative when directed out. mass. balance by: . = 0 is expressed mathematically (2.25) cv �(m) dmdm . fs cv + (2.25) = 0 (2.25) + �( m) fs dm (2.25) . fs = 0 dtcv + �(m) (2.25) dt dt+ �(m)fs = 0 dm . dtcv (2.25) + �(m)fs = 0 thethe second term for thethe control of Fig. is: en la figura 2.5 es: donde elwhere segundo término para el for volumen devolume control que se 2.5 muestra dt where second term control volume of where the second term for the control volume of Fig.Fig. 2.52.5 is: is: where the second term for the control .volume. of Fig. is: . 2.5 . .=m . m . m �( m) − − . . . .is:2m. 2 fs 3 1 �( m) = m − m − where the second term for the control of Fig. 2.5 fs 3 1 �(m) = m − m − m .volume . . . �(m)fsfs = m33 − m11 − m22 . . . . entreexit El operador dedifference diferencia “Δ” en caso representa la diferencia los flujos de salida yflows entrada, The difference operator “�”este here signifies the difference between and entrance flows and y el operator entrance �(signifies m)fs = m1 − m 3 −difference 2 between TheThe difference operator “�”“�” herehere signifies themthe difference between exitexit andand entrance flows andand subíndice indica que el término se aplica a todas las corrientes que circulan. the subscript “fs” indicates that the term applies to all flowing streams. The“fs” difference operator “�” here signifies the difference between exit and entrance flows and subscript indicates term applies to flowing all flowing streams. .the.the the the subscript “fs”“fs” indicates thatthat term applies to all streams. Cuando laWhen relación de flujo de masa está dada por la ecuación (2.24a), la ecuación (2.25) se convierte When the mass flowrate m is given by Eq. (2.24a), Eq. (2.25) becomes: the “fs” indicates that the term applies to all flowing streams. . Thesubscript difference operator “�” here signifies the difference between exit and entrance flows and mass flowrate is given by Eq. (2.24a), (2.25) becomes: When the the mass flowrate m. ismgiven by Eq. (2.24a), Eq.Eq. (2.25) becomes: en: When the flowrate is given by Eq. to (2.24a), Eq. (2.25) becomes: the subscript “fs”mass indicates thatmthe term applies all flowing streams. . dmdm cv cv When the mass flowrate m isdm given by�(ρu Eq. (2.24a), 0 (2.25) becomes: (2.26) A) = Eq. cv + + �(ρu (2.26) dm (2.26) A)fsfsA)=fs 0= 0 (2.26) dtcvdt+ �(ρu (2.26) + �(ρu A)fs = 0 dt dm dtcv In this form the mass-balance equation is often called equation. = 0continuity (2.26) + �(ρu A) fsthe In this mass-balance isllama often called equation. De estaIn forma, la form ecuación del balance deequation masa ecuación decontinuity continuidad. this form the the mass-balance equation isse often called the the continuity equation. dtas flow process characterized steady state is an important special case for which thisThe form the mass-balance equation is as often called the continuity equation. The flow process characterized steady state is an important special case for whichen el ElInproceso de flujo caracterizado como un estado estacionario es un caso especial e importante, The flow process characterized as steady state is an important special case for which conditions within thedel control volume do not change time. TheThe control volume then conprocess characterized asisdo steady statewith iscon an important special case for which In thisThe formflow the mass-balance equation often called the continuity equation. conditions within the control volume not change with time. control volume then concual lasconditions condiciones dentro volumen de control no cambian el tiempo. Por lo tanto el volumen de conwithin the control volume do not change with time. The control volume then contains auna constant mass of fluid, andand the first or accumulation term of Eq. (2.25) is zero, reducing conditions within the control volume do not change time. The control volume then con-es cero, The flow process characterized asprimer steady statewith isdean important special case for which tains a constant mass of fluid, the first or accumulation term of Eq. (2.25) is zero, reducing trol contiene masa constante de fluido y el término acumulación de la ecuación (2.25) tains a constant mass of fluid, and the first or accumulation term of Eq. (2.25) is zero, reducing Eq. (2.26) to: tains aecuación constant mass fluid, volume and the first or accumulation term The of Eq. (2.25)volume is zero,then reducing conditions within the of control do not change with time. control con(2.26) con lo que la Eq.Eq. (2.26) to: to:(2.26) se reduce a: �(ρu A)fsA)=fs 0= 0 term of Eq. (2.25) is zero, reducing Eq. to: mass of fluid, and the first tains(2.26) a constant or accumulation �(ρu �(ρu A)fs = 0 �(ρu A)fs = 0 Eq. (2.26) to: The term “steady state” does not necessarily imply thatthat flowrates are constant, merely that thethe term “steady state” does necessarily flowrates constant, merely TheThe term “steady state” does notnot necessarily imply that are are constant, merely thatthat the �(ρu A)imply = 0 flowrates fsof inflow of mass is exactly matched by the outflow mass. The term “steady state” does not necessarily imply that flowrates are constant, merely thatconstantes, the inflow of mass is exactly matched by the outflow of mass. Elinflow término “estado estacionario” noby implica necesariamente que las relaciones de flujo sean . . of mass is exactly matched the outflow of mass. When there is but a single entrance and a single exit stream, the mass flowrate m is the inflow of mass is exactly matched by the outflow of mass. . The term “steady state” does not necessarily imply that flowrates are constant, merely that When is hacia but a dentro single entrance a single exit stream, mass flowrate is the sino sólo queWhen el flujo dethere masa es exactamente igual derrame dethe masa. there is but a single entrance andand a single exital stream, the mass flowrate m. ismthe same for both streams; When there is corriente butthen, amatched single entrance and acorriente single exit stream, the mass flowrate mdeismasa the es la inflow of mass isuna exactly by theyoutflow of mass. same for both streams; then, Cuando sólo hay que entra una que sale, la relación de flujo same for both streams; then, . ρ u A and − ρa single u A = 0 stream, the mass flowrate m is the sameambas for both streams; then, When there is butpor a single exit misma para corrientes; esto, entrance ρ22uρ222Au222 A −2ρ−11uρ111Au111 A =1 0= 0 ρ 2 u 2 A 2 − ρ1 u 1 A 1 = 0 same for both streams; then, ρ 2 u 2 A 2 − ρ1 u 1 A 1 = 0 02-SmithVanNess.indd 46 8/1/07 12:49:29 2.12. and Balances for Systems 2.12.Mass Mass andEnergy Balances forOpen Open Systems 2.12. Balances de masa yEnergy energía para sistemas abiertos 2.12. Mass and Energy Balances for Open Systems Mass and Energy Balances forfor Open Systems 2.12. Mass and Energy Balances Open Systems 2.12.2.12. Mass and Energy Balances for Open Systems 4747 47 47 4747 47 .. oror const==ρρ 2u 2 2AA 2 2==ρρ 1u 1 1AA 11 o 2u 1u .m==const . .m . or m ==const const =2ρ u2ρ22A1A =1ρ u111AA1 1 or m = = ρA or m or m = const=const ρ2= u 2=ρA 2A 1A 2u 2= 2u21= 11u 2uρ 1uρ Because specific volume reciprocal ofof density, Because specific volumeis isthe reciprocal density, Dado que el volumen específico es elthe recíproco de ladensity, densidad, Because specific volume is the reciprocal ofdensity, Because specific volume the reciprocal ofof density, Because specific volume is the reciprocal Because specific volume is theisreciprocal of density, . . uu1 1AA1 1 uu2 2AA2 2 uuAA == =uAu 1uAA1 A uAu222uAAA uuAA A.m 1 2u= . .u 1.m 2= 22 u A 1u 1= 1=1=== VV 2=2== VV m= ==11V2V m = m m= V VV V1 V1VV 1 2 V2VV 1V 2V2 Esta forma de la ecuación de continuidad es de uso frecuente. This form of the continuity equation finds frequent use. This form of the continuity equation finds frequent use. This form ofthe the continuity equation finds frequent use. form the continuity equation frequent This of continuity equation finds frequent use. This This form ofform theofcontinuity equation findsfinds frequent use. use. (2.27) (2.27)(2.27) (2.27) (2.27) (2.27) (2.27) The Energy Balance TheGeneral General Energy Balance El balance energético general The General Energy Balance The General Energy Balance The General Energy Balance The General Energy Balance Because Becauseenergy, energy,like likemass, mass,isisconserved, conserved,the therate rateofofchange changeofofenergy energywithin withinthe thecontrol controlvolume volume Because energy, like mass, isconserved, conserved, the rate of change ofcambio energy within the control volume Ya que la energía senet conserva, alis igual que lathe masa, lacontrol rapidez de energía dentro del volumen de Because energy, like mass, is conserved, rate of change ofde energy within the control volume Because energy, like the rate of change of energy within the control volume Because energy, like mass, ismass, conserved, the rate of change of energy within the control volume equals the net rate of energy transfer into the control volume. Streams flowing into and out ofof equals the rate of energy transfer into the volume. Streams flowing into and out equals the net rate of energy transfer into the control volume. Streams flowing into and out of control es igual a la rapidez neta de transferencia de energía en el volumen de control. Las corrientes que equals the net rate of energy transfer into the control volume. Streams flowing into and out of equals the net rate of energy transfer into the control volume. Streams flowing into and out of equals the net rate of energy transfer into the control volume. Streams flowing into and out of the thecontrol controlvolume volumehave haveassociated associatedwith withthem themenergy energyininitsitsinternal, internal,potential, potential,and andkinetic kinetic the control volume have associated with them energy insystem. internal, potential, and circulan hacia dentro ycontribute hacia fuera del volumen de control se asocian con energía en sus formas interna, pothethe control volume have associated with them energy in internal, potential, and kinetic control volume have associated with them energy inits itsits internal, potential, and the control volume have associated with them energy in internal, potential, and kinetic forms, and all toto the energy change ofofits the system. Each unit mass of akinetic stream forms, and all contribute the energy change the Each unit mass of akinetic stream 1 1 2 change forms, and all contribute to the energy change of the system. Each unit mass of a stream tencial ycarries cinética, y todas contribuyen al cambio de energía del sistema. Cada unidad de masa de una forms, and all contribute to the energy of the system. Each unit mass of a stream forms, and all contribute to the energy change of the system. Each unit mass of a stream forms, and all contribute to the energy change of the system. Each unit mass of a stream 2 UU++12 uu ++zg, carrieswith withitita atotal totalenergy energy zg,where whereuuisisthe theaverage averagevelocity velocityofofthe thestream, stream,z ziscorrienis 2 + zg, 1+12 u 1 + 2 where 2+ carries with it a total energy U where u is the average velocity of the stream, carries with it a total energy U u + zg, where u is the average velocity of the stream, zstream is te lleva consigo una energía total donde u es la velocidad promedio de la corriente, carries with it a total energy U u + zg, where u is the average velocity of the stream, z zisis z es su carries with it a total energy U + u + zg, u is the average velocity of the stream, z is itsitselevation acceleration ofofgravity. Thus, each 2 2 2and 2 level, elevationabove abovea adatum datum level, andggisisthe thelocal local acceleration gravity. Thus, each stream . .acceleration 2is its elevation above datum level, local acceleration ofgravity. gravity. Thus, each stream itsits elevation above a de datum and g1gis the local gravity. Thus, each stream elevación sobre un nivel referencia yis g+and es aceleración local deenergy laofgravedad. Así, cada corriente transporelevation aataat datum level, and local acceleration of Thus, each stream its elevation above aabove datum level, and g(U the acceleration of gravity. Thus, each stream 2is transports energy the rate (U u1laglocal +the zg) net transported into the system transports energy thelevel, rate + +the The net energy transported into the system .m.The .zg) .m. 12 2 2u.�� �energy � �transported 1 1 1 2 2 �� � transports energy atthe the rate + u + zg) m. The net energy transported into the system transports energy rate ++ u + zg) m. The net energy into the system ta energía en proporción de Por lo tanto, la energía neta transportada hacia adentro del transports energy atthe rate u + zg) m. The net transported into the system transports energy at theatrate (U +(U2(U u(U + zg) m. The net energy transported into the system . . 2���� 2 2−� � �m � , wherethe �zg �zg �� �+ bybythe UU++112 �u1 u2 .2+ theflowing flowingstreams streamsisistherefore therefore −��� theeffect effectofofthe theminus minus . fsfs, where . �.m 2 1 1 1 2 2 2 sistema por las corrientes que circulan es−� donde el efecto del signo menos con by the flowing streams is therefore U+ +u2zg u+ +zg zg where the effect ofthe the minus byby the flowing streams is is therefore −� ,fswhere thethe effect the minus the flowing streams therefore mm , ,,where effect of minus by the flowing streams is therefore −� U−� + UU u+ + mzg ,mwhere the effect of theofminus 2u+ fsof fsrate 2 out. sign The within signwith with“�” “�”isistotomake makethe theterm termread read2inin−− out. The rate offsenergy energyaccumulation accumulation withinthe the .. sign with “�” is to make the term read in − out. The rate of energy accumulation within the “Δ” sign essign hacer que término se lea entrada-salida. La rapidez de acumulación laand energía del voluwith “�” the term read −addition out. The rate of energy within thethe with “�” istoincludes tomake make the term − out.rate The rate of energy accumulation within sign with “�” isvolume toelis make the term read inread − in out. The energy accumulation within thedentro control this quantity inin totoof the heat transfer rate work rate: . de control volume includes this quantity in addition the heat transfer rate and work rate: .Q.Q .accumulation control volume includes this quantity in addition tothe the heat transfer rate Qand and work rate:del trabajo: mencontrol de control incluye esta además de la de transferencia de yrate: la rapidez volume includes this quantity inin addition the heat transfer rate Qcalor and work rate: control volume includes this quantity addition to heat transfer rate Q work rate: control volume includes this cantidad quantity in addition to rapidez thetoheat transfer rate Q and work �� � � �� �� .. d(mU d(mU)cv )cv 1� 1 2 2+ �� � �. + � �m �� � �..m �� � �� u zg = −� U + u + zg workrate rate = −� U + .Q.Q++work d(mU ) d(mU ) d(mU ) d(mU )cv dt fsfs. + 222 . . . cv cvcv 1 2 1 1 2 1 =−� U+ +u2zg u+ +zg zgm +Q+ Q+ + work rate Q work rate ==−� u+ work rate razón de trabajo u+ + mzg +mm Qfs+ + work rate = dt −� U−� + U2 U fs+ 2 fs fs 2 dt dtdtdt Q̇ Q̇Q̇ Q̇ Q̇ Q̇Q̇ u2 PP V,V,U,U,HH P P PP V,U, U,HH H V, V, V, U, HU, V, U, H u1 u1u1 u1 u1uu11 u 2u 2 u 2u 2uu22 Actual Actual Actual Perfil de Actual Actual Actual velocity velocity velocity velocidad velocity velocity velocity profile profile profile real profile profile profile Control Control Volumen Control Control Control Control volume volume de control volume volume volume volume P u 2u 2 u 2u 2uuu222 Ẇs ẆẆ ss Ẇ ẆsẆẆ s ss Figure Figure2.6: 2.6:Control Controlvolume volumewith withone oneentrance entranceand andone oneexit. exit. Figura 2.6: Volumen de control con una entrada y one una salida. Figure 2.6: Control volume with one entrance and one exit. Figure 2.6: Control volume with one entrance and one exit. Figure 2.6: Control volume with one entrance and exit. Figure 2.6: Control volume with one entrance and one exit. The Thework workrate ratemay mayinclude includework workofofseveral severalforms. forms.First, First,work workisisassociated associatedwith withmoving moving The work rate may include work of several forms. First, work is associated with moving The work rate may include work of several forms. First, work is associated with moving The work rate may include work of several forms. First, work is associated with moving The work rate may include work of several forms. First, work is associated with moving the flowing streams through entrances and exits. The fluid at any entrance or exit has a aset the flowing streams through entrances and exits. The fluid at any entrance or exit has setofof con el rapidez del trabajo puede incluir el trabajo defluid varias formas. Primero, el trabajo está asociado the flowing streams through entrances and exits. The fluid at any entrance or exit has a set theLa flowing streams through entrances and exits. The fluid at any entrance or exit has a set of the flowing streams through entrances and exits. The fluid at any entrance or exit has a set ofof the flowing streams through entrances and exits. The at any entrance or exit has a set of average properties, P, V , U , H , etc. Imagine that a unit mass of fluid with these properties average properties, P, V , U , H , etc. Imagine that a unit mass of fluid with these properties movimiento deproperties, las corrientes que circulan aImagine través las ymass las salidas. El fluido enproperties cualquier entrada average properties, UH etc. Imagine that unit of fluid with these properties average properties, V, ,V U ,etc. ,H etc. aentradas unit mass ofof fluid with these properties average P,P, ,,,U , ,H , ,etc. Imagine that a aunit fluid with these average properties, P, VP, ,U HV Imagine thatde athat unit mass ofmass fluid with these properties 02-SmithVanNess.indd 47 8/1/07 12:49:41 48 CHAPTER 2. The First Law and Other Basic Concepts CHAPTER 2. 2. The The First First Law and and Other Basic Basic Concepts Concepts 48 CHAPTER 48 CAPÍTULO 2. LaLaw primeraOther ley y otros conceptos básicos CHAPTER 2. The First Law and Other Basic Concepts 48 exists at an entrance or exit, as shown in Fig. 2.6 (at the entrance). This unit mass of fluid is exists at an an entrance or exit, as as shown shown in Fig. Fig.P,2.6 2.6 (at H, theetc. entrance). This unit mass of of fluid fluid is is o salidaexists tiene at un conjunto deor propiedades promedio, V, a(at U, Suponga quethe una masa unitaria de fluido entrance exit, in the entrance). This unit mass acted upon by additional fluid, here replaced by piston which exerts constant pressure exists at an by entrance or una exit, as here shown in Fig. 2.6 the entrance). This unit mass ofpressure fluid is Esta acted upon by additional fluid, hereoreplaced replaced bycomo a(at piston which exerts exerts the constant pressure con estas propiedades existe en entrada una salida, se muestra en la figura 2.6 (en la entrada). acted upon additional fluid, by a piston which the constant P. Theupon workbydone by this fluid, pistonhere in moving theby unit mass through the entrance is P V ,pressure and the acted additional replaced aque piston which exerts the constant .by P. masa The work work done by this thissobre piston in moving the unit unit mass through the entrance entrance ispistón V,, and and the the preunidad P. de del fluido actúa el in fluido adicional, aquí es sustituido por unis que ejerce The done piston moving the mass through the PPVquantities, work rate is (P V ) m. Because “�” denotes the difference between exit and entrance . . by P. The work done this piston indenotes moving the unit massbetween through the and entrance isa P V , and the work rate is (P (P V))m. m. Because “�” denotes the difference between exit and entrance quantities, sión constante P. El trabajo hecho por este pistón en movimiento, de la unidad de masa través de work rate is V Because “�” the difference exit entrance quantities, . the system when all entrance and exit sections are taken into accountlais entrada the netrate work done on work is (P V ) m. Because “�” denotes the difference between exit and entrance quantities, · .dedone the net work work done on the the(PV)m system whenque all entrance entrance andlaexit exit sectionsentre are taken taken into account account is y de es PV, ythe la net rapidez trabajo es . Dado “Δ” denota diferencia las cantidades de salida system when all and sections are into is −�[(P V )m] . on .. fs the net V work done on the system when cuando all entrance and exit sections are taken.into account is −�[(P V))m] m] . fs entrada,−�[(P el trabajo neto realizado en el sistema se consideran todas las secciones de entrada y . . s . In addition de sali. fs form of work is the shaft work indicated in Fig. 2.6 by rate W Another . −�[(P V ) m] . · Another form of of work work is is the the shaft shaft work work indicated indicated in in Fig. Fig. 2.6 2.6 by by rate rate W W.ss.. In In addition addition da es –Δ[(PV)m ]fs.be fsassociated form work Another may with isexpansion or contraction ofinthe control volume and there . may Another form es of el work thepor shaft work indicated Fig. 2.6laby rate WẆ In addition s .. Además, work may betrabajo associated with expansion or contraction of the the control volume and there may Otra forma de señalado la flecha en la figura 2.6 por rapidez el trabajo work may be associated with expansion or contraction of control volume and there s by W . . may be stirring work. These forms of work are all included in a rate term represented The . work maycon be la associated with expansion orall contraction of volume andby there may be stirring work. These forms forms of work are are all included in athe ratecontrol term represented by Wtrabajo . The The de agi. se puede asociar expansión o la contracción del volumen de control y ahí se considera be stirring work. These of work included in a rate term represented W . preceding equation may now beofwritten: be stirring work. These forms work are all included in a rate represented by W . The preceding equation may now be beestán written: tación. preceding Todas estas formas de trabajo incluidas en un término determ la rapidez representado por Ẇ. La equation may now written: �� � � preceding equation may now be written: . . d(mU )cv escribir ecuación anterior ahora se puede como: . . �� � �� + Q. − �[(P V )m] + W. 1 2 d(mU))cv cv = −� �� U + 21 u 2 + zg� m . − �[(P V )m] . d(mU .. �fs + Q .. fs = −� −� ��U U+ + 12uu2 + + zg zg �m m +W W dt )cv = . �[(P V )m] d(mU . fsfs + Q. − . fsfs + 1 2 2 dt dt = −� U + 2 u + zg m fs + Q − �[(P V )m]fs + W dt in accord with the definition of enthalpy, H = U + P V , leads to: Combination of terms Combination of of terms terms in in accord accord with with the the definition definition of of enthalpy, enthalpy, H H= =U U+ + PPVV,, leads leads to: to: Combination La combinación de términos de con the la��definición UU + .PV, lleva a: � � HH=.= Combination of termsd(mU in acuerdo accord definitionde of entalpía, enthalpy, + Pnos V , leads to: )cv with . � �� + Q. + W. �� 1 2 d(mU))cv . cv = −� �� H + 21 u 2 + zg� m . . d(mU fs . � +Q+W + zg zg �m m = −� −� ��H H+ + 12uu2 + dt )cv = d(mU . fs + Q. + W. dt dt = −� H + 212 u 2 + zg m fsfs + Q + W which is usually written: dt lo que por lo regular se escribe which is usually usually written:como: which is written: which is usually written: � � �� d(mU )cv ��H + 1 u 2 + zg�� m. �� = Q.. + W.. �� d(mU ) (2.28) (2.28) + � cv . +W . d(mU .. �fs = Q + zg � m (2.28) + � ��H H+ + 1212uu22 + dt ))cv fs = Q. + W. (2.28) d(mU cv + � 1 2 zg m. fs 2 dt dt (2.28) + � H + 2 u + zg m fs = Q + W dt de energía cinética de los balances de energía es la magnitud media de La velocidad u en los términos The velocity u in the kinetic-energy terms energy balances is the bulk-mean velocity · /rA. of . u=m The velocity velocity in the kinetic-energy terms of energy balances is the the en bulk-mean velocity un perla velocidad, como sethe define por lakinetic-energy ecuación, Los fluidos que circulan tuberíasprofile, exhiben The uu in the terms of energy is velocity as defined by equation, u = m/ρ A. Fluids flowing in balances pipes exhibit abulk-mean velocity as . . The velocity u in the kinetic-energy terms of energy balances is the bulk-mean as defined by the equation, u = m/ρ A. Fluids flowing in pipes exhibit a velocity profile, as fil de velocidad, como se equation, muestra en figura 2.6, elthe cual se incrementa elavalor cero envelocity las paredes (la as defined by the u la =from m/ρ A. Fluids flowing inno-slip pipes desde exhibit velocity profile, as . zero shown in Fig. 2.6, which rises at wall (the condition) to a maximum at as defined by the equation, u = m/ρ A. Fluids flowing in pipes exhibit a velocity profile, as shown in Fig. 2.6, which rises from zero at the wall (the no-slip condition) to a maximum at de un condición de no deslizamiento) hasta llegar a un máximo en el centro de la tubería. La energía cinética shown in Fig. 2.6, which rises from zero at the wall (the no-slip condition) to a maximum at the center of the pipe. Therises kinetic energy ofthe a fluid in a pipe depends on itstovelocity profile. shown in Fig. 2.6, which from zero at wall (the no-slip condition) a maximum the center of the the pipe. The kinetic energy of aa fluid fluid in pipe depends on its its velocity velocity profile. fluido en una tubería depende de sukinetic perfil de velocidad. Parain eland del flujo laminar, el perfil es deattipo pathe center of pipe. The energy of aacaso pipe depends on profile. For the case of laminar flow, the profile is parabolic, integration across the pipe shows the center of the pipe. The kinetic energy of a fluid in a pipe depends on its velocity profile. For the case of laminar flow, the profile is parabolic, and integration across the pipe shows rabólico, y the la integración a través dethe la tubería muestra el término de energía For case of laminar flow,should profile is parabolic, and integration across cinética the pipe shows In fully developed turbulent flow, thedebería more ser en that the kinetic-energy term properly be u 22. que For case ofun laminar flow, the completamente profile is be parabolic, anddeveloped integration across the pipe 2. En In fully fully developed turbulent flow, theshows more that the kinetic-energy kinetic-energy term should properly be u2 ..desarrollado, sentidothat estricto u flujo turbulento que es el caso más común en la prácIn turbulent flow, the more the term should properly u common case in practice, the velocity across the portion of the pipe isflow, not the far more from 2 . major In fully developed turbulent that the kinetic-energy term should properly be u common case in practice, the velocity across the major portion of the pipe is not far from 2 tica, la common velocidad a través de la parte principal de la tubería no se aleja mucho de lo uniforme, y la expresión case in practice, the velocity across the major portion of the pipe is not far from uniform, and the expression u 2/2, as used in thethe energy equations, is more nearly correct. common casethe inexpression practice, the velocity across major portion of the pipe is not far from uniform, and the expression u2 /2, /2, asenergía, used ines themucho energy equations, is more more nearly correct. u2/2, como seAlthough emplea en las ecuaciones de más correcta. uniform, and u as used in the energy equations, is nearly correct. Eq. (2.28) isu 2an energy balance of reasonable generality, it has correct. limitations. uniform, theEq. expression /2, as usedbalance inenergía the energy equations, is more tiene nearly Although Eq. (2.28)es isun an energy balance of reasonable reasonable generality, it has hassus limitations. Aunque la and ecuación (2.28) balance de en general razonable, limitaciones. En Although (2.28) is an energy of generality, itcontrol limitations. In particular, it reflects the tacit assumption that the center of mass of the volume is Although Eq. (2.28) is an energy balance of reasonable generality, it has limitations. In particular, it reflects the tacit assumption that the center of mass of the control volume is isDe este particular, refleja la suposición tácita de que el centro de masa del volumen de control es estacionario. In particular, it reflects the tacit assumption that the center of mass of the control volume stationary. Thus no terms for kineticand potential-energy changes ofofthe fluid in the control In se particular, ittérminos reflects thefor tacit assumption that the center ofymass thedel control volume is stationary. Thus no terms terms for kineticand potential-energy changes of the the fluid in the the control modo, no incluyen para elkineticcambio de las energías cinética potencial fluido encontrol el volumen de stationary. Thus no and potential-energy changes of fluid in volume are included. For virtually all applications of interest to chemical engineers, Eq. (2.28) stationary. Thus no terms for kineticand potential-energy changes of the fluid in the control volume are included. For virtually all applications of interest to chemical engineers, Eq. (2.28) control.volume En realidad, paramany todas las aplicaciones de interéskineticpara losand ingenieros químicos, la ecuación (2.28) es are included. For virtually allapplications, applications of interest topotential-energy chemical engineers, Eq. (2.28) is adequate. For (but not all) changes in the volume are included. virtually all applicationskineticoflos interest chemical engineers, Eq.yin (2.28) is adequate. For(aunque manyFor (but not all) all) applications, kineticandtopotential-energy potential-energy changes inpotencial the adecuada. Para muchas no todas las aplicaciones), ento: las energías cinética en is adequate. For many not applications, and changes the flowing streams are also(but negligible, and Eq. (2.28)kineticthencambios simplifies is adequate. For many (but not all) applications, and potential-energy changes in the flowing streams are also negligible, and Eq. (2.28) then simplifies to: las corrientes flujo también despreciables, por esto la ecuación flowingdestreams are alsoson negligible, and Eq.y (2.28) then simplifies(2.28) to: se simplifica a: flowing streams are also negligible, . . to: d(mUand )cv Eq. (2.28) . then simplifies . +W . d(mU))cv (2.29) . fs = Q cv + �(H m) . . d(mU . =Q Q. + +W W. (2.29) (2.29) + �(H �(Hm) m) dt )cv + fs = (2.29) d(mU . fs dt dt (2.29) + �(H m)fs = Q + W dt 48 Example 2.12 Example 2.12 Example 2.12 Ejemplo 2.12 Show that Eq. 2.12 (2.29) reduces to Eq. (2.3) for the case of a closed system. Example Show that Eq. (2.29) reduces to Eq. (2.3) for the case of a closed system. Show that Eq. (2.29) reduces to Eq. (2.3) for the case of a closed system. Demuestre que la Eq. ecuación se reduce a la ecuación (2.3)ofena el caso system. de un sistema cerrado. Show that (2.29)(2.29) reduces to Eq. (2.3) for the case closed 02-SmithVanNess.indd 48 8/1/07 12:49:48 2.12. 2.12. Mass Mass and and Energy Energy Balances Balances for for Open Open Systems Systems 49 49 49 2.12. Balances de masa y energía para sistemas abiertos Solution Solution 2.12 Solución 2.12 2.12 The second Eq. is streams, and El segundo de laof ecuación (2.29) se omitein la absence ausenciaof deflowing corrientes de flujo, The término second term term of Eq. (2.29) (2.29) is omitted omitted inenthe the absence of flowing streams, andy por lo equation is then multiplied by dt: tanto la the ecuación se multiplica por dt: the equation is then multiplied by dt: .. .. d(mU = Q dt + W dt d(mU))cv cv = Q dt + W dt Integration time Integration over time gives: gives: Integrando a travésover del tiempo dado: = �(mU �(mU))cv cv = �� tt22 tt11 �� .. Q dt + Q dt + tt22 tt11 .. W W dt dt tt = Q + W �U �U Q+ ΔU t = = Q +W W The Q and W terms are defined by the integrals The Q and W terms are defined by the integrals of of the the preceding preceding equation. equation. Los términos Q y W se definen mediante integrales de la ecuación anterior. o or or Equation Equation (2.29) (2.29) may may be be applied applied to to aa variety variety of of processes processes of of aa transient transient nature, nature, as as illusillusLa ecuación (2.29) se puede aplicar a una variedad de procesos de naturaleza transiente (fenómeno trated trated in in the the following following examples. examples. momentáneo que sucede en un sistema previo a lograr una condición de estado estacionario), como se muestra en los siguientes ejemplos. Example 2.13 An An evacuated evacuated tank is is filled filled with with gas gas from from aa constant-pressure constant-pressure line. line. What What is is the the relation relation Ejemplo 2.13 tank between the enthalpy of the gas in the entrance line and the internal energy of the gas between the enthalpy of the gas in the entrance line and the internal energy of the gas in the between gas the Se llena gas unNeglect tanque heat vacío desde una línea the a presión constante. incon the tank? tank? Neglect heat transfer transfer between the gas and and the tank. tank.¿Cuál es la relación entre la entalpía del gas en la línea de entrada y la energía interna del gas en el tanque? Ignore la transferencia de calor entre el gas y el tanque. Solution Solution 2.13 2.13 The The tank tank with with its its single single entrance entrance serves serves as as the the control control volume. volume. Because Because there there is is .. Soluciónno 2.13 no expansion expansion work, work, stirring stirring work, work, or or shaft shaft work, work, W W= = 0. 0. If If kinetickinetic- and and potentialpotential- energy changes are Eq. becomes: energy changes are negligible, negligible, Eq. (2.29) (2.29) becomes: El tanque con una sola entrada sirve como volumen de control. Debido a que no hay trabajo de · expansión, de agitación o en la flecha, W = 0. Si los cambios en las energías cinética y potencial d(mU ) .. �� d(mU )tank tank − H ��m = 00 − H = m son despreciables, la ecuación (2.29) será: dt dt d (mU ) tanque where stream −H ′ m ′ = and 0 the where the the prime prime (�) (�) identifies identifies the the entrance entrance stream and the minus minus sign sign is is required required dt mass balance is: because it is an entrance stream. The because it is an entrance stream. The mass balance is: donde la prima (′) identifica la corriente de entrada y se requiere el signo menos porque se tiene dm .. � dmtank m una corriente de entrada. El balance de masa es: tank = m� = dt dt dmtanque m ′ = yields: Combining Combining these these two two balance balance equations equations yields: dt d(mU ) tank tank d(mU )tank − Hde dm � dm Al combinar los resultados de las dos ecuaciones balance: tank − H � dt = = 00 dt dt dt d (mU ) tanque dmtanque − H′ =0 dt dt 02-SmithVanNess.indd 49 8/1/07 12:49:54 50 CHAPTER 2. The First Law and ley Other Basic Conceptsbásicos 50 CAPÍTULO LaLaw primera y otros CHAPTER 2. The2. First and Other Basicconceptos Concepts 50 5050 CHAPTER CHAPTER2.2.The TheFirst FirstLaw Lawand andOther OtherBasic BasicConcepts Concepts CHAPTER 2. The First Law and Other Basic Concepts 50 Multiplicando por dtby e integrando a través over del tiempo (observe es constante) se obtiene: Multiplying dt and integrating time (noting thatque H � H′ is constant) gives: Multiplying by dt and integrating over time (noting that H� �� is constant) gives: Multiplying Multiplyingbybydtdtand andintegrating integratingover overtime time(noting (notingthat thatHH isisconstant) constant)gives: gives: Δ(mU) –time H′Δm =00 H � is constant) gives: Multiplying by dt and integrating over− (noting that H � �m �(mUtanque )tank tanque tank = − H� ���m tank =0 �(mU )tank �mtank �(mU �(mU)tank )tank−−HH��m tank==00 � (m − HH �m Whence m�(mU tank tank 2 U2 −)m 1 U− 1 = 2 =m01 ) � Whence m 22UU22−– m 11U11 ==HH′� (m De donde –−mm1)1))) � (m22− Whence Whence mmm 2U 2U 2 2−−mm 1U 1U 1 1==HH(m 2 2 −mm 11 � (m conditions where subscripts 1 and 2mdenote initial and final in the tank. U − m U = H Whence 2 2 initial 1 1and final conditions 2 − m1) where subscripts 1 and 2 denote in the tank. donde los subíndices 1 ymass denotan condiciones inicial yconditions final Debido a que la masa where where subscripts subscripts 121and andin 22denote denote initial and and final conditions intanque. inthe thetank. tank. Because the the tankinitial initially isfinal zero, m 1 =en0;elthen, Because the mass in2the tank initially isfinal zero,conditions m 1 = 0; then, where subscripts 1 and denote initial and in the tank. en el tanque es inicialmente cero, m = 0; entonces, Because Becausethe themass massininthe thetank initiallyisiszero, zero,mm1 1==0;0;then, then, 1tankinitially � Because the mass in the tank initially m 1 = 0; then, U2 =isHzero, � = H U H� � � UUU2222===HH′ =H U2heat a result showing that in the absence of transfer the energy of the gas contained a result showing that inenthe absencede of heat transfer de thecalor, energy of the gas contained un resultado que muestra que, ausencia laof energía en el added. gas contenido awithin aresult result showing showing that that the the absence absence ofoftransferencia heat heat transfer theenergy energy ofthe thegas gas contained contained the tank at theinin end of the process istransfer equal tothe enthalpy of the gas within the tank at the end of the process is equal to the enthalpy of the gas added. awithin result showing that the absence ofentalpía heat transfer the energy ofof the gasgas contained en el tanque althe final delatat proceso es igual a la deltoto gas agregado. within the tank tank the thein end end ofof the theprocess process isisequal equal the the enthalpy enthalpy ofthe the gas added. added. within the tank at the end of the process is equal to the enthalpy of the gas added. Example 2.14 2.14 Example Example Example 2.14 2.14 heated tank for hot water contains 190 kg of liquid water at An insulated, electrically Ejemplo 2.14 electrically Example 2.14 heated An◦insulated, tank for hot water contains 190 kg of liquid water at An Aninsulated, insulated, electrically heated heated tank tankfor hot hotwater water contains containsfrom 190 190kg kgoftank ofliquid liquid atat 60 C when aelectrically power outage occurs. Iffor water is withdrawn the at water awater steady 60◦ C when a power outage occurs. If water is withdrawn from the tank at a steady . . ◦C ◦insulated, −1 An electrically heated tankitIffor hot water contains 190 kg tank of liquid water Un tanque aislado calienta agua eléctricamente 190 kg deofagua líquida 60 °Catcuando 60 60 Cof when when a0.2 apower power outage outage occurs. occurs. If water water isthe iswithdrawn withdrawn from from the tank atat steady steady , how long will take forcontiene temperature the water inaaa the tank rate m. = que kg s−1 ◦ Cofwhen , how long will it take for the temperature of the water in the tank rate m = 0.2 kg s . . −1 −1 ◦ ◦ 60 a power outage occurs. If water is withdrawn from the tank at a steady ocurre rate una interrupción del suministro eléctrico. Siwater el agua se retira del tanque aC, una , ,how howAssume long long will willcold itittake take for for the thetemperature temperature of ofthe the water inin the thetank tankestable rate of ofmm ==0.2 0.2 kg kg to drop from 60 toss35 C? enters the tank at 10 andproporción negligible ◦water .–1 −1◦◦,◦C? to drop from 60kg tos35 Assume cold water enters the tank at 10 andinen negligible ◦ C, ◦C, how longliquid willcold itwater take for the temperature of the water the tank rate oflosses = 0.2 de m· = to 0.2 kg sm , ¿cuánto tiempo debe transcurrir para que la temperatura del agua el tanque todrop drop from from 60 60toto 35 35tank. C? C?Assume Assume cold water water enters the the tank tank at at 10 10 C, and and negligible negligible heat from the For let enters C = C = C, independent of T and P. disV ◦ C? Assume ◦ C, and heat losses from the tank. For el liquid water let Centers C PPtanque =tank C, independent of Tlasand P. V = el to 60 to 35tank. cold water 10°C, minuyaheat dedrop 60 afrom 35from °C? Suponga que agua fría entra aat10 y que pérdidas de heat losses losses from the the tank.For Forliquid liquid water water let letCC ==CCPthe C,independent independent ofofnegligible TTand and P.P. V Ven P==C, heat losses from the tank. For liquid water let Clíquida = supone C, independent and P. indeV = C Pse calor desde el tanque son despreciables. Para el agua que C V =ofCTP = C son Solution 2.14 pendientesSolution de T y P . 2.14 . 2.14 . Solution Solution 2.14 . = 0. Additionally, assume perfect mixing of the contents of the Here, .Q.. = W Solution 2.14 .. = Here, Q = W 0. Additionally, assume perfect mixing of the contents of the Solución 2.14 Here, Here,Q Q = WW 0. 0.Additionally, Additionally, assume perfect mixing mixing ofoftank the thecontents contents ofthe the tank; this implies that the propertiesassume of the perfect water leaving the are thoseofof the . =implies . ==that tank; this the propertiesassume of the water leaving theoftank are those of of the the ·W Here, Q· the =implies =that 0. the Additionally, perfect mixing the contents tank; tank; this this implies that the properties properties of of the the water water leaving leaving the the tank tank are are those those of of the the water in tank. With the mass flowrate into the tank equal to the mass flowrate En este water caso, Q Wtank. = 0. Además, se supone unainto mezcla perfecta detolos contenidos del tanque; in =the Withthe theproperties mass flowrate theleaving tank equal theare mass flowrate tank; this implies that of theinto water theand tank those of and the water water in inthe the tank. tank.With With the the mass flowrate flowrate the thetank tank equal to tothe themass mass flowrate flowrate out, m is constant; moreover, the differences between inlet outlet kinetic cv esto implica que las propiedades delmass agua que deja into el tanque sonequal las mismas que las deland agua en el out, m is constant; moreover, the differences between inlet and outlet kinetic cv the tank. With the mass flowrate into the tank equal to the mass flowrate water in out,mmcvla is constant; constant; moreover, moreover, the thedifferences differences between between inlet inletand and outlet outlet kinetic and andde flujo potential energies can be neglected. Equation (2.29) istanque therefore written: cvis tanque.out, Como relación de flujo de masa hacia el interior delis es igual akinetic la relación potential energies can be neglected. Equation (2.29) therefore written: out, m is constant; moreover, the differences between inlet and written: outlet kinetic and potential energies energies bebeneglected. neglected. Equation Equation isistherefore therefore written: de masapotential hacia cv afuera, mcv can escan constante; por otra parte, (2.29) se(2.29) pueden despreciar las diferencias entre las dU Equation potential energies can be neglected. (2.29) is therefore written: . energías cinética y potencial de entrada y de+salida. PorHlo) tanto, . = 0 la ecuación (2.29) se escribe: m(H − m dU dU mdU . . − H11) = 0 dt + m(H m(H −−HH mmdU dt ++m(H 1 )1 )==00 . + m(H −H m dtdtrefer 1 ) = 0 of the tank and H is the where unsubscripted quantities to the contents dt refer to where unsubscripted quantities the contents of the tank and H11 is the the where whereunsubscripted unsubscripted quantities quantities refer refertoto the the contents contents ofofthe theCtank tank andHH specific enthalpy ofcarecen the water entering the tank. With C = C, 1 1isisthe P =and donde las cantidades queof de entering subíndices se refieren y H1 es la specific enthalpy the water thethe tank. With al CofVVcontenido = Ctank = del C, tanque where quantities refer the to contents the and specific specificunsubscripted enthalpy enthalpyofofthe the water waterentering entering thetank. tank. With WithCC = =CCPPP==C, C, H1 is the V V entalpíaspecific específica deldU agua quedT entra entering en el tanque. DadoWith que C CV ==CCP == C,C, enthalpy water the tank. V P dT dU of=the = C(T − T ) C and H − H 1 dT dU dU CdT and H − H1 = C(T − T11) dt == dt y = = C(T C(T − − T T ) = C C and and H H − − H H dt dt 11 1 1) dT dU dtdt = C dtdt = C(T − T and H − H 1 1) The energy balance thendtbecomes, on rearrangement, dt Theenergético energy balance then becomes, on rearrangement, El balance será en ese caso, después de una reordenación, The Theenergy energybalance balancethen thenbecomes, becomes,ononrearrangement, rearrangement, m dT The energy balance then becomes, on rearrangement, dt = −mm . dT dTT m dt = − m . TdT − dtdt==−− m − T11 . . T dT T−−TT 11 dt = − mm . Tarbitrary Integration from t = 0 (where T = Ttiempo to time yields: 0) m T − T1 time Integrando desde t =from 0 (donde = T0) en unT arbitrario t sett yields: obtiene: Integration t = 0 T(where T = ) to arbitrary 0 Integration Integrationfrom fromt t==00(where (whereTT==TT arbitrarytime yields: 0 )0 )toto �timet tyields: �arbitrary � � m0 ) to T − T1�� Integration from t = 0 (where T = T arbitrary time t yields: � � m T − T t =− m 1 . ln −TT T t = −mm 1 1� . ln �TTT−− t t==−−m − TT111 . .lnln T00 − 0 0−−TT 11 t = − mm . ln TT T0 − T1 m 02-SmithVanNess.indd 50 8/1/07 12:50:02 51 2.12.Mass Massand andEnergy EnergyBalances Balancesfor forOpen OpenSystems Systems 51 2.12. Mass and Energy Balances for Open Systems 51 2.12. 2.12. Mass and Energy Balances for Open Systems 51 2.12. Balances de masa y energía para sistemas 51 2.12. Mass and Energy Balances for Openabiertos Systems Substitution of numerical values into this equation gives, for the conditions of this Substitution of numerical values into this equation gives, for the conditions of this Substitution of numerical values into this equation gives, for the conditions of this problem, problem, Substitution of numerical values intoecuación gives, para for the of de thiseste proSustituyendo los valores numéricos en esta se�� obtiene, lasconditions condiciones � equation problem, �this Substitution of numerical values into� this equation 190 35− −10 10� gives, for the conditions of this 190 35 − 10 blema, problem, 190 35 658.5s s ==− −−190 ln lnln� 35 − 10 � = ==658.5 problem, tt t= 0.2 �60 60− 10� 658.5 s 0.2 60 −−10 0.2 t = − 190 ln 35 − 10 10 = 658.5 s 60 − 10temperature t minutes = − 0.2for lnthe = 658.5 in s the Thus,ititittakes takesabout about11 11 minutes for the water temperature thetank tankto dropfrom from Thus, takes about 11 water tank totodrop drop from Thus, minutes for the water temperature ininthe 0.2 60 − 10 ◦ ◦ C. 60 to 35 ◦ C. 60 to 35 Thus, it takes about 11 minutes for the water temperature in the tank to drop from C. about 11 minutes for the water temperature in the tank to drop from 60 to 35 Thus, it ◦takes Así, le toma C.de 11 minutos a la temperatura del agua en el tanque disminuir de 60 a 35 °C. 60 tocerca 35 ◦ 60 to 35 C. 51 Energy Balancespara forSteady-State Steady-State Flow Processes Energy Balances for Steady-State Flow Processes Balances energéticos los procesosFlow de flujo en estado estacionario Energy Balances for Processes Energy Balances for Steady-State Flow Processes Flowprocesses processes forwhich which theaccumulation accumulationterm termof Eq.(2.28), (2.28),d(mU d(mU))cv ) /dt, /dt,is zeroare aresaid saidtoto Flow processes for which the accumulation term ofofEq. Eq. (2.28), d(mU /dt, isiszero zero are said Energy Balances for Steady-State Flow Processes Flow for the cvcv Los procesos de flujo para los cuales el término de acumulación de la ecuación (2.28), d(mU) /dt estocero, se cv occurprocesses steadystate. state. Asdiscussed discussed withrespect respect themass massbalance, balance, this means that the mass occur atatsteady steady state. As discussed with respect to the mass balance, this means that the mass Flow for which the accumulation term to oftothe Eq. (2.28), d(mU )this /dt, is zero are said to cv at As means that the mass dice queoccur ocurren en estado estacionario. Alwith igual que con el análisis del balance de ismasa, esto significa Flow processes for which the accumulation term of Eq. (2.28), d(mU ) /dt, zero are said to que cv of the system within the control volume is constant; it also means that no changes occur with of the system within the control volume is constant; it also means that no changes occur with occur at steady state. As discussed with respect to the mass balance, this means that the mass the system within the control volume is constant; it also means that no changes occur with la masaof del sistema dentro del volumen de control es constante; también significa que no ocurre ningún camoccur steady state.the As discussed with respect to the mass balance, means that the mass time in theproperties properties thefluid fluid within the control norat entrances and exits. No time inat the properties ofofthe the fluid within the control volume nor atatthat itsitsthis entrances and exits. No of thein system within control volume is constant; itvolume also means no changes occur with the of within the control volume nor its entrances and exits. No bio contime el tiempo en las propiedades del fluido dentro del volumen de control ni en sus entradas ni salidas. No of theinsystem within the control is it also means noThe changes occur with expansion ofthe thecontrol control volume ispossible possible underthese these circumstances. The only work the expansion of the control volume iswithin possible under these circumstances. only work ofof the time theof properties of the fluidvolume theconstant; control volume nor at that its entrances and exits. No expansion volume is under circumstances. The only work of the hay expansión del volumen de control bajo estas circunstancias. El único trabajo del proceso es el de la flecha time in the properties of the fluid the under control volume nor atbecomes: its entrances exits. No process shaft work, and the general energy balance, Eq. (2.28), becomes: process isisshaft shaft work, and the general energy balance, Eq. (2.28), expansion of thework, control volume iswithin possible these circumstances. The onlyand work of the process is and the energy balance, Eq. (2.28), becomes: y el balance energético general, de la general ecuación (2.28), será:these expansion of the control volume is possible under circumstances. The only work of the process is shaft work, and the general energy balance, Eq. (2.28), becomes: �� �� �� ��� Eq. (2.28), process is shaft work, and the �� general energy balance, becomes: . . . . � .W ..m�. ==Q.Q++W (2.30)(2.30) ����H HH+ ++112u12u2u2 2+ ++zg zgzg� m m (2.30) � (2.30) . �fsfsfs= Q. + W. ss s 21 2 �� � � H + 21 u 2 + zg m. fs = Q. + W. s (2.30) � H + 2 u + zg m fs = Q + Ws (2.30) Although “steady state” does not necessarily imply “steady flow,” the usual application Although “steady state” does not necessarily imply “steady flow,” the usual application ofof ecuastate” does not necessarily imply“flujo “steady flow,” la theaplicación usual application of AunqueAlthough el “estado“steady estacionario” no implica necesariamente estable”, usual de esta this equation is to steady-state, steady-flow processes, because such processes represent the this equation is to steady-state, steady-flow processes, because such processes represent the Although “steady state” does not necessarily imply “steady flow,” the usual application of this equation is to steady-state, steady-flow processes, because such processes represent the ción es Although para procesos en8 8estado estacionario y de flujoimply estable, ya queflow,” tales procesos elof modelo “steady state” does not necessarily “steady the usualrepresentan application industrial norm. 8 industrial norm. this equation is to steady-state, steady-flow processes, because such processes represent the 8 industrial norm. industrial. this equation is 8specialization tospecialization steady-state,results steady-flow processes, because such processes represent the further results when thecontrol control volume hasbut but oneentrance entrance and one y una AAfurther further when the control volume has but one entrance and one industrial norm. A when the volume one and one Un resultado másspecialization cuando el volumen dehas control tiene sólo una entrada 8especializadoresults . obtiene .se industrial norm. . exit. The same mass flowrate m then applies to both streams, and Eq. (2.30) reduces to: exit. The same mass flowrate m then applies to both streams, and Eq. (2.30) reduces to: A further specialization results when the control volume has but one entrance and one exit. The same mass flowrate m then applies to both streams, and Eq. (2.30) reduces to: salida. Por lo Atanto se aplica la misma. relación de the flujo de masa a ambas corrientes, y la ecuación specialization when control volume has one reduces entrance and one (2.30) exit. Thefurther same mass flowrate m.results then and Eq.but (2.30) to: �� streams, �� applies to both se reduce a: . . . . � � . . exit. The same mass flowrate m then applies tozgzg both and .W Eq. (2.30) reduces to: (2.31) .mstreams, (2.31) +112u12u2u2 2+ ++ m ==Q.Q++W ��� HH+ + zg � m (2.31) � H . = Q. + W. ss s 21 2 � � (2.31) (2.31) � H + 21 u 2 + zg m. = Q. + W. s + zg m =case Q +and Ws “�” (2.31) �omitted H + 2inuinthis wheresubscript subscript“fs” “fs”has hasbeen been omitted this simple case and “�”denotes denotesthe thechange change from where subscript “fs” has been simple denotes the change from where this simple case and “�” from . en esteincaso . omitted donde elwhere subíndice “fs” ha sido omitido simple y “Δ” denota el cambio de la entrada a la salida. . entrance to exit. Division by m gives: entrance to exit. Division by m gives: subscript “fs” has been omitted in this simple case and “�” denotes the change from entrance to· exit. Division by m. omitted gives: in this simple case and “�” denotes the change from where subscript “fs” has been Dividiendo entre m se obtiene: entrance to exit. Division by m. gives: .. ..W. entrance to exit. Division by ���m gives: ��� Q Q..Q W W ss 2 1 2 1 ���H HH+ ++12u2u2u + ++zg zgzg� = ==Q.. .+ ++ ..s. = ==Q QQ+ ++W WW � ss s .m W .m s 21 2 m m � � m. s = Q + Ws � H + 21 u 2 + zg = m Q. + W � H + 2 u + zg = m. + m. = Q + Ws m m �u 22 2 �u �u or �H + (2.32a) + g �z = or �H + (2.32a) + g �z = Q ++W WW o (2.32a) 2 or �H + �u (2.32a) + g �z = QQ+ ss s 2222 + g �z = Q + Ws or �H + �u (2.32a) or �H + 2 + g �z = Q + Ws (2.32a) 2 Esta ecuación es la expresión matemática deexpression la primera ley para un law proceso en estado estacionario y flujo estaThis equation is the mathematical expression of the first law for a steady-state, steady-flow This equation is the mathematical of the first for a steady-state, steady-flow This equation is the mathematical expression of the first law for a steady-state, steady-flow ble entreThis unaequation entrada yisuna salida. Todos los términos representan energía por unidad demass masa del fluido. process between one entrance and one exit. All terms represent energy per unit mass of fluid. process between one entrance and one exit. All terms represent energy per unit of fluid. the mathematical expression of the first law for a steady-state, steady-flow process between one entrance and one exit. All of terms represent energy per unit mass of fluid. This equation isthe the mathematical expression the firstthe law for steady-state, Enprocess todas las ecuaciones de balance energético escritas hasta ahora sea supone que lasteady-flow unidad de In all of energy-balance equations so far written, the energy unit is presumed to be theenergía between one entrance and one exit. All terms represent energy per unit mass of fluid. In all of the energy-balance equations so far written, energy unit is presumed to be the In between all of theone energy-balance equations so farterms written, the energy unitper is unit presumed to be the process and one exit. All represent energy mass of fluid. es el joule, deIn acuerdo con elentrance sistema deequations unidades SI. Para el sistema inglés deisunidades de ingeniería, joule, in accord with the SI system of units. For the English engineering system of units, the los joule, in accord with the SI system of units. For the English engineering system of units, all of the energy-balance so far written, the energy unit presumed to be the joule, In in accord the SI system of units.soFor English of units, of thewith energy-balance equations far the written, the engineering energy unit issystem presumed to be the the joule, in all accord with the SI system of units. For the English engineering system of units, the joule, in accord with the SI system of units. For the English engineering system of units, the 88 Anexample exampleof steady-stateprocess processthat thatis notsteady steadyflow flowis waterheater heaterin whichvariations variationsin flowrate rateare are An example ofofaa asteady-state steady-state process that isisnot not steady flow isisaa awater water heater ininwhich which variations ininflow flow rate are 8 An 8 An compensated exactly compensated bychanges changes inthe therate rate ofheat transfer sothat that throughout remain constant. exactly by changes inprocess the rate ofque heat transfer so that temperatures throughout remain constant. Un ejemplo de un proceso de estado estacionario notransfer tiene flujo estable es unheater calentador de agua en el cual las variaciones en el example of a by steady-state that isheat not steady flow istemperatures atemperatures water in which variations in flow rate are exactly compensated in of so throughout remain constant. 8 An example of a steady-state process that is not steady flow is a water heater in which variations in flow rate are caudal sonexactly compensadas exactamente con los cambios la rapidez de calor, de modo remain que las constant. temperaturas permanezcan compensated by changes in the rate ofenheat transferdesotransferencia that temperatures throughout compensated by changes in the rate of heat transfer so that temperatures throughout remain constant. constantesexactly en todas partes. 8 02-SmithVanNess.indd 51 8/1/07 12:50:10 52 CHAPTER CHAPTER 2. 2. The The First First Law Law and and Other Other Basic Basic Concepts Concepts 52 52 CAPÍTULO 2. La primera ley y otros conceptos básicos kinetickinetic- and and potential-energy potential-energy terms, terms, wherever wherever they they appear, appear, require require division division by by the the dimensional dimensional términosconstant de las energías cinética y1.8). potencial, dondequiera que aparezcan, requieren la división entre la consg (Secs. 1.4 and In this event Eq. (2.32a), for example, is written: c constant gc (Secs. 1.4 and 1.8). In this event Eq. (2.32a), for example, is written: tante dimensional gc (secciones 1.4 y 1.8). En este caso, por ejemplo, la ecuación (2.32a) se escribe: 2 �u �u 2 + gg �z = Q + Ws �H + �H + 2gc + gc �z = Q + Ws 2gc gc (2.32b) (2.32b)(2.32b) the usual for �H and the kinetic energy, and are Aquí, laHere, unidad para la energía cinética, energía energy, potencial y work el trabajo Here, thehabitual usual unit unit forΔH �Hy Q andesQ Qelis is(Btu); the (Btu); (Btu); kinetic energy,lapotential potential energy, and work are se ex−1 –1 usually expressed as (ft lb ). Therefore the factor 778.16(ft lb )(Btu) must be used with the −1 f f presan generalmente como (pie lb ). Por lo tanto, el factor 778.16(pie lb )(Btu) se debe utilizar con f usually expressed as (ft lbff). Therefore the factor 778.16(ft lbf )(Btu) must be used with the los térappropriate terms to put them all in consistent units of either (ft lb ) or (Btu). f minos apropiados para poner a todos en unidades convenientes en (pie lb ) o en (Btu). appropriate terms to put them all in consistent units of either (ft lbff ) or (Btu). In many applications, kineticand potential-energy terms are omitted, they are En muchas aplicaciones se omiten los términos de las energías cinética y potencial porque In many applications, kinetic- and potential-energy terms are omitted, because because theyson aredespre9 For such cases, Eqs. (2.32a) and (2.32b) reduce to: 9 En compared with other terms. 9 ciables negligible en comparación con otros términos. estos casos las ecuaciones (2.32a) y (2.32b) se reducen a: negligible compared with other terms. For such cases, Eqs. (2.32a) and (2.32b) reduce to: (2.33) (2.33) (2.33) �H �H = =Q Q+ +W Wss Esta expresión de la primera ley para un proceso en estado estacionarioprocess y flujo estable es análoga a la ecuación This This expression expression of of the the first first law law for for aa steady-state, steady-state, steady-flow steady-flow process is is analogous analogous to to Eq. Eq. (2.3) (2.3) (2.3) para un proceso sin flujo. Sin embargo, más que la energía interna,energy la entalpía esthermodynamic una propiedad termofor a nonflow process. However, enthalpy rather than internal is the for a nonflow process. However, enthalpy rather than internal energy is the thermodynamic dinámica de gran importancia. property property of of importance. importance. A Calorimeter Enthalpy Measurements Calorímetro flujo parafor mediciones la entalpía A Flow Flow de Calorimeter for Enthalpy de Measurements The of (2.32) (2.33) to the solution of problems requires enThe application application of Eqs. Eqs. (2.32) (2.32)yand and (2.33) the solution of practical practicalprácticos problems requires enLa aplicación de las ecuaciones (2.33) paratolaits solución de problemas requiere de valores de thalpy values. Because H is a state function, values depend only on point conditions; once thalpy values. Because H is a state function, its values depend only on point conditions; once entalpía.determined, Porque H es una función de estado, sus valores dependen sólo de condiciones puntuales; una vez they may be for subsequent use for the same sets of conditions. To determined, they may be tabulated tabulated for subsequent use forvez the que same sets of el conditions. To this thisde condeterminados pueden ser tabulados para el uso consecutivo cada se tenga mismo conjunto end, Eq. (2.33) may be to laboratory processes designed for enthalpy measurements. end, Eq. (2.33) may be applied applied to se laboratory processes measurements. diciones. Con este fin, la ecuación (2.33) puede aplicar a losdesigned procesosfor de enthalpy laboratorio diseñados específicamente para medir la entalpía. Section 1 Section 1 Sección 1Heater Heater T2 T2 T2 P2 P2 P2 Calentador Constant Constant temperature temperature Baño a bath bath temperatura constante Supply Supply Suministro Valve Valve Válvula Discharge Discharge Descarga Applied Applied emf emf fem aplicada Section 2 Section 2 Sección 2 Figure Figure 2.7: 2.7: Flow Flow calorimeter. calorimeter. Figura 2.7: Calorímetro de flujo. A A simple simple flow flow calorimeter calorimeter is is illustrated illustrated schematically schematically in in Fig. Fig. 2.7. 2.7. Its Its essential essential feature feature is an electric resistance heater immersed in a flowing fluid. The design provides for minimal is an electric resistance heater immersed in a flowing fluid. The design provides for minimal Un calorímetro de flujo simple se ilustra en forma esquemática en la figura 2.7. Su característica esencial es un calentador de resistencia eléctrica sumergido en un líquido que circula. El diseño proporciona para 9 Exceptions are applications to nozzles, metering devices, wind tunnels, and hydroelectric power stations. 9 Exceptions are applications to nozzles, metering devices, wind tunnels, and hydroelectric power stations. 9 Las excepciones son las aplicaciones a las toberas, a los aparatos de medición, a los túneles de viento y a las estaciones hidroeléctricas. 02-SmithVanNess.indd 52 8/1/07 12:50:13 53 2.12. Balances de masa y energía para sistemas abiertos velocidad y cambios de elevación mínimos de la sección 1 a la sección 2, haciendo despreciables los cambios en las energías cinética y potencial del fluido. Además, como no se hace trabajo en la flecha del sistema, la ecuación (2.33) se reduce a ΔH = H2 – H1 = Q. La rapidez de transferencia de calor al fluido se determina por la resistencia del calentador y por la corriente que pasa a través de éste. En la práctica se necesita poner atención a un gran número de detalles, pero en principio la operación del calorímetro de flujo es simple. Las medidas del rendimiento térmico y la relación de flujo permiten el cálculo del cambio ΔH entre las secciones 1 y 2. Como ejemplo, se determinan fácilmente las entalpías del H2O como líquido y como vapor. El baño a temperatura constante se llena de una mezcla de hielo molido y agua para mantener una temperatura de 0 °C. Se suministra agua líquida al aparato y el serpentín que lleva el agua a través del baño a temperatura constante es lo suficientemente extenso, de modo que el líquido emerge a la temperatura del baño que es esencialmente de 0 °C. La temperatura y la presión en la sección 2 se miden con instrumentos adecuados. Los valores de la entalpía del H2O para diversas condiciones en la sección 2 están dados por: H2 = H1 + Q donde Q es el calor agregado por unidad de masa del flujo de agua. La presión varía en cada una de las experimentaciones, pero en el intervalo aquí encontrado tiene un efecto despreciable sobre la entalpía del agua que entra, y para propósitos prácticos H1 es constante. Los valores absolutos de la entalpía, al igual que los valores absolutos de la energía interna son desconocidos. Por lo tanto, se le puede asignar a H1 un valor arbitrario como base para los demás valores de la entalpía. Estableciendo H1 = 0 para el agua líquida a 0 °C obtenemos: H2 = H1 + Q = 0 + Q = Q Los valores de la entalpía se pueden tabular para las temperaturas y presiones que existen en la sección 2, para una gran cantidad de mediciones. Además, las mediciones del volumen específico realizadas para estas mismas condiciones se pueden agregar a la tabla, junto con los valores correspondientes de la energía interna calculados por medio de la ecuación (2.11), U = H – PV. De esta manera, las tablas de propiedades termodinámicas se compilan sobre toda la gama útil de condiciones. El uso más extenso posible para esta tabulación es para el H2O y se conoce como las tablas de vapor.10 La entalpía se puede tomar igual a cero para algunos otros estados del líquido a 0 °C. La elección es arbitraria. Las ecuaciones termodinámicas, tales como las ecuaciones (2.32) y (2.33) se aplican a los cambios de estado, para los cuales las diferencias de entalpía son independientes de la localización del punto cero. Sin embargo, una vez que se elige un punto cero arbitrario para la entalpía, no se puede hacer una elección arbitraria para la energía interna, debido a que los valores de la energía interna pueden calcularse a partir de los de la entalpía mediante la ecuación (2.11). Ejemplo 2.15 Para el calorímetro de flujo recién analizado se toman los siguientes datos con el agua como líquido de prueba: Relación de flujo = 4.15 g s –1 t1 = 0 °C t 2 = 300 °C P2 = 3 bar 10 Las tablas de vapor se dan en el apéndice F. Las tablas para otras sustancias se encuentran en la literatura. Un análisis de compilaciones de las propiedades termodinámicas aparece en el capítulo 6. 02-SmithVanNess.indd 53 8/1/07 12:50:14 54 CHAPTER 2. The First Law Other Basic Concepts CHAPTER 2. First Law and and Basic Concepts CHAPTER 2. The The2. First and Other Other Basicconceptos Conceptsbásicos CAPÍTULO La Law primera ley y otros 54 54 54 Rate of heat addition from resistance heater = 12,740 W of from = Rapidez Rate de adición deaddition calor desde calentadorheater de resistencia 12 740 W Rate of heat heat addition fromelresistance resistance heater = 12,740 12,740=W W The The water water is is completely completely vaporized vaporized in in the the process. process. Calculate Calculate the the enthalpy enthalpy of of steam steam at at The water is completely vaporized in liquid the process. the enthalpy El agua300 se ◦evapora el proceso. Calcule la entalpía a 300 °Cofysteam 3 bar, at conside3totalmente bar based en on 0 for water atCalculate 0◦◦C. del vapor C and and bar on H H= = 300◦◦C andel33agua bar based based = 00 for for liquid liquid water water at at 00◦C. C. rando H300 = 0C para líquidaon a 0H°C. Solution Solution 2.15 Solución 2.15 2.15 Solution 2.15 2 son If �z anddespreciables, �u 222 are negligible are zero, H and s and 2y = Q, Si Δz y Δu y si W and y Hif1 W son cero,H1entonces Hthen If H 2 = Q, If �z �z and and �u �u are are negligible negligible Sand and if if W Wss and and H H11 are are zero, zero, then then H22 = = Q, Q, and and −1 12,740 J s −1 12,740 J ss−1 = 3,070 J g−1 H −1 2 = 12,740 J −1 H = gg ss−1 H22 = = 4.15 = 3,070 3,070 JJ gg−1 4.15 −1 4.15 g s Example 2.16 Example 2.16 Example 2.16 Ejemplo 2.16 Air at 1 bar and 25◦◦C enters a compressor at low velocity, discharges at 3 bar, and Air at Air at at 11 bar bar and and 25 25◦C C enters enters aa compressor compressor at at low low velocity, velocity, discharges discharges at 33 bar, bar, and and −1 enters a to a velocity of 600 ss−1 at the Se introduce aire a 1 barin ywhich 25 °C it un compresor a baja velocidad, sem descarga a 3initial bar ycondientra en una enters aa nozzle nozzle in which ititaexpands expands to aa final final velocity of m at initial condienters nozzle inand which expands tothe final velocity of a600 600 m s−1240 at the the initial condi–1 tions of pressure temperature. If work of compression is kJ per kilogram tobera en la cual se expande a una velocidad final de 600 m s las condiciones iniciales de presión tions of pressure and IfIf the work tions of pressure and temperature. temperature. theduring work of of compression compression is is 240 240 kJ kJ per per kilogram kilogram of air, how much heat must be removed compression? y temperatura. Si el trabajo de compresión es de 240 kJ por kg de aire, ¿cuánto calor debe eliminarse of of air, air, how how much much heat heat must must be be removed removed during during compression? compression? durante la compresión? Solution 2.16 Solution Solución 2.16 2.16 Solution 2.16 Because the air returns to its initial conditions of T and P, the overall process Because air to conditions TT el and P, overall process Dado que el aire the regresa a sus condiciones iniciales de Tof proceso no produce ningún Because the air returns returns to its its initial initial conditions ofy P, and P, the thetotal overall process produces no change in enthalpy of the air. Moreover, the potential-energy change produces no change in enthalpy of the air. Moreover, the potential-energy cambio en la entalpía del aire. Por otra parte, elair. cambio en la energía potencial del change aire se supone produces no change in enthalpy of the Moreover, the potential-energy change of the air is presumed negligible. Neglecting also the initial kinetic energy of the of air is negligible. also initial kinetic of que es insignificante. Ignorando también Neglecting la energía cinética aire,energy escribimos of the the air is presumed presumed negligible. Neglecting also the theinicial initial del kinetic energy of the thela ecuaair, we write Eq. (2.32a) as: air, write ción (2.32a) como: air, we we write Eq. Eq. (2.32a) (2.32a) as: as: 2 uu222 Q = u2 − W s Q = Q = 222 − −W Wss 2 The kinetic-energy term is as The kinetic-energy term is evaluated evaluated as follows: follows: Thede kinetic-energy term evaluated follows:forma: El término energía cinética seisevalúa de laassiguiente � 2 2 �� � 11 � m m 11 2 2 2 m m 180,000 1 600 m22 m �2 = 1uu222 = = = 180,000 600 22 u22 = 22 600 ss = 180,000 ss22 2 2 s 2 kg s m 2 kg −1 = 180 kJ kg−1 m 2 = 180,000 · = 180,000 m kg −1 = 180 kJ kg−1 m kg = 180,000 N = N m kg −1 = 180 kJ kg−1 = 180,000 180,000 ss222 ·· kg = 180,000 N m kg kg kg s −1 –1 Then Q ==180 240 −60 kJ kg −1 En tal caso 180− 240 = = –60 Then Q − = −60 kJ kg Then QQ= = 180 180 −– 240 240 = −60kJ kJkg kg−1 Heat in the amount of 60 kJ must be removed per kilogram of air compressed. Heat in amount kJ be kilogram Se deben eliminar kJ de of calor kilogramo de aireper comprimido. Heat in the the60 amount of 60 60 por kJ must must be removed removed per kilogram of of air air compressed. compressed. Example 2.17 Example 2.17 Example 2.17 Ejemplo 2.17 ◦ Water at 200( ◦F) is pumped from a storage tank at the rate of 50(gal)(min)−1 −1. Water at 200( ◦F) is pumped from a storage tank at the rate of 50(gal)(min) −1. The The Water at 200( F) °F) is pumped from aatstorage tank atlathe rate of 50(gal)(min) . –1The motor for the pump supplies work the rate of 2(hp). The water goes through a Se bombea agua a 200(0 de un tanque de almacenaje en proporción de 50(gal)(min) . El motor for the pump supplies work at the rate of 2(hp). The water goes through aa motor motor for the pump supplies work at the rate of 2(hp). The water goes through −1 heat exchanger, giving up heat at the rate of 40,000(Btu)(min) , and is delivered to a de la bomba suministra trabajo con un valor de 2(hp). El agua pasa a través de un intercambiador de −1 heat giving up heat rate of 40,000(Btu)(min) is to −1,, and heat exchanger, exchanger, giving upproporción heat at at the the rate of 40,000(Btu)(min) and is adelivered delivered to aatanque –1, y se second storage tank at an elevation 50(ft) above the first tank. What is the temperature calor, proporcionando calor a una de 40 000(Btu)(min) entrega un segundo second storage tank elevation 50(ft) above the first What is the temperature second storage tank at attoan an elevation 50(ft) above thetanque. first tank. tank. What temperature of the water delivered the second tank? de almacenaje a una elevación de 50(pies) sobre el primer ¿Cuál esislathe temperatura del agua of the water delivered to the second tank? of the water delivered to the second tank? entregada al segundo tanque? 02-SmithVanNess.indd 54 8/1/07 12:50:18 2.12. 2.12.Mass Massand andEnergy EnergyBalances Balancesfor forOpen OpenSystems Systems 5555 55 2.12. Balances de masa y energía para sistemas abiertos Solución 2.17 2.17 Solution Solution 2.17 Éste esThis un proceso en estado estacionario yprocess flujo establewhich para el que(2.32b) se aplica la ecuación (2.32b). Thisisisa asteady-state, steady-state,steady-flow steady-flow processfor for whichEq. Eq. (2.32b)applies. applies. The The Las velocidades inicial y final delofagua eninlosthetanques detanks almacenaje son insignificantes, y se initial and final velocities water storage are negligible, and the initial and final velocities of water in the storage tanks are negligible, and the 2/2g . Los puede omitir el 2término Δu términos restantes se expresan en unidades de (Btu)(lb )–1 c m 2 term �u /2g may be omitted. The remaining terms are expressed in units of c c may be omitted. The remaining terms are expressed in units of term �u /2g −1 ◦ mediante el uso factoresuse de conversión apropiados. A 200(°F) la densidad del agua es −1through ◦ F)the ) de (Btu)(lb mm through useofofappropriate appropriateconversion conversionfactors. factors.AtAt200( 200(F) thedenden(Btu)(lb –3, )y 1(pie) 3 es equivalente −3 3 3is equivalent 60.1 (lbsity )(pie) a 7.48(gal); así, la rapidez de flujo de masa es: −3 m of water is 60.1(lb )(ft) , and 1(ft) to 7.48(gal); thus the mass mm )(ft) , and 1(ft) is equivalent to 7.48(gal); thus the mass sity of water is 60.1(lb flowrate is: flowrate is: –1 (50)(60.1/7.48) = 402(lbm)(min)−1 (50)(60.1/7.48) )(min) mm (50)(60.1/7.48)==402(lb 402(lb )(min)−1 De donde Q −40,000/402 = –40 000/402 = –99.50(Btu)(lbm))–1 −1 Whence QQ= mm )−1 Whence = −40,000/402==−99.50(Btu)(lb −99.50(Btu)(lb −1 , the shaft work is: Because isisequivalent toto42.41(Btu)(min) Dado que 1 (hp)1(hp) es equivalente a 42.41 (Btu)(min)–1, el−1 trabajo en lawork flecha Because 1(hp) equivalent 42.41(Btu)(min) , the shaft is:es: −1 = =(2)(42.41)/(402) )–1 WW (2)(42.41)/(402) = ==0.21(Btu)(lb sW mm (2)(42.41)/(402) 0.21(Btu)(lb )−1 s s= –2−2 −2 IfIfthe local taken value, 32.174(ft)(s) , the Si el valor local devalue g seoftoma elasthe valor estándar, 32.174(pie)(s) , el término de la energía the local value ofg gisiscomo takenas thestandard standard value, 32.174(ft)(s) , thepotentialpotentialenergy term potencial se convierte en: energy termbecomes: becomes: −2 −2 32 .174(pie)(s)−2 50 (pie) 32.174(ft)(s) 50(ft) ggg 32.174(ft)(s) 50(ft) Δ = z �z = −1 −2 −2 ⋅· · −1 �z = −1 32 .32.174(lb 174(lbmm)()(ft)(lb pie )( lb ) ( s ) 778 . 16 ( pie ( Btu−1 )−1 −1 −2 ggcgc 32.174(lb ) (s) 778.16(ft f))(Btu) f f ) (s) f )(Btu) )(ft)(lb 778.16(ftlblb lb c m f f ==0.06(Btu)(lb )−1 mm 0.06(Btu)(lb )−1 Equation (2.32b) now yields �H Equation (2.32b) yieldsΔH: �H: : La ecuación (2.32b) ahoranow produce gg = −99.50 + 0.21 − 0.06 = −99.35(Btu)(lbm )−1−1 �H s s−− �z �H==QQ++WW gcgc �z = −99.50 + 0.21 − 0.06 = −99.35(Btu)(lbm ) ◦ ◦ is: value for enthalpy ofofagua liquid water 200( The value forthe theentalpía enthalpy liquid waterat 200(F) F) El valorThe en steam-table lasteam-table tabla de vapor para la del líquida aat200(°F) es:is: −1 )−1 HH 1 11== 168.09 (Btu)(lbmmm ))–1 H =168.09(Btu)(lb 168.09(Btu)(lb Thus, Thus, De esta forma, and and y �H 2 2−−HH 1 1==HH 2 2−−168.09 �H==HH 168.09==−99.35 −99.35 ΔH = H2 – H1 = H2 – 168.09 = –99.35 HH )−1 mm 2 2==168.09 168.09−−99.35 99.35==68.74(Btu)(lb 68.74(Btu)(lb )−1 –1 H 168.09 –this 99.35 = 68.74 (Btu)(lb The enthalpy isisfound 2 = having m) the Thetemperature temperatureofofwater water havingthis enthalpy foundfrom from thesteam steamtables: tables: La temperatura que el agua tiene a esta entalpía se encuentra en las tablas de vapor: ◦◦ t t==100.74( F) 100.74(F) t = 100.74(°F) are small compared with Q, and for practical InInthis s sand c )�z and(g/g (g/g thisexample exampleWW c )�z are small compared with Q, and for practical purposes they could be neglected. theyy could be neglected. En este purposes ejemplo W (g/gc)Δz son pequeños en comparación con Q, y para fines prácticos se s pueden despreciar. 02-SmithVanNess.indd 55 8/1/07 12:50:22 56 CAPÍTULO 2. La primera ley y otros conceptos básicos PROBLEMAS 2.1. Un recipiente no conductor lleno con 25 kg de agua a 20 °C tiene un agitador, el cual se mueve debido a la acción de la gravedad sobre una pesa con masa de 35 kg. La pesa cae lentamente a una distancia de 5 m accionando el agitador. Suponiendo que todo el trabajo hecho en la pesa se transfiere al agua y que la aceleración local de la gravedad es 9.8 m s–2, determine: a) b) c) d) e) La cantidad de trabajo realizado en el agua. El cambio en la energía interna del agua. La temperatura final del agua, para la cual CP = 4.18 kJ kg–1 °C–1. La cantidad de calor que debe extraerse del agua para que regrese a su temperatura inicial. El cambio en la energía total del universo debido a: 1) el proceso de bajar el peso, 2) el proceso de enfriamiento del agua para que regrese a su temperatura inicial, y 3) ambos procesos juntos. 2.2. Trabaje de nuevo con el problema 2.1 para un recipiente aislado que cambia de temperatura junto con el agua, y que tiene una capacidad térmica equivalente a 5 kg de agua. Trabaje el problema con: a) El agua y el recipiente como el sistema; b) considere sólo al agua como el sistema. 2.3. Un huevo, inicialmente en reposo, se deja caer sobre una superficie de concreto y se rompe. Si se considera al huevo como el sistema, a) b) c) d) e) ¿Cuál es el signo de W? ¿Cuál es el signo de ΔEP? ¿A qué es igual ΔEK? ¿A qué es igual ΔU t? ¿Cuál es el signo de Q? Al configurar este proceso, suponga que pasa el tiempo suficiente para que el huevo roto regrese a su temperatura inicial. ¿Cuál es el origen de la transferencia de calor del inciso e)? 2.4. Un motor eléctrico bajo carga fija toma 9.7 amper a 110 volts, liberando 1.25(hp) de energía mecánica. ¿Cuál es la intensidad de transferencia de calor desde el motor en kW? 2.5. Un mol de gas en un sistema cerrado se somete a un ciclo termodinámico de cuatro etapas. Use la información que se proporciona en la tabla siguiente para determinar los valores numéricos de las cantidades faltantes, es decir, “complete los espacios en blanco”. 02-SmithVanNess.indd 56 Paso ΔU t/J 12 23 Q/J W/J –200 ? –6 000 ? –3 800 ? 34 ? –800 300 41 4 700 ? ? 12 341 ? ? –1 400 8/1/07 12:50:23 Problemas 57 2.6. Explique la factibilidad de enfriar su cocina durante el verano abriendo la puerta de un refrigerador accionado eléctricamente. 2.7. Un laboratorio de renombre reporta coordenadas del punto cuádruple de 10.2 Mbar y 24.1 ºC para el equilibrio en las cuatro fases de las formas alotrópicas sólidas del exótico producto químico bmiasmone. Evalúe la afirmación. 2.8. Un sistema cerrado sin reacción química contiene a las especies 1 y 2 en equilibrio vapor/líquido. La especie 2 es un gas muy ligero, esencialmente insoluble en la fase líquida. La fase vapor contiene ambas especies. Se agregan algunas moles adicionales de la especie 2 al sistema, el cual regresa a su T y P iniciales. Como resultado del proceso, ¿el número total de moles del líquido se incrementa, disminuye o no cambia? 2.9. Un sistema compuesto por cloroformo, 1,4-dioxano y etanol existe como un sistema de dos fases vapor/líquido a 50 °C y 55 kPa. Se encuentra que después de agregar cierta cantidad de etanol puro, el sistema regresa al equilibrio de dos fases a su T y P iniciales. ¿En qué aspecto el sistema cambia? ¿En qué aspecto no cambia? 2.10. Para el sistema descrito en el problema 2.9: a) ¿Cuántas variables de la regla de la fase, además de T y P, se deben seleccionar para fijar las composiciones de ambas fases? b) Si la temperatura y la presión siguen siendo las mismas, ¿puede la composición global del sistema cambiar (por la adición o eliminación de materia) sin afectar las composiciones de las fases líquida y vapor? 2.11. Un tanque que contiene 20 kg de agua a 20 °C cuenta con un agitador que proporciona trabajo al agua a la intensidad de 0.25 kW. ¿Cuánto tiempo transcurre para incrementar la temperatura del agua a 30 °C si no hay pérdida de calor desde el agua? Para el agua, CP = 4.18 kJ kg–1 °C–1. 2.12. Se agrega a un sistema cerrado 7.5 kJ de calor a medida que su energía interna disminuye 12 kJ. ¿Cuánta energía se transfiere como trabajo? Para un proceso que ocasiona el mismo cambio de estado pero cuyo trabajo es cero, ¿cuánto calor se transfiere? 2.13. Un bloque de acero fundido de 2 kg tiene una temperatura inicial de 500 °C. En un tanque de 5 kg de acero perfectamente aislado se tienen 40 kg de agua a 25 °C. Se sumerge el bloque en el agua y se permite al sistema llegar al equilibrio. ¿Cuál es la temperatura final? Ignore cualquier efecto de expansión o contracción y suponga constantes el calor específico del agua que es de 4.18 kJ kg–1 K–1 y del acero 0.50 kJ kg–1 K–1. 2.14. Un fluido incompresible (r = constante) está contenido en un cilindro aislado con un pistón sin fricción hecho a la medida. ¿Puede transferirse energía al fluido en forma de trabajo? ¿Cuál es el cambio en la energía interna del fluido cuando la presión aumenta de P1 a P2? 2.15. Un kg de agua líquida a 25 °C: a) Experimenta un aumento en la temperatura de 1 K. ¿Cuánto es ΔU t, en kJ? 02-SmithVanNess.indd 57 8/1/07 12:50:23 58 CAPÍTULO 2. La primera ley y otros conceptos básicos b) Experimenta un cambio en la elevación Δz. El cambio en la energía potencial ΔEP es igual a la que presenta ΔU t para el inciso a). ¿Cuánto es Δz en metros? c) Se acelera desde el reposo hasta la velocidad final u. El cambio en la energía cinética ΔEK es igual al que presenta ΔU t en el inciso a). ¿Cuánto es u, en m s–1? Compare y analice los resultados de estos tres incisos. 2.16. Un motor eléctrico funciona “caliente” en servicio, debido a sus irreversibilidades internas. Se sugiere que la pérdida de energía asociada sea minimizada por el aislamiento térmico del armazón del motor. Realice un comentario crítico con respecto a esta sugerencia. 2.17. Una hidroturbina funciona con una carga de 50 m de agua. Los conductos de entrada y salida tienen 2 m de diámetro. Estime la potencia mecánica desarrollada por la turbina para una velocidad de salida de 5 m s–1. 2.18. El agua líquida a 180 °C y 1 002.7 kPa tiene una energía interna (en una escala arbitraria) de 762.0 kJ kg–1 y un volumen específico de 1.128 cm3 g–1. a) ¿Cuál es su entalpía? b) El agua se hace llegar al estado de vapor a 300 °C y 1 500 kPa, donde su energía interna es 2 784.4 kJ kg–1 y su volumen específico es 169.7 cm3 g–1. Calcule ΔU y ΔH para el proceso. 2.19. Un cuerpo sólido con temperatura inicial T0 se sumerge en. un baño de agua a la temperatura inicial Tw0. Se transfiere calor del sólido al agua en proporción Q = K · (Tw – T), donde K es una constante Tw y T son los valores instantáneos de las temperaturas del agua y del sólido. Desarrolle una expresión para T en función del tiempo t. Compruebe su resultado para los casos límites, t = 0 y t = ∞. Ignore los efectos de la expansión o contracción y suponga constante el calor específico tanto para el agua como para el sólido. 2.20. Observe una lista de operaciones unitarias comunes: a) Intercambiador de calor de una sola tubería; b) intercambiador de calor de doble tubería; c) bomba; d) compresor de gas; e) turbina de gas; f) válvula reguladora, y g) tobera. Desarrolle una forma simplificada del balance energético general en estado estacionario más apropiado para cada operación. Indique y justifique de manera cuidadosa cualquier suposición que haga. 2.21. El número de Reynolds, Re, es un grupo adimensional que caracteriza la intensidad de un flujo. Para Re grandes, un flujo es turbulento; para Re pequeños, es laminar. Para el flujo en la tubería, Re ≡ urD/m, donde D es el diámetro de la tubería y m es la viscosidad dinámica. . a) Si D y m están fijos, ¿cuál es el efecto al aumentar la relación de flujo de masa m en Re? . b) Si m y m están fijos, ¿cuál es el efecto sobre Re al aumentar D? 2.22. Un líquido incompresible (r = constante) fluye de manera estable a través de un conducto de sección transversal circular con diámetro en aumento. En el punto 1, el diámetro es de 2.5 cm y la velocidad es 2 m s–1; en el punto 2, el diámetro es de 5 cm. 02-SmithVanNess.indd 58 8/1/07 12:50:24 Problems Problemas Problems 59 59 59 (a) Whatvelocidad is the velocity at location 2? a) (a)¿Cuál punto 2? Whatesislathe velocityen at el location 2? −1 ) of the fluid between locations 1 and 2? –1 (b) es What is the kinetic-energy change −1 b) (b)¿Cuál el cambio en la energía cinética ) delfluid fluido entre locations los puntos11and y 2? What is the kinetic-energy change (J kg(J kg ) of the between 2? 2.23. A stream of warmde water iscaliente produced in aproceso steady-flow mixing by combining 2.23. corriente un de mezcla de process flujo al combinar 1.0 2.23.SeAproduce stream una of warm water isagua produced in–1en a steady-flow mixing process byestable combining kg s−1 ofacool water at0.8 25◦kg C with 0.8agua kg s−1 of hotawater at Durante 75◦ C. During mixing, kg1.0 s–1kg1.0 des−1 agua fría 25 °C con s de caliente 75 °C. la mezcla ◦ −1 ◦ of cool water at 25 C with 0.8 kg s of –1 hot water at 75 C. During mixing, se pierde heat is lost to the surroundings at thederate of 30 kJ s−1 . What is the temperature of the calor hacia los alrededores en proporción 30 kJ s . ¿Cuál es la temperatura de la de −1 heat is lost to the surroundings at the rate of 30 kJ s . What is the temperature of −1 thecorriente −1 . –1 K–1 warm-water stream? Assume the specificdel heat of water constant at 4.18 kJ kgkJ agua caliente? Suponga que el calor específico agua es constante e igual a 4.18 kg K . −1 −1 warm-water stream? Assume the specific heat of water constant at 4.18 kJ kg K . 2.24. Gas bled from a tank. heat transfer between the gas and the el tank, 2.24. queisestá deNeglecting un tanque. Ignore la transferencia calor gasshow y el tanque 2.24.Imagine Gas is bled fromsaliendo a tank. gas Neglecting heat transfer between the gasde and the entre tank, show that mass and energy balances produce the differential equation: y that demuestre que los balances energéticos y de masa producen la ecuación diferencial: mass and energy balances produce the differential equation: dU dm dU dm= � = H −U m H� − U m � is the specific enthalpy of the Here, U and m refer to the gas remaining in the tank; � is H the of the del gas que Here, U and m refer to the gas remaining in the tank; H � enthalpy En estegas caso, U y mthe setank. refieren al gas restante en el can tanque; ′ esspecific la entalpía específica leaving Under what conditions one H assume � =HH ?= H ? gas leaving the tank. Under what conditions can one assume H abandona el tanque. ¿Bajo qué condiciones se supone que H ′ = H? ◦ C flows in a straight horizontal pipe in which there is no exchange of 2.25. Water at ◦ C28 2.25. Water at 28 flows in atubería straight horizontal pipeeninlawhich there is no −1 exchange calor of o de tra2.25. Fluye agua 28 °Corenwork una recta, cual no eithera heat with thehorizontal surroundings. Its velocity ishay 14intercambio m s in ade pipe with −1 either heat or work with the surroundings. Its velocity is 14 m s in a pipe with –1 bajo con alrededores. Su of velocidad de 14 m s into en una tubería where con diámetro interno de 2.5 cm anlos internal diameter 2.5 cm es until it flows a section the pipe diameter an internal diameter of 2.5 cm until it flows into section aumenta where the pipe diameter ¿Cuál es el hasta que fluye aincreases. una sección donde el diámetro de laachange tubería repentinamente. abruptly What is the temperature of the water if the downstream abruptly increases. What isagua the si temperature change of abajo the water thecm? downstream cambio en la temperatura elcm]? diámetro estemperature deif3.8 ¿Si el diámetro diameter is 3.8 cm?del If it is 7.5 Whatcorriente is the maximum change for an es de diameter is 3.8 cm? If it is 7.5 cm]? What is the maximum temperature change for an 7.5 cm?enlargement ¿Cuál es el in máximo cambio de temperatura para un alargamiento de la tubería? the pipe? enlargement in the pipe? 2.26. Se comprimen cincuenta(50) kmol porishora de aire defrom P1 = P1.2 bar aP compresor 2.26. Fifty (50) kmol per hour of air compressed 1.2 bar P2bar = en 6.0unbar in 2 =to6.0 1 = 2.26.con Fifty (50) kmol La perpotencia hour of mecánica air is compressed from P1 98.8 = 1.2 barLas to temperaturas P2 = 6.0 baryinlas velocidaflujo estable. transmitida es de a steady-flow compressor. Delivered mechanical powerkW. is 98.8 kW. Temperatures and a steady-flow compressor. Delivered mechanical power is 98.8 kW. Temperatures and des son: velocities are: velocities are: T1 T=1300 K K T2T2= = 520 KK = 300 520 T1 = 300 K –1 T2 = 520 K −1 u1 u=110=m10s−1m s uu2 2= = 3.53.5 mm s–1s−1 u 1 = 10 ms u 2 = 3.5 m s−1 CalculeEstimate la rapidez transferencia de calor delthe compresor. Suponga paraforelair airethat queCC = = 7 R, thederate of heat transfer from compressor. Assume R y que Estimate the rate of heat transfer from the compressor. Assume for air that C P = P72 PR 2 la entalpía de la presión. and es thatindependiente enthalpy is independent of pressure. and that enthalpy is independent of pressure. 2.27. Nitrogen flows at steady state through a horizontal, insulated pipe with insidecon diameter 2.27. Circula nitrógeno en estado estacionario a través de una tubería horizontal, aislada un diámetro 2.27. Nitrogen flows at steady state through a horizontal, insulated pipe with inside diameter A pressure results fromválvula flow through a partially opened valve.unJust interiorofde1.5(in). 1.5 (pulg.). Debido drop al flujo por una parcialmente abierta se produce descenso of 1.5(in). A pressure drop results from flow through a partially opened valve. Just ◦ F), and upstream from valve the pressureesisde100(psia), temperatureesisde 120( en la presión. Antes dethe la válvula la presión 100(psia),the la temperatura 120(°F) y la velo◦ upstream from the valve the pressure is 100(psia), the temperature is 120( F), and –1. Si la−1 the average velocity is 20(ft)(s) . If thea la pressure justla downstream the valve cidad promedio es de 20(pie)(s) presión salida de válvula es defrom 20(psia), ¿cuál es la the average velocity is 20(ft)(s)−1 . If the pressure just downstream from the valve is 20(psia), whatque is the Assume for nitrogen P V /Ty C isPconstant, temperatura? Suponga paratemperature? el nitrógeno PV/T es constante, CVthat = (5/2)R = (7/2)R. (Los is 20(psia), what is the temperature? Assume for nitrogen that P V /T is constant, = están (5/2)R, anden C el (7/2)R.A.) (Values for R are given in App. A.) valoresCde dados apéndice V R P = C V = (5/2)R, and C P = (7/2)R. (Values for R are given in App. A.) 2.28. Water flows through horizontalhorizontal coil heated from the outsidedesde by high-temperature flue gases 2.28. través de unaserpentín que esoutside calentado el exterior mediante 2.28.Circula Water agua flowsathrough a horizontal coil heated from the by high-temperature flue gases. As it passes through the coil the water changes state from liquid at 200 kPaestado degases. combustión de alta through temperatura. Mientras pasachanges a travésstate del serpentín el agua cambia As it◦ passes the coil the water from liquid at−1 200 kPa del –1 ◦ C. Its and 80 C to vapor at 100 kPa and 125 entering velocity is 3 m s and its exit líquido y 80 °C akPa vapor a 125 100◦ C. kPaIts y 125 °C. La velocidad desentrada es de −1 and its and 80a◦ 200 C to kPa vapor at 100 and entering velocity is 3 m exit3 m s y su –1. Determine velocity is 200 mdes−1 . Determine the heat el transferred through por the coil per de unitmasa mass of velocidad de salida es 200 m s calor transferido unidad −1 velocity is 200 m s . Determine the heat transferred through the coil per unit mass of de agua a water. Enthalpies of the inlet and outlet streams are: través serpentín.ofLas las corrientes de entrada y salida son: water.delEnthalpies theentalpías inlet andde outlet streams are: −1 ; Outlet: 2,726.5 kJ kg−1 Inlet: 334.9 kJ kg −1 −1 Inlet: 334.9 kJ kg kg–1 Entrada: 334.9kJ kJkg kg–1; Outlet: salida: 22,726.5 726.5 kJ 02-SmithVanNess.indd 59 8/1/07 12:50:28 60 CHAPTER 2. The First Law and Other Basic Concepts 60 CAPÍTULO 2. La primera ley y otros conceptos básicos 2.29. Steam flows at steady state through a converging, insulated nozzle, 25 cm long and 2.29. Circula estado estacionario través de entrance una tobera convergente aislada, de and 25 cm de largo with anvapor inleten diameter of 5 cm. At athe nozzle (state 1), the temperature y pressure un diámetro de entrada de 5 cm. En la entrada de la tobera (estado 1), la temperatura are 325◦ C and 700 kPa, and the velocity–1is 30 m s−1 . At the nozzle exit (statey la presión son 325 °C y 700 kPa, y la velocidad es de m◦ C s and . En350 la salida de la tobera (estado 2), the steam temperature and pressure are30 240 kPa. Property values are: 2), la temperatura y la presión del−1 vapor son 240 °C y 3503kPa. Los valores de sus propiedades son: H1 = 3,112.5 kJ kg V1 = 388.61 cm g−1 −13 –1 2,945.7 kg−1 V2 = V 667.75 cm3 gcm H1H=2 = 3 112.5 kJ kJ kg–1 g 1 = 388.61 is the kJ velocity nozzle cm exit, 3 gand –1 what is the exit diameter? HWhat = 2 945.7 kg–1 of the steam at V the = 667.75 2 2 = 20.8 C P =de29.1 J mol−1y ◦¿cuál C−1 for nitrogen gas: 2.30.¿Cuál In theesfollowing takedel C Vvapor la velocidad en and la salida la tobera?, es el diámetro de salida? –1 °C–1 para C, contained in aelrigid vessel,gaseoso: is heated to 250◦ C. (a) Threea moles of nitrogen at 30J◦ mol 2.30. Considere CV = 20.8 y CP = 29.1 nitrógeno How much heat is required if the vessel has a negligible heat capacity? If the vessel weighs 100 kg and has a heat capacity of 0.5 kJ kg−1 ◦ C−1 , how much heat a) Tres moles de nitrógeno a 30 °C, contenidas en un recipiente rígido, se calientan a 250 °C. is required? ¿Cuánto calor se requiere si el recipiente tiene una capacidad calorífica insignificante? Si el ◦ is contained in a piston/cylinder arrangement. (b)recipiente Four moles nitrogen at 200 pesaof100 kg y tiene unaC capacidad calorífica de 0.5 kJ kg–1 °C–1, ¿cuánto calor se How much heat must be extracted from this system, which is kept at constant requiere? ◦ C if the heat capacity of the piston and cylinder is nepressure, to cool it to 40 b) Cuatro moles de nitrógeno a 200 °C están contenidas en una combinación de pistón/cilindro. glected? ¿Cuánto calor debe extraerse de este sistema, que se mantiene a presión constante, para enfriar- lo a 40 °C si la capacidad calorífica del pistón y del cilindro se desprecia? 2.31. In the following take C V = 5 and C P = 7(Btu)(lb mole)−1 (◦ F)−1 for nitrogen gas: 2.31. Considere a CV = 5 y CP = 7(Btu)(lb mol)–1◦(°F)–1 para el nitrógeno gaseoso: (a) Three pound moles of nitrogen at 70( F), contained in a rigid vessel, is heated to 350(◦ F). How much heat is required if the vessel has a negligible heat capacity? If a) Tres libras mol de nitrógeno a 70(°F), contenidas en un recipiente rígido, se calientan a 350(°F). it weighs 200(lbm ) and has a heat capacity of 0.12(Btu)(lbm )−1 (◦ F)−1 , how much ¿Cuánto calor se requiere si el recipiente tiene una capacidad calorífica insignificante? Si el heat is required? recipiente pesa 200(lbm) y tiene una capacidad calorífica de 0.12(Btu)(lbm)–1(°F)–1, ¿cuánto ◦ F) is contained in a piston/cylinder arrange(b)calor Foursepound moles of nitrogen at 400( necesita? ment.libras How mol much must be extracted from this system, which is kept at conb) Cuatro deheat nitrógeno a 400(°F) están contenidas en una combinación de pistón/cilin◦ F) if the heat capacity of the piston and cylinder stant pressure, to cool it to 150( dro. ¿Cuánto calor debe extraerse de este sistema, que se mantiene a presión constante, para is neglected? enfriarlo a 150(°F) si se desprecia la capacidad calorífica del pistón y del cilindro? 2.32.Encuentre Find the equation for para the work of a reversible, isothermalisotérmica compression of 1 moldeof1gas 2.32. la ecuación el trabajo de una compresión reversible mol de gas en in combinación a piston/cylinder assembly if thesimolar volumemolar of thedel gasgas is está givendado by por una de pistón/cilindro el volumen V = RT +b P whereb by and R constantes are positivepositivas. constants. donde R son ◦ F) [state 2.33. a 200(psia) y 600(°F) [estado 1] entra en unaaturbina través deauna tubería de 3 pulgadas de 1] enters turbinea through 3-inch-diameter 2.33.Vapor Steam at 200(psia) and 600( −1 . The–1exhaust diámetro con una velocidad de 10(pie)(s) . La descarga desde la turbina se efectúa a través de una pipe with a velocity of 10(ft)(s) from the turbine is carried through a ◦ F) y tubería de 10 pulgadas y está 5(psia) 200(°F) [estado 2].power ¿Cuáloutput es la potencia de 10-inch-diameter pipe de anddiámetro is at 5(psia) anda200( [state 2]. What is the salida la turbina? of thedeturbine? H1 = 1,322.6(Btu)(lbm )−1 H1 = 1 322.6(Btu)(lbm)–1 −1 H2 = 1,148.6(Btu)(lbm ) H2 = 1 148.6(Btu)(lbm)–1 02-SmithVanNess.indd 60 V1 = 3.058(ft)3 (lbm )−1 V = 3.058(pie)3(lb )–1 V21= 78.14(ft)3 (lbmm)−1 V2 = 78.14(pie)3(lbm)–1 8/1/07 12:50:30 Problems Problemas 61 61 61 Problems 2.34. Carbon dioxide gas enters a water-cooled compressor at conditions P11 = 15(psia) and 2.34. Dióxido de carbono entra a un compresor que se enfría con agua en las condiciones P 1 = 15(psia) y T1 = 50(◦◦F), and is discharged at conditions P22 = 520(psia) and T22 = 200(◦◦F). T1 =1 50(°F), y descarga en las condiciones P2 = 520(psia) y T2 = 200(°F). El CO2 que−1 circula −1entra, through a 4-inch-diameter pipe a velocity of15(psia) 20(ft)(s) , The entering COgas 22 flows 2.34.a Carbon enters apulgadas water-cooled compressor at with conditions Pde –1and 1 = través dedioxide una tubería de 4 de diámetro con una velocidad 20(pie)(s) , y se descarga ◦ F), and through and is 50( discharged a 1-inch-diameter pipe. =The shaft work T supplied to◦the at 520(psia) 200( F). al compre1 = de 2 = aT través una tuberíaisdedischarged 1 pulgada de conditions diámetro. ElP2trabajo de flechaand que se proporciona −1 −1 −1 , compressor isCO 5,360(Btu)(mol) . What is the heat-transfer rate from the compressor flows through a 4-inch-diameter pipe with a velocity of 20(ft)(s) The entering –1 2 sor es de 5 360(Btu)(mol) . ¿Cuál es la rapidez de transferencia de calor desde el compresor en −1 −1? in (Btu)(hr) and is discharged through a 1-inch-diameter pipe. The shaft work supplied to the –1? (Btu)(hr) −1 −1 −1 H11 = 307(Btu)(lb V11−1=. What 9.25(ft) (lbm )−1 compressor is 5,360(Btu)(mol) is 33the heat-transfer rate from the compressor m m) m −1 −1 3 −1 in (Btu)(hr) ? –1 3 –1 −1 3(lbm )(lb −1 ) 330(Btu)(lb V22 =V0.28(ft) H1H=22 = 307(Btu)(lb m) m 1 = 9.25(pie) m m) m −1 H1 = 307(Btu)(lb–1 V1 = 9.25(ft)3 (lbm3)−1 –1 m) V2mechanically = 0.28(pie) (lb 2 = 330(Btu)(lb m)Q for m) 2.35.HShow W and reversible nonflow process are given −1 an arbitrary 3 (lb )−1 H2 = that 330(Btu)(lb V2 = 0.28(ft) m) m by: � 2.35. Demuestre que W y Q�para un proceso arbitrario sin flujo, mecánicamente reversible, están dados 2.35.por: Show that W and Q forVandarbitrary reversible are given W = P − �(Pmechanically V) Q = �H nonflow − V dprocess P by: � � d P −reversibly �(P V ) at constant Q =pressure �H − from V danP initial state of 2.36. One kilogram W of = air isVheated 300 K and 1 bar until its volume triples. Calculate W , Q, �U , and �H for the 33 molconstante −1 −1 K−1 −1 de un 2.36. kilogramo deofaire calienta enV forma presión inicial de 300 process. Assume forseisair that Preversibly /T = reversible 83.14 baracm C PPestado = 29of J 2.36.Un One kilogram air heated at constant pressure from anand initial state −1 −1 −1 −1 K300 y1K barK hasta el triple de su volumen. Calcule W, Q, ΔU y ΔH para el proceso. Suponga para el mol . and 1 bar until its volume triples. Calculate W–1, Q, �U , and �H for the 3 mol–1 K–1 y C = 29 J mol –1 3 −1 −1 aire que PV/T = 83.14 bar cm K . P process. Assume for air that P V /T = 83.14 bar cm mol K ◦ and C P = 29 J 2.37. mol The−1 conditions of a gas change in a steady-flow process from 20 ◦C and 1,000 kPa K−1 . ◦ ◦ to condiciones 60 C and 100 Devise a reversible nonflow (any number 2.37. Las dekPa. un gas cambian en un proceso deprocess flujo estable de 20 °Cofysteps) 1 000 for kPa a 60 °C y accomplishing of state, calculate �U andnúmero �H process the este camkPa. Diseñe this un reversible flujo (cualquier de◦the etapas) paraonlograr 2.37.100 The conditions of proceso achange gas change in and a sin steady-flow process from for 20 C and 1,000 kPa basis of mol of kPa. gas. gascalcule that PΔU V /T is constant, C VV = of (5/2)R, bio de y,100 con baseAssume en 1 mol dethe gas, y ΔH para proceso. Asuma para to 60◦estado C 1and Devise afor reversible nonflow process (anyelnumber steps) and for el gas que C PP = PV/T es (7/2)R. constante, CVchange = (5/2)R y CP =and (7/2)R. accomplishing this of state, calculate �U and �H for the process on the basis of 1 mol of gas. Assume for the gas that P V /T is constant, C V = (5/2)R, and 2.38.a)C (a)Un= An incompressible fluid (ρ r==constant) through a pipe of tubería constantcon cross2.38. fluido incompresible (con constante)flows circula a través de una un área de sec(7/2)R. P sectional area. If the flow is steady, show that velocity u and volumetric flowrate ción transversal constante. Si el flujo es estable, demuestre que la velocidad u y la relación qflujo are constant. volumétrico qfluid son constantes. 2.38. (a)deAn incompressible (ρ = constant) flows through a pipe of constant crossA corriente chemically gas is stream flows steadily through pipe of aconstant crosssectional area. If the flow steady, show that velocity u aand volumetric b)(b)Una dereactive gas químicamente reactivo fluye de manera estable travésflowrate de una tubería con area.transversal Temperature andtemperatura pressure vary pipevarían length. Which of the qsectional are constant. área de sección fija. La y.lawith presión con la longitud de la tubería. . following are necessarily constant: m,through n, q,constantes: u? a pipe ofm·constant de las quantities siguientes cantidades necesariamente , n·, q, u? cross(b)¿Cuál A chemically reactive gas streamson flows steadily sectional area. Temperature and pressure vary with pipe length. Which of the . .estimating pressure drop owing 2.39.ElThe mechanical-energy balance provides a basis for 2.39. balance de laquantities energía mecánica proporciona un fundamento following are necessarily constant: m, n, q, u? para calcular la caída de presión que to friction in fluid flow. For steady flow of an incompressible in afluido horizontal pipe resulta de la fricción en el flujo del fluido. Para un flujo establefluid de un incompresible en una of constant cross-sectional area, it may be written, 2.39.tubería The mechanical-energy provides a basis estimating pressure drop owing horizontal de área balance de sección transversal fija,for puede establecerse que: to friction in fluid flow. For steady fluid in a horizontal pipe �Pflow of 2 an incompressible + be written, f FF ρu 22 = 0 of constant cross-sectional area, it may �L D �P 2 2 11 11 011 proporciona wherefF fes Fanning friction the following expression for donde el the factor de fricción defactor. Fanning. la siguiente expresión para fF F + Churchill fChurchill F is F ρu = gives �L D f Fcorresponde for turbulentalflow: que flujo con turbulencia: F �� � � Churchill11 where f F is the Fanning friction factor. the following expression for −2 � gives �0.9 −2 0.9 � 7 f F for turbulent flow:f = 0.3305 ln 0.27 + F F D Re � � � �0.9 ��−2 � 7 f F = 0.3305 ln 0.27 + 11 D Re 11AIChE AIChE J., J., vol. vol. 19, 19, pp. pp. 375–376, 375–376, 1973 1973 11 AIChE J., vol. 19, pp. 375-376, 1973. 11 AIChE J., vol. 19, pp. 375–376, 1973 02-SmithVanNess.indd 61 8/1/07 12:50:37 62 CHAPTER 2. The First Law and Other Basic Concepts 62 CAPÍTULO 2. La primera ley y otros conceptos básicos Here,Re Reesiselthe Reynolds number (see Pb.el2.21), and 2.21) �/D isy /D the dimensionless pipe Aquí, número de Reynolds (véase problema es la aspereza adimensional de flowturbulento obtains forpara Re > laroughness. tubería. SeTurbulent obtiene flujo Re3,000. > 3 000. Consider elthe flow liquid water at 25◦ C.a 25 For°C. one of cada the sets given Considere flujo delofagua en estado líquido Para uno of de conditions los conjuntos de condiciones . −1 −1 ). Assume · below, determine m (in kg s ) m and kPa m(en �/D = 0.0001. dados a continuación, determine (en�P/�L kg s–1), y(in∆P/∆L kPa m–1). Suponga que /D = 0.0001. ◦ C, ρ = 996 kg m−3 –1 sm –1−1 For el liquid at a2525 = 9.0 s−1 . Verify Para aguawater líquida °C, r = 996 kg m–3, yand m =µ9.0 × 10×–410 kg−4mkg . Verifique si el flujo tiene that the flow is turbulent. turbulencia. (a) D = 2 cm, u = 1 m s−1 a) D = 2 cm, u = 1 m s–1 −1 (b) D = 5 cm, u = 1 m s b) D = 5 cm, u = 1 m s–1 −1 2 cm, ms c) (c)D D = 2=cm, u =u5 = m 5s–1 −1 (d) D = 5 cm, u = 5 –1 d) D = 5 cm, u = 5 m s m s 2.40.Un A sistema system de of propano propane yand n-butane exists in equilibrio two-phasedevapor/liquid equilibrium aat10 bar y 323 2.40. n-butano existe en un dos fases vapor/líquido 10 bar and 323 K. The mole fraction of propane is about 0.67 in the phase de 0.40 en K. La fracción molar del propano es aproximadamente 0.67 en la fase vaporvapor y alrededor and about 0.40 in the liquid phase. Additional pure propane is added to the system, la fase líquida. Se agrega propano puro adicional al sistema, que de nuevo lleva al equilibrio a las which is equilibrium andpresentes. P, with both liquid vapor mismas T brought y P con again ambastofases, líquidaaty the de same vaporTaún ¿Cuál es eland efecto de agregar el phases still present. What is the effect of the addition of propane on the mole fractions propano sobre las fracciones molares de propano en las fases líquida y de vapor? of propane in the vapor and liquid phases? 2.41. Seis especies químicas se encuentran presentes en cantidades significativas en un sistema de frac2.41.cionamiento Six chemical aremetano, present etano, in significant amounts in a light-ends despecies petróleo: propano, isobutano, n-butano ypetroleum n-pentano.fracUna mezcla de tionation system: methane, ethane, propane, isobutane, n-butane, and n-pentane. A estas especies existe en equilibrio vapor/líquido en un recipiente cerrado. ¿De cuántas variables mixture of these species exists in vapor/liquid equilibrium in a closed vessel. On how de la regla de fase dependen las composiciones de las fases además de T y P? many phase-rule variables in addition to T and P do the compositions of the phases Sidepend? T y P permanecen iguales, ¿existe alguna manera de que la composición del contenido total del recipiente pueda modificarse (agregando o eliminando material) sin afectar las composiciones de If fases? T and P are to remain the same, is there any way the composition of the total conlas tents of the vessel can be changed (by adding or removing material) without affecting compositions 2.42. Sethe introduce etilenoofa the unaphases? turbina a 10 bar y 450 K, y se vacía a 1 (atm) y 325 K. Para m· = 4.5 kg s–1, determine el costo C de la turbina. Establezca las suposiciones que considere necesarias. 2.42. Ethylene enters a turbine at 10 bar and 450 K, and exhausts at 1(atm) and 325 K. For . you make. m = 4.5 kg s−1 , determine the cost C of the–1turbine. State any assumptions · Datos: H1 = 761.1 H2 = 536.9 kJ kg−1 C/$ = (15 200)(|W.|/kW)0.573 Data: H1 = 761.1 H2 = 536.9 kJ kg C/$ = (15, 200)(|W |/ kW)0.573 2.43. Para incrementar la temperatura de una casa, la calefacción debe modelarse como un sistema abier2.43.to,The of a home to its temperaature must be modeled as anuna open system, ya heating que la expansión delincrease aire doméstico a presión constante produce fuga de aire hacia los because expansion of the household air at constant pressure results in leakage air las mismas exteriores. Suponiendo que las propiedades molares del aire que abandona la casaofsean to the outdoors. Assuming that the molar properties of air leaving the home are the que las que corresponden al aire en el hogar, demuestre que los balances molar y de energía produsame as those of the air diferencial: in the home, show that eneregy and mole balances yield the cen la siguiente ecuación following differential equation: . dU dn +n Q = −P V dt dt . Here, ·Q is the rate of heat transfer to the air in the home, and t is time. Quantities P, Aquí, es la de calor para el aire en la casa, y t es el tiempo. Las cantidades V , n,Qand Urapidez refer tode thetransferencia air in the home. P, V, n y U se refieren al aire dentro de la casa. . 2.44. (a) Water flows through the nozzle of a garden hose. Find an expression for m in terms 2.44. a) Elofagua fluye a través de la boquilla de una manguera de jardín. Encuentre expresión para nozzle line pressure P1 , ambient pressure P2 , inside hose diameter D1 , and una m· en términos de la presión de la línea P1, la presión ambiental P2, el diámetro interno de la 02-SmithVanNess.indd 62 10/1/07 14:06:09 Problemas 63 manguera D1 y el diámetro de la boquilla de salida D2. Suponga que se tiene un flujo estable, así como operación isotérmica y adiabática. Para el agua líquida modelada como un fluido incompresible, H2 – H1 = (P2 – P1)/r para una temperatura constante. b) De hecho, el flujo no puede ser realmente isotérmico: esperamos que T2 > T1, obedeciendo a la fricción del fluido. Por lo tanto, H2 – H1 = C(T2 – T1) + (P2 – P1)/r, donde C es el calor específico del agua. De modo direccional, ¿de qué forma la incorporación del cambio de temperatura podría afectar el valor de m· como fue calculado en el inciso a)? 02-SmithVanNess.indd 63 8/1/07 12:50:39 Capítulo 3 Propiedades volumétricas de fluidos puros Las cantidades de trabajo y calor que se necesitan para llevar a cabo procesos industriales se calculan a partir del conocimiento de propiedades termodinámicas, tales como la energía interna y la entalpía. Para los fluidos, esas propiedades con frecuencia son evaluadas a partir de medidas del volumen molar como una función de la temperatura y la presión, y proporcionan relaciones de presión/volumen/temperatura (PVT), las cuales se expresan en forma matemática como ecuaciones de estado. La ecuación menos compleja, PV = RT, proporciona el modelo realista más simple del comportamiento de un fluido. Además las ecuaciones de estado sirven para la medición de los fluidos y la dimensión de recipientes y tuberías. En este capítulo se describe, en primer lugar, la naturaleza general del comportamiento PVT de fluidos puros; de ahí, se continúa con un tratamiento detallado del gas ideal; a continuación, la atención se enfoca en ecuaciones de estado más realistas, que proporcionan los fundamentos para la descripción cuantitativa del comportamiento de los fluidos reales; por último, se presentan las correlaciones generalizadas que permiten la predicción del comportamiento PVT de fluidos, de los cuales no se tiene información experimental. 3.1 COMPORTAMIENTO PVT DE SUSTANCIAS PURAS Las líneas 1-2 y 2-C de la figura 3.1 representan las condiciones de presión y temperatura de una sustancia pura en donde existen las fases líquida y sólida en equilibrio con una fase de vapor. Estas líneas de presión de vapor en función de temperatura caracterizan las relaciones de equilibrio sólido/vapor (línea 1-2) y líquido/ vapor (línea 2-C). La relación de equilibrio sólido/líquido está representada en la línea 2-3. Las tres líneas despliegan las condiciones de P y T, en las cuales es posible coexistir en dos fases y separa en el diagrama las regiones de una sola fase. Así, la línea 1-2, la curva de sublimación, separa las regiones de sólido y de gas; la línea 2-3, la curva de fusión, separa las regiones de sólido y líquido; la línea 2-C, la curva de vaporización, separa las regiones de líquido y de gas. El punto C se conoce como el punto crítico; sus coordenadas Pc y Tc corresponden a la presión y temperatura más altas a las cuales se observa que una especie química pura existe en equilibrio vapor/líquido. Las tres líneas se encuentran en el punto triple, donde las tres fases coexisten en equilibrio. De acuerdo con la regla de la fase, ecuación (2.7), el punto triple es invariante (F = 0). Si el sistema existe a lo largo de cualquiera de las líneas de dos fases de la figura 3.1, éste es univariante (F = 1), mientras que en las regiones de una sola fase es divariante (F = 2). 64 03-SmithVanNess.indd 64 8/1/07 12:51:57 65 3.1. Comportamiento pvt de sustancias puras Es posible representar los cambios de estado mediante líneas en el diagrama PT: un cambio isotérmico por una línea vertical y un cambio isobárico por una línea horizontal. Cuando una línea así cruza una frontera de fase, se presenta un cambio súbito en las propiedades del fluido a T y P constantes; por ejemplo, la vaporización para la transición de líquido a vapor. 3 Presión Pc Curva de fusión A Región de fluido C Región líquida Curva de vaporización B Región sólida Región gaseosa 2 1 Punto triple Figura 3.1: Diagrama PT para una sustancia pura. Región de vapor Curva de sublimación Tc Temperatura Es evidente que el agua en un matraz abierto es un líquido que está en contacto con el aire a través de un menisco. Si el matraz se encuentra sellado y el aire se extrae, el agua se vaporiza para reemplazar el aire, y el H2O llena el matraz. Aunque la presión en el matraz se reduce, todo parece igual. El agua líquida reside en la parte inferior del matraz porque su densidad es mucho mayor que la del vapor de agua, y las dos fases están en equilibrio en las condiciones representadas por un punto de la curva 2-C de la figura 3.1. Las propiedades del líquido y del vapor son muy diferentes. No obstante, si la temperatura aumenta de tal forma que el estado de equilibrio avanza de manera ascendente a lo largo de la curva 2-C, las propiedades de las dos fases se hacen cada vez más parecidas; así, en el punto C se hacen idénticas y el menisco desaparece. Una consecuencia es que las transiciones de líquido a vapor es posible que sucedan a lo largo de trayectorias que no cruzan la curva de vaporización 2-C, es decir, de A hasta B. Por lo tanto, la transición de líquido a gas es gradual y no incluye etapa de vaporización. La región que existe a temperaturas y presiones mayores que Tc y Pc se marca por las líneas discontinuas en la figura 3.1; éstas no representan fronteras de fase, sino más bien los límites establecidos por los significados concedidos con las palabras líquido y gas. En general, se considera una fase líquida si se produce vaporización a partir de la reducción de presión a temperatura constante. Se considera gas una fase si se produce una condensación a partir de la reducción de la temperatura a presión constante. Debido a que ninguno de estos procesos puede iniciarse más allá de la región de las líneas punteadas, se le llama región de fluido. La región gaseosa algunas veces está dividida en dos partes, como se indica mediante la línea vertical discontinua de la figura 3.1. A un gas a la izquierda de esta línea, que puede ser condensado por compresión a temperatura constante o por enfriamiento a presión constante, se le llama vapor. Un fluido que existe a una temperatura mayor que Tc se conoce como supercrítico. Un ejemplo es el aire atmosférico. 03-SmithVanNess.indd 65 8/1/07 12:51:58 66 CAPÍTULO 3. Propiedades volumétricas de fluidos puros Diagrama PV La figura 3.1 no proporciona información alguna acerca del volumen, tan sólo despliega las fronteras de fase en un diagrama PT. En un diagrama PV [figura 3.2a)] estas fronteras, a su vez, serán regiones donde dos fases (sólido/líquido, sólido/vapor y líquido/vapor) coexisten en equilibrio. Estas regiones están separadas por curvas limítrofes que representan fases únicas, cuyas cantidades relativas determinan los volúmenes molares (o específicos) en puntos intermedios. Aquí, el punto triple de la figura 3.1 es una línea horizontal, donde coexisten las tres fases a una sola temperatura y presión. Sólido/líquido C Fluido P Gas Líquido/vapor Vapor Sólido/vapor Vc C Pc Pc Q Líquido P Sólido Pc Líquido Tc N Va p or Tc T Tc Tc J K Líquido/vapor B D T1 Tc T2 Tc Vc V a) V b) Figura 3.2: Diagramas PV para una sustancia pura. a) Se muestran las regiones sólida, líquida y gaseosa. b) Se muestran las regiones de líquido, de líquido/vapor y de vapor con isotermas. La figura 3.2b) muestra las regiones de líquido, de líquido/vapor y de vapor en un diagrama PV, con cuatro isotermas sobrepuestas. Las isotermas en la figura 3.1 son líneas verticales, y a temperaturas mayores que Tc no cruzan una frontera de fase. En la figura 3.2b) la isoterma marcada con T > Tc es, por lo tanto, uniforme. Las líneas señaladas con T1 y T2 corresponden a temperaturas subcríticas y se componen de tres segmentos. El segmento horizontal de cada isoterma representa todas las posibles combinaciones de líquido y de vapor en equilibrio, variando desde 100% líquido en el extremo izquierdo hasta 100% vapor en el derecho. El lugar de estos puntos extremos es la curva en forma de domo marcada con BCD, donde la mitad izquierda (de B a C) representa líquidos de una sola fase en sus temperaturas de vaporización (ebullición) y la mitad derecha (de C a D), vapores de una sola fase a sus temperaturas de condensación. Los líquidos y vapores representados por BCD se conocen como saturados, y las fases coexistentes se conectan mediante el segmento horizontal de la isoterma a la presión de saturación específica para ésta. También conocida como la presión de vapor, está dada por un punto en la figura 3.1, donde una isoterma (línea vertical) cruza la curva de vaporización. La región de dos fases líquido/vapor se encuentra debajo del domo BCD, la región de líquido subenfriado se localiza a la izquierda de la curva de líquido saturado BC y la región de vapor sobrecalentado está si- 03-SmithVanNess.indd 66 8/1/07 12:51:59 3.1. Comportamiento pvt de sustancias puras 67 tuada a la derecha de la curva de vapor saturado CD. El líquido subenfriado existe para temperaturas por abajo y, el vapor sobrecalentado en temperaturas por arriba del punto de ebullición para una presión determinada. Las isotermas en la región de líquido subenfriado son de pendiente muy pronunciada, porque el volumen de los líquidos cambia muy poco con grandes variaciones en la presión. Los segmentos horizontales de las isotermas en la región de dos fases se vuelven progresivamente más cortos a temperaturas altas, y al final se reducen a un punto en C. Por esto, la isoterma crítica, marcada como Tc, exhibe una inflexión horizontal en el punto crítico C en la parte superior del domo, donde las fases líquida y de vapor no pueden distinguirse entre sí. Comportamiento crítico Se obtiene cierta idea acerca de la naturaleza del punto crítico a partir de una descripción de los cambios que ocurren cuando se calienta una sustancia pura en un tubo recto, sellado y de volumen constante. Estos procesos se indican en la figura 3.2b) mediante las líneas discontinuas verticales. También es posible trazar en el diagrama PT de la figura 3.3, donde la curva de vaporización (figura 3.1) aparece como una línea continua, y las líneas discontinuas son trayectorias a volumen constante en las regiones de una sola fase. Si el tubo se llena, ya sea con líquido o vapor, el proceso de calentamiento produce cambios que se sitúan a lo largo de las líneas discontinuas, por ejemplo, el cambio de E a F (líquido subenfriado) y el de G a H (vapor sobrecalentado). Las líneas verticales correspondientes de la figura 3.2b) no se muestran, pero se encuentran a la izquierda y a la derecha de BCD, respectivamente. V2l Vc Líquido C l V1 P F Q N (J, K) G E V1v H V2v Figura 3.3: Diagrama PT para un fluido puro donde se muestra la curva de presión de vapor y las líneas a volumen constante en las regiones de una sola fase. Vapor T Si el tubo se llena sólo de manera parcial con líquido (el resto es vapor en equilibrio con el líquido), el calentamiento provoca, al principio, los cambios descritos por la curva de la presión de vapor (línea continua) de la figura 3.3. Para el proceso indicado con la línea JQ de la figura 3.2b), de manera inicial, el menisco está cerca de la parte superior del tubo (punto J), y el líquido se expande lo suficiente debido al calentamiento hasta que llena completamente el tubo (punto Q). En la figura 3.3 el proceso describe una trayectoria que va de (J, K) hasta Q, y con calentamiento adicional se desvía de la curva de presión de vapor siguiendo la línea del volumen molar constante V2l . El proceso indicado por la línea KN de la figura 3.2b) comienza con un nivel inferior del menisco en el tubo (punto K), y el calentamiento origina que el líquido se vaporice hasta el punto en que el menisco retro- 03-SmithVanNess.indd 67 8/1/07 12:52:00 68 68 68 68 68 CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3. Properties of CHAPTER 3.3.Volumetric Volumetric Properties of Pure Pure CHAPTER 3.3. Volumetric Properties ofofFluids Pure CHAPTER Volumetric Properties PureFluids Fluids cede hacia el fondo del tubo (punto N). En la figura 3.3 el proceso se indica mediante una trayectoria que va bottom of tube N 3.3 the process traces atraces (J, K ))(J, to With bottombottom of the theof tube (point N).). On On Fig. 3.3 the process tracestraces a path pathaafrom from (J, K(J, to KN N With the tube NN).Fig. Fig. 3.3 the NN. . With of the(point tube(point (point ).elOn On Fig. 3.3 theprocess process path from K)..)to to With de (J, K) a N. bottom Con calentamiento adicional, proceso continúa a lo largo de lapath líneafrom del molar consvv. volumen vv. heating the path continues along the line of constant molar volume V furtherfurther heating the path continues along the line of constant molar volume V . vfurther heating the path continues along the line of constant molar volume V 2 2 further heating the path continues along the line of constant molar volume V22 . tante V 2. For aa unique filling of tube, particular intermediate meniscus level, heating unique fillingfilling of the theof tube, with particular intermediate meniscus level, the the heating For aaunique tube, aaparticular intermediate meniscus level, the For unique filling ofthe thewith tube,aawith with particular intermediate meniscus level, the heating Para un For llenado único del tubo, con una altura intermedia particular del menisco, el proceso deheating calentaprocess follows a vertical line on Fig. 3.2(b) that passes through the critical point C. Physically, process follows a vertical line on Fig. 3.2(b) that passes through the critical point C. Physically, process follows a vertical line on Fig. 3.2(b) that passes through the critical point C. Physically, process follows a vertical on 3.2b) Fig. 3.2(b) that por passes through the critical point C. Physically, miento describe una línea vertical en la line figura que pasa el punto crítico C. Físicamente, el proceso heating does produce much in level the meniscus. As critical point is heatingheating does not not produce much change change in the thein level of theof meniscus. As the theAs critical point point ispointisis does not change the level the meniscus. the does not produce much change theof level the meniscus. As the critical no produce unheating gran cambio enproduce el nivel much del menisco. Ainmedida queofel proceso se acerca alcritical punto crítico, el approached, the meniscus becomes indistinct, then hazy, and finally disappears. On Fig. 3.3 approached, the meniscus becomes indistinct, then hazy, and finally disappears. On Fig. 3.3 approached, the meniscus becomes indistinct, then hazy, and finally disappears. On Fig. approached, the meniscus indistinct, thendesaparece. hazy, and finally disappears. Fig.3.3 3.3 menisco se torna indistinguible, despuésbecomes es nebuloso y al final En la figura 3.3, laOn trayectoria the first the curve, proceeding from (J, K ))(J, to the path path first follows the vapor-pressure vapor-pressure curve, curve, proceeding from point point (J, K(J, toKthe the critical path first follows vapor-pressure proceeding ))tocritical the the pathfollows first follows the vapor-pressure proceeding from point to the critical primero siguethe la curva de presión dethe vapor, procediendocurve, del punto (J, K) alfrom puntopoint crítico C,Kdonde se critical introdupoint C, where it enters the single-phase fluid region, and follows V , the line of constant point point C, where it enters the single-phase fluid region, and follows V , the line of constant c c C, where it enters the single-phase fluid region, and follows V , the line of point C, where enters the single-phase region, and follows Vcc , igual the line of constant constant ce a la región fluida de una solait fase y sigue a Vc, la líneafluid de volumen molar constante al volumen crímolar equal to the critical volume of fluid. molar volume volume equal equal to theto critical volume of the theof fluid. volume molarvolume volume equal tothe thecritical critical volume ofthe thefluid. fluid. tico del fluido.molar Single-Phase Region Single-Phase Region Region Regiones Single-Phase de una sola fase For regions of diagram where aa single phase exists, Fig. implies aa relation conFor the the regions of the theof diagram wherewhere single phasephase exists, Fig. 3.2(b) 3.2(b) implies relation con- conFor the the aasingle Fig. implies aarelation For theregions regions of thediagram diagram where single phaseexists, exists, Fig.3.2(b) 3.2(b) implies relation connecting P, V , and T . Expressed analytically, as f (P, V, T ) = 0, such a relation is known as P, V , and T . Expressed analytically, as f (P, V, T ) = 0, such a relation is known as aas necting P, V , and T . Expressed analytically, as f (P, V, T ) = 0, such a relation is Para lasnecting regiones del diagrama donde sólo existe una fase, la figura 3.2b) implica una relación que conecta P, necting P, V , and T . Expressed analytically, as f (P, V, T ) = 0, such a relation isknown known as PVT of state. It relates pressure, molar or specific volume, and temperature for a PVTse equation of state. It relates pressure, molar or specific volume, and temperature for a aaequation PVT equation of state. It relates pressure, molar or specific volume, and temperature for a V y T, laaacual expresa de manera analítica mediante f (P, V, T) = 0; una relación así se conoce como ecuación PVT equation of state. It relates pressure, molar or specific volume, and temperature for a pure homogeneous in equilibrium states. The ideal-gas of P V ,,homogéneo pure homogeneous fluid in equilibrium states. The ideal-gas equation of state, state, Pstate, V= = RT has pure homogeneous fluid The equation of PPRT VV = RT de estado PVT. Relaciona lafluid presión, elin volumen molarstates. o específico y equation la temperatura para un fluido pure homogeneous fluid inequilibrium equilibrium states. Theideal-gas ideal-gas equation ofstate, =has RT, ,has has approximate validity for the low-pressure gas region of Fig. 3.2(b), and is discussed in detail validity for the low-pressure gas region of Fig. 3.2(b), and is discussed in detail approximate validity for the low-pressure gas region of Fig. 3.2(b), and is discussed in puro en approximate estados de equilibrio. La ecuación de estado de gas ideal, PV = RT, tiene una validez aproximada para approximate validity for the low-pressure gas region of Fig. 3.2(b), and is discussed indetail detail Sec. 3.3. indel Sec. 3.3. inin Sec. 3.3. la regiónin gas de baja Sec. 3.3.presión de la figura 3.2b), y se discutirá con detalle en la sección 3.3. An of solved for any one of the three quantities ,,P, or An equation equation of state state may be solved for any one ofone thetres three quantities P, or as An equation of state may be solved for any of the three VTT or as Una ecuación de puede resolverse para cualquiera de las cantidades, P,P, V oVVT, como una Anestado equation ofmay statebe may be solved for any one of the threequantities quantities P, V, ,as or TTfunas a function of the other two. For example, if V is considered a function of T and P, then of the other two. For example, if V is considered a function of T and P, then aa function of the other two. For example, if V is considered a function of T and P, then ción de alasfunction otras dos. Por ejemplo, si se considera a V como una función de T y P, entonces V = V(T, P), y function of the other two. For example, if V is considered a function of T and P, then V P), and V= =V VVV(T, (T, P), and ==V P), � V(T, (T, P),and and � � � � � � � � � ∂∂V V �∂∂VV � ∂∂V V �∂∂VV � ddV d P (3.1) (3.1) V= =ddVV == dT dT + +dT d P (3.1) (3.1) dP dT (3.1) ∂∂TT PP∂∂TT ∂∂+ P P+ TT∂∂PP T d P PP T The partial derivatives in this equation have definite physical meanings, and related to The partial derivatives in this equation have definite physical meanings, and yare are related to two twotototwo The partial derivatives in this equation have definite physical meanings, and are related The partial derivatives in this equation have definite physical meanings, and are related two Las derivadas parciales en esta ecuación tienen significados físicos bien definidos están relacionadas con properties, commonly tabulated for liquids, and defined as follows: properties, commonly tabulated for liquids, and defined as follows: properties, commonly tabulated for liquids, and defined as follows: properties, commonly tabulated for liquids, and defined as follows: dos propiedades que comúnmente se tabulan para los líquidos, y que se definen como: � � � �� �� 11 ∂∂V 1V1 ∂∂VV • Volume expansivity: β ≡ (3.2) • Volume expansivity: (3.2) (3.2) ••Volume expansivity: • Coeficiente de expansión volumétrica:β ≡ Vββ≡≡∂ T (3.2) (3.2) Volume expansivity: V ∂VTV PP∂∂TT P P � � � �� �� 11 ∂∂V 1V1 ∂∂VV •• Isothermal compressibility: κ ≡ − (3.3) Isothermal compressibility: κ ≡ − (3.3) (3.3) • Isothermal compressibility: κ ≡ − • Isothermal compressibility: κV (3.3) • Compresibilidad isotérmica: (3.3) ∂∂V P V≡ − PV TT∂∂PP T T In Equations (3.1) (3.3) In combination combination Equations (3.1) through (3.3) yield: yield: In Equations (3.1) through (3.3) yield: La combinación de las ecuaciones (3.1)through a(3.1) (3.3) produce la ecuación: Incombination combination Equations through (3.3) yield: ddV V ddVV = κκdT dd − P (3.4) (3.4) = ββ dT dT − P−κκddPP (3.4) (3.4) ==− β βdT (3.4) V V VV Las isotermas para la fase de laphase partephase izquierda deside la figura 3.2b) son are de muy pronunThe for the liquid on the left Fig. very and The isotherms isotherms forlíquida the liquid phase on theon left side of Fig. 3.2(b) arependiente very steep and The for the the left side of Fig. are very The isotherms isotherms for the liquid liquid phase on the leftof side of3.2(b) Fig. 3.2(b) 3.2(b) aresteep very steep steep and and ciada y closely muy cercanas entre sí. De esta manera, (∂V/∂T) y (∂V/∂P) y, por tanto, b y k son pequeños. Este and (∂ V /∂ P) and hence both β and κ are small. spaced. Thus both (∂ V /∂ T ) P T and (∂ V /∂ P) and hence both β and κ are small. closely spaced. Thus both (∂ V /∂ T ) P T closely and (∂ (∂VVT/∂ /∂P) P)TT and and hence hence both both ββ and and κκ are are small. small. closely spaced. spaced. Thus Thus both both (∂ (∂VVP/∂ /∂TT))PP and comportamiento característico debehavior losof (lejos de la región delregion) puntoregion) crítico) sugiere una idealización, This behavior liquids (outside the critical suggests an This characteristic characteristic behavior oflíquidos liquids (outside the critical region) suggests an idealization, idealization, This of (outside the suggests an This characteristic characteristic behavior of liquids liquids (outside the critical critical region) suggests an idealization, idealization, comúnmente utilizada en mecánica de fluidos, que se conoce como el fluido incompresible, para el queboth b yk commonly employed in fluid mechanics and known as the incompressible fluid, for which both commonly employed in fluid mechanics and known as the incompressible fluid, for which both commonly commonlyemployed employedininfluid fluidmechanics mechanicsand andknown knownas asthe theincompressible incompressiblefluid, fluid,for forwhich which both son cero. ningún fluido real es incompresible, pero la idealización es muy útil porque con frecuenββ De and κ are zero. No real fluid is truly incompressible, but the idealization is useful, because andhecho, κ are zero. No real fluid is truly incompressible, but the idealization is useful, because ββand andκκ are arezero. zero. No Noreal realfluid fluidisistruly trulyincompressible, incompressible,but butthe theidealization idealizationisisuseful, useful,because because cia proporciona unaamodelo bastante realista delmodel comportamiento del para finespurposes. prácticos. No existe itit provides sufficiently realistic model of behavior for many practical No provides sufficiently realistic model of liquid liquid behavior forlíquido many practical purposes. No ititprovides aasufficiently realistic of behavior for purposes. No provides sufficiently realistic model ofliquid liquid behavior formany manypractical practical purposes. No ningunaequation ecuación de estado para un fluido incompresible, porque V es independiente de T y P. of state exists for an incompressible fluid, because V is independent of T and P. equation of state exists for an incompressible fluid, because V is independent of T and P. equation independent of TT and P. equationof ofstate stateexists existsfor foran anincompressible incompressiblefluid, fluid,because becauseVV isis independent of and P. Para líquidos, b casi esalways positiva (es positive una(liquid excepción agua líquida entre 0C °C) k es nece◦an For ββ siempre is positive water between 00◦◦C ◦CCexcepFor liquids liquids is almost almost always positive (liquid waterelwater between C and and 4◦◦and Cyis is444an excepFor ββisisalmost always (liquid 00◦◦C4C isisyan Forliquids liquids almost always positive (liquid waterbetween between and anexcepexcepsariamente positiva. En condiciones no cercanas al punto crítico b y k son funciones débiles de la temperatution), and κ is necessarily positive. At conditions not close to the critical point, β and κ are tion), and κ is necessarily positive. At conditions not close to the critical point, β and κ are tion), tion),and andκκ isisnecessarily necessarilypositive. positive. At Atconditions conditionsnot notclose closetotothe thecritical criticalpoint, point,ββ and andκκ are are 03-SmithVanNess.indd 68 8/1/07 12:52:03 3.1. Behavior of of Pure Substances 6969 3.1.PVT PVT Behavior Pure Substances 69 69 3.1. PVT PVT Behavior Behavior of of Pure Pure Substances Substances 69 3.1. 69 3.1. PVT Behavior of Pure Substances 3.1. PVT Behavior of Pure Substances 3.1. Comportamiento de sustancias puras Thus for small changes in T and P little error is69 69 weak functions ofpvt temperature and pressure. weak functions of temperature and pressure. Thus Thus for for small small changes changes in in TT and and P P little little error error69 is 3.1. PVT Behavior of Pure Substances weak functions of temperature and pressure. is weak functions of temperature and pressure. Thus for small changes in T and P little error is weak functions of temperature and pressure. Thus for small changes in T and P little error is introduced if they areare assumed constant. Integration Eq. (3.4) then yields: introduced if they they are assumed constant. Integration of Eq. (3.4) then yields: weak functions of temperature and pressure. Thusof for small changes in T and P little error is introduced if assumed constant. Integration of Eq. (3.4) then yields: introduced ifeste they are assumed constant. Integration of Eq. (3.4) then yields: yields: if they are assumed constant. Integration of Eq. (3.4) then ra y la introduced presión. De modo, para cambios pequeños de T y P se introduce error is si se les weak functions of temperature and pressure. Thus for small changes in T un andpequeño P little error introduced if they are assumedVconstant. Integration of Eq. (3.4) then yields: 2V 222 pressure. Thus for small changes in T and P little error is V weak functions temperature and = β(T − T ) − κ(P − P ) (3.5) ln ln V considera constantes. Laofintegración de la ecuación (3.4) produce: V 2 1 2 1 = β(T − T ) − κ(P − P ) (3.5) introduced if they are assumed constant. Integration of Eq. (3.4) then yields: 22 = β(T222 − T111) − κ(P222 − P111) (3.5) ln Vconstant. = β(T β(T − TT11)) − − of κ(P −(3.4) (3.5) ln κ(P PP11))then yields: (3.5) ln 1V introduced if they are assumed Integration Eq. 22 − 22 − 1121 = V V = β(T − T ) − κ(P − P ) (3.5) ln V 1 2 1 2 1 1 V 2 V21 =than This is is a less restrictive approximation the assumption an incompressible fluid. This a less restrictive approximation than the fluid. β(T T1assumption ) − κ(P2of−of P1an ) incompressible (3.5) (3.5) ln V This is less restrictive approximation than the assumption of an incompressible fluid. 2− This is is aaa less less restrictive restrictive approximation approximation than theTassumption ofPan incompressible fluid. fluid. (3.5) This than the ) − κ(P2 −of ) incompressible ln V1 = β(T 2− 1assumption 1an This is a less restrictive approximation than the assumption of an incompressible fluid. V1 Thisaproximación is a less restrictive the assumption an incompressible Ésta es una menosapproximation restrictiva quethan la suposición de unof fluido incompresible.fluid. This is a less restrictive approximation than the assumption of an incompressible fluid. Example 3.1 Example 3.1 Example 3.1 3.1 ◦ ◦◦ Example For liquid acetone at at 2020 C◦ C and 1 bar, Example 3.1 For liquid acetone and 1 bar, For liquid acetone at 20 C and bar, For liquid liquid acetone acetone at 20 20◦◦◦C C and and 111 bar, bar, For at Example 3.1 −3 ◦ −1 −6−6 −1−1 Ejemplo 3.1 For liquid acetone at 20 C and 1 bar, −3 ◦ −1 −6 −1 −3 ◦ −1 Example 3.1 C κ = 6262 ×× 1010 bar ββ = 1.487 × 10 −3 ◦ −1 −6 −1 C κκ = = 1.487 × 10 ◦ = 62 × 10−6 bar−1 β = 1.487 × 10−3 −3 ◦◦C−1 −1 −6 bar −1 3 333−1−1 −1 VV == 1.287 cmcm g33 g −1 1.287 V = 1.287 cm gg−1 −1 For liquid at 20◦ C and 1 bar, C κ = 62 × 10 bar V = 1.287 cm =acetone 1.487 × × 10 10−3 κ = 62 × 10 bar V = 1.287 cm g ββ = 1.487 3 −6 bar−1 −1 liquid acetone at 20 y◦C and Para For la For acetona líquida a×20 °C 1−1 bar, 1 bar, C κ = 62 × 10 V = 1.287 cm g β = 1.487 10 acetone, find: For acetone, find: −3 ◦ C−1 −6 bar−1 −1 For acetone, find: κ = 62 × 10−6 V = 1.287 cm33 g−1 β = 1.487 × 10−3 For acetone, acetone, find: For find: ◦ C−1 −1 ◦ κ = 62 × 10 bar V = 1.287 cm g β = 1.487 × 10 ◦ ◦ For acetone, find: (a)(a) The value of of (∂ (∂ P/∂ T )TV ) at 2020 C◦ C and 1 bar. The value P/∂ and 1 bar. V at (a) The value of (∂ P/∂ at 20 C and bar. (a) The The value value of (∂ (∂P/∂ P/∂TTT)))VVVV at at 20 20◦◦◦C C and and 111 bar. bar. For (a) acetone, find: of Para la For acetona, encuentre: ◦ C◦◦◦ and 1 bar to (a) The value of (∂ P/∂ T ) at 20 C and 1 bar. acetone, find: V byby (b)(b) The pressure generated heating at at constant VV from 2020 C and 1 bar to The pressure generated heating constant from ◦ C bar to (b) The pressure generated by heating at constant V from 20 ◦◦C (a) value of (∂ P/∂ T ) at 20 C and 1 bar. (b) The pressure generated by heating at constant V from 20 C and and 111 bar bar to to V ◦ (b) The pressure generated by heating at constant V from 20 ◦◦◦C. ◦ C and 30 a) El valor deC. (∂P/∂T) 20 °CTy)1 (a) The value P/∂ at by 20◦heating C and 1at bar. (b)3030 pressure generated constant V from 20 and 1 bar to Vofa(∂ 30 C. V bar. ◦ ◦ 30◦C. C. ◦ C and 1 bar to (b) 30 The pressure generated by heating at constant V from 20 b) La(c) presión generada por calentamiento a V from constante °C 1 hasta 30 °C. ◦ C◦◦◦desde ◦ C◦bar C. ◦◦◦C and 1V20 bar to yto 020 and 1010 The change in in volume forfor a by change 20 C and 1 bar 0 and bar. (c) The change volume a change from 20 1bar. bar to (b) pressure generated heating at constant from ◦ C and 1 bar to 0 C and 10 bar. (c) The change in volume for a change from 20 ◦ ◦C ◦ ◦10 30 C. c) El cambio de volumen para una modificación de 20 °C y 1 bar a 0 °C y bar. C and 1 bar to 0 C and 10 bar. (c) The The change in volume for a change from 20 C and 1 bar to 0 C and 10 bar. (c) change in volume for a change from 20 ◦ C. ◦ ◦ 30 (c) The change in volume for a change from 20 C and 1 bar to 0 C and 10 bar. (c) The change in volume for a change from 20◦◦ C and 1 bar to 0◦◦ C and 10 bar. Solución 3.1 (c) The change Solution 3.1 Solution 3.1 in volume for a change from 20 C and 1 bar to 0 C and 10 bar. Solution 3.1 Solution 3.1 a) LaSolution derivada (∂P/∂T) determina con la aplicación de la ecuación (3.4) para elthe caso en el que (a) The derivative P/∂ T )T determined byby application of of Eq.Eq. (3.4) to to the case 3.1 V(∂se(∂ V )is (a) The derivative P/∂ determined application (3.4) case V is V (a) The derivative (∂ P/∂ is determined by application of Eq. (3.4) to the case V (a) Theyderivative derivative (∂P/∂ P/∂ is determined determined by by application application of of Eq. Eq. (3.4) (3.4) to to the the case case The (∂ TTTd)))V V es constante dV = 0: V is V Solution 3.1 for(a) which V is constant and = 0: for which V is constant and V = 0: (a) The derivative (∂ P/∂and T )VddddV is determined by application of Eq. (3.4) to the case for which V is constant and V == 0: 0: Solution 3.1 for which which V is is constant constant and V= 0: for V (a) The derivative (∂ P/∂and T )Vd V is determined by(V application of Eq. (3.4) to the case for which V is constant = 0: constante) β dT κ determined dκκPdd P == 0 (const V )V β dT − 00 by (const )) of Eq. (3.4) to the case (a) derivative (∂ P/∂ )V− is application ββTdT dT − P = (const V for The which V is�constant and d V = 0: dT − κ d P = 0 (const V ) β − κ d P = 0 (const V ) �� � for which V is constant andβ d V− 1.487 = 0: −3−3 � � dP × =× 0 10 −3(const V ) ∂� 10 −3 �P∂∂ P � β dT � β κ 1.487 P 1.487 × 10 −3 −1 −3 −1 or =−= = 2424 bar −1 β= dTβββ κ 1.487 d1.487 P =× 0−6 (const Vbar )◦ C◦◦◦◦◦−1 P �= × 10 10 �∂∂P or = C or = = = 24 bar C o −1 −3 −6 (const −6 ∂�T∂∂ T κ β dT − κ d P = 0 V ) 62 × 10 −6 or = = = 24 bar C−1 or = = = 24 bar C P 1.487 × 10 β κ 62 × 10 V�V T κ V 62 × 10 ◦ −1 −6 −6 V = κκ = 62 −3 ∂ T or = 24 bar C ∂ T 62 × 10 × 10 P �VV 1.487 ×1010 β � ∂∂ T −6−3 −1 κ = 1.487 62 ×× orIf β and κ are assumed = 24 bar ◦◦ C−1 ∂P V = 10 β ◦ C◦◦◦ temperature −6 (b)(b) constant in the 10 interval, then thethe b) Si se supone que b y k son constantes en el intervalo de temperatura de 10 °C, por lo tanto la If β and κ are assumed constant in the 10 C temperature interval, then ∂ T κ or = = = 24 bar C 62 × 10 (b) If β and κ are assumed constant in the 10 C temperature interval, then the ◦ ◦ V −6 (b) If If ββderived and κκ in are assumed in the 10◦C C temperature temperature interval, interval, then then the the (b) and are constant in the 10 ∂assumed Tmay κconstant 62 × 10 equation (a) be written (V = const): V ecuación deducida en a) se puede escribir como (V = constante): equation derived in (a) may be written (V = const): (b) If β and κ are constant 10 C temperature interval, then the equation derived in (a) may be written (V const): equation derived in assumed (a) may may be be writtenin (Vthe const): equation derived in (a) written (V === const): (b) If β and κ are constant in(Vthe 10◦◦ C temperature interval, then the equation derived in assumed (a) may βbe written = const): β β (b) If β and κ are assumed constant in the 10 temperature interval, then the �P == == (24)(10) =C 240 barbar ββwritten �P �T (24)(10) = 240 equation derived in (a) may be�T (V = const): �P = �T = (24)(10) = 240 bar κbe �P = �T = = (V (24)(10) = 240 240 bar bar �T (24)(10) = κβ equation derived in (a)�P may= written = const): κ �P = κβκ �T = (24)(10) = 240 bar = P1= +1 κ+ �P === 1(24)(10) + 240 == 241 bar and P2P y P �P 1+ 240 = 241 bar and 222 = = P + �P = + 240 = 241 bar and P �P �T 240 bar == P1111β +�T �P= =(24)(10) + 240 240= =240 241bar bar and P + �P = 111 + = 241 bar and PP�P 22 = κ and P2 = P1 κ+ �P = 1 + 240 = 241 bar Direct substitution into Eq.Eq. gives: c) La (c) sustitución directa en la (3.5) da: P(3.5) �P = 1 + 240 = 241 bar and Pecuación (c) Direct substitution into (3.5) gives: 2 = 1+ (c) Direct substitution into Eq. (3.5) gives: (c) Direct Direct substitution substitutionPinto into Eq. (3.5) gives: (c) (3.5) P1 + �Pgives: = 1 + 240 = 241 bar and 2 = Eq. (c) Direct V substitution into Eq. (3.5) gives: 2V 222 −3−3 −6−6 V −3 −6 (1.487 ×× 10 )(−20) −− (62(62 ×× 1010 )(9) == −0.0303 ln ln −3 −6 V2= V (1.487 10 )(−20) −0.0303 (c) Direct substitution into Eq. (3.5) gives: 2 = = (1.487 × 10 )(−20) − (62 × 10 )(9) = −0.0303 ln −3 −6)(9) −3 V1V = (1.487 (1.487 ×Eq. 10−3 )(−20) − (62 (62 × × 10 10−6 )(9) = = −0.0303 −0.0303 ln 10 )(−20) − )(9) (c) Directln substitution into× (3.5) gives: 1121 = V −6 V121 = (1.487 × 10 )(−20) − (62 × 10 )(9) = −0.0303 ln V V −3 −6 V V2V 3 333−1−1 222ln V21 = (1.487 × 10−3 )(−20) − (62 × 10−6 )(9) = −0.0303 V −1 0.9702 and = (0.9702)(1.287) 1.249 cmcm g33 gg−1 −1 V2= V = V (0.9702)(1.287) = 1.249 2ln V0.9702 × 10 V2)(−20) − (62 × 10 = )(9) = −0.0303 222 = = 0.9702 and V = (0.9702)(1.287) = 1.249 cm 1 = (1.487and −1 V1V = 0.9702 and V = (0.9702)(1.287) = 1.249 cm y = 0.9702 and V = (0.9702)(1.287) = 1.249 cm gg−1 22 1121 V1 V 3 V = 0.9702 and V = (0.9702)(1.287) = 1.249 cm g V 1 2 1 V2 �V = V − V = 1.249 − 1.287 = −0.038 cm3 g333−1−1 −1 3 −1 Then, −1 V 2V 1V Then, �V = 1.249 1.287 = −0.038 gg−1 2− 1= 0.9702 and V2 =− (0.9702)(1.287) =cm 1.249 cm g Then, �V = V − V = 1.249 − 1.287 = −0.038 cm −1 33 g 21 = �V Then, V �V = =V V2222 − −and V1111 = = 1.249 1.249 − 1.287 = −0.038 cm g Then, V − 1.287 = −0.038 cm V1 = 0.9702 V = (0.9702)(1.287) = 1.249 cm3 g−1 2 − 1.287 = −0.038 cm3 g−1 Then, V1 �V = V2 − V1 = 1.249 En tal caso, Then, �V = V2 − V1 = 1.249 − 1.287 = −0.038 cm3 g−1 Then, �V = V2 − V1 = 1.249 − 1.287 = −0.038 cm3 g−1 03-SmithVanNess.indd 69 8/1/07 12:52:12 70 70 70 3.2 ECUACIONES DE ESTADO VIRIALES 3.2 VIRIAL EQUATIONS OF STATE 3.2 VIRIAL EQUATIONS OF STATE CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3.3.Volumetric Properties of Pure CHAPTER 3. Volumetric Properties of Pure Fluids Las isotermas para gases y vapores, que se localizan arriba y a la derecha de CD en la figura 3.2b), son curvas Isotherms for gases and vapors, lying above and to the right of C D in Fig. 3.2(b), are relarelativamente simples para las que V disminuye a medida que P se incrementa. Aquí, el producto PV para una Isotherms forcurves gases for andwhich vapors, aboveasand to the right of Cthe D product in Fig. 3.2(b), relatively simple V lying decreases P increases. Here, P V forare a given T determinada es mucho más constante que cualquiera de sus integrantes, y por tanto más fácil de representar tively simple curvesmore for which decreases as P increases. Here, the product P Vmore for a easily given T should be much nearlyVconstant than either of its members, and hence de manera analítica como una función de P. Esto sugiere expresar a PV para una isoterma, por medio de T should beanalytically much moreasnearly constant of its members,Pand more easily represented a function of P.than Thiseither suggests expressing V forhence an isotherm by a un desarrollo en serie de potencias de P: represented as a function of P. This suggests expressing P V for an isotherm by a power seriesanalytically in P: power series in P: P V = a + b P + c P 22 + · · · 2 If b′, ≡ B ��, ′,c etc., ≡ aCen��, tal etc.,caso, then, P V = a + b P + c P + · · · Si b ≡ aB c ≡a aC If b ≡ a B � , c ≡ aC � , etc., then, P V = a(1 + B �� P + C �� P 22 + D �� P 33 + · · ·) (3.6) (3.6) � � 2 � 3 P V = a(1 + B P + C P + D P + · · ·) (3.6) ��, Cson ��, etc., donde a,where B ′, Ca,′, B etc., constantes para una y especie química are constants for atemperatura given temperature and a givendeterminadas. chemical species. � , etc., Enwhere principio, derecho de ecuación (3.6) es infinite una serie infinita. Dechemical cualquier modo, en la práctiB � ,elClado are constants for given and a given species. Ina,principle, the right sidela of Eq.a(3.6) istemperature an series. However, in practice a finite ca se emplea un número finito de In términos. hecho, la información muestra a presiones In of principle, the right side an infinite However, inque practice a finite number terms is used. fact,ofPEq. VDe T(3.6) dataisshow that atseries. low PVT pressures truncation after twobajas el truncamiento después de is dosused. términos con proporciona satisfactorios. number of terms In fact, Pfrecuencia V T data show that at resultados low pressures truncation after two terms often provides satisfactory results. terms often provides satisfactory results. Ideal-Gasde Temperatures; Universal Gas Constant Temperaturas gases ideales; constante universal de los gases Ideal-Gas�� Temperatures; Universal Gas Constant �� Parameters B , C , etc., in Eq. (3.6) are species dependent and functions of temperature, but � , etc., Los parámetros B ′,a BC en ecuación son de and laof temperatura y dependen de la Parameters Cetc., in Eq. (3.6)(3.6), are species dependent functions of temperature, butespecie, parameter is� ,′,found byla experiment to be the funciones same function temperature for all chemical pero, enspecies. forma experimental, seby demuestra que elof parámetro a es la misma de la parameter a isisfound by experiment to be the same function of temperature for alltemperatura chemical This shown measurements volume as a function offunción P for various gases at para todas lasconstant especiestemperature. químicas. Esto muestra mediciones del como una función species. This is shown bysemeasurements of de volume as a of function forfour various gases atde P paFigure 3.4, fora través example, is a plot P Vvolumen vs.ofPPfor gases at the ra diferentes gases atemperature temperatura Porlimiting ejemplo, desame PV en constant temperature. Figure 3.4,The for example, islaafigura plotP of Pes VPuna vs. P forthe four gases at triple-point ofconstante. water. value of V3.4as → gráfica 0 is forfunción allthe of de P, para cuatro gases en la temperatura del punto del agua. Todos el mismo valor límite de triple-point of water. limiting value of Pbecomes: Vlos as gases P →tienen 0 is the same for all of the gases. Intemperature this limit (denoted by The thetriple asterisk), Eq. (3.6) PV conforme P →In0.this En limit este límite (denotado por∗el asterisco), labecomes: ecuación 3.6 será: the gases. (denoted by the asterisk), Eq. (3.6) (P V )∗ = a = f (T ) (P V )∗ = a = f (T ) Figura 3.4: (PV)*, el valor límite limiting value ofVes)P∗V as de PV conforme P →(P0, Figure 3.4: , the P → 0, independent independiente delis gas. limiting value of P V asof the gas. P → 0, is independent of the gas. 0 0 H2 PV/cm3 bar mol1 Figure 3.4: (P V )∗∗, the 3 bar 3 bar �1 �1 PV/cm PV/cm molmol H22 H N22 N Air Air N 2 �1 � 22,711.8 22,711.8 cm33 bar bar mol mol3�1 (PV)*t*t � (PV) 22 711.8 cm bar mol1 (PV)* cm t (PV)*t � 22,711.8 cm3 bar mol�1 Air O22 O O2 H2 N2 Aire O2 � 273.16 273.16 K K� � triple triple point point of of water water TT � T 273.16 K punto triple del agua T � 273.16 K � triple point of water 0 P P P This property gases basis for establishing an absolute temperature scale. All that Todo Esta propiedad de losof gases esiselthe fundamento para establecer una escala de temperatura absoluta. This es property of gases is the basis establishing an absolute scale. thatespecíis requiere required is la theasignación arbitrary assignment functional relationship ) and the assignment lo que se arbitraria deoffor lathe relación funcional f (T) y temperature laf (T asignación de unAll valor issolo required thelaarbitrary themás functional f (T )internacional andthe theone assignment a specific value to a single point onofthe scale. The relationship simplest procedure, adopted fico a unof puntoisde escala. Elassignment procedimiento simple, adoptado a nivel para definir la of a specific value toes:a the single pointscale on the scale. internationally to define Kelvin (Sec. 1.5): The simplest procedure, the one adopted escala Kelvin (sección 1.5), internationally to define the Kelvin scale (Sec. 1.5): 03-SmithVanNess.indd 70 8/1/07 12:52:17 3.2. Virial VirialEquations EquationsofofState State 71 71 3.2. 71 71 Virial Equations State 3.2. Virial of State 3.2. 3.2. Ecuaciones deEquations estadoofviriales 71 3.2. Virial Equations of State 71 Makes(P (PVV)∗)∗∗∗directly directlyproportional proportionaltotoTT, ,with withRRasasthe theproportionality proportionalityconstant: constant: •• Makes • Makes (P V(P)∗de proportional to Tto, with R asRthe proportionality constant: • Makes Vdirectly )manera directly T , with as the constant: • Establecer (PV)* queproportional sea directamente proporcional aproportionality T, con R como la constante de propor• Makes (P V )∗ directly proportional to T , with R as the proportionality constant: ∗ cionalidad: (PVV)∗)∗∗ ==aa≡≡RT RT (3.7) (P (3.7) (P V(P)∗V= (3.7) ) a=≡a RT ≡ RT (3.7) ∗ (P V ) = a ≡ RT (3.7) (3.7) Assignsthe thevalue value273.16 273.16KKtotothe thetemperature temperatureofofthe thetriple triplepoint pointofofwater water(denoted (denotedby by •• Assigns • Assigns the the value 273.16 K toKthe temperature of the triple point of water (denoted by by • Assigns value 273.16 to the temperature of the triple point of water (denoted subscript t):273.16 subscript t): • Asignar el valor de K a la temperatura del punto triple delpoint agua of (denotado por el subíndice t): • subscript Assigns the of the triple water (denoted by t): value subscript t): 273.16 K to the temperature ∗ ∗ ∗ (PVV)t)∗t ==RR××273.16 273.16KK (3.8) (P (3.8) subscript t): (P V(P)∗tV= K K (3.8) )t R=×R273.16 × 273.16 (3.8) (3.8) ∗ t (P V )t = R × 273.16 K (3.8) DivisionofofEq. Eq.(3.7) (3.7)by byEq. Eq.(3.8) (3.8)gives: gives: Division El resultado de la Eq. ecuación (3.7) por la (3.8) nos da: Division of la Eq. (3.7) bydeEq. (3.8) gives: Division ofdivisión Eq. (3.7) by (3.8) gives: Division of Eq. (3.7) by Eq. (3.8) gives: /K (PVV)∗)∗∗ TT/K (P (P V(P)∗V )∗∗∗ ==T /K T /K (P∗VV= 273.16 K (P )t)∗t∗t =T273.16 /K K K (P V V (P (P))∗tV = )t 273.16 273.16 K ∗ (P V )t 273.16 K (PVV)∗)∗∗ (P /K==273.16 273.16 (3.9) (3.9) o (P V(P)∗V )∗∗∗ oror TT/K (3.9) ∗VV))∗t (P or or T /K = 273.16 (3.9) T /K = 273.16 (3.9) (P (P V ) t ∗ ∗ t or T /K = 273.16 (P V(P)t∗V )t (3.9) (P V )t Equation (3.9) establishes the Kelvin temperature scale throughout thetemperature temperature range for Equation establishes the Kelvin temperature throughout the range for La ecuación (3.9)(3.9) establece la escala Kelvin de temperaturascale en todo el intervalo de temperatura para el que se Equation (3.9) establishes Kelvin temperature scale throughout the the temperature range for for Equation (3.9) establishes the Kelvin temperature scale throughout temperature range ∗)∗∗the are experimentally accessible. which values of (P V are experimentally accessible. which values of (P V ) tieneEquation acceso experimental a los valores de temperature (PV)*. ∗ ∗ (3.9) establishes the Kelvin scale throughout the temperature range for experimentally accessible. which values of (P experimentally accessible. which values of V(P) Vare ) are The state thelimiting limiting condition where → deserves discussion. The ∗aaare El estado deof un gas lagas condición límite,accessible. donde P →where 0 merece cierto análisis.discussion. A medida que disminuThe state ofof)en gas atatthe condition PP → 00deserves The experimentally which values (P V The state of a gas at the limiting condition where P → 0 deserves discussion. The The state of a gas at the limiting condition where P → 0 deserves discussion. The molecules making up a gas become more and more widely separated as pressure is decreased, ye la presión sobre un gas, las moléculas del mismo se separan cada vez más y el volumen de las moléculas se molecules making up a gas become more and more widely separated as pressure is decreased, The making state of up a gas thebecome limiting condition where Pseparated → 0 deserves discussion. The molecules aup gas become more andand more widely assmaller pressure is decreased, molecules making aat gas more more widely separated as pressure is decreased, and the volume ofthe the molecules themselves becomes asmaller smaller and fraction ofthe thetotal total convierte en una fracción cada vez más pequeña del volumen total ocupado por el gas. Además, las fuerzas and the volume of molecules themselves becomes a and smaller fraction of molecules making up a gas become more and more widely separated assmaller pressure is of decreased, and the volume of the molecules themselves becomes a smaller andand smaller fraction the total and the volume of the molecules themselves becomes a smaller fraction of es the totalvez más volume occupied bythe the gas. Furthermore, the forces attraction between molecules become de atracción entre lasofmoléculas se vuelven más pequeñas debido a que lasmaller distancia entre ellas cada occupied by gas. Furthermore, the forces ofofattraction between molecules become andvolume the volume thethe molecules themselves becomes aofsmaller andbetween fraction of the total volume occupied by gas. Furthermore, the forces attraction molecules become volume occupied by the gas. Furthermore, the forces of attraction between molecules become eversmaller smaller because theconforme increasing distances betweenthem them (Sec.16.1). 16.1). thelimit, limit,indetermigrande (sección 16.1). Enthe el límite, Pthe → 0, las moléculas están separadas por ever because ofofthe increasing distances between (Sec. InIndistancias the asas volume occupied by Furthermore, forces of attraction between molecules become ever smaller because of gas. the increasing distances between them (Sec. 16.1). Inbecome the limit, as as ever smaller because of the increasing distances between them (Sec. 16.1). In the limit, P → 0, the molecules are separated by infinite distances. Their volumes negligible nadas. Sus volúmenes se vuelven despreciables en comparación con el volumen total dellimit, gas, yaslas fuerzas P smaller → 0, the molecules separateddistances by infinite distances. Their volumes become negligible ever because ofare theare increasing between them (Sec. 16.1). In the P → 0, the molecules separated by distances. Their volumes become negligible Pcompared → 0, the molecules are separated by infinite distances. Their volumes become negligible with the totalvolume volume ofthe theinfinite gas, and the intermolecular forces approach zero. Theseestableintermoleculares tienden a cero. Estas condiciones definen un estado de gas ideal, y la ecuación (3.9) compared with the total of gas, and the intermolecular forces approach zero. These P → 0, the molecules are volume separated by infinite distances. Their forces volumes become negligible compared with the total volume of the gas, and the intermolecular approach zero. These compared with the total of the gas, and the intermolecular forces approach zero. These conditions define an ideal-gas state, and Eq. (3.9) establishes the ideal-gas temperature scale. ce lacompared escala dewith temperatura del gas ideal. La constante deestablishes proporcionalidad Rapproach de latemperature ecuación (3.7) se conoce conditions define an ideal-gas and Eq.the (3.9) the ideal-gas scale. theantotal volume ofstate, theand gas, and intermolecular forces zero. These conditions define ideal-gas state, Eq. (3.9) establishes the the ideal-gas temperature scale. conditions define an ideal-gas state, and Eq. (3.9) establishes ideal-gas temperature scale. The proportionality constant R in Eq. (3.7) is called the universal gas constant. Its numerical como constante universal de los gases. Su valor numérico se determina mediante la ecuación (3.8) a The proportionality constant R in Eq. (3.7) is called the universal gas constant. Its numerical conditions define anconstant ideal-gas Eq. establishes the ideal-gas temperature scale. partir de The proportionality Rstate, inREq. (3.7) is(3.9) called the the universal constant. Its numerical The proportionality constant inand Eq. (3.7) is called universal constant. Its numerical value isdetermined determined bymeans means of Eq. (3.8) from experimental Pgas VTTgas data: información PVT experimental: value is by of Eq. (3.8) from experimental P V data: Thevalue proportionality constant RofinEq. Eq. (3.7) is from called the universal Its numerical value is determined by means (3.8) from experimental P VPTgas is determined by means of Eq. (3.8) experimental Vdata: T constant. data: value is determined by means of Eq. (3.8) from experimental P V T data: ∗ ∗ (PVV))∗t (P (P V(P)∗tV )t∗tt RR== 273.16 K R =R =(P273.16 V )∗tK K 273.16 K R = 273.16 273.16 K pressure, Because dataen cannot fact betaken taken zero pressure, datataken taken finite are Because PPVVTTPVT data cannot ininfact be atat zero data atat finite pressures are Ya que la información realidad no taken es a unadata presión cero, toma apressures presiones Because P VPTVdata cannot in fact be at zero pressure, taken at finite pressures arevalue Because Ttodata cannot in fact be posible taken attomarse zero pressure, data taken atsefinite pressures arefinitas y extrapolated the zero-pressure state. Determined as indicated by Fig. 3.4, the accepted extrapolated to the zero-pressure state. Determined as indicated by Fig. 3.4, the accepted value después se extrapola althe estado dein3presión cero. Determinado como se en3.4, la 1figura 3.4,value elare valor Because P V∗∗to T the data cannot fact bestate. taken at zero as pressure, data taken at finite pressures extrapolated zero-pressure state. Determined indicated byindica Fig. 3.4, the accepted extrapolated to zero-pressure Determined asfollowing indicated by Fig. the accepted value acepta−1 −1 −1 V)22 )∗∗t is711.8 is22,711.8 22,711.8 cm333mol bar mol leading the value R: *(P 3 bar –1mol 111 ofof(P bar , ,leading totoas the following value ofofR: do de (PV) es cm , lo cual nos lleva al siguiente valor de ∗Vis 3cm −1 1R: t −1 extrapolated to the zero-pressure state. Determined indicated by Fig. 3.4, the accepted value t t of (P V ) 22,711.8 cm bar mol , leading to the following value of R: of (PtV )t is 22,711.8 cm bar mol , leading to the following value of R: of (P V )∗t is 22,711.8 cm3 bar mol−1 , 3leading to−1the following value of R:1 −1 barmol mol−1 22,711.83cm cm333 bar 22,711.8 3 −1 K −1 −1 −1 = 83.1447 cm −1 −1 barmol mol−1 bar bar molmol cm cm = 83.1447 3cm333 bar RR22,711.8 == 22,711.8 K−1 −1 K −1−1 3 −1 273.16 K = 83.1447 cm cm R =R 22,711.8 bar bar molmol = 83.1447 = K−1 273.16 K bar mol cm 273.16 K K 273.16 = 83.1447 cm3 bar mol−1 K−1 R= 273.16 K Through the use of conversion factors, R may beexpressed expressedininvarious variousunits. units.Commonly Commonlyused used Through the use of conversion factors, R may be Through theare use of conversion factors, R may be expressed in various units. Commonly usedused Through the use of conversion factors, R may be expressed in various units. Commonly values given in Table A.2 of App. A. values arede given in Table A.2factors, of App. A. Mediante elare uso factores deA.2 conversión, se puede expresar en unidades. En general, Through the use ofTable conversion may be expressed in diversas various units. Commonly usedse usan los values given in of App. A.RR A. values are given in Table A.2 of App. valores dados la tabla A.2 del A. values areen given in Table A.2apéndice of App. A. 1 http://physics.nist.gov/constants. 11http://physics.nist.gov/constants. 1 http://physics.nist.gov/constants. 1 http://physics.nist.gov/constants. http://physics.nist.gov/constants. http://physics.nist.gov/constants. 1 http://physics.nist.gov/constants. 03-SmithVanNess.indd 71 8/1/07 12:52:20 72 CHAPTER 3.3.Volumetric Volumetric Properties of Pure Pure Fluids CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3. CHAPTER Properties of CHAPTER 72 72 Dos formas de laof ecuación Two Forms Forms of the Virial Virialvirial Equation Two the Equation A useful useful auxiliarytermodinámica thermodynamicauxiliar property ismediante defined by by the equation: equation: Se define una propiedad útilis la the ecuación: A auxiliary thermodynamic property defined PV Z≡ ≡ PV Z RT RT RT (3.10) (3.10) (3.10) This dimensionless dimensionless ratio is called called the compressibility factor. With this definition definition and with Esta relación adimensional ratio se llama factorthe de compressibility compresibilidad.factor. Con esta definición y con aand = RT [ecuación This is With this with a = RT [Eq. (3.7)], Eq. (3.6) becomes: (3.7)], la ecuación (3.6) será: a = RT [Eq. (3.7)], Eq. (3.6) becomes: Z= = 11 + +B B���� P P+ +C C ���� P P2222 + +D D���� P P3333 + + ·· ·· ·· Z (3.11) (3.11) (3.11) 2 2 An alternative expression for Z is also in common use: An alternative alternative expression for is also also in common common use: 22 An expression is in Una expresión alternativa tambiénfor deZZuso común para Z es:use: Z= = 11 + + Z C B D C + D B+ C D + ··· B + V 222 + 3 + ··· V V V V2 V 333 (3.12) (3.12) (3.12) � C′,��, etc., � etc., Ambas Both ecuaciones conocen are como expansiones viriales, y los parámetros B ′, CB y B,and C, D, etc., of these equations known as virial expansions, and the parameters D Both of these theseseequations equations are known as virial virial expansions, expansions, and the parameters parameters B′,���,,,DC C�� ,, D D���,,, etc., etc., and Both of are known as and the B and � se llaman coeficientes viriales. Los parámetros B ′ Parameters y B son los B segundos coeficientes viriales; C ′ y C son los B, C, D, etc., are called virial coefficients. B are second virial coefficients; ��� and B, C, D, etc., are called virial coefficients. Parameters B and B are second virial coefficients; B,� C, D, etc., are called virial coefficients. Parameters B and B are second virial coefficients; tercerosC viriales, etc. Para ciertoetc. gasFor los aacoeficientes viriales son funciones C are third virial coefficients; given gas the virial coefficients are functions ��� and Ccoeficientes and C C are are third third virial coefficients; etc. For given gas gas the the virial coefficients coefficients areúnicamente functions de la C and virial coefficients; etc. For a given virial are functions temperatura. of temperature only. of temperature only. of temperature only. Los dosThe conjuntos ecuaciones (3.12) estánasrelacionados de la manera sitwo sets setsdeof ofcoeficientes coefficientsen in las Eqs. (3.11) and and(3.11) (3.12)yare are related follows: The two coefficients in Eqs. (3.11) (3.12) related as follows: guiente: B B���� = = B B RT RT RT (3.13a) (3.13a) a) C ���� = = C C− −B B2222 C (RT ))2222 (RT (3.13b) (3.13b) b) D− − 3BC 3BC + + 2B 2B3333 �� = D D � D� = (RT ))3333 (RT (3.13c) (3.13c) c) To derive derive these these relations, relations, we we set set Z Z= =P P V /RT /RT in in Eq. Eq. (3.12), and and solve solve for for P. P. This This allows allows elimelimTo Para deducir estas relaciones, establecemos ZV = PV/RT en la(3.12), ecuación (3.12) y resolvemos para P. Esto perination of P on the right of Eq. (3.11). The resulting equation reduces to a power series in 1/V inationPofdePlaonderecha the right (3.11).(3.11). The resulting equation reduces a power series in de 1/V mite eliminar deofterm laEq. ecuación La ecuación resultante setoreduce a una serie potencias which may be compared by term with Eq. (3.12) to yield the given relations. They hold which may be compared term by term with Eq. (3.12) to yield(3.12) the given relations. They hold en 1/V que es posible compararse término a término con la ecuación para lograr las relaciones que se exactly only for the two virial expansions as infinite series, but are acceptable approximations exactlySe only for the theprecisamente two virial virial expansions expansions as infinite series, but but viriales are acceptable acceptable approximations exactly only for two as infinite series, are approximations proporcionan. cumplen sólo para las dos expansiones como series infinitas, pero son for the truncated forms used in practice. for the the truncated truncated forms used in practice. practice. for forms in aproximaciones aceptables paraused las formas truncadas que se emplean en la práctica. Many other other equations equations of state state have have been been proposed proposed for for gases, gases, but but the virial virial equations equations are are Se han Many propuesto muchas otrasofecuaciones de estado para los gases, perothe las ecuaciones viriales son las the only ones firmly based on statistical mechanics, which provides physical significance to the only based on statistical mechanics, which provides physical un significance to únicas que tienenones unafirmly base teórica firme en la mecánica estadística, que proporciona significado físico a the virial coefficients. Thus, for the expansion in 1/V , the term B/V arises on account of the virialviriales. coefficients. Thus, for the in 1/V , theel2term B/VB/V arises ona account oflas inter­ los coeficientes De esta manera, paraexpansion el desarrollo en 1/V, término surge causa de term, on on account of of three-body three-body interactions between between pairs pairs of of molecules molecules (Sec. (Sec. 16.2); 16.2); the C/V C/V 222 term, interactions acciones entre pares de moléculas (sección 16.2); el términothe C/V 2, con motivoaccount de las interacciónes entre un interactions; etc. Because two-body interactions are many times more common than threeinteractions; Because two-body interactions are many more common than que threeconjunto de tres, etc.etc. Ya que las interacciones entre conjuntos de dostimes son mucho más comunes entre tres, body interactions, interactions, and and three-body three-body interactions interactions are are many many times times more more numerous numerous than than four-body y éstas body a su vez son más numerosas que las correspondientes entre conjuntos de cuatro, etc.,four-body las contribuciointeractions, etc., the contributions to Z of the successively higher-ordered terms decrease etc., de theorden contributions to Z of the con successively higher-ordered terms decrease nes a Z interactions, de estos términos superior disminuyen mucha rapidez. rapidly. rapidly. 2 Proposed by H. Kamerlingh Onnes, “Expression of the Equation of State of Gases and Liquids by Means of 222Proposed Propuesto Proposed by H. H. Kamerlingh Kamerlingh Onnes, “Expression “Expression of the the Equation Equation of Stateand of Liquids Gases and and Liquidsofby by MeansCommunicaof by Onnes, of State of Gases Liquids Means of por H. Kamerlingh Onnes, “Expression of the Equation of State ofof Gases by Means Series”, Proposed by H. Kamerlingh Onnes, “Expression of the Equation ofof State of no. Gases and Liquids by Means of Series,” Communications from the Physical Laboratory of the University Leiden, 71, 1901. Series,” Communications from the Physical Laboratory Laboratory of the the University of Leiden, Leiden, no. no. 71, 71, 1901. 1901. Communications Physical of University tions fromSeries,” the Physical Laboratoryfrom of thethe University of Leiden, núm. 71, 1901. of Series,” Communications from the Physical Laboratory of the University of Leiden, no. 71, 1901. 03-SmithVanNess.indd 72 8/1/07 12:52:24 3.3. The The Ideal Ideal Gas Gas 3.3. 3.3. El3.3. gas ideal The 3.3. The Ideal Ideal Gas Gas 3.3 73 73 73 73 73 3.3 GAS THE IDEAL GAS EL 3.3 THEIDEAL IDEAL GAS 2, ,,etc., Because the terms terms B/V C/Vde22,,laetc., etc., of the the virial virial expansion [Eq. (3.12)] arise on account account of the B/V C/V of (3.12)] arise on of Ya que Because los términos B/V, C/V expansión virialexpansion [ecuación [Eq. (3.l2)] surgen a causa de las interaccio22, etc., of the virial expansion [Eq. (3.12)] arise on account of Because the terms B/V , C/V Because the terms B/V , C/V , etc., of the virial expansion [Eq. (3.12)] arise on account molecular interactions, the virial coefficients B, C, etc., would be zero were no such interacmolecular interactions, the virial coefficients B, C, etc., would be zero were no such interacnes moleculares, los coeficientes viriales B, C, etc., serían cero donde no existen estas interacciones. of Con esto, molecular interactions, the coefficients B, molecular interactions, the virial virial coefficients B, C, C, etc., etc., would be be zero zero were were no no such such interacinteractions to exist, and the virial virial expansion would reduce reduce to: would tions to exist, the expansion would to: la expansión virial seand reduce a: tions tions to to exist, exist, and and the the virial virial expansion expansion would would reduce reduce to: to: = 11 o PPPV = RT RT Z= = 1 or = RT ZZ or VV = ZZ = or P = 11 or PV V= = RT RT De hecho, las interacciones moleculares existen y influence ejercen influencia sobrebehavior el comportamiento observaMolecular interactions do in in fact fact exist, exist, and influence the observed observed behavior of real real gases. gases. Molecular interactions do and the of do de gases reales. Conforme disminuye la presión a temperatura constante, V aumenta y las contribuciones de Molecular interactions do in fact exist, and influence the observed behavior of real gases. Molecular interactions do intemperature, fact exist, andVVinfluence observed behavior of gases. As pressure pressure is reduced reduced at constant constant temperature, increasesthe and the contributions contributions of real the terms, terms, As is at increases and the the 2, D/V 3, etc., en la ecuación (3.12) disminuyen. Para P → 0, Z se aproxima a uno, pero 2 3 2 3 los términos B/V, C/V As pressure is reduced at constant temperature, V increases and the contributions of the terms, As pressure reduced temperature, increases the contributions terms, D/V ...atin inconstant Eq. (3.12), (3.12), decrease.VFor For → 0, 0,and approaches unity,of notthe because B/V C/V is ,, D/V ,, ... Eq. decrease. PP → ZZ approaches unity, not because B/V ,, C/V 22, D/V 33,coeficientes no por of algún viriales sino porque tiende infinito. Thus Por loin en elas ... (3.12), decrease. For 0, unity, not B/V ,, cambio C/V , en D/V ,virial ... in in Eq. Eq. (3.12), decrease. For P P → 0, ZZ aapproaches approaches unity, not because B/V C/V of any change in los the virial coefficients, but because VV→ becomes infinite. Thus intanto, the because limit aslímite, a any change in the coefficients, but because V becomes infinite. the limit medidaof que P → 0, la ecuación de estado se reduce a la misma forma simple que para el caso hipotético any change in the virial coefficients, but because V becomes infinite. Thus in the limit as of any0, in the virial coefficients, V becomes Thus in the limit as donP→ → 0,change the equation equation of state state reduces to tobut the because same simple simple form as asinfinite. for the the hypothetical hypothetical case of of P the of reduces the same form for case de B = C = · · · = 0; es decir, P → 0, the equation of state reduces to the same simple form as for the hypothetical case of P= →C = C0,= =the = 0; 0; i.e., i.e.,of state reduces to the same simple form as for the hypothetical case of BB ·· ·· ··equation = B B= =C C= = ·· ·· ·· = = 0; 0; i.e., i.e., → 11 or → RT RT ZZZ→ PPVV → → → 1 or o PV RT ZZ → or P → 11 or PV V→ → RT RT We know know from from the the phase phase rule rule that that the the internal internal energy energy of of aa real real gas gas is is aa function function of of We De la regla dewell la fase sabemos querule la energía interna de un gas real unafrom función tanto de la We know the phase that internal energy of aaesreal gas is aa function of Weas know from the phase rule that the the internal energy ofresults real gas isforces function ofpresión pressure as as of of temperature. This pressure dependency results forces between pressure well from as temperature. This pressure dependency from between como depressure la temperatura. Esta dependencia de la presión aparece como resultado de las fuerzas entre las as well as of temperature. This pressure dependency results from forces between pressure as wellIf of forces temperature. dependency results forces the molecules. Ifas such forces did did not not This exist,pressure no energy energy would be be required requiredfrom to alter alter the between averagemo­lécu­ the molecules. such exist, no would to the average las. Si estas fuerzas no existieran, no se necesitaría energía alguna para alterar la distancia intermolecular the molecules. If such forces did not exist, no energy would be required to alter the average the molecules. distance, If such forces did not no exist, no energy be required alter volume the average intermolecular distance, and therefore therefore no energy energy would would be required required to bring bringtoabout about volume and prointermolecular and would be to and medio y, en consecuencia, no se requeriría energía para originar cambios de volumen y presión en intermolecular distance, and therefore no energy would be required to bring about volume and intermolecular distance, therefore no energy would be required tothe bring aboutof andun gas a pressure changes in aa gas gasand at constant constant temperature. We conclude conclude that in in the absence ofvolume molecular pressure changes in at temperature. We that absence molecular temperatura constante. Concluimos que en ausencia de interacciones moleculares, la energía interna del gas depressure changes in a gas at constant temperature. We conclude that in the absence of molecular pressure changes in a gas energy at constant thatonly. in theThese absence of molecular interactions, the internal internal energy of aatemperature. gas depends dependsWe on conclude temperature only. These considerations interactions, the of gas on temperature considerations pende exclusivamente de la temperatura. Estas consideraciones acerca del comportamiento de un gas interactions, the internal energy of a gas depends on temperature only. These considerations interactions, theof energy of gaswhich depends on temperatureforces only. exist These considerations of the behavior behavior ofinternal hypothetical gasa in in which no intermolecular intermolecular forces exist and of aa real real gas gas hipoté­ of the aa hypothetical gas no and of tico en of el que no existen fuerzas moleculares, y de un gas real en el límite conforme la presión tiende the behavior of a hypothetical gas in which no intermolecular forces exist and of a real gas of the behavior of a hypothetical gaszero in which no intermolecular existgas andas a real gas a cero, in the limit limit as as pressure pressure approaches zero lead to to the the definition of offorces an ideal ideal gas asofone one whose in approaches lead definition an whose conducen a la definición de un gas ideal como aquel cuyo comportamiento macroscópico se caracteriza in the limit as pressure approaches zero lead to the definition of an ideal gas as one whose in the limit as pressure zero macroscopic behavior is approaches characterized by:lead to the definition of an ideal gas as one whose por: macroscopic behavior is characterized by: macroscopic behavior is characterized by: macroscopic behavior is characterized by: • La ecuación de estado The equation equation of state: state: •• The of •• The equation The equation of of state: state: = RT RT (ideal (ideal gas) (3.14) (3.14) (gas ideal) PPVV = gas) (3.14) P (ideal (3.14) PV V= = RT RT (ideal gas) gas) (3.14) • An internal energy that is a function of temperature only: • Una •energía internaenergy que esthat unaisfunción sóloofdetemperature la temperatura: An internal a function only: •• An An internal internal energy energy that that is is aa function function of of temperature temperature only: only: U= =U U(T (T )) (ideal (ideal gas) (3.15) (3.15) U gas) (3.15) (gas ideal) U (ideal (3.15) U= =U U(T (T )) (ideal gas) gas) (3.15) Implied Property Relations for an Ideal Gas Relaciones una propiedad implícitas paraGas un gas ideal Impliedde Property Relations for an Ideal The definition definition of of heat heat capacity capacity at at constant constant volume, volume, Eq. Eq. (2.16), (2.16), leads leads for for an an ideal ideal gas gas to to the the The The definition of heat capacity at constant volume, Eq. (2.16), leads for an ideal gas to the The definition of heat capacity at constant volume, Eq. (2.16), leads for an ideal gas to thea la conconclusion that C is a function of temperature only: La definición de capacidad térmica a volumen constante, ecuación (2.16), para un gas ideal conduce conclusion that C VV is a function of temperature only: conclusion that C is a function of temperature only: conclusion that C is a function of temperature only: V clusión de que CV es una Vfunción exclusiva� �de la� �temperatura: ∂U � dU(T (T )) ∂U dU � � � = C ≡ = dU (T )) (3.16) =C CVV (T C VV ≡ ∂U (3.16) (T ∂U dU (T )) = ∂ T dT C (3.16) CVV ≡ ≡ ∂ T VV = = dT = (T )) (3.16) (3.16) =C CVV (T ∂∂TT VV dT dT The defining defining equation equation for for enthalpy, enthalpy, Eq. Eq. (2.11), (2.11), applied applied to to an an ideal ideal gas, gas, leads leads to to the the conclusion conclusion The The defining for Eq. (2.11), applied an ideal gas, leads The H defining equation forof enthalpy, Eq.(2.11), (2.11), applied to angas ideal gas,conduce leads to to athe the conclusion that Hque also isequation function ofenthalpy, temperature only: aplicada La ecuación define a la entalpía, ecuación atoun ideal, la conclusion conclusión de que that also is aa function temperature only: that H also is of only: thates Huna alsofunción is aa function function oflatemperature temperature only: H también sólo de temperatura: H≡ ≡U U+ + PPVV = =U U(T (T )) + + RT RT = =H H(T (T )) (3.17) H (3.17) H (3.17) H≡ ≡U U+ +P PV V= =U U(T (T )) + + RT RT = =H H(T (T )) (3.17) (3.17) 03-SmithVanNess.indd 73 8/1/07 12:52:27 74 74 74 74 CHAPTER 3. Volumetric Properties of Pure Fluids CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER Properties of CHAPTER 3. 3.3.Volumetric Volumetric Properties of Pure Pure Fluids The heat capacity at constant pressure byecuación Eq. (2.20), likecomo CV , C isVa, es function of sólo P , defined La capacidad calorífica a presión constante CP, C definida por la (2.20) una función The heat capacity at constant pressure C by Eq. (2.20), like C aa function of The heat capacity at constant pressure C defined by Eq. (2.20), like C is function of PP,, defined VV,, is temperature only: de la temperatura: � � temperature temperature only: only: ∂ H d H (T ) � � � = C P ≡ �∂∂H (3.18) ddH H H(T (T)) = C P (T ) C (3.18) CPP ≡ ≡ ∂T P = = dT = (T)) (3.18) (3.18) =C CPP(T ∂∂TT PP dT dT A useful relation between C P and C V for an ideal gas comes from differentiation of Eq. (3.17): useful C and C for ideal from Eq. Arelación useful relation relation CPPun and CVVideal for an an ideal gas gas comes from differentiation differentiation of Eq. (3.17): (3.17): Una útilA entre Cbetween y CV para gas proviene decomes la derivada de la ecuación of (3.17): Pbetween dH dU C P = ddH (3.19) H = dU dU + R = C V + R C (3.19) CPP = = dT = + RR (3.19) (3.19) = dT + + RR = =C CVV + dT dT dT dT This equation does not imply that C P and C V are themselves conEsta ecuación no implica que C C enthat síC constantes para un gas ideal, This does not imply that and C conP ybut V sean This equation equation does not imply that Cmismas andvary CVV are are themselves conPPthey stant for an ideal gas, only withthemselves temperature in sino que varían únicamente con la temperatura de tal manera que su diferencia es igual stant for an ideal gas, but only that they vary with temperature stant afor anthat ideal gas, but onlyisthat they with temperature in ina R. such way their difference equal to Rvary . such a way that their difference is equal to R . such a way that their difference is equal to R . Para cualquier gasgas ideal las(3.16) ecuaciones (3.16)lead y (3.18) For anycambio change de of estado state ofen anun ideal Eqs. and (3.18) to: nos conducen a: For For any any change change of of state state of of an an ideal ideal gas gas Eqs. Eqs. (3.16) (3.16) and and (3.18) (3.18) lead lead to: to: � dU = C V dT (3.20a) a) �U = �� C V dT (3.20b) b) dU dU = =C CVV dT dT (3.20a) (3.20a) �U �U = = C CVV dT dT (3.20b) (3.20b) � d H = C P dT (3.21a) a) �H = �� C P dT (3.21b) b) ddH H= =C CPP dT dT (3.21a) (3.21a) �H �H = = C CPP dT dT (3.21b) (3.21b) both the internal energy an son idealfunciones gas are functions temperatureDU para V of Ya que Because tanto la energía interna como CV deand unCgas ideal sólo de laof temperatura, Because both the internal energy and C of an ideal gas are functions of temperature Because both the internal energy and C of an ideal gas are functions of temperature VVEq. only, �U for an ideal gas is always given by (3.20b), regardless of the kind of process un gas ideal siempre se da por la ecuación (3.20b), sin considerar la clase de proceso que ocasione el cambio. only, �U for an ideal gas is always given by Eq. (3.20b), regardless of the kind of only, �U for an ideal gas is always given by Eq. (3.20b), regardless of the kind of process process causing the change. This is demonstrated in Fig. 3.5, which shows a graph of internal energy Esto se prueba en la figura 3.5, la cual exhibe una gráfica de la energía interna como una función del volumen causing the This is in which a graph of energy causing the change. change. This is demonstrated demonstrated in Fig. Fig. 3.5, which shows shows graph of internal internal as alafunction of molar volume with temperature as3.5, parameter. Because U is independent of V, molar con temperatura como parámetro (cantidad que es constante bajoacierto conjunto deenergy condiciones, as aa function of molar volume with temperature as parameter. Because U is independent of VV as function of molar volume with temperature as parameter. Because U is independent of a plot of U vs. V at constant temperature is a horizontal line. For different temperatures, U,, de U pero es posible que sea diferente bajo otras condiciones). Ya que U es independiente de V, una gráfica aa plot of VV at temperature is horizontal line. different temperatures, U plot of U U vs. vs. at constant constant temperature is aaeach horizontal line. For For different temperatures, U has different values, with a separate line for temperature. Two such lines are shown in en función de V a temperatura constante es una línea horizontal. A diferentes temperaturas, U tiene valores has has different different values, values, with with aa separate separate line line for for each each temperature. temperature. Two Two such such lines lines are are shown shown in in distintos, con una línea separada para cada temperatura. En la figura 3.5 se muestran dos de estas líneas, una U2 U U22 Figure 3.5: Internal energy changes for an ideal Figure Figure 3.5: 3.5: Internal Internal energy energy changes changes for for an an ideal ideal gas. Figura 3.5: Cambios en la energía interna de un gas gas. gas. ideal. U U U U1 U U11 b bb c cc d dd b U2 c U1 2 d T2 T1 TT1 a aa U T2 TT2 1 T1 a V VV V Fig. 3.5, one for temperature T1 and one for a higher temperature T2 . The dashed line connectFig. 3.5, for and temperature TT22..the The dashed connectFig.points 3.5, one one for temperature temperature and one one for for aa higher higher temperature Thetemperature dashed line lineincreases connecting a and b representsTTa11 constant-volume process for which ing points a and b represents a constant-volume process for which the temperature increases ing points a and b represents a constant-volume process for which the temperature increases from T1 to T2 and the internal energy changes by �U = U2 − U1 . This change in internal � para la temperatura y la the otra para unaenergy temperatura superior . La que une a los puntos a 2= from TTT221 and internal by �U U in from TT1is to and the(3.20b) internal changes by. The �UTdashed = U2línea −U Udiscontinua . This This change change in internal internal 11.connecting 1 to 2− energy given by Eq. asenergy �U =changes C V dT lines points a and c � � y b representa un proceso a volumen constante para el que la temperatura aumenta de T a T y laccenergía 1 energy is (3.20b) as = dT .. The lines points aa2and energy is given given byd Eq. Eq. (3.20b)other as �U �U = C CVVnot dToccurring The dashed dashed lines connecting connecting points and and points a andby represent processes at constant volume but which also interna cambia por DU = U – U . Este cambio en la energía interna es conocido por la ecuación (3.20b) 2represent 1 and aa and other not constant but also and points points and dd represent other processes not occurring occurring constant volume but which which also como lead from an initial temperature T1 processes to a final temperature T2at .atThe graphvolume shows that the change DU = ∫ lead C dT. Las líneas discontinuas que unen a los puntos a y c, así como a los puntos a y d, representan V from an temperature TT22.. The shows change lead an initial initial temperature T11 to to aaasfinal final temperature The graph graph shows that the change in U from for these processes is the T same for temperature the process, andthat it isthe therefore � constant-volume otros procesos que no ocurren a volumen constante, pero que también conducen de una temperatura inicial T1 in U for these processes is the same as for the constant-volume process, and it is therefore in U for these processes is the same as for the constant-volume process, and it is therefore given by the same equation, namely, �U = �� C V dT . However, �U is not equal to Q for a la temperatura final T . La gráfica muestra que el cambio en U para estos procesos es el mismo que 2 equation, given given by by the the same same equation, namely, namely, �U �U = = C CVV dT dT.. However, However, �U �U is is not not equal equal to to Q Q for for para el 03-SmithVanNess.indd 74 8/1/07 12:52:32 75 75 75 proceso a volumen constante, y esto es consecuencia de la misma ecuación, a saber, DU = ∫ CV dT. No obstante, DU no processes, es igual a Q para estos procesos, queonQTdepende T2, sino también de la trathese because Q depends notya only butsólo alsode onTthe of the process. 1 y path 1 and T2 no these processes, because Q depends not only on T11 and T22 but also on the path of the process. yectoriaAn delentirely proceso. A la entalpía H de un gas ideal seenthalpy le aplicaHunofanálisis analogous discussion applies to the an idealcompletamente gas. (See Sec. análogo. 2.11.) (Véase An entirely analogous discussion applies to the enthalpy H of an ideal gas. (See Sec. 2.11.) la sección 2.11.) The ideal gas is a model fluid described by simple property relations, which are freThe ideal is a model fluid described by simple property whichsimple, are freElquently gas ideal es approximations ungas modelo de fluido mediante relaciones de relations, unacalculations, propiedad good when descrito applied to actual gases. In process gases atque con quently good approximations when applied to actual gases. In process calculations, gases at frecuencia son una aproximación cuando se aplican aideal, los gases reales. equations En los cálculos de los procesos, pressures upbuena to a few bars may often be considered and simple then apply. pressures up to a few bars may often be considered ideal, and simple equations then apply. los gases con presión hasta de unos pocos bar con frecuencia se consideran ideales, y en tal caso se les aplican ecuaciones sencillas. 3.3. El gas ideal 3.3. The Ideal Gas 3.3. The Ideal Gas Equations for Process Calculations for Ideal Gases Equations for Process Calculations for Ideal Gases Ecuaciones para cálculos de procesos: gases Process calculations provide work and heat quantities. Theideales work of a mechanically reversible Process calculations provide work and heat quantities. The work of a mechanically reversible closed-system process is given by Eq. (1.2), here written for a unit mass or a mole: closed-system process is given bycantidades Eq. (1.2), de here written for a unit mass orpara a mole: Los cálculos de procesos proporcionan trabajo y calor. El trabajo un proceso mecánicamente reversible en un sistema cerrado se conoce por la ecuación (1.2), y en este caso se escribe para un mol d W = −P d V d W = −P d V o una unidad de masa: For an ideal gas in any closed-system process, the first law as given by Eq. (2.6) written for a For an ideal gas in any closed-system process, the first law as given by Eq. (2.6) written for a dWEq. = –PdV unit mass or a mole, may be combined with (3.20a) to give: unit mass or a mole, may be combined with Eq. (3.20a) to give: Para un gas ideal en cualquier proceso de sistema cerrado, la primera ley se conoce por la ecuación (2.6), Q + d Wcombinarla = C V dT escrita para una masa unitaria o un mol, y esddposible Q + d W = C VV dT con la ecuación (3.20a) para dar: Substitution for d W and solution fordQ d Q+ dW yields equation valid for an ideal gas in any = Can V dT Substitution for d W and solution for d Q yields an equation valid for an ideal gas in any mechanically reversible closed-system process: mechanically reversible closed-system La sustitución para dW y la solución para dQ process: conducen a una ecuación que es válida para un gas ideal en cualquier proceso mecánicamente reversible de=sistema + P dV dQ C V dTcerrado: d Q = C VV dT + P d V dQ = CV dT + PdV This equation contains the variables P, V , and T , only two of which are independent. This equation contains the variables P, V , and T , only two of which are independent. Working equations forlas d Qvariables and d WP,depend which variables is Las selected as Esta ecuación contiene V y T,on pero sólopair dos of sonthese independientes. ecuaciones de Working equations for d Q and d W depend on which pair of these variables is selected as upon which variable is eliminated by Eq. Withcomo P = independiente; RT /V , trabajo independent; para dQ y dWi.e., dependen de cuál par de estas variables se (3.14). seleccione es decir, independent; i.e., upon which variable is eliminated by Eq. (3.14). With P = RT /V , de qué variable se elimine mediante la ecuación (3.14). Con P = RT/V, dV d Q = C VV dT + RT d V d Q = C V dT + RT V V (3.22) (3.22) dV d W = −RT d V d W = −RT V V (3.23) (3.23) With V = RT /P and with C VV given by Eq. (3.19), the equations for d Q and d W become; V y=con RTC/P and with C Vpor given by Eq. (3.19), forpara d Q and W become; Con V =With RT/P, la ecuación (3.19),the lasequations ecuaciones dQ yd dW se convierten en: V determinada dP d Q = C PP dT − RT d P d Q = C P dT − RT P P (3.24) (3.24) dP d W = −R dT + RT d P d W = −R dT + RT P P (3.25) (3.25) T el = P V /R,es thesimplemente work is simply = −Py dcon V , and withvez C V given again (3.19),(3.19), Con T =With PV/R, dW dd=W conoce porby la Eq. ecuación V otra With T = trabajo P V /R, the work is simply W–P=dV, −P d V , C and with C Vsegiven again by Eq. (3.19), V CP CV (3.26) (3.26) d Q = C VV V d P + C PP P d V (3.26) d Q = R V d P + R P dV R R Es posible aplicar estas ecuaciones para gases ideales a diversas clases de procesos, como los descritos These equations may be applied fordeducción ideal gases various kindsesofcerrado processes, a continuación. Las suposiciones implícitas en su sonto el sistema y queas el deproceso es These equations may be applied for ideal gases toque various kinds of processes, as described in what follows. The assumptions implicit in their derivation are that the system is mecánicamente reversible. scribed in what follows. The assumptions implicit in their derivation are that the system is closed and the process is mechanically reversible. closed and the process is mechanically reversible. 03-SmithVanNess.indd 75 8/1/07 12:52:34 76 76 76 767676 76 76 76 76 76 76 76 CAPÍTULO 3. Propiedades volumétricas de fluidos puros CHAPTER 3. 3. Volumetric Properties of Pure Fluids CHAPTER CHAPTER Volumetric Volumetric Properties Properties Pure ofPure Pure Fluids Fluids CHAPTER 3.3.Volumetric Volumetric Properties ofof Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Properties of Pure Fluids CHAPTER CHAPTER 3. 3. Volumetric Properties of Pure Fluids CHAPTER CHAPTER 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids Proceso isotérmico Isothermal Process Isothermal Isothermal Process Process Isothermal Process Isothermal Process Isothermal Process Isothermal Process Isothermal Process Isothermal Process Isothermal Process Process ByIsothermal Eqs. (3.20b) and (3.21b), �U = �H ==00= De las ecuaciones (3.20b) y (3.21b), DU = DH By By Eqs. Eqs. (3.20b) (3.20b) and and (3.21b), (3.21b), �U �U �H =�H �H By Eqs. (3.20b) and (3.21b), �U == �H == 0=0000= 0 By Eqs. Eqs. (3.20b) (3.20b) and and (3.21b), (3.21b), �U �U = = �H By Eqs. (3.20b) and (3.21b), �U = By Eqs. (3.20b) and (3.21b), �U = �H = By Eqs. (3.20b) and (3.21b), �U = �H = ByBy Eqs. (3.20b) and (3.21b), �U = �H = 0= By By Eqs. Eqs. (3.20b) (3.20b) and and (3.21b), (3.21b), �U �UV= = �H �H0�H = = 000 P 2V V V P2P2P 2 2 2PP 2 2 By Eqs. (3.22) and (3.24), Q = RT ln = −RT ln V VV= V−RT VVln PP 2222=−RT 222 P2 2−RT By By Eqs. Eqs. (3.22) (3.22) and and (3.24), (3.24), Q = Q = RT RT ln ln = −RT ln ln 2V 2P PP2ln By Eqs. (3.22) and (3.24), Q = RT ln ln 2 2 1P V V De By las ecuaciones (3.22) y (3.24), By Eqs. (3.22) and (3.24), Q = RT = ln 2 2 22 1 Eqs. (3.22) and (3.24), QQRT = RT ln = −RT ln By Eqs. (3.22) and (3.24), Q = RT ln = −RT Eqs. (3.22) and (3.24), Q = ln = −RT ln V V P P By Eqs. (3.22) and (3.24), = RT ln = −RT ln V P ByBy Eqs. (3.22) and (3.24), Q = RT ln = −RT ln 1 1V 1 1= By By Eqs. Eqs. (3.22) (3.22) and and (3.24), (3.24), Q Q= = RT RTVln ln = −RT −RTPln ln1 1P111 1 P1 VV11111 V1 PP111 V11 V P11PP V P V 2 2P V V P V P 2 2 2 2 2 2 By Eqs. (3.23) and (3.25), W = −RT ln = RT ln V VV= PP V= VV2V PP2P 222=RT 222 P2 2 ln By By Eqs. Eqs. (3.23) (3.23) and and (3.25), (3.25), == −RT =−RT −RT ln RT RT ln ln PP2ln By Eqs. (3.23) and (3.25), WW= =W −RT ln−RT 2ln= 2 1P V2V 22RT 22 1V By By Eqs. Eqs. (3.23) (3.23) and and (3.25), (3.25), W W = −RT ln ln = = RT RT ln ln By Eqs. (3.23) and (3.25), W = ln = RT Eqs. (3.23) and (3.25), W −RT ln ln V P P By Eqs. (3.23) and (3.25), W = −RT ln = RT ln De By las (3.23) y (3.25), V P Byecuaciones Eqs. (3.23) and (3.25), W = −RT ln = RT ln 1 1 1 1V By By Eqs. Eqs. (3.23) (3.23) and and (3.25), (3.25), W W= = −RT −RTVln ln =1RT RTPln ln1 P111 1 P1 VV1111 = V PP111 V1from P11 P 1V V11 Eq. P Note that Q = −W , a result that also follows (2.3). Therefore, Note Note that that Q = Q −W = −W , a , result a result that that also also follows follows from from Eq. Eq. (2.3). (2.3). Therefore, Therefore, Note that Q = −W , a result that also follows from Eq. (2.3). Therefore, Note Note that that Q Q = = −W −W , , a a result result that that also also follows follows from from Eq. Eq. (2.3). (2.3). Therefore, Therefore, Note that Q = −W , a result that also follows from Eq. (2.3). Therefore, Note that Q = −W , a result that also follows from Eq. (2.3). Therefore, Note that Q = −W , a result that also follows from Eq. (2.3). Therefore, Note that Q = −W , a result that also follows from Eq. (2.3). Therefore, Note that = = −W −W,, aaresultado result result that that also follows follows from from Eq. (2.3). (2.3).aTherefore, Therefore, ObserveNote que that Q =Q –QW, como dealso la ecuación (2.3).Eq. Debido eso, V P 2V VV P2P2P 2V2= 2V 2PP 2 (const T ) 2V Q = −W = RT ln −RT ln VV PP 222=−RT 222 P(const 2 −RT 2(const QQ = Q= −W = −W = = RT RT ln ln −RT = ln ln (const T ) T)) ) T ) 2V 2P V PP2ln Q = −W = RT ln = ln T(const 2 2 1P V V 2 2 22 (const 1 Q = −W −W = = RT RT ln ln = = −RT −RT ln ln (const (const Q −W = RT ln = −RT QQ== −W = RT ln = −RT ln TT )))TT V V P P Q = −W = RT ln = −RT ln (const V P −W = RT ln = −RT ln (const (T constante) 1V 1 1= 1P1 1 1V 1 Q Q= = −W −W = = RT RTVln ln = −RT −RT ln ln (const (const TTT))) P V P PP11 P VV11111 1 PP11111 1 V11 V (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) (3.27) Isobaric Process Isobaric Isobaric Process Process Isobaric Process Isobaric Process Isobaric Process Isobaric Process Isobaric Process Proceso isobárico Isobaric Process Isobaric Isobaric Process Process By Eqs. (3.20b) and (3.21b), By By Eqs. Eqs. (3.20b) (3.20b) and and (3.21b), (3.21b), By Eqs. (3.20b) and (3.21b), By Eqs. (3.20b) and (3.21b), By Eqs. (3.20b) (3.21b), By Eqs. (3.20b) and (3.21b), By Eqs. (3.20b) and (3.21b), By Eqs. (3.20b) and (3.21b), Byecuaciones Eqs. (3.20b) andand (3.21b), �� � �� � By By Eqs. Eqs. (3.20b) (3.20b) and and (3.21b), De las (3.20b) y (3.21b), (3.21b), ��� �����C V�dT ��� �����C P�dT �U = and �H = �U �U =CC CVdT dTdT and andand �H �H =CCPCC CPdT dT �U ==�U and �H == VdT VdT PdT PdT VC �U = C and �H = = �H = C �H C �U == dT �H == dT VVV dT PPP dT V and �U = C dT and �H = C dT �U�U dT andand �H dT VC PC �U �U = =CCV= C dT and and �H �H = =CCP= C dTP dT y VV dT PP dT and by by Eqs. (3.24) and (3.25), and and Eqs. Eqs. (3.24) (3.24) and and (3.25), (3.25), and bybyby Eqs. (3.24) and (3.25), and by Eqs. Eqs. (3.24) (3.24) and and (3.25), (3.25), and by Eqs. (3.24) and by (3.24) and (3.25), y deand lasand ecuaciones (3.24) yand (3.25), and by Eqs. (3.24) (3.25), and byEqs. Eqs. (3.24) and (3.25), �� (3.25), and and by by Eqs. Eqs. (3.24) (3.24) and and (3.25), (3.25), � � �� �����C P�dT Q = and WW = −R(T TT−1 ))T ) 2−− dT and and and −R(T =−R(T −R(T = Q= =CC CPdT QQ = WWW == −R(T PdT PdT 2− 2T− 1T1− 1 PC 1))T and W = −R(T Q = C and = Q y and W =222− −R(T C dT W QQ == CCP= PPP dT 1− Q = C dT and W = −R(T − TT11111))))) T1 ) dT and W= = −R(T −22222T2T− PC 1 )2T dTP dTand and and W W−R(T = = −R(T −R(T − Q Q= =Q C PP dT Note that Q = �H ,, aa ,result also given by Eq. (2.13). Therefore, Note Note that that Qthat = Q= �H = �H aresult result also also given given by by Eq. Eq. (2.13). (2.13). Therefore, Therefore, Note that Q= = �H ,= aresult result also given by Eq. (2.13). Therefore, Observe que Q ∆H es un resultado también conocido por laTherefore, ecuación (2.13). Por lo tanto, Note Note that that Q Q = �H �H ,�H ,result aaresult result also also given given by by Eq. Eq. (2.13). (2.13). Therefore, Note Q , a result also given by Eq. (2.13). Therefore, Note that Q = �H , a also given by Eq. (2.13). Therefore, Note that Q = �H , a result also given by Eq. (2.13). Therefore, Note that Q = �H , a also given by Eq. (2.13). Therefore, Note Note that that Q Q= = �H �H,, aa result result also also given given � by by Eq. Eq. (2.13). (2.13). Therefore, Therefore, � � ��� �����C P�dT Q = �H = (const P) (3.28) Q = Q �H = �H = =CC CPdT dTdT (const (const P) P) P) (3.28) (3.28) (3.28) (P constante) Q=Q = �H = �H (const P) (3.28) PdT PdT PC Q = = �H �H = = C dT (const (const P) P) (3.28) (3.28) Q = = C (const (3.28) QQ �H = C dT (const P) (3.28) P P P P Q = �H = C dT (const P) (3.28) = �H = C dT (const P) (3.28) Q Q= = �H �H = = PCCP dT dT (const (const P) P) (3.28) (3.28) PP Isochoric (Constant-V ))Process Process Isochoric Isochoric (Constant-V (Constant-V )Process Process Isochoric (Constant-V Proceso isocórico (V constante) Isochoric (Constant-V Process Isochoric (Constant-V )))Process Isochoric (Constant-V ) Process Isochoric (Constant-V )))Process Isochoric (Constant-V Process Isochoric Isochoric (Constant-V (Constant-V ) Process Process Equations (3.20b) and (3.21b) again apply: Equations Equations (3.20b) (3.20b) and and (3.21b) (3.21b) again again apply: apply: Equations (3.20b) and (3.21b) again apply: Equations (3.20b) and (3.21b) again Equations (3.20b) and (3.21b) again apply: Equations (3.20b) and (3.21b) again apply: Equations (3.20b) and (3.21b) again apply: Equations (3.20b) and (3.21b) again apply: Se aplican de nuevo las ecuaciones (3.20b) yapply: (3.21b): Equations (3.20b) and (3.21b) again apply: � �� � Equations Equations (3.20b) (3.20b) and and (3.21b) (3.21b) again apply: ���again � � apply: � � ��� �����C P�dT � ���C V dT �U = and �H = �U �U =CC CVdT dTdT and andand �H �H =CCPCC CPdT dT �U ==�U and �H == VdT VdT PdT PdT VC �U = C and �H = = �H = y C �H C �U == dT and �H == dT VVV dT PPP dT V �U = C dT and �H = C dT �U�U dT andand �H dT VC PC �U �U = =CCV= C dT and and �H �H = =CCP= C dTP dT VV dT PP dT By Eq. (3.22) and the basic equation for work, By By Eq. Eq. (3.22) (3.22) and the the basic basic equation equation for for work, work, Eq. (3.22) and the basic equation for work, De By laBy ecuación (3.22) yand la ecuación fundamental delwork, trabajo, By Eq. Eq. (3.22) (3.22) and and the basic basic equation equation for work, work, By Eq. (3.22) and the basic equation for Eq. (3.22) and the basic equation for work, By Eq. (3.22) and the basic equation for work, ByBy Eq. (3.22) and thethe basic forfor work, ��equation � By By Eq. Eq. (3.22) (3.22) and and the the basic basic equation equation for for work, work, � � ����� � � � � � � ��P d V = 0 � � � Q= dTdT and = − �P ��C VdT y and andandWW WW == − =− − PdPPdVPdV dVVP= V0d=0= = Q= = C QQ = C and =W − = VCVdT VdT VC Q = C dT and W = − and 000 Q Q = C dT W = dT and W = − P QQ == C V V V V dT and W = − PP VV00= = Q = C dT = dV Vddd= VV dT dT andand and W W W− = =− −Pd− P d= V = =V000= 0 Q Q= =C V C C V Note that Q = �U ,, aa ,result also given by Eq. (2.10). Therefore, Observe que Qthat ∆U es,�U también un resultado conocido porTherefore, la ecuación (2.10). Por lo tanto, Note Note that Qthat = Q= �U = aresult result also also given given by by Eq. Eq. (2.10). (2.10). Therefore, Therefore, Note that Q= = �U aresult result also given by Eq. (2.10). Note Note that that Q Q = �U �U ,,result aaresult result also also given given by by Eq. Eq. (2.10). (2.10). Therefore, Therefore, Note Q = �U , a result also given by Eq. (2.10). Therefore, Note that Q = �U , a also given by Eq. (2.10). Therefore, Note that Q = �U , a result also given by Eq. (2.10). Therefore, Note that Q = �U , a also given by Eq. (2.10). Therefore, Note Note that that Q Q= = �U �U,, aa result result also also given given by by Eq. Eq. (2.10). (2.10). Therefore, Therefore, �� � ��� �����C V�dT (3.29) (V constante) Q = �U = (const V ))V ) (3.29) = Q= �U = �U =CC CVdT dTdT (const (const V)V (3.29) (3.29) QQ = �U ==�U (const V (3.29) VdT VdT VC Q Q = �U �U = = C dT (const (const V ) ) (3.29) (3.29) Q = = C (const V ) (3.29) QQ == �U = C dT (const V ) (3.29) V V V V Q = �U = C dT (const V ) (3.29) �U = C dT (const V ) (3.29) Q Q= = �U �U = = V CCV dT dT (const (const VV)) (3.29) (3.29) VV 03-SmithVanNess.indd 76 8/1/07 12:52:52 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3. The Ideal Gas 3.3.The TheIdeal IdealGas Gas 3.3. El3.3. gas ideal 77 7777 7777 77 77 77 7777 77 Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Adiabatic Process; Constant Heat Capacities Proceso adiabático: capacidades caloríficas constantes Adiabatic Adiabatic Process; Process; Constant Constant Heat Heat Capacities Capacities An An adiabatic adiabatic process process is is one one for for which which there there is is no no heat heat transfer transfer between between the the system system and and its its AnAn adiabatic process is is one for which there is is no transfer between thethe system andand its An adiabatic process isisone one for which there isisno no heat transfer between the system and itsitsits An adiabatic process isone one for which there isheat heat transfer between the system and adiabatic process for which there heat transfer between system its An adiabatic process for which there nono heat transfer between the system and surroundings; i.e., ddQ 0. Each of Eqs. (3.22), (3.24), and (3.26) may therefore be set equal An adiabatic process is= one for which there isisno no heat transfer between the system and its surroundings; i.e., Qis =one 0.0. Each ofof Eqs. (3.22), (3.24), and (3.26) may therefore bebe set equal An An adiabatic adiabatic process process is one for for which which there there is no heat heat transfer transfer between between the the system system and and its its surroundings; i.e., d Q = 0. Each of Eqs. (3.22), (3.24), and (3.26) may therefore be set equal surroundings; i.e., d Q = 0. Each of Eqs. (3.22), (3.24), and (3.26) may therefore be set equal surroundings; i.e., d Q = Each Eqs. (3.22), (3.24), and (3.26) may therefore set equal surroundings; i.e., d Q = 0. Each of Eqs. (3.22), (3.24), and (3.26) may therefore be set equal Un proceso adiabático esddaquel en el que no hay(3.22), transferencia de calor entre sistemathe y set sus alrededores; surroundings; i.e., Q= =C0. 0. Each of Eqs. (3.22), (3.24), and (3.26) mayel therefore be setequal equal to zero. Integration with and C constant then yields simple relations among variables surroundings; i.e., Q Each of Eqs. (3.24), and (3.26) may therefore be V P to zero. Integration with C and C constant then yields simple relations among the variables V P surroundings; surroundings; i.e., i.e., d d Q Q = = 0. 0. Each Each of of Eqs. Eqs. (3.22), (3.22), (3.24), (3.24), and and (3.26) (3.26) may may therefore therefore be be set set equal equal to zero. Integration with C and C constant then yields simple relations among the variables to zero. Integration with C and C constant then yields simple relations among the variables to zero. Integration with C and C constant then yields simple relations among the variables to zero. Integration with C and C constant then yields simple relations among the variables esto es,to dQ = 0. Por tanto, es cada then una de las ecuaciones (3.22), (3.24)the y (3.26) a cero. La V mechanically P igualar to zero. Integration with C and constant then yields simple relations among the variables VV V Vand PP P P TT ,,zero. P, and VV ,lo valid for reversible adiabatic compression or expansion of ideal Integration with Cposible CC constant yields simple relations among variables V andC and ,valid for mechanically reversible adiabatic compression or expansion ofof ideal to zero. Integration Integration with with C C CPPreversible constant then then yields yields simple simple relations relations among among the the variables V Vand Pconstant T ,TTto P, and V ,VVvalid for mechanically adiabatic compression or expansion ofvariables ideal , Tzero. P, and ,V for mechanically reversible adiabatic compression or expansion of ideal ,P, P, and ,valid valid for mechanically reversible adiabatic compression or expansion ideal para ,gases. P, and , valid for mechanically reversible adiabatic compression or expansion of ideal integración con C y C constantes produce relaciones sencillas para las variables T, P y V, válida T , P, and V , valid for mechanically reversible adiabatic compression or expansion of ideal V P For example, Eq. (3.22) becomes: P, and V valid for mechanically reversible adiabatic compression ororexpansion expansion ofofideal ideal For example, Eq. (3.22) becomes: TTgases. T,,gases. ,P, P, and and V Vexample, ,, ,valid valid for for mechanically mechanically reversible reversible adiabatic adiabaticde compression compression or expansion of ideal gases. For example, Eq. (3.22) becomes: gases. For example, Eq. (3.22) becomes: For Eq. (3.22) becomes: gases. example, Eq. (3.22) becomes: compresión oFor expansión adiabática, mecánicamente reversible gases ideales Por ejemplo, la ecuación gases. For example, Eq. (3.22) becomes: gases. For example, Eq. (3.22) becomes: gases. gases. For For example, example, Eq. Eq. (3.22) (3.22) becomes: becomes: dT d V R (3.22) será: dT d V R dTdT dRVdRdV R RR dT VdVV dT= − dT dT ddV VV RR = − == −− = T = =− −−C VV d T C dT dT R = − T C V T TTT ==C VVV CC VdV VC V V V V − − TT CCV V VV T C V V gives: Integration with C VV constant constant gives: Integration with constant gives: Integration with C constant gives: Integration with CVC constant gives: Integration with CVconstant gives: Integration with Vconstant V gives: Integration with C VVconstante De la integración con CC se obtiene: constant gives: Integration with C V � � constantgives: gives: Integration Integrationwith withCCV Vconstant �� R/C R/CVV �� TT22� � VVR/C 1� � ��� R/C R/C R/C V VR/C 1� VV V V � � T2 TT22TT V V V = V R/C 1 1 2 1 V 1 V = � � � � 2 = V11 R/C R/C T VV 2 = == VVVV TT2T11= 2 T 2 2 1 1 = T1 TT11TT V2VV2V V 1= TT11 = V2V22 2 T V 1 1 2 2 Similarly, Eqs. (3.24) and (3.26) lead to: Similarly, Eqs. (3.24) and (3.26) lead to: Similarly, Eqs. (3.24) andand (3.26) lead to: Similarly, Eqs. (3.24) and (3.26) lead to: Similarly, Eqs. (3.24) and (3.26) to: conducen a: Similarly, Eqs. (3.26) lead to: Similarly, Eqs. (3.24) and (3.26) lead to: De manera similar, las(3.24) ecuaciones (3.24) ylead (3.26) Similarly, Eqs. (3.24) and (3.26) lead to: � R/C � � � Similarly, Similarly,Eqs. Eqs.(3.24) (3.24)and and(3.26) (3.26) to: �� C PPleadto: R/Clead CPP/C /CVV �� � �� �� � �PPR/C TT22� � PP22� � VVC1� 2� � � �� ��� R/C C/C /C R/C CPC /C R/C C /C P PVV/C V PR/C P� 2 1V PPP PV P P � � � VV T P P T2 TT22T2 2= P� P V P V and = PP2P P V R/C P C PP/C 2� 2 1 2 2�2� 2 1 2 1 P V 2 1 V = and = � � � � 2 1 R/C R/C CC /C /C T P P V P P P P VV 2 2 2 1 y = and = = and = = and = T P P V = and = = PPP and = 112= 112 1 2 T P V T T P P V V 1 2 2 2 2 2 1 1 and T1 TT11TT P1PP1P P1PP1P V2VV2V P P= V 1= 1= and and 1= P1P11 1 P1P11 = V2V22 2 TTmay T1 1 also P P V 1 1 1 1 1 2 2 These equations be expressed as: These equations may also bebe expressed as: These equations may also be expressed as:as: These equations may also be expressed as: These equations may also expressed as: These equations may be Estas ecuaciones también esalso posible expresarlas como: These equations may also beexpressed expressed as: These equations may also be expressed as: These Theseequations equationsmay mayalso alsobebeexpressed expressedas: as: γγ−1 (1−γ )/γ −1 (1−γ )/γ = T V = const. (3.30a) T P const. (3.30b) P VVγγγγγ γ= const. (3.30c) const. (3.30a) TPTP const. (3.30b) const. (3.30c) )/γ −1 (1−γ )/γ γ −1 (1−γ )/γ γγV −1 )/γ γ= −1 )/γ constante (3.30a) constante (3.30b) constante (3.30c) T VTTγT const. (3.30a) T PTT(1−γ = const. (3.30b) P VPPγP const. (3.30c) == const. (3.30a) == const. (3.30b) == const. (3.30c) TV = const. (3.30a) P(1−γ = const. (3.30b) = const. (3.30c) VV−1 = const. (3.30a) P (1−γ = const. (3.30b) VVP= (3.30c) T V = const. (3.30a) T P = const. (3.30b) P VγγV= =const. const. (3.30c) γ −1 (1−γ )/γ T V = const. (3.30a) T P = const. (3.30b) P V = const. (3.30c) γ −1 γ −1 (1−γ (1−γ )/γ )/γ γ TTVV ==const. const. (3.30a) (3.30a) TTPP ==const. const. (3.30b) (3.30b) PPVV ==const. const. (3.30c) (3.30c) C PP CC γγC≡ (3.31) PC C PPC (3.31) PP C γ ≡ (3.31) ≡ (3.31) γ≡ ≡ (3.31)(3.31) C γγ γ≡ (3.31) ≡ (3.31) VVP C C CPPC γ ≡ (3.31) C C C V V V C V γ γ≡≡C VV (3.31) (3.31) CCV V Equations (3.30) are restricted in application to ideal with conLas ecuaciones (3.30) restringen su aplicación a gases ideales congases capacidades caloríficas Equations (3.30) are restricted inin application toto ideal gases with conEquations (3.30) are restricted in in application toto ideal gases with conEquations (3.30) are restricted in application to ideal gases with conEquations (3.30) are restricted application ideal gases with conEquations (3.30) are restricted gases with conEquations (3.30) are restricted inapplication application toideal ideal gases with constant heat capacities undergoing mechanically reversible adiabatic Equations (3.30) are restricted in application to ideal gases with conconstantes que son sometidos a expansión o compresión adiabática mecánicamente restant heat capacities undergoing mechanically reversible adiabatic Equations Equations (3.30) (3.30) are are restricted restricted inin application application toto ideal ideal gases gases with with conconstant heat capacities undergoing mechanically reversible adiabatic stant heat capacities undergoing mechanically reversible adiabatic stant heat capacities undergoing mechanically reversible adiabatic stant heat capacities undergoing mechanically reversible adiabatic stant heat capacities undergoing mechanically reversible adiabatic expansion or compression. stant heat capacities undergoing mechanically reversible adiabatic versible. expansion or compression. stant stant heat heat capacities capacities undergoing undergoing mechanically mechanically reversible reversible adiabatic adiabatic expansion oror compression. expansion or compression. expansion compression. expansion compression. expansion oror compression. expansion or compression. expansion expansionor orcompression. compression. Para gases ideales, el trabajo de cualquier proceso adiabático en sistema cerrado se conoce por: For ideal gases, the work of any adiabatic closed-system process is given by: For ideal gases, the work any adiabatic closed-system process by: ForFor ideal gases, thethe work of of any adiabatic closed-system process is given by:by: For ideal gases, the work ofof any adiabatic closed-system process isis given by: For ideal gases, the work of any adiabatic closed-system process isgiven given by: ideal gases, work adiabatic closed-system process is For ideal gases, the work ofany any adiabatic closed-system process isgiven given by: For ideal gases, the work of any adiabatic closed-system process is given by: dW = dU = C dI V For Forideal idealgases, gases,the thework workofofany any adiabatic adiabatic closed-system closed-system process process is is given given by: by: ddW = dU =C VV dT dT dU CC d WddW =dW dU =dU C dT == dU == CdT dT = = = = VdT VC dW WW =dU dU = C VV VdT dT d W = dU = C V dT dT d d W W = = dU dU = = C C V V constant C VV ,, W = �U = C �T (3.32) Para CVFor constante, (3.32) For constant CVC �T (3.32) V�T For constant W =�U =�T �T (3.32) ForFor constant C VC WW =W �U =�U C (3.32) For constant C,C W == �U == C (3.32) constant ,V, V, , = (3.32) VC V For constant C W= =�U �U = C �T (3.32) VVC VV V�T For constant C , W = �U = C �T (3.32) V V For Forconstant constant C CV V, , of WW =V�U �U ==CCV V�T �Telimina (3.32) (3.32) Alternative forms Eq. (3.32) result ifif= C is eliminated in favor the heat-capacity ratio γγ :: capaciAlternative forms Eq. (3.32) result C iseliminated eliminated of the heat-capacity ratio Veliminated Se obtienen formas alternativas de la ecuación (3.32) se Cof en favor de la ratio relación Vthe Alternative forms of of Eq. (3.32) result if C is in favor of of the heat-capacity γratio :γγde Alternative forms ofof Eq. (3.32) result Cif is inin favor of the heat-capacity ratio Alternative forms of Eq. (3.32) result iscuando eliminated infavor favor of the heat-capacity Alternative forms (3.32) result ififVifC is in heat-capacity ratio :γ: γ: : Vis Alternative forms ofEq. Eq. (3.32) result C iseliminated eliminated infavor favor of the heat-capacity ratio VVC V Alternative forms of Eq. (3.32) result if C eliminated in favor of the heat-capacity ratio γ V isiseliminated dades caloríficas γforms : formsofofEq. Alternative Alternative Eq. (3.32) (3.32) result result if if C C eliminated in in favor favor of of the the heat-capacity heat-capacity ratio ratio γ γ:: : V V C C R R + R P V C C R R + R P V C=VC R RRRR R CC C+ + RRR= γγC≡ 1+ or C VV = RRRR PC V +R+ PPC VVC =R PP V+ C + RR= CC RR R γ ≡ = 1== + or or = ≡ == 111+ +1+ oror CC = γ≡ C −R 11 γγ γ≡ 1= V= ≡≡C ==C = ++C oror C VC C ==γγ R VVP= VV VV VVC C C+ C CPPC CVVCC R += R V= V γ ≡ = = 1 + or C V C C C γ − 1− C C γ C C γ− C C C γ − V V V V C V C V V == V C VV VV γ −11−11 V V V γ γ≡≡ = = 1 1 + + or or C C = = o V V C C C γ − V V V CC CC CC γ γ−−111 33 Where by definition, 3 33 3 Where byby definition, Donde por definición, Where by definition, Where by definition, Where definition, Where by definition, Where by definition, 33 Where by definition, 33 Where Wherebybydefinition, definition, VV VV VV 33If C and C are constant, γ is necessarily constant. For an ideal gas, the assumption of constant γ is equivalent If3 CVV and CPP are constant, γ is necessarily constant. For an ideal gas, the assumption of constant γ is equivalent 3 If 33CIf C constant, γ isγγnecessarily constant. For an ideal gas, the assumption of constant γ isγγequivalent If3VIf and are constant, necessarily constant. For anan ideal gas, the assumption ofof constant equivalent If are constant, γis isnecessarily necessarily constant. For anideal ideal gas, the assumption ofconstant constant is equivalent CCand and CCare are constant, necessarily constant. For an ideal gas, the assumption of constant isisγis equivalent Pand VC PC C constant, γisisis constant. For gas, the assumption equivalent Vand Pare V 3the to assumption that the themselves are constant. This is only the /C V P VV ≡ to 3the assumption that the heat heat capacities themselves are For constant. This isthe theassumption only way way that that the ratio ratioγγC CisPPequivalent /C ≡ γγ If C and CPC are constant, γcapacities necessarily constant. an ideal gas, of constant 3the Vassumption Pthat If IfCassumption C and CC are constant, constant, γcapacities γis isnecessarily necessarily constant. constant. For For an an ideal ideal gas, gas, the assumption assumption of of constant constant γCC γconstante is is equivalent the assumption that the heat capacities themselves areare constant. This is the only way that the ratio Csea /C ≡ to the assumption the heat capacities themselves are constant. This only way that the ratio /C ≡V≡ to that the heat capacities themselves are constant. This isthe the only way that the ratio C to to the assumption the heat capacities themselves constant. This isismonotonic the only way that the ratio /C ≡ γγ≡γ γ Pequivalent Vdifference Vand Pthat Pare Pand V PC V/C to that the heat themselves are constant. This is the only way that the ratio C /C P V and the C − C = R can both be constant. Except for the gases, both C and actually Si Cto ythe C son constantes, necesariamente es constante. Para un gas ideal, la suposición de que a la P Vγ V the P P V P V and the difference C − C = R can both be constant. Except for the monotonic gases, both C C actually assumption thatP the−heat capacities themselves are constant. This is monotonic the only way that both the ratio C P /C ≡equivale γ V P V V and the difference C C = R can both be constant. Except for the gases, C and C actually to tothe the assumption assumption that that the the heat heat capacities themselves themselves are are constant. constant. This This is isthe the only only way way that that the ratio ratio CC /C /C ≡ ≡ andand the difference C PCC − R= can both beson constant. Except for the monotonic gases, both C the and C actually and the difference − C= can both be constant. Except for the monotonic gases, both and CC actually the difference − C = RRcapacities can both be constant. Except for the monotonic gases, both CC and actually P P V V V P V P V P V and the difference C − C = R can both be constant. Except for the monotonic gases, both C and C actually P V P V P V P V increase with temperature, but the ratio γ is less sensitive to temperature than the heat capacities themselves. P V P V creencia de que las capacidades caloríficas mismas constantes. Ésta es la única manera en que la relación C / C ≡ y laγ γdiferencia increase with temperature, γ is be lessconstant. sensitiveExcept to temperature than the heat capacities P C VV actually and the difference C P − C Vbut=the R ratio can both for the monotonic gases, both Cthemselves. and P and and the thedifference difference CC −Cbut C = = Rratio Rcan both be be constant. constant. Except Except forforthan the the monotonic monotonic gases, gases, both boththemselves. Cthemselves. C andCC actually increase with temperature, but the ratio γ can isγγboth less sensitive to temperature the heat capacities themselves. increase with temperature, but the ratio isγis less sensitive to temperature than the heat capacities with temperature, the ratio is less sensitive totemperature temperature than the heat capacities temperature, the less sensitive to temperature the heat capacities PExcepto P− Vbut Vbut Pthemselves. Pand V Vactually with temperature, the ratio γis less sensitive to the heat capacities C – C increase =increase Rincrease seanwith constantes. para los gases monotónicos, en realidad Cthan ythan C aumentan con la themselves. temperatura, pero la relación γ γ γ P increase with temperature, but the ratio γ is less sensitive to temperatureP thanVthe heat capacities themselves. increase increasewith withtemperature, temperature,but butthe theratio ratioγ γisisless lesssensitive sensitivetototemperature temperaturethan thanthe theheat heatcapacities capacitiesthemselves. themselves. es menos sensible a la temperatura que a las mismas capacidades caloríficas. 03-SmithVanNess.indd 77 V γ 8/1/07 12:53:09 78 78 78 78 78 CHAPTER CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids CHAPTER 3. 3.3.Volumetric Volumetric Properties of Pure Pure Fluids CHAPTER Properties of CHAPTER CAPÍTULO Propiedades volumétricas deFluids fluidos puros RR �T �T W R �T �T W= =C CVV �T �T = = γR W= =C CVVV �T �T = = γ− W − 11 − 11 γγ − Because RT = RT = expression may written: 1V Como RT V es posible escribirla como: Because = 2PP= VP112and and RT22expresión = PP22VV22,, this this expression may be be written: 1 = P1VRT 1 y11 RT 2, esta Because RT =P P1111V V111 and and RT222 = =P P222V V222,, this this expression expression may may be be written: written: Because RT RT 111 = RT − RT V − P V P RT22 − RT11 = P22 V22 − P11 V11 W (3.33) RT2 − − RT RT11 = P V2 − −P P11V V11 P2V W= = RT (3.33) (3.33) W= = γγ22 − = 22 γγ22 − (3.33) W (3.33) − 11 1 = − 111 1 − 11 − 11 γγ − γγ − Equations (3.32) and are compression expansion proEquations (3.32) and (3.33) (3.33) are general general for adiabatic compression and expansionadiabático proLas ecuaciones (3.32) y (3.33) son generales parafor unadiabatic proceso de compresiónand y expansión en Equations (3.32) and whether (3.33) are general for adiabatic compression and expansion proEquations (3.32) and (3.33) are general for adiabatic compression and expansion processes in a closed system, reversible or not, because P, V , and T are state funccesses in a closed system, whether reversible or not, because P, V , and T are state funcun sistema cerrado, sea o no reversible, ya que P, V y T son funciones de estado, independientes de la trayeccessesindependent in aa closed closed system, system, whether reversible or2 not, not, becauseunknown. P, V V ,, and and TT are are state state of funccesses in whether reversible or because P, functions, of However, and are usually tions, independent of path. path. However, and unknown. deElimination Elimination of VV22 (3.33) toria. De cualquier modo, por lo general T2 y VTT se VV conocen. La eliminación V2 de la ecuación 2 are usually 222no tions, independent of path. However, T and V are usually unknown. Elimination of V2 tions, independent of path. However, T and V are usually unknown. Elimination of V 2 2 2 2 from Eq. by (3.30c), valid only for reversible leads the 2 from Eq. (3.33) (3.33) by Eq. Eq.que (3.30c), validsólo only2para for mechanically mechanically reversible processes, processes, leads to the22 mediante la ecuación (3.30c), es válida procesos mecánicamente reversibles, nosto conduce a la from Eq. (3.33) by Eq. (3.30c), valid only for mechanically reversible processes, leads to the from Eq. (3.33) by Eq. (3.30c), valid only for mechanically reversible processes, leads to the expression: expression: expresión: expression: expression: � � � � � � �� �� �(γ �(γ �P � �P � � � �� �� (γ−1)/γ −1)/γ (γ−1)/γ −1)/γ � � � RT PP11VV11 � 1 2 �(γ � � P22 � � � RT P (γ−1)/γ −1)/γ − 1 = (γ−1)/γ −1)/γ − 1 1 2 (γ (γ −1)/γ (γ −1)/γ (3.34) W = RT111 P222 P222 V111 P111V RT P P − 1 = − 1 (3.34) (3.34) W = γP PP11 PP11 − 11 = = γγ − − 11 (3.34) W= = γ− − − (3.34) W − 11 − 11 − 11 P111 − 11 P111 γγ − P γγ − P The result is the PP and given by Theelsame same result is obtained obtained when the relation relation between and VVpor given by Eq. Eq. (3.30c) (3.30c) isseused used Se obtiene mismo resultado cuando la relación entrebetween P y V conocida la ecuación (3.30c)is usa para la ��when Thethe same result is is W obtained when the relation(3.34) between P and and V for given bygases Eq. (3.30c) (3.30c) is used used The same result obtained when the relation between P V given by Eq. is for integration, = − P d V . Equation is valid only ideal with constant � � for the d VLa. Equation valid only for ideal gases with con constant integración de integration, la expresiónW W= = –−∫ P P dV. ecuación (3.34) (3.34) is sólo es válida para gases ideales capacidades for the the integration,adiabatic, W= =− − P P ddV V .. Equation Equation (3.34) is is valid valid only only for for ideal ideal gases gases with with constant constant for integration, W (3.34) heat capacities reversible, heatconstantes, capacities in adiabatic, mechanically reversible, closed-system closed-system processes. caloríficas yinen procesosmechanically adiabáticos mecánicamente reversiblesprocesses. de sistema cerrado. heat capacities capacities in adiabatic, mechanically reversible, closed-system processes. heat in adiabatic, mechanically reversible, closed-system processes. applied to gases, (3.30) yield approxWhen applied to real real gases, Eqs. (3.30) through through (3.34) often oftenfrecuencia yield satisfactory satisfactory approxCuandoWhen se aplican a gases reales, lasEqs. ecuaciones (3.30) a (3.34) (3.34) con proporcionan aproximaWhen appliedthe to real real gases, Eqs. Eqs. (3.30) (3.30) through (3.34) often often yield satisfactory approxWhen applied to gases, through (3.34) yield satisfactory approximations, provided deviations from ideality are relatively small. For monatomic gases, imations, provided they deviations ideality are small. For monatomic gases, ciones satisfactorias, siempre cuando lasfrom desviaciones de lorelatively ideal sean relativamente pequeñas. Para gases imations, provided the the deviations deviations from1.4 ideality are relatively relatively small. For For simple monatomic gases, provided from ideality are small. monatomic gases, γimations, values of for gases and for polyatomic γ= = 1.67; 1.67; approximate valuesaproximados of γγ are are 1.4de forγdiatomic diatomic gases and 1.3 1.3son for de simple polyatomic monoatómicos, γ =approximate 1.67; los valores para gases diatómicos 1.4 para gases diatómiγ = 1.67; approximate values of γ are 1.4 for diatomic gases and 1.3 for simple polyatomic γ = 1.67; approximate values of γ are 1.4 for diatomic gases and 1.3 for simple polyatomic such as .. como CO2, SO2, NH3 y CH4. 2 ,, SO gases suchgases as CO CO SO22,, NH NH33,simples, , and and CH CH4tales cos y degases 1.3 para poliatómicos gases such such as as CO CO2222,, SO SO222,, NH NH333,, and and CH CH4444.. gases Whence Whence Whence Whence De donde, Polytropic Polytropic Process Process Polytropic Process Proceso politrópico Because polytropic means “turning many ways,” polytropic process suggests a model of some Because polytropic means “turning many ways,” polytropic process suggests a model of some Because polytropic polytropic means “turning “turning many ways,” ways,” polytropic processbysuggests suggests model of of some some Because means many polytropic process aa model With itit is as represented empirical versatility. With δδ asignifica a constant, constant, is defined defined as aa process process represented by the thepolitrópicos empirical equation: equation: Debidoversatility. a que politrópico “cambios de muchas maneras”, los procesos sugieren un moversatility. With With δδ aa constant, constant, itit is is defined defined as as aa process process represented represented by by the the empirical empirical equation: equation: versatility. delo con algo de versatilidad. Con d como una constante, éste se define como un proceso representado por la PPVV δδδ = (3.35a) ecuación empírica: = constant constant (3.35a) PV V δδ = = constant constant (3.35a) P (3.35a) d PV =(3.30a) constante (3.30b) are readily derived: (3.35a) For For an an ideal ideal gas gas equations equations analogous analogous to to Eqs. Eqs. (3.30a) and and (3.30b) are readily derived: For an an ideal ideal gas gas equations equations analogous analogous to to Eqs. Eqs. (3.30a) (3.30a) and and (3.30b) (3.30b) are are readily readily derived: derived: For Es fácil deducir las ecuaciones análogas a las ecuaciones (3.30a) y (3.30b) para un gas ideal: δ−1 = (1−δ)/δ = TT VV δ−1 constant (3.35b) TT PP(1−δ)/δ = constant (3.35b) = constant constant (3.35c) (3.35c) δ−1 (1−δ)/δ δ−1 (1−δ)/δ δ−1 = Vd–1 = constant constant (3.35b) TT P P(1−δ)/δ = constant constant (3.35c) TT V (3.35b) TV = constante (3.35b) TP(1–d)/d = = constante (3.35c) (3.35c) �� When the relation between P and V is given by Eq. (3.35a), evaluation When the relation between P and V is given by Eq. (3.35a), evaluation of of �� PP ddVV yields yields When the relation relation between P dada and V Vpor is given given by Eq. Eq.(3.35a), (3.35a), la evaluation of deP P∫ddPV V dV yields When the P and is by (3.35a), evaluation of yields Cuando la relación entre between P y Vδ:está la ecuación evaluación produce la Eq. Eq. (3.34) (3.34) with with γγ replaced replaced by by δ: Eq. (3.34) with replaced by by δ:d: Eq. (3.34) with γγ replaced δ: ecuación (3.34) con γ reemplazada por � � �� � �(δ−1)/δ �P � � � (δ−1)/δ � � RT 1 2 � � � � P2 (δ−1)/δ RT1 (δ−1)/δ − 1 (δ−1)/δ (3.36) W = P222 RT111 P − 1 (3.36) W = δRT PP11 − 11 (3.36) (3.36) W= = δ− − (3.36) W − 11 δ − 1 P δ−1 P111 Moreover, Moreover, for for constant constant heat heat capacities, capacities, the the first first law law solved solved for for Q Q yields: yields: Moreover, for constant constant heat capacities, capacities, thela first law solved solved for Q Q yields: yields: Moreover, for heat the first law for Además, para capacidades caloríficas constantes, primera ley resuelta para Q produce: � � � �� � (δ−1)/δ � � �� � � (δ �(δ−1)/δ � PP22 � (δ − − γγ )RT )RT11 � (δ−1)/δ − 1 (δ−1)/δ (3.37) Q P222 (δ−1)/δ (δ − − γγ )RT )RT111 P −1 (3.37) Q= = (δ(δ − 1)(γ − 1) P − 11 (3.37) (3.37) Q= = (δ − 1)(γ − 1) − (3.37) Q P11 (δ − − 1)(γ 1)(γ − − 1) 1) P111 (δ P 03-SmithVanNess.indd 78 8/1/07 12:53:21 79 3.3. El gas ideal Los procesos descritos en esta sección corresponden a las cuatro trayectorias que se muestran en la figura 3.6 para valores específicos de d: • Proceso isobárico: mediante la ecuación (3.35a), d = 0. • Proceso isotérmico: por la ecuación (3.35b), d = 1. • Proceso adiabático: d = γ. • Proceso isocórico: mediante la ecuación (3.35a), dV/dP = V/Pd; para V constante, d = ±∞. 0 1 P Figura 3.6: Trayectorias de procesos politrópicos caracterizados por valores específicos de d. V Proceso irreversible Todas las ecuaciones desarrolladas en esta sección fueron deducidas para procesos mecánicamente reversibles en sistemas cerrados para gases ideales. Sin embargo, las ecuaciones que proporcionan cambios de propiedad (dU, dH, ∆U y ∆H) son válidas para gases ideales sin considerar el proceso. se aplican de igual modo a procesos reversibles e irreversibles tanto en sistemas cerrados como abiertos, ya que los cambios en las propiedades dependen sólo de los estados inicial y final del sistema. Por otra parte, una ecuación para Q o W, a menos que sea igual al cambio en una propiedad, se somete a las restricciones en su deducción. El trabajo de un proceso irreversible se calcula con un procedimiento de dos pasos. Primero, se determina W para un proceso mecánicamente reversible que alcanza el mismo cambio de estado que el proceso irreversible real. Segundo, este resultado se multiplica o se divide por una eficiencia para obtener el trabajo real. Si el proceso produce trabajo, el valor absoluto para el proceso reversible es muy grande y debe multiplicarse por una eficiencia. Si el proceso requiere trabajo, el valor para el proceso reversible es muy pequeño y debe dividirse entre la eficiencia. Las aplicaciones de los conceptos y de las ecuaciones de esta sección se ilustran en los siguientes ejemplos. En particular, el trabajo de los procesos irreversibles se trata en el ejemplo 3.4. Véase el problema 3.13. 03-SmithVanNess.indd 79 8/1/07 12:53:23 80 CAPÍTULO 3. Propiedades volumétricas de fluidos puros Ejemplo 3.2 Se comprime aire a partir de la condición inicial de 1 bar y 25 °C hasta alcanzar un estado final de 5 bar y 25 °C mediante tres procesos diferentes y mecánicamente reversibles en un sistema cerrado: a) Calentamiento a volumen constante seguido por un enfriamiento a presión constante. b) Compresión isotérmica. c) Compresión adiabática seguida por enfriamiento a volumen constante. Suponga que el aire es un gas ideal con capacidades caloríficas constantes, CV = (5/2)R y C P = (7/2)R. Calcule el trabajo requerido, el calor transferido y los cambios en la energía interna y la entalpía del aire para cada proceso. 10 8 Figura 3.7: Diagrama para el ejemplo 3.2. P/bar c 6 2 4 a b 2 1 0 5 10 15 20 25 V 103/m3 Solución 3.2 Elija como sistema 1 mol de aire. Para R = 8.314 J mol–1 K–1, CV = 20.785 CP = 29.099 J mol–1 K–1 Los estados inicial y final del aire son idénticos a los del ejemplo 2.9, donde: V1 = 0.02479 V2 = 0.004958 m3 Ya que T es la misma al principio y al final del proceso, para todos los casos: DU = DH = 0 a) El proceso en este caso es exactamente el del ejemplo 2.9b), para el cual: Q = –9 915 J y 03-SmithVanNess.indd 80 W = 9 915 J 8/1/07 12:53:24 3.3. El 3.3. gas ideal The Ideal Gas 3.3. 3.3. The The Ideal Ideal Gas Gas 3.3. The Ideal Gas 81 81 81 81 81 b) Aplicamos la ecuación la compresión isotérmica un gas (b) Equation (3.27)(3.27) for thepara isothermal compression of ande ideal gas ideal: applies: (b) Equation (3.27) for the isothermal compression of an ideal (b) Equation Equation (3.27) (3.27) for for the the isothermal isothermal compression compression of of an an ideal ideal gas gas applies: applies: (b) gas applies: P1 1 Q = −W = RT ln P (8.314)(298.15) ln 111 = −3,990 J PP111 = Q (8.314)(298.15) 2 = Q= = −W −W = = RT RT ln ln P = (8.314)(298.15)ln ln 55 = = −3,990 −3,990 JJJ Q = −W = RT ln = (8.314)(298.15) ln = −3,990 P PP222 55 (c) The initial step of adiabaticadiabática compression takes the air it to its final c) La etapa inicial la compresión toma el aire y lo lleva afinal un volume volumenof (c) initial step of adiabatic compression takes the air it volume of 3de (c) The The initial step of (3.30a), adiabaticthe compression takes thepoint air to its its final volume offinal de (c) The initial step of adiabatic compression takes the air ititis:to to its final volume of 0.004958 m . By Eq. temperature at this 3 3 0.004958 m . Usando la ecuación (3.30a), la temperatura en este punto es: 3 3 0.004958 m . By Eq. (3.30a), the temperature at this point is: 0.004958 m m .. By By Eq. Eq. (3.30a), (3.30a), the the temperature temperature at at this this point point is: is: 0.004958 � �γ −1 � �0.4 � � � � �V �γγγ−1 � 0.02479 �0.4 � −1 = (298.15) � 0.4 = 567.57 K −1 VV1111� 0.02479 T �� = T1 � 0.02479 0.4 0.02479 �� = T1 V T = (298.15) = V 0.004958 = TT11 V2 = (298.15) (298.15) 0.004958 = 567.57 567.57 K K TT = = = 567.57 K 0.004958 VV222 0.004958 For this step, Q = 0, and by Eq. (3.32), the work of compression is: = 0, and (3.32), work compression For this this step, Qpor = la 0, ecuación and by by Eq. Eq. (3.32), the workdeof ofcompresión compression is: For this step, Q = 0, and by Eq. (3.32), the work of compression is: En esta For etapa Q step, = 0 y,Q (3.32), el the trabajo es:is: W = C V �T = (20.785)(567.57 − 298.15) = 5,600 J W �T = = (20.785)(567.57 (20.785)(567.57 − − 298.15) 298.15) = = 5,600 5,600 JJJ W= =C CVVV �T �T = (20.785)(567.57 − 298.15) = 5,600 W = C For the constant-volume step, no work is done; the heat transfer is: For constant-volume step, no work done; For the the constant-volume step, noefectúa work is istrabajo; done; the the heat transfer transfer is: is: For the constant-volume step, no work is done; the heat transfer is: Para la etapa a volumen constante no se la heat transferencia de calor es: � Q = �U = C V (T2 − T � ) = −5,600 J Q (T222 − −T = −5,600 −5,600 JJJ Q= = �U �U = =C CVVV(T (T − TT��))) = = −5,600 Q = �U = C Thus for process (c), for process (c), De esta Thus manera, el proceso c), Thus for para process (c), Thus for process (c), WW == 5,600 J J and 5 600 y QQ == –5−5,600 600 J J W and Q W= = 5,600 5,600 JJJ and Q= = −5,600 −5,600 JJJ W = 5,600 and Q = −5,600 Aunque Although los cambios la propiedad ∆U y�U ∆H and son cero cada for proceso, y W dependen de theenproperty changes �H para are zero each Q process, Q Although the changes �U and �H are zero for each process, Q Although theQproperty property changes �U and �H are zero for each process, Q Ya que the property �U �H are zero process, Q la trayectoria, aquí = –W. Lachanges figura proceso en each un diagrama PV. and Although W pero are path-dependent, but here3.7 Q muestra =and −W .cada Figure 3.7for shows each process and W are path-dependent, but here Q = −W 3.7 each process and W arediagram. path-dependent, but here Qfor =each −W..of . Figure Figure 3.7 shows shows each process and are path-dependent, but here Q = −W Figure 3.7 shows each process el trabajo para cada uno de estos procesos mecánicamente reversibles es conocido por W = –∫ P on aW PV Because the these mechanically reversible � work on a P V diagram. Because the work for each of these mechanically reversible on aa PPde diagram. Because the work for each of these mechanically reversible on VVcada diagram. for each of these reversible ��� Pwork dV, el trabajo proceso es proporcional área total debajo de las trayectorias en el diagraprocesses is given byBecause W = −the d V ,al the work for eachmechanically process is proportional processes is given by W − P dddV ,,, the work for each is proportional processes is given by W= = − on the work forfrom each1process process islos proportional processes by W = − PP the VVde work for each process proportional ma PV que vatotal de 1is a given 2. Los tamaños relativos estas áreas corresponden ais valores numéricos to the area below the paths Pthe V diagram to 2. The relative sizes to area below paths on diagram from 1 to to the the total total area below the theto paths on the the P diagram from to 2. 2. The The relative relative sizes sizes the total area below the paths the PPV VV values diagram 2. The relative sizes de W. to of these areas correspond the on numerical offrom W . 11 to of of these these areas areas correspond correspond to to the the numerical numerical values values of of W W... of these areas correspond to the numerical values of W Ejemplo 3.3 3.3 Example Example 3.3 Example 3.3la siguiente secuencia de procesos mecánicamente reversibles en un sisteExample 3.3 Un gas ideal experimenta An ideal gas undergoes the following sequence of mechanically reversible processes An gas undergoes ma cerrado: Anaideal ideal gas undergoes the the following following sequence sequence of of mechanically mechanically reversible reversible processes processes An ideal gas undergoes the following sequence of mechanically reversible processes in closed system: in in a closed system: system: in aa closed closed system: a) Desde un estado inicial de 70 °C y◦ 1 bar, se comprime adiabáticamente hasta la temperatura de (a) From an initial state of 70◦ C and 1 bar, it is compressed adiabatically to 150◦◦ C. ◦ ◦ ◦ ◦ From an initial state of 70 C and 1 bar, it is compressed adiabatically to 150 C. 150 (a) °C. (a) From From an an initial initial state state of of 70 70 C C and and 11 bar, bar, itit is is compressed compressed adiabatically adiabatically to to 150 150 C. C. (a) ◦ (b) It is then from 150 to °C 70 C at constant pressure. b) A continuación secooled enfría de 150 a 70 constante. (b) It is then cooled from 150 to 70◦◦a◦Cpresión at constant pressure. (b) ItIt is is then then cooled cooled from from 150 150 to to 70 70 C C at at constant constant pressure. pressure. (b) Finally, it is expanded isothermally to its state. c) Por (c) último, se expande isotérmicamente hasta su original estado original. (c) (c) Finally, Finally, it is expanded expanded isothermally isothermally to to its its original original state. state. (c) Finally, itit is is expanded isothermally to its original state. Q, para �U , cada and �H of procesos the three yprocesses andcompleto. for the entire CalculeCalculate W, Q, ∆UW y ,∆H uno for de each los tres para el ciclo Tomecycle. CV = (3/2)R Calculate W ,,, Q, ,,, and �H for Calculate W(3/2)R Q, �U �U and �H for each each of of the the three three processes processes and and for for the the entire entire cycle. cycle. Calculate Q, �U and for each of the three processes and for the entire cycle. C V =W and C P�H = (5/2)R. y C P = Take (5/2)R. Take Take C CVVV = = (3/2)R (3/2)R and and C CPPP = = (5/2)R. (5/2)R. Take C = (3/2)R and C = (5/2)R. 03-SmithVanNess.indd 81 8/1/07 12:53:31 82 CHAPTER CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids CHAPTER 3. Volumetric Properties of Pure Fluids 82 82 82 82 82 CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CAPÍTULO 3. Propiedades volumétricas de fluidos puros 3 3 70°C 3 70°C 70°C 3 70°C 3 70°C Figure Figure 3.8: 3.8: Diagram Diagram for for Ex. Ex. 3.3. 3.3. Figure 3.8: Diagram for Ex. 3.3. Figure 3.8: Diagram for Ex. 3.3. FiguraFigure 3.8: Diagrama para el 3.8: Diagram forejemplo Ex. 3.3.3.3. PP P P P b 22 b 150°C b 2 150°C 150°C b 2 150°C b 23 b 2 150°C 150 °C 70 °C a a a c c c P a a c c c a 70°C 70°C 1 70°C 1 1 70°C 1 70°C 1 VV V V V 1 70 °C V Solution 3.3 Solution 3.3 Solución 3.3 Solution 3.3 −1 −1 –1, −1 K −1, R Para Solution R =For 8 314 mol For R= =J 8.314 8.314 mol 3.3–1 JJKmol −1 K−1 , For R = 8.314 Solution 3.3 J mol K , −1 −1 −1 K−1 , C 12.471 C For R = 8.314 mol CV J= = 12.471 CP = = 20.785 20.785 JJ mol mol−1 K K−1 −1 K−1 , C VVJ = 12.471 C PP = 20.785 J mol−1 K−1 For R = 8.314 mol −1 K is shown on 3.8. Take as aa basis 11−1 mol 12.471 C Pfigura = 20.785 J mol V = en The cycle is C shown on aa P PV V diagram diagram in Fig. 3.8. Take as basis mol of gas. El cicloThe estácycle representado PVin enFig. la 3.8. Tome como baseof 1 gas. mol −1 The cycle isCshown onuna diagrama P V diagram in Fig. Take as a basis mol of gas. de gas. C P 3.8. = 20.785 J mol K1−1 V = 12.471 a) Para (a) un gas ideal que se somete a una compresión adiabática, Q = 0, de donde an gas adiabatic compression, Q (a) For For an ideal ideal gas undergoing undergoing adiabatic compression, Qa= = 0; 0; whence whence The cycle shown a P V diagram in Fig. 3.8. Take asQ 1 mol of gas. (a) For an isideal gas on undergoing adiabatic compression, =basis 0; whence The cycle is shown on a P V diagram in Fig. 3.8. Take as a basis 1 mol of gas. W = − JJ (a) For an ideal�U gas= adiabatic compression, Q == whence �U =undergoing W= =C CV �T �T = (12.471)(150 (12.471)(150 − 70) 70) =0;998 998 W = C VV �T = (12.471)(150 − 70) J (a) For an ideal�U gas= undergoing adiabatic compression, Q ==0;998 whence �U = W = C �T = (12.471)(150 − 70) = 998 J �T = (20.785)(150 − 70) = 1,663 J �H = C V = (20.785)(150 − 70) = 1,663 J �H = CPP �T �U == W C=P C = (12.471)(150 − 70) = 998J J �H �T = (20.785)(150 − 70) = 1,663 V �T from Eq. (3.30b): Pressure is found found from Eq. (3.30b): Pressure P22 is La presión P2 se P puede encontrar a partir de la ecuación (3.30b): �H = C �T = (20.785)(150 − 70) = 1,663 J PEq. (3.30b): from Pressure P2 is found �H = C �T = (20.785)(150 − 70) = 1,663 J P � � � � 2� �γγ/(γ /(γ −1) 2.5 Eq.−1) (3.30b): �150 + 273.15 �2.5 Pressure P2 is found � TTfrom 2 �γ /(γ −1) = (1) � 150 + 273.15 �2.5 = 1.689 bar P = P 2 1 + 273.15 Eq. (3.30b): Pressure PP22 is=found P1 TTfrom = (1) 150 = 1.689 bar 2 P2 = P1 � T11 �γ /(γ −1) = (1) � 70 70 + + 273.15 273.15 �2.5 = 1.689 bar 70 + 273.15 T 150 + 273.15 21 P2constant-pressure = P1 � T2 �γ /(γ −1) = (1) � 150 + 273.15 �2.5 = 1.689 bar (b) process, T (b) For For this this constant-pressure process, 1 P = P = (1) 70 + 273.15 = 1.689 bar 1 (b) For this2 constant-pressure process, T1 constante: 70 + 273.15 b) Para este proceso a presión = Q (b) For this constant-pressure process, �T = (20.785)(70 (20.785)(70 − − 150) 150) = = −1,663 −1,663 JJ Q= = �H �H = =C CP �T = (20.785)(70 − 150) = −1,663 J Q = �H = C PP �T (b) For this constant-pressure process, (20.785)(70 150)==−998 −1,663 Q =�U �H= �T (12.471)(70 −− 150) J J P �T== �U ==C CVC V �T = (12.471)(70 − 150) = −998 J �T==(12.471)(70 (20.785)(70−−150) 150)==−998 −1,663 Q =�U �H==CC J J V P�T W = �U − Q = −998 − (−1,663) = 665 J �U == (12.471)(70 − 150) = W= = C�U −Q −998 − (−1,663) = −998 665 J J V �T W= =C�U − Q = −998 − (−1,663) = 665 J �U V �T = (12.471)(70 − 150) = −998 J (c) process, (c) For For ideal ideal gases gases undergoing an isothermal process,=�U �U and �H �H are are zero; zero; W undergoing = �U − Qan = isothermal −998 − (−1,663) 665 and Jand (c) (3.27) For ideal gases undergoing an isothermal process,= �U �H are zero; Eq. yields: W = �U − Q = −998 − (−1,663) 665 J Eq. (3.27) yields: Eq. For (3.27) yields: (c) ideal gases undergoing an isothermal process, �U and �H are zero; c) Para (c) gases ideales que seundergoing someten a an un isothermal proceso isotérmico, ∆H �H son cero; la ecuación P P For ideal gases process, ∆U �U y1.689 and are zero; Eq.Q(3.27) yields: P2 1.689 P3 = 1,495 JJ = = P22 = P33 = = RT RT ln ln P = (8.314)(343.15) (8.314)(343.15) ln ln 1.689 = 1,495 Q = −W −W = RT RT ln ln P (3.27) produce: Eq. Q (3.27) yields: = −W = RT ln P11 = RT ln P11 = (8.314)(343.15) ln 11 = 1,495 J 1 P21 1.689 P31 Q = −W = RT ln P3 = RT ln P2 = (8.314)(343.15) ln 1.689 = 1,495 J Q = −W = RT ln P1 = RT ln P1 = (8.314)(343.15) ln 1 = 1,495 J P1 P1 1 03-SmithVanNess.indd 82 8/1/07 12:53:49 3.3. The The Ideal Ideal Gas Gas 3.3. 3.3. The Ideal Gas The 3.3. El3.3. gas ideal 3.3. The Ideal Ideal Gas Gas 83 83 83 83 83 83 For the the entire cycle, cycle, For entire For the entire cycle, Para todo ciclo, For entire Forelthe the entire cycle, cycle, Q= = 000 − − 1,663 1,663 + + 1,495 1,495 = = −168 −168 JJJ Q Q = − 1,663 + 1,495 = −168 Q Q= = 00 − − 1,663 1,663 + + 1,495 1,495 = = −168 −168 JJ W = = 998 998 + + 665 665 − − 1,495 1,495 = = 168 168 JJJ W W = 998 + 665 − 1,495 = 168 W W = = 998 998 + + 665 665 − − 1,495 1,495 = = 168 168 JJ �U = 998 − 998 + 0 = 0 �U �U = = 998 998 − − 998 998 + + 000 = = 000 �U �U = = 998 998 − − 998 998 + + 0= =0 �H = 1,663 − 1,663 + = 000 �H = 1,663 − 1,663 + �H = = 1,663 − − 1,663 + + 000 = = �H �H = 1,663 1,663 − 1,663 1,663 + 00 = = 00 The property property changes changes �U �U and and �H �H both both are are zero zero for for the the entire entire cycle, cycle, because because the the The The property changes �U and �H both are zero for the entire cycle, because the The property changes �U and both are zero for the entire cycle, the Los cambios en la final propiedad ∆U yidentical. ∆H�H son cero para elthat ciclo completo, yathe quebecause los estados The property changes �U and �H both are zero for the entire cycle, because the inicial initial and final states are identical. Note also that Q = −W for the cycle. This initial and states are Note also Q = −W for cycle. This initial and final final states are are identical. Note also thatelQ Q= = −W −W for the cycle. cycle. This initial and states identical. Note also that the This y final son idénticos. Observe también que==Q W para Estefor resultado surge de la priinitial and final states are identical. Note that Qciclo. = −W for the cycle. This follows from the first law law with �U 0.= – also follows from the first with �U 0. follows from the first law with �U = 0. follows from the first law with �U = 0. mera leyfollows con ∆Ufrom = 0.the first law with �U = 0. Example 3.4 Example 3.4 Example 3.4 Ejemplo 3.4 If the processes of Ex. Ex. 3.3 3.3 are are carried carried out out irreversibly irreversibly but but so so as as to to accomplish accomplish exactly exactly If the processes If the processes of of Ex. 3.3 are carried out irreversibly but so as to accomplish exactly IfIf the the processes processes of of Ex. Ex. 3.3 3.3 are are carried carried out out irreversibly irreversibly but but so so as as to to accomplish accomplish exactly exactly the same samedel changes of state— the same same changes changes in P, P, T U and H H— — then then different differentlos misthe changes state— the in the same changes of state— the same changes in P, TT ,,,, U U and H — then different Si los procesos ejemploof 3.3 son irreversibles, pero de modo se,,,, and consigan the same changes of state— the changes in P, T U H then different the same changes of state— the same sameQ changes in P, que T ,step U , and and H— — exactamente then different values of Q and W result. Calculate Q and W if each step is carried out with an an values of Q and W result. Calculate and W if each is carried out with valuesde ofestado Q and and(los W mismos result. cambios Calculate QP, and and WH),ifif por each step se is producen carried out out with an mos cambios enQ U yW lo tanto diferentes values of Q step with values of of Q 80%. and W W result. result. Calculate Calculate Q T, and W if each each step is is carried carried out with an anvalores efficiency of 80%. efficiency efficiency ofQ80%. 80%. de Q y efficiency W. Calculeof y W si cada etapa se lleva a cabo con una eficiencia de 80%. efficiency of 80%. Solution 3.4 Solución 3.4 Solution 3.4 Solution 3.4 Solution 3.4 If the the same same changes of state statede asestado in Ex. Ex.que 3.3 en are carried carried out3.3 by irreversible irreversible processes, If changes of as in 3.3 are out by If the same changes of state as in Ex. 3.3 are carried out by irreversible processes, Si se realizan los mismos cambios ejemplo medio deprocesses, procesos irreverIf the same changes of state as in 3.3 are out by irreversible processes, If the same changes offor state assteps in Ex. Ex. 3.3 areelcarried carried out bypor irreversible processes, the property changes for the steps are identical with those of Ex. 3.3. However, the property changes the are identical with those of Ex. 3.3. However, the property changes for the steps are identical with those of Ex. 3.3. However, sibles, los cambios en la propiedad para las etapas son idénticos a los del ejemplo 3.3. No obstanthe property changes for the the property the steps steps are are identical identical with with those those of of Ex. Ex. 3.3. 3.3. However, However, the values values ofychanges Q and Wfor change. the of Q and W change. the values of Q and W change. te, los valores de Q W se modifican. the the values values of of Q Q and and W W change. change. (a) For mechanically reversible, adiabatic compression, compression, W== = 998 J. If If the process (a) For mechanically reversible, adiabatic W 998 process (a) For For mechanically reversible, adiabatic compression, W = 998 J. If the process a) Para (a) una compresión adiabática, mecánicamente reversible, W 998 J. J. Si elthe proceso tiene 80% mechanically reversible, adiabatic compression, W = 998 J. the process (a) For mechanically reversible, adiabatic compression, W = 998 J. If If the process is 80% efficient compared with this, W = 998/0.80 = 1,248 J. This step cannot is 80% efficient compared with this, W = 998/0.80 = 1,248 J. This step cannot is 80% 80%enefficient efficient compared with this, W = 998/0.80 998/0.80 = J. 1,248 J. This This step cannot de eficiencia comparación con esto, W = 998/0.80 = 1 248 En este caso no es posible que is compared with this, W = = 1,248 J. step cannot is 80% efficient with this, W = 998/0.80 = 1,248 J. This step cannot here beadiabática. adiabatic.compared By the first law, here be adiabatic. By the first law, here be adiabatic. By the first law, esta etapa sea Por la primera ley, here be adiabatic. By the first law, here be adiabatic. By the first law, Q= = �U �U − −W W= = 998 998 − − 1,248 1,248 = = −250 −250 JJJ Q Q = �U − W = 998 − 1,248 = −250 Q Q= = �U �U − −W W= = 998 998 − − 1,248 1,248 = = −250 −250 JJ (b) The The work for the thede mechanically reversible coolingreversible process is ises665 665 J. For For the el pro(b) work for mechanically cooling process J. the b) El trabajo para el proceso enfriamientoreversible mecánicamente de 665 J. Para (b) The work for the mechanically reversible cooling process is 665 J. For the (b) The work for the mechanically reversible cooling process is 665 J. For the (b) The work for the mechanically reversible cooling process is 665 J. For the irreversible process, W = 665/0.80 = 831 J, and irreversible process, W = 665/0.80 = 831 J, and ceso irreversible, = 665/0.80 = 831 J, y = 831 irreversibleWprocess, process, W= = 665/0.80 831 J, and and irreversible irreversible process, W W = 665/0.80 665/0.80 = = 831 J, J, and Q= = �U �U − −W W= = −998 −998 − − 831 831 = = −1,829 −1,829 JJJ Q Q = �U − W = −998 − 831 = −1,829 Q Q= = �U �U − −W W= = −998 −998 − − 831 831 = = −1,829 −1,829 JJ c) A medida que el trabajo es by realizado por elin en the esta etapa, el trabajo (c) As As work is done done by the system system insistema this step, step, the irreversible irreversible workirreversible in absolute absoluteen valor (c) work is the this work in (c) As work is done by the system in this step, the irreversible work in absolute (c) As work is by the system in this step, the (c) As work is done done by the system in this step,J:J: the irreversible irreversible work work in in absolute absolute absolutovalue es menor que el trabajo reversible de –1 495 value is less than the reversible work of −1,495 J: is less than the reversible work of −1,495 value is is less than than the reversible reversible work of of −1,495 J: J: value value is less less than the the reversible work work of −1,495 −1,495 J: W= = (0.80)(−1,495) (0.80)(−1,495) = = −1,196 −1,196 JJJ W W = (0.80)(−1,495) = −1,196 W W= = (0.80)(−1,495) (0.80)(−1,495) = = −1,196 −1,196 JJ Q= = �U �U − −W W= = 000 + + 1,196 1,196 = = 1,196 1,196 JJJ Q Q = �U − W = + 1,196 = 1,196 Q Q= = �U �U − −W W= = 00 + + 1,196 1,196 = = 1,196 1,196 JJ For the the entire entire cycle, cycle, �U �U and and �H �H are are zero, zero, with with For For the entire cycle, �U and �H are zero, with entire and are zero, Para el For ciclo completo, ∆ U �U y�U ∆H son�H cero, For the the entire cycle, cycle, and �H arecon zero, with with Q= = −250 −250 − − 1,829 1,829 + + 1,196 1,196 = = −883 −883 JJJ Q Q = −250 − 1,829 + 1,196 = −883 Q Q= = −250 −250 − − 1,829 1,829 + + 1,196 1,196 = = −883 −883 JJ W= = 1,248 1,248 + + 831 831 − − 1,196 1,196 = = 883 883 JJJ W W = 1,248 + 831 − 1,196 = 883 W W= = 1,248 1,248 + + 831 831 − − 1,196 1,196 = = 883 883 JJ A summary summary of of these these results results and and those those for for Ex. Ex. 3.3 3.3 is is given given in in the the following following table; table; A A summary of these results and those for Ex. 3.3 is given in the following table; A summary of these results and those for Ex. 3.3 is given in the following table; A summary of these results and those for Ex. 3.3 is given in the following table; En la siguiente tabla se proporciona un resumen de estos resultados y los correspondientes para values are in joules. values values are are in in joules. joules. values in values are in joules. joules. el ejemplo 3.3,are donde los valores se proporcionan en joules. 03-SmithVanNess.indd 83 8/1/07 12:54:02 84 CAPÍTULO 3. Propiedades volumétricas de fluidos puros Mecánicamente reversible, ejemplo 3.3 ∆U ∆H a) 998 1 663 b) –998 c) Suma Irreversible, ejemplo 3.4 W ∆U ∆H Q W 0 998 998 1 663 –250 1 248 –1 663 –1 663 665 –998 –1 663 –1 829 831 0 0 1 495 –1 495 0 0 1 196 –1 196 0 0 –168 168 0 0 –883 883 Q Es un ciclo que requiere trabajo y produce una cantidad igual de calor. La característica más sorprendente de la comparación mostrada en la tabla es que el trabajo total requerido cuando el ciclo se compone de tres etapas irreversibles es más de cinco veces el trabajo total requerido cuando las etapas son mecánicamente reversibles, aun cuando cada etapa irreversible se supone con 80% de eficiencia. Ejemplo 3.5 Se confina una cantidad de gas nitrógeno en un cilindro vertical mediante un pistón sin fricción. La parte superior del pistón está abierta hacia la atmósfera. El peso del pistón hace que la presión del nitrógeno sea 0.35 bar mayor que la de la atmósfera de los alrededores, que es de 1 bar y 27 °C. De este modo, el nitrógeno se encuentra inicialmente a una presión de 1.35 bar y se encuentra en equilibrio mecánico y térmico con respecto a sus alrededores. El pistón es empujado dentro del cilindro, de modo que comprime el nitrógeno hasta una presión de 2.7 bar. A esta presión, se permite que el nitrógeno llegue al equilibrio térmico con la atmósfera circundante a 27 °C. En este punto, el pistón se fija en su sitio mediante seguros. Los seguros se retiran liberando al pistón de su restricción, y con el tiempo el aparato regresa al equilibrio mecánico y térmico con sus alrededores. Discuta la aplicación de la termodinámica a este proceso. Suponga que el nitrógeno en estas condiciones es un gas ideal. Solución 3.5 Cuando los seguros que sostienen el pistón sin fricción son removidos, dicho pistón se mueve rápidamente hacia arriba, y debido a su inercia al ir más allá de su posición de equilibrio. Esta expansión inicial se aproxima a un proceso adiabático reversible, ya que se produce poca turbulencia de un solo recorrido del pistón y porque la transferencia de calor es relativamente lenta. Sin embargo, la oscilación consecutiva del pistón introduce la irreversibilidad como resultado de la agitación y la turbulencia tanto del gas como de la atmósfera. Este proceso sigue durante un tiempo considerable, durante el cual se presenta transferencia de calor en una cantidad suficiente para regresar el nitrógeno a su temperatura inicial de 27 °C a una presión de 1.35 bar. No es posible especificar la trayectoria de un proceso irreversible, y esto hace imposible el cálculo tanto de Q como de W. A diferencia del calor y el trabajo, es posible calcular los cambios de la propiedad del sistema, ya que dependen únicamente del estado inicial y final, y éstos son conocidos. Tanto ∆U como ∆H para el proceso de expansión son cero, porque las temperaturas 03-SmithVanNess.indd 84 8/1/07 12:54:03 The Ideal Gas Gas 3.3. The Ideal 3.3. 3.3. El gas ideal 85 85 85 ◦ C. The inicial final son 27 °C. primera aplica tanto para procesos reversiarey zero, because theLa initial and ley final temperatures are 27 first are zero, because the initial andsefinal temperatures are 27◦ C.irreversibles The law firstapplies lawcomo applies bles, to y nos lleva a que: irreversible as well as to as reversible processes, and itand becomes: to irreversible as well to reversible processes, it becomes: == Q +WW de donde == –W−W �U DU = +Q Q =QQ −W �UQ +==W00 = 0 whence whence Aunque no es posible calcular Qcan ni W, sus absolutos sonvalues los values mismos. Eltheresultado Although neither Q norQ W their absolute are the Although neither nor W be cancalculated, be valores calculated, their absolute aresame. same. del proceso es la elevación del pistón y la atmósfera, así como una disminución en la compensación The process results in elevation of theofpiston and the and aand compenThe process results in elevation the piston andatmosphere, the atmosphere, a compenen la sating energía interna dethe lainatmósfera circundante. decrease in internal energy of theofsurrounding atmosphere. sating decrease the internal energy the surrounding atmosphere. Ejemplo 3.6 Example 3.63.6 Example En una que contiene una parcialmente cerrada, circulavalve. aire con una rapidez Air tubería flows athorizontal a at steady rate through a válvula horizontal pipe pipe to a to partly closed The Air flows a steady rate through a horizontal a partly closed valve. The uniforme. La tubería que deja la válvula es lo suficientemente larga con respecto a la tubería de entrapipe pipe leaving the valve is enough larger thanthan the entrance pipe pipe that that the kinetic-energy leaving the valve is enough larger the entrance the kinetic-energy da, de modo que el cambio de energía cinética del aire a medida que fluye a través de la válvula change of the flows through the valve is negligible. The The valvevalve and and connecting change ofair theas airit as it flows through the valve is negligible. connecting es despreciable. Tanto la válvula como las tuberías de conexión se encuentran bien aisladas. ◦ CLas pipespipes are well insulated. The The conditions of the fromfrom the valve are 20 are well insulated. conditions of air theupstream air upstream the valve are 20◦ Ccondiciones del aire corriente arriba de la válvula son de 20 °C y 6 bar, y la presión corriente abajo es de 3 and and 6 bar, and and the downstream pressure is 3 bar. If airIfisair regarded as an gas, gas, 6 bar, the downstream pressure is 3 bar. is regarded asideal an ideal bar. what Si el aire se considera como un gas ideal, ¿cuál es la temperatura del aire a cierta distancia cois theistemperature of the distance downstream fromfrom the valve? what the temperature ofair thesome air some distance downstream the valve? rriente abajo de la válvula? Solución 3.6 3.6 3.6 Solution Solution El flujo a través de una válvula parcialmente cerrada se conoce como un proceso de estrangulaFlowFlow through a partly closed valvevalve is known as a throttling process. The system is is through a partly closed is known as a throttling process. The system ción. El sistema se aísla, lo cual hace que se ignore a Q; además, son despreciables los cambios insulated, making Q negligible; moreover, the potential-energy and kinetic-energy insulated, making Q negligible; moreover, the potential-energy and kinetic-energy de energías potencial y cinética. Ya que no hay trabajo de flecha, Ws = 0. Por tanto, la ecuación changes are negligible. Because no shaft workwork is accomplished, Ws =W0. Hence, changes are negligible. Because no shaft is accomplished, 0. Hence, s = (2.32) se reduce a ∆H = 0. Así, para un gas ideal, Eq. (2.32) reduces to: �H = 0. = Thus, for anfor ideal gas, gas, Eq. (2.32) reduces to: �H 0. Thus, an ideal � T2 � T2 de donde �H �H = = C P dT C P=dT0 = 0 whence whence T2 =TT21= T1 T1 T1 The result that �H =0= is general for a for throttling process, because the assumpThe result that �H 0 is general a throttling process, because the assumpEl resultado de que ∆H = 0 es general para un proceso de estrangulación, ya que son válidas las tionstions of negligible heat transfer and potentialand kinetic-energy changes are usuof negligible heat transfer and potentialand kinetic-energy changes are ususuposiciones de que la transferencia de calor y los cambios en las energías potencial y cinética son ally valid. If theIffluid is an is ideal gas, no temperature change occurs. The throttling ally valid. the fluid an ideal gas, no temperature change occurs. The throttling despreciables. Si el fluido es un gas ideal, no ocurre cambio en la temperatura. El proceso de esprocess is inherently irreversible, but this immaterial to thetocalculation, because process is inherently irreversible, butisthis is immaterial the calculation, because trangulación es inherentemente irreversible, pero esto no tiene importancia en el cálculo, ya que Eq. (3.21b) is valid for anfor ideal gas whatever the process. Eq. (3.21b) is valid an ideal gas whatever the process. la ecuación (3.21b) es válida para un gas ideal sin importar el proceso. Example Ejemplo 3.7 3.73.7 Example −1 −1 inejemplo Ex. the flow rate rate of isair 1 mol and if, both upstream and un downstream If in 3.6 Ex.3.6 3.6la the flow ofair the is es 1 smol s s–1and if both upstream and downstream Si enIfel rapidez dethe flujo del aire 1 mol y si la tubería tiene diámetro interno de pipes have an inner diameter of 5 cm, what is the kinetic-energy change of the air pipes have an inner diameter of 5 cm, what is the kinetic-energy change of theand air and del 5 cm, tanto en la entrada como en la salida de la válvula, ¿cuál es el cambio en−1la energía cinética −1 . what is its temperature change? For air, C = (7/2)R and M = 29 g mol . what is its temperature change? For air, C = (7/2)R and M = 29 g mol –1 P aire y cuál es su cambio en la temperatura? ParaP el aire, C P = (7/2)R y M = 29 g mol . 03-SmithVanNess.indd 85 8/1/07 12:54:36 86 86 86 86 86 86 86 86 86 CHAPTER CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Fluids CHAPTER 3. Volumetric Properties of Pure CHAPTER 3. Volumetric Properties of Pure Pure Fluids Fluids CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3.3.Volumetric Properties of Pure CHAPTER 3. Volumetric Properties of Pure Fluids Solución 3.7 Solution 3.7 Solution 3.7 Solution 3.7 Solution 3.7 Solution Solution 3.7 3.7 3.7 By Eq. (2.24b), Por laSolution ecuación (2.24b), By Eq. Eq. (2.24b), (2.24b), Solution 3.7 By .. . nV nn.. . nV .. . = uu = nV n . . = = . nV n nV n = nV u = Aρ A n. = Aρ A = By Eq. (2.24b), u = . By Eq. (2.24b), u = Aρ A = nV By Eq. (2.24b), u = Aρ n . . Aρ A A �� π ��u = Aρ A n = 2nV By Eq. (2.24b), π 2 π D 22 = � π �u(5=×Aρ −2 −3 = By Eq. (2.24b), A 2 −2 −3 m where A = ) = 10 π π � � � � 2 2 where A = = ) = 1.964 1.964 × × 10 10−3 m22 D (5 × 10 donde where −2 � � Aρ A π π A = π4π = ) = 1.964 × 10 m D (5 × 10 4 2 2 −2 −3 π D22 = 4 � (5 where = = � 4π = 1.964 1.964 × × 10 10−3 m222 (5 × × 10 10−2 −2))22 = −3 m where AA 44 D where A= =π 44 D2 = �π 44 � (5 × 10−2 )2 = 1.964 × 10−3 m2 4π 4π where A = volume × 10 −3is: m2 D 2 = as (5 ×by 10the The upstream molar equation 2 = 1.964 −2))ideal-gas The upstream molar volume as4 given given by the ideal-gas equation is: 1.964 × del 10 gas mideal es: where A = 4arriba, D =que ×by 10the El volumen molar corriente se(5 conoce porideal-gas la=ecuación The upstream molar volume as given equation is: 4 4 The upstream molar volume as given by the ideal-gas equation is: The upstream molar volume as given by the ideal-gas equation is: The upstream molar volume as given by the ideal-gas equation is: RT (83.14)(293.15) 11 The upstream molar volume as given by the ideal-gas equation 3 RT (83.14)(293.15) −6 −3 −1 −6 −3is: −1 = × m × 10 V RT (83.14)(293.15) 1 = The upstream molar as given by ideal-gas is: = RT = volume = 4.062 4.062equation × 10 10−3 m33 mol mol−1 × the 10−6 V11 = RT (83.14)(293.15) (83.14)(293.15) 1 = = = 4.062 × 10 m mol × 10 V P 6 1 3 1 −6 −3 −1 1 33 mol−1 RT (83.14)(293.15) −6 −3 P 6 1 1 = = = 4.062 × 10 m × 10 V −6 −3 −1 = 4.062 × 10 m mol × 10 VV111 = P1 = 6 = RT = = 4.062 × 10 m mol × 10 (83.14)(293.15) P 6 1 P 6 11 3 −6 −3 −1 1 P 6 RT (83.14)(293.15) × −3 10 −6 = 4.062 × 10 −3 m 3 mol −1 V1 = 1 = −3 (1)(4.062 × 10 m mol × −3 10)) = 4.062 ×−1 V1 = P1 = 6 (1)(4.062 × 10 10 −1 Then, 2.069 m ss−1 )= (1)(4.062 × 10 P1 uu11 = 6 −3 −3 Then, = = 2.069 m −3 ) (1)(4.062 × 10 −3 ) (1)(4.062 × 10 Then, u = = 2.069 m s −3 1.964 10 −1 (1)(4.062 1.964 × ×× 1010 −3−3 ) = En tal caso, Then, = = 2.069 2.069 m m ss−1 −1 Then, uuu1111 = 1.964 × 10 Then, = (1)(4.062 −3 −3 ) = 2.069 m s−1 ×10 10 1.964 × 10 −3 −3 1.964 × 1.964 × 10 ) (1)(4.062 × 10 Then, u = = 2.069 m s 1 If the is changed from upstream Then, u 1 = 1.964 = 2.069 m s−1 If the downstream downstream temperature temperature is little little changed from the the upstream temperature, temperature, × 10−3 If the downstream temperature is little changed from the upstream temperature, −3 1.964 × 10 then to a good approximation: If the downstream temperature is little changed from the upstream temperature, If the downstream temperature is little changed from the upstream Si la temperatura corriente abajo cambia un poco de la correspondiente a la temperature, corriente arriba, por then to a good approximation: If the downstream temperature is little changed from the upstream temperature, then to a good approximation: then to a good approximation: to a good approximation: If the downstream temperature is little changed from the upstream temperature, lo tantothen para una buena aproximación: then a good approximation: If thetodownstream temperatureand is little changed from 4.138 the upstream temperature, −1 −1 uu22 = then to a goodV 22 = Vapproximation: = 2V 2V11 and = 2u 2u11 = = 4.138 4.138 m m sss−1 = 2V and u2 = 2u = m then to a goodV approximation: 2 1 1 −1 −1 = 2V and = 2u = 4.138 m VV y uuu222 = V222 = = 2V 2V111 and and = 2u 2u111 = = 4.138 4.138 m m sss−1 The rate of change in kinetic energy is therefore: V = 2V and u = 2u = 4.138 m s−1 1 The rate rate of of change change in2Vkinetic kinetic energy is therefore: therefore: The energy V22 =in and is u22 = 2u11 = 4.138 m s−1 1 The rate of changedein in kinetic energy is therefore: Debido The aThe esto, laof rapidez de la energía cinética es: rate change kinetic energy is rate incambio kinetic energy is therefore: therefore: ..of change .. 11 22 11 22 �( u ) = n M �( u ) The ratem of change in kinetic energy is therefore: . . m.of �( M �( �(energy 21 u 2 ) = 21 u 2 ) .. �( nn...M The ratem change in kinetic 1221 u22) = 1221 u22) is therefore: m �( = M �( 2 2 m m. �( �(2212uuu 2))) = = nnn. M M �( �(2212uuu 2))) −3 (4.138 (4.13822 − − 2.069 2.06922))) = 0.186 J s−1 1×210 −3) (4.138 −1 (1 × 29 − 2.069 2 2 = 0.186 J s−1 m. �( 121u 22) = n M �( u ) 2 2 . = (1 (1 × × 29 2921× × 210 10−3 ))(4.138 − 2.069 (4.138 = = 0.186 J s−1 22 2.069 m �( 2 u ) = = (1 nM �( u 10 ) −3 −3 −1 − 2.0692))) = (4.1382 − 2× = (1 × 29 × 10 ) = 0.186 J s −3 −1 × 29 ) 0.186 J s 2 2 2 = (1 × 29 × 10 ) (4.138 2 − 22 2.069 2) = 0.186 J s−1 2 −3 − 2.069 ) (4.138 = (1 × 29 × 10 ) = 0.186 J s In work, (2.31), becomes: −3 ) the In the the absence absence of of heat heat transfer and work, the energy energy balance,=Eq. Eq. (2.31), becomes: = transfer (1 × 29and × 10 0.186 J s−1 2 balance, In the absence of heat transfer and work, the energy balance, Eq. (2.31), becomes: 2 In the absence of heat transfer and work, the energy balance, Eq. (2.31), becomes: In the absence of heat transfer and work, the energy balance, Eq. (2.31), becomes: In thedeabsence of heatde transfer work, the Eq. (2.31), becomes: .. .. elenergy .. balance, En ausencia transferencia calor y11and de balance de11energía, ecuación (2.31), será: 22 trabajo, 22 + u ) m = m �H + m �( u ) = 0 In the absence of heat�(H transfer and work, the energy balance, Eq. (2.31), becomes: . . . 2 2 �(H + u ) m = m �H + m �( u ) = 0 1 1 + 122211and u222)m + m...balance, �( 122211u222) Eq. = 0(2.31), becomes: ... = m ... �H In the absence of heat�(H transfer work, the energy �(H m= = m �H + m �( = �(H ... �H .. 222uuu1 )))22= ..+ .. C �(H + + 222uuu11 )))m =m m �H + +m m. �( �( = 000 22m C PP �T .)) = 1 212u 1 2112u nC �T + m �( )) = + m �( m C . . .+ .. C P P �T 2 2 + m �( u nC �T + m �( u = m �(H u ) m = m �H + m �( u ) = 0 000 . . . P C . . . 2 2 ) = nC + m )= �T�(H + m..+�( m.. C M .m .. �( 21 u 21 u1221 u)22= P �T 12211u MPPP �T . �H )222m + m �( 0 00 2 1 u ) = nC �T + m �( u ) = �T + m �( m u ) = nC �T + m �( u ) = + m �( m P 2 2 M �T + m �(22 u ) = nC m. C M . .PP �T + m. �(2122 u2 ) = 0 M MPP �T + m.. �( 121u 22) = nC C �T + m �( u ) = m 1 . . . 2 . P �T 21 u 2 ) = 00 2 = −0.186 Whence (1)(7/2)(8.314)�T m =− nC �T + m �(22 u ) = mM = �( −0.186 Whence (1)(7/2)(8.314)�T = − m... P�( �( 2112 uuu+ 2))) m 2 = −0.186 Whence (1)(7/2)(8.314)�T = − m �( 1 2 M . �(121uu22)) = −0.186 Whence (1)(7/2)(8.314)�T = − m Whence (1)(7/2)(8.314)�T = − 22 u ) = = −0.186 −0.186 Whence (1)(7/2)(8.314)�T = −m m. �( �(2K and �T = −0.0064 2 and �T = ==−0.0064 −0.0064 K Whence (1)(7/2)(8.314)�T −m. �( 12K and �T 1u 2) = −0.186 De donde Whence (1)(7/2)(8.314)�T −m �(K and �T = −0.0064 K and �T −0.0064 2 u ) = −0.186 and �T = == −0.0064 K andClearly, �T = −0.0064 K temperature change Clearly, the the assumption assumption of of negligible negligible temperature change across across the the valve valve is is andClearly, �T = −0.0064 K the assumption of negligible temperature change across the valve is justified. Even for an upstream pressure of 10 bar and a downstream pressure Clearly, the assumption of negligible temperature change across the valve Clearly, the assumption of negligible temperature change across the valve is justified. Even for an upstream pressure of 10 bar and a downstream pressure Clearly, the for assumption of negligible temperature change across thepressure valve is is justified. Even an upstream pressure of 10 and a downstream y DT = the –0.0064 K bar of 1 bar and for the same flow rate, temperature change is only −0.076 K. justified. Even for an upstream pressure of 10 bar and a downstream pressure justified. Even for an upstream pressure of 10 bar and a downstream pressure Clearly, the assumption of negligible across the valve is of 1 bar and for the same flow rate, the temperature change is only −0.076 K. justified. Even for an upstream pressure of 10 bar and a downstream pressure of 1 bar and for the same flow rate, the temperature change is only −0.076 K. Clearly, the assumption of negligible temperature change across the valve is We that, except for very unusual conditions, �H = is of bar and for the same flow rate, the temperature change is only −0.076 K. of 111conclude bar for the same flow rate, the temperature change is −0.076 K. justified. Even an upstream pressure of 10 bar and a downstream pressure We conclude that, except for very unusual conditions, �H =despreciable is aaa satisfactory satisfactory of bar and and forfor the same flow rate, the temperature change is000 only only −0.076 K. de la We conclude that, except for very unusual conditions, �H = is satisfactory justified. Even for an upstream pressure of 10 bar and a downstream pressure Es evidente, se justifica la suposición del cambio de temperatura a través energy for aexcept throttling process. We conclude that, for very unusual conditions, �H = is satisfactory We except very unusual conditions, �H = 00 only is of 1conclude barbalance and that, for samefor flow rate, the temperature change −0.076 K. energy balance forthe throttling process. We that, for very unusual conditions, �H =is is aaa satisfactory satisfactory balance for aaexcept throttling process. of 1conclude bar and the same flow rate, the is0corriente only −0.076 válvula.energy Aun para unafor presión corriente arriba detemperature 10 bar y unachange presión abajoK.de 1 bar energy balance for a throttling process. energy balance for a throttling process. We conclude that, except for very unusual conditions, �H = 0 is a satisfactory energy balancethat, for a throttling process. We conclude very unusual conditions,es�H = 0 –0.076 is a satisfactory para la misma relación de except flujo, elfor cambio en la temperatura de sólo K. Concluimos energy balance for a throttling process. energy balance for a throttling process. que, excepto para muy pocas condiciones inusitadas, ∆H = 0 es un balance de energía satisfactorio para un proceso de estrangulación. 03-SmithVanNess.indd 86 8/1/07 12:54:59 3.4. Aplicación de las ecuaciones viriales 3.4. of Equations 3.4. Application Application of the the Virial Virial Equations 3.4. Application of the Virial Equations 87 87 87 87 3.4 APLICACIÓN DE LAS VIRIALES 3.4 APPLICATION OFECUACIONES THE VIRIAL EQUATIONS 3.4 3.4 APPLICATION APPLICATION OF OF THE THE VIRIAL VIRIAL EQUATIONS EQUATIONS Las dos formas de la expansión virial conocidas en las ecuaciones (3.11) y (3.12) son series infinitas. Para The The two two forms forms of of the the virial virial expansion expansion given given by by Eqs. Eqs. (3.11) (3.11) and and (3.12) (3.12) are are infinite infinite series. series. For For propósitos su of usothe es virial práctico sólo cuando convergencia rápida, esto es,series. cuandoFor dos o tres Thetécnicos, two forms expansion givenlaby Eqs. (3.11) es andmuy (3.12) are infinite engineering engineering purposes purposes their their use use isis practical practical only only where where convergence convergence isis very very rapid, rapid, that that is, is, where where términos son suficientes para una aproximación razonable a los valores de las series. Esto ocurre para gases y engineering purposes their use is practical only where convergence is very rapid, that is, where two two or or three three terms terms suffice suffice for for reasonably reasonably close close approximations approximations to to the the values values of of the the series. series. This This vaporesistwo de presiones bajas a moderadas. or three terms suffice for reasonably close approximations to the values of the series. This is realized realized for for gases gases and and vapors vapors at at low low to to moderate moderate pressures. pressures. is realized for gases and vapors at low to moderate pressures. 1.00 1.00 1.00 0.75 Z� � PV/RT PV/RT Z Z� PV/RT Z PV/RT 1.00 0.75 0.75 0.75 150 (F) 0.50 0.25 0.50 0.50 0.50 �150 �150 �150 (�F) (�F) (�F) 0 ( F) 400(F) 440000((��FF)) 250(F) 00(�F) 2254 500((��FF)) 250(�F) 100(F) 110000((��FF)) 100(�F) F) 0 ( F)) 00((��FF) 0 (� ) (�FF) 5500((��F) � � 50 � ) )) ( F 0 � ((�FF ) 0 10 0 F 0 � 0 1 ( ��1 00 1 � 5 0 0.25 0.2501 000 2 000 3 000 1000 2000 0.25 0 1000 2000 0 P(psia) 1000 2000 P(psia) P(psia) P(psia) Figura 3.9: Gráfica del factor de compresibilidad Figure 3.9: Figure 3.9: Compressibility-factor Compressibility-factor para el metano. Figure 3.9: Compressibility-factor graph graph for for methane. methane. graph for methane. 3000 3000 3000 La figura 3.9 muestra una gráfica del factor de compresibilidad para el metano. Los valores del factor Figure Figure 3.9 3.9 shows shows aa compressibility-factor compressibility-factor graph graph for for methane. methane. Values Values of of the the compressibilcompressibilFigure 3.9 shows a compressibility-factor graph for methane. Values of the compressibilde compresibilidad Z (como se calculan a partir de la información PVT del metano mediante la ecuación que ity ity factor factor ZZ (as (as calculated calculated from from PPVVTT data data for for methane methane by by the the defining defining equation equation ZZ = = PPVV/RT /RT)) ity factor Z (as calculated from P V T data for methane by the defining equation Z = P V /RT ) la define Z = PV/RT) están graficados en función de la presión para varias temperaturas constantes. Las isoare are plotted plotted vs. vs. pressure pressure for for various various constant constant temperatures. temperatures. The The resulting resulting isotherms isotherms show show graphgraphare plotted vs. pressure various constant temperatures. The resulting show graphtermas ically resultantes muestran defor manera gráfica lo que se intenta representar en isotherms formaisotherms analítica con la expanically what what the the virial virial expansion expansion in in PP isis intended intended to to represent represent analytically. analytically. All All isotherms origiorigiically what the virial expansion in P is intended to represent analytically. All isotherms origisión virial en P. Todas las isotermas se originan en el valor Z = 1 para P = 0, y a bajas presiones las isotermas nate nate at at the the value value ZZ = = 11 for for PP = = 0, 0, and and the the isotherms isotherms are are nearly nearly straight straight lines lines at at low low pressures. pressures. nate at the value Z = 1 for P = 0, and the isotherms are nearly straight lines at low pressures. son líneas casi rectas. Así, la tangente a una isoterma en P = 0 es una buena aproximación de la isoterma Thus Thus the the tangent tangent to to an an isotherm isotherm at at PP = = 00 isis aa good good approximation approximation of of the the isotherm isotherm from from PP → → 00 Thus the tangent to an isotherm at P = 0 is a good approximation of the isotherm from P → 0 desde Pto → 0 para alguna presión finita. Al derivar la ecuación (3.11) para cierta temperatura se obtiene: to some some finite finite pressure. pressure. Differentiation Differentiation of of Eq. Eq. (3.11) (3.11) for for aa given given temperature temperature gives: gives: to some finite pressure. Differentiation of Eq. (3.11) for a given temperature gives: � � � � �∂∂ZZ � � � � 2 = ∂Z = BB � + + 2C 2C �PP + + 3D 3D �PP 2 + + ······ ∂∂PP TT = B � + 2C � P + 3D � P 2 + · · · ∂P T � � � � �∂∂ZZ � � = from which, ∂ Z = BB �� from which, ∂∂PP TT;P=0 = B a partir from de la which, cual, ∂ P T ;P=0 ;P=00 � equation the ZZ = 11también + result also truncating Thus the the de equation of the tangent tangent line = + BB ��P, P,esaa un result also given given by truncating Así, la Thus ecuación la rectaof tangente es: Z =line 1 +isis B ′P, que resultado que seby obtiene al truncar la Thus the equation of the tangent line is Z = 1 + B P, a result also given by truncating Eq. (3.11) to two terms. Eq. (3.11) to two terms. ecuación (3.11) a dos términos. Eq. (3.11) to two terms.form of this equation results from substitution for B �� by Eq. (3.13a): A A more more common of thisesta equation results from substitution by Eq. (3.13a): Una forma máscommon común deform expresar ecuación es sustituyendo B ′ porfor la B ecuación A more common form of this equation results from substitution for B � by Eq.(3.13a): (3.13a): PPVV BBPP ZZ = (3.38) = PV = (3.38) (3.38) = 11 + + BP Z = RT (3.38) RT = 1 + RT RT RT RT This aa direct ZZ and often This equation equation expresses direct proportionality proportionality between and P, P, and andseisisaplica often applied applied toa tempeEsta ecuación expresaexpresses una proporcionalidad directa entre between Z y P, y con vaporesto This equation expresses a direct proportionality between Z frecuencia and P, At andhigher is oftena applied to at temperatures up pressures. vapors at subcritical subcritical temperatures up to to their theirAsaturation saturation pressures. At higher temperatures temperatures raturas vapors subcríticas hasta sus presiones de saturación. temperaturas altas se proporciona una aproximación vapors at subcritical temperatures up to their saturation pressures. At higher temperatures 03-SmithVanNess.indd 87 8/1/07 12:55:09 CHAPTER 3.3.Volumetric Properties of Pure CAPÍTULO Propiedades volumétricas deFluids fluidos puros 88 88 it provides a reasonable for gases up to a pressure of several with the que razonable para los gases hasta unaapproximation presión de varios bar, incrementando el intervalo de bars, presión a medida range increasing as the temperature increases. aumentapressure la temperatura. Equation (3.12) as well may betruncarse truncatedatodos twotérminos terms for application at lowa bajas pressures: La ecuación (3.12) también es posible para su aplicación presiones: PV B =1+ (3.39) (3.38) RT V De cualquier modo, ecuación (3.38) es más conveniente y suand aplicación es tan exacta la (3.39). De However, Eq.la(3.38) is more convenient in application is normally at least as como accurate as este modo, cuando Thus la ecuación virial se trunca a dos términos to setwo prefiere la Eq. ecuación Eq. (3.39). when the virial equation is truncated terms, (3.38)(3.38). is preferred. El segundo virial B depende desubstance la sustancia y de una and función de la temperatura. Los valores The coeficiente second virial coefficient B is dependent a function of temperature. 5 Además, es posible experimentales están disponibles para varios calcularestimation el segundoof coeficiente Experimental values are available forgases. a number of gases.5 Moreover, second virial cuando virial no hay información disponible, como analiza en la sección 3.6. coefficients is possible where no se data are available, as discussed in Sec. 3.6. Para presiones que están arriba del intervalo de aplicabilidad de labut ecuación peropresabajo de la For pressures above the range of applicability of Eq. (3.38) below (3.38), the critical presiónsure, crítica, ecuación virialtruncated truncadatoa three tres términos con provides frecuencia proporciona excelentes resultados. thelavirial equation terms often excellent results. In this case En este Eq. caso, en la the ecuación (3.12)inla1/V expansión en 1/V estomuy que en la ecuación Así, cuan(3.12), expansion , is far superior Eq. superior (3.11). Thus when the virial(3.11). equation do la ecuación virialtosethree trunca a tres la forma es: is truncated terms, thetérminos, appropriate form apropiada is: Z= Z= PV B C =1+ + 2 RT V V2 (3.40) (3.40) 0 0 100 �200 200 0 C B B B 4,000 4 000 2,000 2 000 0 0 �2,000 2 000 �100 �300 C C �2 C/cm C/cm66mol mol�2 100 B/cm3 mol1 Figura 3.10: Coeficientes Figure 3.10: Virialviriales B y C de la progresión densidad para coefficientsenBlaand C el nitrógeno. for nitrogen. �1 B/cm B/cm33mol mol�1 100 300 �4,000 4 000 0 100 100200 T/K 200300 T/K 300400 C/cm6 mol2 This equation be solved directly for butpero is cubic in volume. SolutionLa forsolución V is para Esta ecuación se puedecan resolver directamente parapressure, la presión, es cúbica en el volumen. readily accomplished byun an esquema iterative scheme, in Ex.se3.8. V se obtiene fácilmente usando iterativo as deillustrated cálculo, como ilustra en el ejemplo 3.8. 400 Values of C, like those of B, depend on the gas and on temperature. However, much less is known virial coefficients than aboutdel second data forse sabe Los valoresabout de C,third al igual que los de B, dependen gas yvirial de lacoefficients, temperatura.though Sin embargo, number of gases are found in the literature. virial beyond the third are mucho amenos sobre los terceros coeficientes virialesBecause que acerca de coefficients los segundos coeficientes viriales, aun cuando rarely es posible encontrar en la literatura información que rara vez se conocen known and because the virial alguna expansion with morepara thanvarios threegases. terms Ya becomes unwieldy, los coeficientes más allá de los terceros, y ya que la expansión virial es muy difícil de manejar debido its use isviriales uncommon. a que tiene más de tres términos, su uso no es muy común. 55 J. H. Dymond and E. B. Smith, The Virial Coefficients of Pure Gases and Mixtures, Clarendon Press, Oxford, 1980. J. H. Dymond y E. B. Smith, The Virial Coefficients of Pure Gases and Mixtures, Clarendon Press, Oxford, 1980. 03-SmithVanNess.indd 88 8/1/07 12:55:13 3.4. Application of the Virial Equations 3.4. Application of the Virial Equations 3.4. Aplicación de las ecuaciones viriales 89 89 89 Figure 3.10 illustrates the effect of temperature on the virial coefficients B and C for La figura 3.10 3.10 ilustra el efecto the de laeffect temperatura sobre losoncoeficientes viriales B y BC and para el for nitrógeno; Figure temperature virialthe coefficients nitrogen; althoughillustrates numerical values areofdifferent for otherthe gases, trends are similar.C The aunque nitrogen; los valores numéricos son diferentes para otros gases, las tendencias son similares. La curva although numericalthat values are different for other gases, trends are similar. Thede la ficurve of Fig. 3.10 suggests B increases monotonically with Tthe ; however, at temperatures gura 3.10 sugiere que B aumenta en forma monotónica con T; no obstante, a temperaturas mucho mayores que curve Fig. than 3.10 shown suggests B increases monotonically with Tdecreases. ; however,The at temperatures much of higher B that reaches a maximum and then slowly temperature las mostradas B alcanza un máximo y en seguida disminuye lentamente. La dependencia de la temperatura de much higherofthan B reaches a maximum and then slowly decreases. The temperature dependence C isshown more difficult to establish experimentally, but its main features are clear: C C es más difícil de establecer de manera experimental, pero sus características principales son claras: C es dependence C istemperatures, more difficultpasses to establish experimentally, its main features C is negative atoflow through a maximum atbut a temperature nearare theclear: critical, negativais a bajas temperaturas, pasa por un máximo a una temperatura cercana a la crítica, y después disminunegative at low temperatures, passes through aT maximum at a temperature near the critical, and thereafter decreases slowly with increasing . ye de forma lenta conforme T aumenta. and thereafter decreases slowly withby increasing T .known as extended virial equations, is illusA class of equations inspired Eq. (3.12), Hay unaA de ecuacionesinspired inspiradas en la (3.12), que se como ecuaciones 6 class of equations by Eq. (3.12), known asconoce extended virial equations,viriales is illus-extenditrated byclase the Benedict/Webb/Rubin equation: 6 6 das. Éstas se ilustran mediante la ecuación de Benedict/Webb/Rubin: trated by the Benedict/Webb/Rubin equation: RT b RT − a B0 RT − A0 − C0 /T 2 P = RT + B00 RT − A002 − C00/T 22 + b RT 3− a V V V P= + + V V 22 � V 33 � γ −γ aα c + aα6 + 3c 2 � 1 + γ 2 � exp −γ2 + V 66 + V 33 T 22 1 + V 22 exp V 22 V V T V V where A0 , B0 , C0 , a, b, c, α, and γ are all constant for a given fluid. This equation and its A000,,a,Bb, C0a a,γ b, c,constantes α, and γ are all forpetroleum a given Esta fluid. This equation and its 00, c, 0, y donde Awhere son paraare unconstant fluido y susindustries modificaciones, modifications, despite their complexity, used indeterminado. the andecuación natural-gas 0, B0, C modifications, despite their complexity, are used in the petroleum and natural-gas industries a pesar de complejidad, se utilizan en las industrias del petróleo y delgases. gas natural para hidrocarburos ligeros forsu light hydrocarbons and a few other commonly encountered forotros light gases hydrocarbons and aencontrados. few other commonly encountered gases. y algunos comúnmente Example 3.8 Example 3.8for the virial coefficients of isopropanol vapor at 200◦ C are: Ejemplo 3.8values Reported Reported values for the virial coefficients of isopropanol vapor at 200◦◦C are: Los valores reportadosBpara los coeficientes de isopropanol a 200 °C son: C vapor = −26,000 cm6 mol−2 = −388 cm3 mol−1 viriales del 66 −1 −2 −1 −2 C = −26,000 cm B = −388 cm33 mol 3 mol–1 C 6 molmol –2 B = –388 cm = –26 000 cm ◦ Calculate V and Z for isopropanol vapor at 200 C and 10 bar by: Calculate V and Z for isopropanol vapor at 200◦◦C and 10 bar by: Calcule V y(a) Z para vapor deequation; isopropanol 200 °C y 10 bar mediante: Theelideal-gas (b)a Equation (3.38); (c) Equation (3.40). (a) The ideal-gas equation; (b) Equation (3.38); (c) Equation (3.40). a) La ecuación del gas ideal; b) La ecuación (3.38); c) La ecuación (3.40). Solution 3.8 Solución 3.8 Solution 3.8 La temperatura absoluta es T = 473.15 y el valor de la constante The absolute temperature is T K, = 473.15 K, apropiado and the appropriate valuede oflos thegases gas es R = 3 bar −1 K−1 The absolute temperature T mol = 473.15 K,. and the appropriate value of the gas 83.14 cm mol . constant is –1 RK =–183.14 cm3isbar −1 K−1 −1. constant is R = 83.14 cm33 bar mol−1 a) Para (a) un gas Z =gas, 1, yZ = 1, and For ideal, an ideal (a) For an ideal gas, Z = 1, and RT (83.14)(473.15) V = RT = (83.14)(473.15) = 3,934 cm3 mol−1 −1 P 10 V = = = 3,934 cm33 mol−1 P 10 (b) Solving Eq. (3.38) for V gives: b) Resolviendo paraEq. V de la ecuación (3.38) se obtiene: (b) Solving (3.38) for V gives: RT V = RT + B = 3,934 − 388 = 3,546 cm3 mol−1 −1 V = P + B = 3,934 − 388 = 3,546 cm33 mol−1 P 6 M. Benedict, G. B. Webb, L. C. Rubin, J. Chem. Phys., vol. 8, pp. 334–345, 1940; vol. 10, pp. 747–758, 1942. 66M. Benedict, G. B. Webb, L. C. Rubin, J. Chem. Phys., vol. 8, pp. 334–345, 1940; vol. 10, pp. 747–758, 1942. M. Benedict, G. B. Webb y L. C. Rubin, J. Chem. Phys., vol. 8, pp. 334-345, 1940; vol. 10, pp. 747-758, 1942. 03-SmithVanNess.indd 89 8/1/07 12:55:20 90 CHAPTER 3. 3.3.Volumetric Properties of Pure Pure CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER CHAPTER 3. Volumetric Volumetric Properties Properties of of Pure Fluids Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids PV V V 3,546 P V 3,546 = 0.9014 Whence, Z= = PV = = V = = 3,546 0.9014 De donde, Whence, Z V V 3,546 Whence, Z = PRT = = = 0.9014 RT RTV/P /P 3,934 = 3,934 V = RT Whence, Z = PRT RT /P = 3,546 3,934 = 0.9014 Whence, Z = RT = RT /P = 3,934 = 0.9014 RT Eq. /P as:(3.40) 3,934 se escribe como: (c) To To facilitate facilitate iteration, write (3.40) c) Para facilitar el proceso de iteración, laRT ecuación (c) iteration, write (3.40) (c) To facilitate iteration, write Eq. Eq. (3.40) as: as: (c) To facilitate iteration, write Eq. (3.40) � � as: � � (c) To facilitate iteration, write Eq. (3.40) � � as: B RT C� � B RT C B RT C 1 + = + V i+1 = P �11 + V Bi + C222 � +V +V Vi+1 i+1 = RT P Bii + V Ci = RT Vi+1 P 1+ V V V Vi+1 = P 1 + Vi + Viii22 P Vi Vthe i first iteration, i = 0, and where subscript denotes the iteration iteration number. For subscript iii denotes the number. the 0, donde elwhere subíndice i denota el número de iteración. ParaFor la primera iteración, = 0, where subscript denotes the iteration number. For the first first iteration, iteration, iii = = 0, yand and where subscript i denotes the iteration� number. For the first iteration, i = 0, and � � � where subscript i denotes the iteration�number. For the � first iteration, i = 0, and RT � C� B RT �11 + C� B + V11 = = RT + C + B V C22 B +V V = RT P 1+ V V P V11 = RT P 1 + VB00 + VC02 V1 = P 1 + V00 + VV00022 P V0 V0 where V V00 = = 3,934, 3,934, the the ideal-gas ideal-gas value. value. Numerically, Numerically, where where V = 3,934, the ideal-gas value. Numerically, 0 donde Vwhere = 3 934, el valor del gas ideal. En forma numérica, 0 V = 3,934, the ideal-gas Numerically, � � � value. � where V00 = 3,934, the ideal-gas � value. 26,000 � 388Numerically, 26,000 388 � � 26,000 388 − = 3,539 3,539 = 3,934 �11 − − V11 = V 26,000222 � = 388 − = 3,539 − (3,934) 3,934 1 − 3,934 V = 3,934 26,0002 = 3,539 388 − (3,934) V11 = 3,934 1 − 3,934 3,934 (3,934) V1 = 3,934 1 − 3,934 − (3,934) 2 = 3,539 3,934 The second second iteration iteration depends depends on this this result: (3,934) The The second iteration depends on on this result: result: The second iteration on result: �this � depends � � La segunda depende de este resultado: � � The iteración second iteration depends on this result: � � � � 388 RT � C� 26,000 �� B RT C 388 26,000 B � � 388 RT C 26,000 B V22 = = 3,934 �11 + + = 3,495 3,495 = + 2� = − �1 + V 388 − C22 = 3,934 26,000222 � = B + V = RT 3,934 1 + 3,539 = 3,495 +V +V − (3,539) P 11 + P 388 − (3,539) C112 = 3,934 1 + 3,539 26,0002 = 3,495 V22 = RT P 1+ V VB111 + V 3,539 (3,539) V V2 = P 1 + V1 + V112 = 3,934 1 + 3,539 − (3,539) 2 = 3,495 P V 3,539 (3,539) 1 Iteration continues until until1 theVdifference difference Vi+1 −V Vii is is insignificant, insignificant, and and leads leads after after i+1 − Iteration V Iteration continues continues until the the difference V − V i is insignificant, and leads after i+1 7 7 five iterations to the final value, Iteration continues until the difference V − V is insignificant, and leads after i+1 7 five to final value, five iterations iterations to the theuntil finalthe value, Iteration continues difference Vii isinsignificante, insignificant, yand leads de after La iteración continúa hasta diferencia Vi V+i+1 Vi sea después cinco ite7 1–− five iterations to the que finallavalue, 7 iterations the final, final value, 3 −1 racionesfive se obtiene el to valor V= = 3,488 cm cm3 mol−1 −1 V V = 3,488 3,488 cm3 mol mol−1 V = 3,488 cm33 mol–1 −1 = cmwith molthis result, the ideal-gas value is = 33,488 488 cm mol from which which Z = = 0.8866. In InVVcomparison from from which Z Z = 0.8866. 0.8866. In comparison comparison with with this this result, result, the the ideal-gas ideal-gas value value is is 13% too high and Eq. (3.38) gives a value 1.7% too high. from which Z = 0.8866. In comparison with this result, the ideal-gas value 13% too high and Eq. (3.38) gives a value 1.7% too high. a partir from del cual Z =Z0.8866. En comparación con1.7% este this resultado, del gas idealis 13% too high and (3.38) a value too high. which = Eq. 0.8866. Ingives comparison with result, el thevalor ideal-gas value is es 13% too high(3.38) and Eq. (3.38) gives value1.7% 1.7% too high. mayor y13% la ecuación proporciona unaavalor 13% too high and Eq. (3.38) gives value 1.7%mayor. too high. 90 90 90 90 90 3.5 ECUACIONES CÚBICAS DE 3.5 CUBIC CUBIC EQUATIONS OF ESTADO STATE 3.5 3.5 CUBIC EQUATIONS EQUATIONS OF OF STATE STATE 3.5 CUBIC EQUATIONS OF STATE Si una ecuación de estado representa el comportamiento 3.5 CUBIC EQUATIONS OF STATEPVT para líquidos y vapores, debe abarcar un amplio If de antemperaturas equation of of state is to to represent the PV V T ser behavior of both both liquids liquids and vapors,grandes it must must intervalo y presiones. Aun así,the no P debe tan compleja paraand presentar If is of vapors, If an an equation equation of state state is to represent represent the PVT T behavior behavior of bothcomo liquids and vapors, it it mustdificulencompass a wide range of temperatures and pressures. Yet it must not be so complex asel to to If an equation of state is to represent the P V T behavior of both liquids and vapors, it must tades numéricas o analíticas para su aplicación. Las ecuaciones polinomiales que son cúbicas en volumen encompass aa wide range it not complex as encompass wide range of temperatures and pressures. Yet it must must not be be sovapors, complex as to If an equation of state is of to temperatures represent the and P V Tpressures. behaviorYet of both liquids andso it must present excessive numerical or analytical difficulties in application. Polynomial equations that encompass a wide range of temperatures and pressures. Yet it must not be so complex as to molar ofrecen un compromiso entre la generalidad y la simplicidad adecuadas para muchos propósitos. De present or in equations present excessive excessive numerical or analytical analytical difficulties difficulties in application. application. Polynomial equationsasthat that encompass a widenumerical range of temperatures and pressures. Yet it mustPolynomial not be so complex to are cubic in molar volume offer a compromise between generality and simplicity that is suitable present excessive numerical or analytical difficulties in application. Polynomial equations that hecho, las ecuaciones cúbicas son las más sencillas, pero capaces de representar el comportamiento tanto de are in volume aa compromise between and simplicity is are cubic cubic in molar molarnumerical volume offer offer compromise between generality andPolynomial simplicity that that is suitable suitable present excessive or analytical difficulties ingenerality application. equations that to many purposes. Cubic equations are in fact the simplest equations capable of representing cubic in molar volume offer a compromise between generality and simplicity that is suitable líquido are como de vapor. to Cubic equations are the equations capable representing to many many purposes. Cubic offer equations are in in fact factbetween the simplest simplest equations capable of of representing are cubicpurposes. in molar volume a compromise generality and simplicity that is suitable both liquid and vapor vapor behavior. to many purposes. Cubic equations are in fact the simplest equations capable of representing both liquid and behavior. both liquid and vapor behavior. to many purposes. Cubic equations are in fact the simplest equations capable of representing both liquid and vapor behavior. liquid and vapor behavior. Losboth esquemas de iteración, incorporados en los paquetes de software, llevan a cabo esta iteración de manera inadvertida y rutinaria. 777Iteration Iteration schemes, schemes, incorporated in in software packages, packages, accomplish this this iteration routinely routinely and unobserved. unobserved. Iteration schemes, incorporated incorporated in software software packages, accomplish accomplish this iteration iteration routinely and and unobserved. 7 Iteration schemes, incorporated in software packages, accomplish this iteration routinely and unobserved. 7 Iteration schemes, incorporated in software packages, accomplish this iteration routinely and unobserved. 03-SmithVanNess.indd 90 8/1/07 12:55:29 91 3.5. Ecuaciones cúbicas de estado 3.5. Cubic Equations of State 91 La ecuación de estado de van der Waals The van der Waals Equation of State La primera ecuación de estado cúbica que se considera práctica fue propuesta por J.8 D. van der Waals en 1873: The first practical cubic equation of state was proposed by J. D. van der Waals in 1873: P= RT a − 2 V −b V (3.41) (3.41) En este Here, caso, aa yand b son constantes positivas; when cuando sonare cero, se the recupera la ecuación gas ideal. b are positive constants; they zero, ideal-gas equationdel is recovered. Conocidos losvalues valoresofde a y bb para fluido particular, es posible calcular como una Given a and for a un particular fluid, one can calculate P as aPfunction of función V for de V para valores diferentes deTT.. La figura 3.11isesa un diagrama PVdiagram esquemático, quethree muestra isotermas. Está various values of Figure 3.11 schematic PV showing suchtres isotherms. sobrepuesto el “domo”isque los estados states de líquido y de vapor saturados. Para lavapor. isoterma Superimposed therepresenta “dome” representing of saturated liquid and saturated ForT1 > Tc, la presión una función que decrece monótonamente conforme aumenta el volumen molar. La isoterma the es isotherm T1 > T , pressure is a monotonically decreasing function with increasing molar c crítica (marcada como Tc) contiene inflexión horizontal C, característica crítico. Para la volume. The critical isothermuna (labeled Tc ) contains the en horizontal inflection del at Cpunto characteristic isotermaofTthe Tc, la presión en forma Trápida en la región de líquido subenfriado conforme V aumenta; 2 < critical point. decrece For the isotherm 2 < Tc , the pressure decreases rapidly in the subcooleddespués,liquid cruzaregion la línea de increasing líquido saturado, a un mínimo, se eleva a un máximo a continuación with V ; afterllega crossing the saturated-liquid line, it goesythrough a mini- decrece, cruza la línea saturadoand y continúa hacia abajo dentro la región de vapor sobrecalentado. mum, risesdetovapor a maximum, then decreases, crossing the de saturated-vapor line and continuing Las isotermasinto experimentales no exhibenregion. esta transición uniforme de líquido saturado a vapor saturado; downward the superheated-vapor más bien, contienen un segmento horizontal dethis la región de transition dos fases donde el líquidoliquid y el vapor Experimental isotherms do notdentro exhibit smooth from saturated to saturados coexisten diferentes presión de saturación o dethevapor. Este comportamiento, saturateden vapor; rather,proporciones they containaalahorizontal segment within two-phase region where que se muestra mediante la línea discontinua de la figurain3.11, no esproportions analítico, yatesthe aceptado como conducta saturated liquid and saturated vapor coexist varying saturation or una vapor inevitable no realista las ecuaciones en laline región de dos fases. pressure. Thisdebehavior, shown de by estado the dashed in Fig. 3.11, is nonanalytic, and we accept Enasrealidad, comportamiento PV predicho en esta región, mediante ecuaciones inevitableelthe unrealistic behavior of equations of state in the two-phase region. de estado cúbicas apropiadas, noActually, es del todo ficticio. Cuando la presión disminuye en un líquido saturado sin que hayaissitios de the P V behavior predicted in this region by proper cubic equations of state formación de núcleos de vapor en un experimento controlado de manera cuidadosa, la vaporización no ocurre not wholly fictitious. If pressure is decreased on a saturated liquid devoid of vapor-nucleation y la fasesites líquida persiste sólo a presiones que razonablemente están por debajo de la presión de vapor. De in a carefully controlled experiment, vaporization does not occur, and liquid persists alone manera to similar, el aumento en la presión en un vapor saturado en un experimento apropiado no ocasiona pressures well below its vapor pressure. Similarly, raising the pressure on a saturated vapor condensación y el vapor persistedoes a presiones que, de manera razonable, poralone arribatode la presión de vapor. in a suitable experiment not cause condensation, and vapor están persists pressures well Estos estados de no equilibrio o metaestables para el líquido sobrecalentado para el vapor subenfriado se above the vapor pressure. These nonequilibrium or metastable states ofysuperheated liquid and aproximan por aquellas de la isoterma queportions se encuentran región de dos fases subcooled vapor partes are approximated by PV those of the PenVlaisotherm which lie inadyacentes the two- a los estados phase de líquido y de vapor to saturados. region adjacent the states of saturated liquid and saturated vapor. Las ecuaciones cúbicas de estado tienen raícesroots, para el las be cuales es posible Cubic equations of state have threetres volume of volumen, which twodemay complex. Phys-que dos sean complejas. Los valores de V físicamente significativos siempre son reales, positivos y mayores ically meaningful values of V are always real, positive, and greater than constant b. For an que la constante b. Para at una a T > Tc,tolaFig. figura 3.11 muestra la solución para de cualquier isotherm T isoterma > Tc , reference 3.11 shows that que solution for V at anyV positive valuevalor of positivo de P P da sólo una de estas raíces. Para la isoterma crítica (T = T ), esto también es cierto, excepto c yields only one such root. For the critical isotherm (T = Tc ), this is also true, except at a la presión the crítica donde existenwhere tres raíces a Vroots, lasequal isotermas T < isotherms Tc, la ecuación c. Paraall at T puede < Tc , exhibir critical pressure, thereiguales are three to Vc .a For una o tres raíces reales, dependiendo de la presión. Aunque estas raíces son reales y positivas, no hay estados the equation may exhibit one or three real roots, depending on the pressure. Although these físicos estables para la parte de una isoterma que está entre líquido y vapor saturados (debajo del “domo”). roots are real and positive, they are not physically stable states for the portion of an isotherm saturado(líquido) y V saturado(vapor), son estados estables, conectaSólo laslying raícesbetween para P =saturated P saturadoliquid , es decir, V and saturated vapor (under the “dome”). Only for the vapor or dos porsaturation la parte horizontal desatlaare isoterma real. Para otras indican mediante las líneas ho­ pressure P the roots, V sat (liq) and presiones V sat (vap),(que stablesestates, lying at the ends of saturado), la raíz más pequeña es un volumen líquido o rizontales de la figura 3.11 arriba y abajo de P the horizontal portion of the true isotherm. For other pressures (as indicated by the horizontal “semejante un líquido”, y la3.11 másabove grande es below un volumen o “semejante vapor”. La tercera raíz, que linesa shown on Fig. and P sat ),de thevapor smallest root is a al liquid or “liquid-like” está entre los otros valores, no es importante. volume, and the largest is a vapor or “vapor-like” volume. The third root, lying between the other values, is of no significance. Johannes Diderik van der Waals (1837-1923). Físico holandés ganador del premio Nobel de Física en 1910. 8 Johannes Diderik van der Waals (1837–1923), Dutch physicist who won the 1910 Nobel Prize for physics. 03-SmithVanNess.indd 91 8/1/07 12:55:31 92 CAPÍTULO 3. Propiedades volumétricas de fluidos puros CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids CHAPTER 92 92 C C C Figura 3.11: Isotermas proporcionadas por ecuaciónas Figure 3.11: 3.11:una Isotherms Figure Isotherms as cúbica degiven estado. by a cubic equation of of given by a cubic equation state. state. T1 Tc P T �T T11 � Tcc PP Psaturado Tc T Tcc sat PPsat T2 Tc T �T T22 � Tcc Vsat urado(líquido) sat (liq) VVsat (liq) Vsat urado(vapor) sat (vap) VVsat (vap) V VV Una ecuación estado cúbica of genérica A Generic GenericdeCubic Cubic Equation State A Equation of State Desde laSince introducción de la ecuación de van Waals se han propuesto varias cubic ecuaciones de estado cúbicas. the introduction introduction of the the van van derder Waals equation, several hundred hundred equations of state state Since the of der Waals equation, several cubic equations of Todas son casos la ecuación: have beenespeciales proposed99de All are special special cases cases of of the the equation: equation: have been proposed .. All are (V − − η) η) RT θθ(V − P= = RT − P 2 + κ V + λ) V − b (V − b)(V 2 V − b (V − b)(V + κ V + λ) Here, b, b, θθ,, κ, κ, λ, λ, and and ηη are are parameters parameters which which in in general general depend depend on on temperature temperature and and (for (for mixmixHere, Aquí, b,tures) q, k, composition. l y h son parámetros que enequation general dependen de la temperatura y de la composición (para las Although this appears to possess great flexibility, it has inherent tures) composition. Although this equation appears to possess great flexibility, it has inherent 10 mezclas). Aunque esta ecuación parece tener gran flexibilidad, posee limitaciones inherentes debido limitations because because of of its its cubic cubic form. form.10 It It reduces reduces to to the the van van der der Waals Waals equation equation when when ηη = = b, b, a su limitations 10 Ésta se reduce forma cúbica. a la ecuación de van der Waals cuando h = b, q = a y k = l = 0. = a, a, and and κκ = = λλ = = 0. 0. θθ = Una clase importante de ecuaciones cúbicas results resultafrom de lathe ecuación anterior haciendo lasasdesignaAn important class of cubic cubic equations equations preceding equation with the the An important class of results from the preceding equation with asciones: signments: signments: = bb ηη = = a(T a(T )) θθ = = (� (� + + σσ )b )b κκ = = �σ �σ bb22 λλ = It is is thus thus transformed transformed into into an an expression expression general general enough enough to to serve serve as as aa generic generic cubic cubic equation equation of of It Para una revisión, véase J. O. Valderrama, Ind. Eng. Chem. Res., vol. 42, pp. 1603-1618, 2003. M. M. 9Abbott, AIChe J., vol. 19, pp. 596-601, 1973; Adv. in Chem. Series 182, K. C. Chao y R. L. Robinson, Jr., eds., pp. 47-70, For aWashington, a review, review, see see D. J. O. O. Valderrama, Ind. Eng. Eng. Chem. Chem. Res., Res., vol. vol. 42, 42, pp. pp. 1603–1618, 1603–1618, 2003. 2003. 9 For J. Ind. Am. Chem. Soc., C.,Valderrama, 1979. 10 10 M. M. Abbott, AIChE J., vol. 19, pp. 596–601, 1973; Adv. in Chem. Series 182, K. C. Chao and R. L. Robinson, 10 M. M. Abbott, AIChE J., vol. 19, pp. 596–601, 1973; Adv. in Chem. Series 182, K. C. Chao and R. L. Robinson, Jr., eds., eds., pp. pp. 47–70, 47–70, Am. Am. Chem. Chem. Soc., Soc., Washington, Washington, D.C., D.C., 1979. 1979. Jr., 03-SmithVanNess.indd 92 8/1/07 12:55:36 3.5. Equations of 3.5. Ecuaciones cúbicas de estado 3.5. Cubic Equations of State 3.5. Cubic Cubic Equations ofState State 3.5. 3.5. Cubic Cubic Equations Equations of of State State 93 93 93 93 93 93 Así, ésta se transforma en una general bastante útil assignment como una ecuación de estado cúbica genérica, state, which to all here of parameters: state, which reduces to all others of interest here upon assignment of appropriate parameters: state, whichreduces reduces toexpresión allothers othersof ofinterest interest hereupon upon assignment ofappropriate appropriate parameters: state, which reduces to all others of interest here upon assignment of appropriate parameters: state, which reduces to all ecuaciones others of interest here pero uponbajo assignment of appropriate parameters: la cual se reduce a todas las otras de interés, la designación de parámetros apropiados: RT a(T RT a(T a(T))) RT − a(T P (3.42) PP = = (3.42) − RT a(T )) + σ b) −(V + �b)(V = V RT (3.42) P = (3.42) − − b VV − − (V + �b)(V + b) P=V (3.42) (3.42) −bbb − (V (V + +�b)(V �b)(V + +σσσb) b) V − b (V + �b)(V + σ b) For For given equation, and are pure numbers, the same for all substances, whereas paramForaaaagiven givenequation, equation,����and andσσσ are arepure purenumbers, numbers,the thesame samefor forall allsubstances, substances,whereas whereasparamparamFor given equation, are pure numbers, the same for all substances, whereas paramPara una ecuación conocida, and y s σσson números puros, iguales para todas las of sustancias, mientras que los For a given equation, � and are pure numbers, the same for all substances, whereas parameters a(T ) and b are substance dependent. The temperature dependence a(T eters a(T and are substance dependent. The temperature dependence of a(T isisspecific specific to etersa(T a(T)))and andbbbare aresubstance substancedependent. dependent. The Thetemperature temperaturedependence dependenceof ofa(T a(T))))is specificto to eters is specific to parámetros a(T) y b dependen de la sustancia. La dependencia de la temperatura de a(T) se especifica para eters a(T ) and b are substance dependent. The temperature dependence of a(T ) is specific to each each equation of state. For the van der Waals equation, a(T = isisaaaasubstance-dependent substance-dependent eachequation equationof ofstate. state. For Forthe thevan vander derWaals Waalsequation, equation,a(T a(T)))) = = aaaa is substance-dependent each equation of state. For the van der Waals equation, a(T = is substance-dependent cada ecuación de estado. Para la ecuación de van der Waals, a(T) = a, es una constante que depende del tipo each equation of state. For the van der Waals equation, a(T ) = a is a substance-dependent constant, constant, and = = 0. constant,and and���= =σσσ = =0. 0. constant, 0. de sustancia, y and = s ��= = 0. σσ = constant, and = = 0. Determination Determination of Equation-of-State Parameters Determinationof ofEquation-of-State Equation-of-StateParameters Parameters Determination of Equation-of-State Parameters Determinación de los de la ecuación de estado Determination ofparámetros Equation-of-State Parameters The The constants in an equation of state for particular substance may be evaluated by fit to Theconstants constantsin inan anequation equationof ofstate statefor foraaaparticular particularsubstance substancemay maybe beevaluated evaluatedby byaaaafit fitto to The constants in an equation of state particular substance may be evaluated by fit to Las constantes enPuna ecuación de estado para for unaaasustancia particular es posible evaluarlas mediante The constants in an equation of state for particular substance may be evaluated by a fit toun ajusavailable V T data. For cubic equations of state, however, suitable estimates are usually available PPV VVT TT data. data. For cubic equations of state, however, suitable estimates are usually available P data. For For cubic cubic equations equations of of state, state, however, however, suitable suitable estimates estimates are are usually usually available te de lafound información disponible. Noequations obstante, of para lasc .however, ecuacionesthe de estado cúbicas, lasusually estimaciones available PV TPVT data. Forcritical cubic state, suitable estimates are found from values for the constants TTccccand and PPccc...Because Because the critical isotherm exhibits foundfrom fromvalues valuesfor forthe thecritical criticalconstants constantsTT andP Becausethe thecritical criticalisotherm isothermexhibits exhibitsaaaa found from values for the critical constants and P Because critical isotherm exhibits c c adecuadas usualmente se encuentran a partir de los valores para las constantes críticas T y P . Ya que la found frominflection values foratthe critical constants Tmay andimpose Pc . Because the critical isotherm a isoterc cexhibits cmay horizontal the horizontal the critical point, we impose the mathematical conditions: horizontalinflection inflectionat thecritical criticalpoint, point,we wemay impose themathematical mathematicalconditions: conditions: horizontal inflection atatthe the critical point, we may impose the mathematical conditions: ma crítica exhibe una inflexión horizontal en el punto crítico, podemos imponer las condiciones matemáticas: horizontal inflection at the critical point, we may impose the mathematical conditions: � �� 2 � �� � � � ��∂ P � �� � � � � ∂∂∂∂2222P ∂ P P � ∂∂PP� P P 2 = (3.43) = (3.44) = (3.43) = (3.44) P P2 =0000 (3.43) =0000 (3.44) = (3.43) = (3.44) ∂∂∂∂∂V ∂∂∂∂∂V VV TTTT;cr =0 (3.43) (3.44) VV22222 T ;cr = 0 V V ;cr ;cr ;cr T ;cr T ;cr ∂ V TT ;cr ∂ V TT ;cr ;cr ;cr T ;cr where the “cr” denotes point. of (3.42) donde el subíndice “cr” denota punto the crítico. Derivando la ecuación (3.42) produce expresiones para amwhere the subscript “cr” denotes the critical point. Differentiation of Eq. (3.42) yields expreswhere thesubscript subscript “cr”el denotes thecritical critical point.Differentiation Differentiation ofEq. Eq. (3.42)yields yieldsexpresexpreswhere the subscript “cr” denotes the critical point. Differentiation of Eq. (3.42) yields expreswhere the subscript “cr” denotes the critical point. Differentiation of Eq. (3.42) yields expres, T = T , and V = V sions for both derivatives, which may be equated to zero for P = P c c c bas derivadas, las cuales se pueden igualar a cero para P = P , T = T y V = V . La ecuación de estado , T = T , and V = V sions for both derivatives, which may be equated to zero for P = P sions for both derivatives, which may be equated to zero for P = P , T = T , and V = V c cP = Pccc,c T = Tccc, and V = Vccc.... puede sions for both derivatives, which may be equated to zero for = Tcthree , andequations V, V=, VTcc,. a(T ) y sions forlas both derivatives, which may be equated tocritical zero for P =cinco Pcc , T The equation of state may itself be written for the conditions. These escribirse para condiciones críticas. Estas tres ecuaciones contienen constantes: The equation of state may itself be written for the critical conditions. These The equation of state may itself be written for the critical conditions. Thesecthree threePequations equations c c c c The equation of state may itself be written for the critical conditions. These three equations The equation of state may itself be written for the critical conditions. These three equations five ), several ways to these ccc,,,,V b. Entrecontain las diferentes manerasP tratar más adecuada eliminación de Vc para obcontain five constants: PPde V TTcccc,,estas ,,a(T a(T ),),and and b. Of the several ways to treat these equations, contain fiveconstants: constants: Vcccc,,,,TT a(Tcccecuaciones, andb. b. Of Oflathe the several wayses tolatreat treat theseequations, equations, c contain five constants: P V a(T ), and b. Of the several ways to treat these equations, c cc , a(Tcc ), and b. Of the several ways to treat these equations, contain five constants: Pcc ,a(T Vcc ,) Tyof to relating . the is tener expresiones que relacionen bV aV yyield Tyield razón es que Pc y Tca(T soncccc)))conocidas con másTT to yield expressions relating a(T and to PPccc and and TTcprecisión the most suitable isiselimination elimination of VVcP to expressions relating a(T )and andbbbbto to P and themost mostsuitable suitable elimination of c of c. Laexpressions cc cto cc... yield expressions relating a(T to P and the most suitable is elimination cc ) and yieldaccurately expressions relating a(T and b to Pccc and Tccc . the most suitable is elimination of Vccc tomore reason is that P and T are usually known than V . c c c que Vc.The The reason is that P and T are usually more accurately known than V . The reason is that P and T are usually more accurately known than V . The reason is that Pcccc and Tcccc are usually more accurately known than Vcccc. The reason is thatequivalente Pc but and Tc are usually more accurately known than Vc .for An straightforward, is illustrated the der Un procedimiento pero más directo seprocedure ilustra para ecuación de der Puesto que An equivalent, but more straightforward, procedure isisla illustrated for the van der Waals Anequivalent, equivalent, butmore more straightforward, procedure illustrated forvan thevan vanWaals. derWaals Waals An equivalent, but more straightforward, procedure is illustrated for the van der Waals An equivalent, but more straightforward, procedure is illustrated for the van der Waals Because V = V for each of the three roots at the critical point, c V = Vc equation. para cada una de las tres raíces en el punto crítico, equation. Because V = V for each of the three roots at the critical point, equation. Because V = V for each of the three roots at the critical point, equation. Because V = Vcccc for each of the three roots at the critical point, equation. Because V = Vc for each of the three roots at the critical point, 3 (V (V − VVccc))))3333= = (V − −V =0000 (V − V = (V − Vccc )3 = 0 3 2 2 − V 333= 0 or V (A) or VV3333− − 3V VV2222+ + 3V VV − − VVc33 = = (A) or −3V 3VcccV +3V 3Vc2222V −V =000 (A) (A) o or V − 3V V + 3V V (A) or V 3 − 3Vccc V 2 + 3Vcccc2c V − Vcccc3c = 0 (A) Equation (3.41), written for (T = T , P = P ) and expanded in polynomial form, becomes: Equation (3.41), written for (T TTccc=c,,,P PP)= = P and expanded in form, becomes: Equation (3.41), written for=(T (T = = =P Pcccc)))and andexpanded expanded in polynomial polynomial form, becomes: La ecuación (3.41), escrita para for (T , PT desarrollada en forma polinomial, es: becomes: Equation (3.41), written P in form, c =y � � Equation (3.41), written for (TTc= = P in polynomial polynomial form, becomes: ��cc ) and expanded ��Tcc , PRT � � RT ab � RTcccc� V 2222+ aaaa V − ab ab = 0 3 RT ab V (B) cc V VV33333− VV22 + − + + VV − − = (B) − bbbb+ + RT + Pa V − ab =000 (B) (B) V − + = (B) P P c c PPccc V + P PPcccV − P PPcccc = 0 V − b+ P (B) c c c Pc Pc Pc where parameters aaaaand dependent, but independent of where parameters and are substance dependent, but independent of temperature. where parameters andbdependientes aresubstance substance dependent, but independent oftemperature. temperature. where parameters and bbbare are substance dependent, but independent of temperature. donde los parámetros a y b son de la sustancia, pero independientes de la temperatura. whereTerm-by-term parameters a and b are substance dependent, but independent of temperature. comparison of Eqs. (A) and (B) provides three equations: Term-by-term comparison of Eqs. (A) and (B) provides three equations: Term-by-term comparison of Eqs. (A) and (B) provides three equations: Term-by-term comparison of Eqs. (A) and (B) provides three equations: Al comparar término a término las ecuaciones (A) y (B) se obtienen tres ecuaciones: Term-by-term comparison of Eqs. (A) and (B) provides three equations: RT ab RT ab RTcccc (C) 3V 222= aaaa ab 3 RT ab 3V (D) V (E) cc 3V = + (C) 3V = (D) V = (E) cc222c = cc3333c3= a ab 3Vcccc= =bbbb+ + RT (C) 3V = (D) V = (E) 3V = + (C) 3V (D) V = (E) P P P c c c c c c c PPccc PPccc PPcccc 3Vc = b + P (C) 3Vc = P (D) Vc = P (E) Pcc Pcc Pcc Solving Eq. for a, Eq. for Solving Eq. (D) for a, combining the result with Eq. (E), and solving for gives: Solving Eq.(D) (D) forpara a,combining combining the resultwith with Eq. (E),and andsolving solving forbbbgives: gives: Al resolver la ecuación (d) a, combinarthe el result resultado con la(E), ecuación (e) y resolver para b se encuentra que: Solving Solving Eq. Eq. (D) (D) for for a, a, combining combining the the result result with with Eq. Eq. (E), (E), and and solving solving for for bb gives: gives: 111 2 bbbb= aaaa= VVccc2222c2 = VVccc = 3P = 311 V =3P 3PccccV V = V = 3P c b = 333Vccc a = 3Pc Vcc 3 03-SmithVanNess.indd 93 8/1/07 12:56:00 94 CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids CHAPTER 94 CHAPTER 3. Volumetric Properties of Pure Fluids 94 CHAPTER 3. Volumetric Properties of Pure Fluids 94 94 CAPÍTULO 3. Propiedades volumétricas de fluidos puros Substitution for b in Eq. (C) allows solution for V Vc , which which can can then then be be eliminated eliminated from from the the Substitution for for bb in in Eq. Eq. (C) (C) allows allows solution solution for for Substitution Vcc,, which can then be eliminated from the equations for a and b: equations forfor a and and b:Eq. (C) allows solution for Vc, which can then be eliminated from the Substitution b inb: equations for Sustituyendo a b en laa ecuación (c) se puede encontrar a V2c, la que después se elimina de las ecuaciones para 27 R 1 RT RTcc R22 TTc222 RTcc and b:33 RT 27 a y b: equations for a V = 3 RTc a= = 27 R Tcc b= = 11 RT c= a b V c Pcc 64 2Pcc 2 Pccc a = 64 b = 818 RT Vc = 838 RT P P T 27 RP a = 64 Pc c b = 8 Pc c Vc = 8 Pc c Although these these equations equations may not not yield yield the the best best possible results, they they provide provide reasonable valval8 Pcmay 64 possible Pc 8 Pc reasonable Although results, Although these equations may be notdetermined, yield the best possible results, they provide reasonable values which can almost always because critical temperatures and pressures (inrazonaues which can almost almost always be determined, because critical temperatures and pressures (in Aunqueues conwhich estasthese ecuaciones no se obtienen los the mejores resultados posibles, proporcionan valores Although equations may notdetermined, yield best possible results, theysíprovide reasonable valcan always be because critical temperatures and pressures (in contrast to extensive P V T data) are often known, or can be reliably estimated. contrast to extensive extensive PV V Tdeterminar, data) are often known, or can can be reliably reliably estimated. bles quecontrast casiwhich siempre pueden ya que lasbecause temperaturas ytemperatures presiones críticas (en contraste ues canse almost always beare determined, critical and pressures (in con la to often known, or be estimated. Substitution forPV VccTin indata) the equation equation for theconocidas critical compressibility factor reduces it imimSubstitution for the for the critical compressibility factor reduces it gran cantidad de información PVT) con frecuencia son o se calculan de manera fiable. contrast to extensive P V T data) are often known, or can be reliably estimated. Substitution Vc in the equation for the critical compressibility factor reduces it immediately to:de V for mediately to: La sustitución para el factor de critical compresibilidad crítico la reduce inmediatamente a: c en Substitution forlaVcecuación in the equation for Pthe compressibility factor reduces it immediately to: Vcc V PccV 33 Z ≡ = P 3 mediately to: c c c = Zc ≡ ≡ RT 8 Z c = 8 c RT P c Vccc RT 83 ≡ = Z c A single value for Z , applicable alike to all substances, results whenever whenever the the parameters parameters of of aa A single single value value for for Z Z cc,, applicable applicable alike alike to to all all substances, substances, results 8 results c manera whenever the parameters a que se ResultaA un solo valor para Zcc, el of cual es apropiado deRT igual para todas las sustancias, cadaofvez two-parameter equation state are found by imposition of the critical constraints. Different two-parameter equation of state state are are found bysubstances, impositionresults of the the critical critical constraints. constraints. Different A single value for Z , applicable alike to all whenever the parameters of two-parameter equation of found by imposition of Different encuentran los parámetros de una ecuación de estado de dos parámetros mediante la imposición de las c values are are found found for for different different equations equations of of state, state, as as indicated indicated in in Table Table 3.1, 3.1, p. p. 98. 98. Unfortunately, Unfortunately,a restricvalues two-parameter equation of state are found by imposition of the critical constraints. Different values are found for different equations of state, as indicated in Table 3.1, p. 98. Unfortunately, ciones críticas. Se encuentran valores diferentes para las distintas ecuaciones de estado, como se indica en la the values values so so obtained obtained do do not not in in general general agree agree with with those those calculated calculated from from experimental experimental values the values values are found for different equations of state, as indicated in Table 3.1, p. 98. Unfortunately, the values so obtained do not in general agree with those calculated from experimental values tabla 3.1. Por desgracia, los valores así obtenidos por lo general no están de acuerdo con los calculados a of TTcc,, P Pcc,, and and V Vcc;; each each chemical chemical species species in in fact fact has has its its own own value value of of ZZcc.. Moreover, Moreover, the the values values of values the values so obtained do not in general agree with those calculated from experimental T , P , and V ; each chemical species in fact has its own value of Z . Moreover, the values partir deof los valores experimentales de T , P y V ; de hecho, cada especie química tiene su propio valor de Z c c c c c various c c substances are almost all smaller than any of the c. given in in Table Table B.1 B.1 of of App. App. B B for for given various substances are almost allZsmaller smaller than any any of the the of T , P , and V ; each chemical species in fact has its own value of . Moreover, the values given in Table B.1 of App. B for various substances are almost all than of c c c c Además, casi todos los valores proporcionados en la tabla B.1 del apéndice B para diferentes sustancias son equation values values given given in in Table Table 3.1. 3.1. equation given An inque Table B.1 of App. for various substances are almost all smaller any equation values given in Table 3.1. más pequeños cualquiera de los B valores obtenidos a partircubic, de las ecuaciones en lathan tabla 3.1.of the analogous procedure applied to the the generic Eq. (3.42), yields expressions for An values analogous procedure applied to generic cubic, Eq. Eq. (3.42), yields expressions for equation given in Table 3.1. An analogous procedure applied to the generic cubic, (3.42), yields expressions for Un procedimiento análogo que se puede aplicar a la cúbica genérica, ecuación (3.42), produce expreparameters a(T ) and b. For the former, c parameters a(Tc)) and andprocedure b. For For the theapplied former,to the generic cubic, Eq. (3.42), yields expressions for Anparámetros analogous parameters a(T b. former, siones para los a(T ) y b. Para la anterior, c c R222TTc222 parameters a(Tc ) and b. For the former, R =� �R a(Tcc)) = Tcc a(T P 2T a(Tc ) = � RP c2 Pcc c ) = � a(T c This result result is extended extended to temperatures temperatures other than the thePde critical by introduction introduction ofintroducción a dimensionless dimensionless Este resultado se puede extender a temperaturas diferentes mediante laof de una funThis is to other than critical by c la crítica This resultα(T is extended to temperatures other than the critical by introduction of aa dimensionless function ) that becomes unity at the critical temperature. Thus ción adimensional a(T ), que es igualunity a uno en lacritical temperatura crítica. Thus Así rr ) rthat function α(T becomes at the temperature. This result isrextended to temperatures other thantemperature. the critical byThus introduction of a dimensionless function α(T ) that becomes unity at the critical function α(Tr ) that becomes unity at the critical temperature. Thus )R222TTc222 α(Trr )R α(T a(T )) = =� � α(T (3.45) (3.45) r )R Tcc a(T (3.45) Pcc 2 2 a(T ) = � α(TrP (3.45) )R T Pc c a(T ) = � (3.45) P Function α(T ) is an empirical expression, specific tocuna a particular particular equation of state state (Table (Table 3.1).3.1). El La función a(Tr)α(T es una empírica, específica para ecuaciónequation de estado particular (tabla rr ) isexpresión Function an empirical expression, specific to a of 3.1). Function α(T )given is an empirical expression, specific to a particular equation of state (Table 3.1). r Parameter b is by: parámetro b se conoce por: by: Parameter isr )given given Function α(T is an by: empirical expression, specific to a particular equation of state (Table 3.1). Parameter bb is RTcc RT Parameter b is given by: b= =� � RT (3.46) b (3.46) Pccc b = � RT (3.46) (3.46) P c b = � Pc (3.46) Pc In these these equations equations � � and and � � are are pure pure numbers, numbers, independent independent of substance substance and and determined determined for for aa In of In these equations �ofand � from are pure numbers, independent of determined for a particular equation state the values assigned to �� and and σsubstance .sustancia,and En estasparticular ecuaciones W y Ψofson números puros e independientes de la que se determinan para una equation state from the values assigned to σ . In these equations � and � are pure numbers, independent of substance and determined for a particular equation of state from the values assigned to � and σ . The modern modern development oflos cubic equations of state state was initiated in in 1949 1949 by by publication publication ecuación de estado particular a partir deof valores asignados a was y s.initiated The development cubic equations of particular equation of state from the values assigned to � and σ . The modern development of cubic equations of state was initiated in 1949 by publication 11 11 ofdesarrollo the Redlich/Kwong Redlich/Kwong (RK) equation:11 Elof moderno de las equation: ecuaciones de estado cúbicas se inició en 1949 con la publicación de la the (RK) The modern development of cubic equations of state was initiated in 1949 by publication of the Redlich/Kwong (RK) equation: 11 ecuación Redlich/Kwong (RK): 11 RT a(T )) of the Redlich/Kwong (RK) equation: RT a(T P= = RT − a(T (3.47) ) P − (3.47) VRT − b V (V +) b) b) P = V − (3.47) − b V (V + a(T (V + b) P = V − b − V−1/2 (3.47) (3.47) −1/2 V − b V (V .+ ) = T . b) where a(T ) is given by Eq. (3.45) with α(T r ) = Trr−1/2 where a(T ) is given by Eq. (3.45) with α(T r . where a(T ) is given by Eq. (3.45) with α(Tr ) = Tr −1/2 –1/2 . ) = T where a(T ) is given by Eq. (3.45) with α(T r r donde a(T) . 11 se conoce por la ecuación (3.45) con a(T ) = T r pp. 233–244, r Otto Redlich Redlich and and J. J. N. N. S. S. Kwong, Kwong, Chem. Chem. Rev., Rev., vol. vol. 44, 44, 1949. 11 Otto pp. 233–244, 1949. 11 Otto Redlich and J. N. S. Kwong, Chem. Rev., vol. 44, pp. 233–244, 1949. 11 Otto Redlich and J. N. S. Kwong, Chem. Rev., vol. 44, pp. 233–244, 1949. 11 Otto Redlich and J. N. S. Kwong, Chem. Rev., vol. 44, pp. 233–244, 1949. Otto Redlich y J. N. S. Wong, Chem. Rev., vol. 44, pp. 233-244, 1949. 03-SmithVanNess.indd 94 8/1/07 12:56:17 3.5. Ecuaciones cúbicas de estado 3.5. 3.5. Cubic Cubic Equations Equations ofofState of State 3.5. Cubic Equations State 3.5. Cubic Equations of State 959595 95 95 Teorema de estados correspondientes; factor acéntrico Theorem Theorem ofofof of Corresponding Corresponding States; States; Acentric Acentric Factor Factor Theorem Corresponding States; Acentric Factor Theorem Corresponding States; Acentric Factor Las observaciones experimentales muestran que los factores de Z compresibilidad Zfluids para diferentes fluidos Experimental Experimental observation observation shows shows that that compressibility compressibility factors factors Z for different for different fluids exhibit exhibit simisimiExperimental observation shows that compressibility factors Zfor different fluids exhibit simiExperimental observation shows that compressibility factors Zfor different fluids exhibit simiexhiben una conducta similar cuando se correlacionan con una función de temperatura reducida T y una r larlarlar behavior lar behavior when when correlated correlated asasaas as afunction function ofofreduced of reduced temperature temperature TrTrand reduced reduced pressure pressure PrP; P r and behavior when correlated afunction reduced temperature reduced pressure ; r; behavior when correlated afunction of reduced temperature TTand r and reduced pressure rPr ; presión reducida P ; por definición, r bybyby definition, by definition, definition, definition, TT TT PP PP and yand PrPr≡ TrTr≡ ≡ P≡ ≡ r≡ r≡ and TTr≡ and P r TcTcTTc PcPcPPc c c These dimensionless dimensionless thermodynamic thermodynamic coordinates coordinates provide provide the the basis basis for the for the simplest simplest form form Estas These coordenadas termodinámicas adimensionales proporcionan las bases para lasimplest forma más simple These dimensionless thermodynamic coordinates provide the basis for the simplest form These dimensionless thermodynamic coordinates provide the basis for the form ofofthe of the theorem theorem of corresponding of corresponding states: states: del teorema de estados correspondientes: ofthe thetheorem theoremofofcorresponding correspondingstates: states: All fluids, fluids, when when compared compared atatthe at the same same reduced reduced temperature temperature and and rereAll fluids, when compared the same reduced temperature and reTodosAll los fluidos, cuando se les compara con la misma temperatura y presión reducidas, All fluids, when compared at the same reduced temperature and reduced duced pressure, pressure, have have approximately approximately the the same same compressibility compressibility factor, factor, duced have approximately the factor, tienen aproximadamente mismo factor de compresibilidad y todos se desvían del comducedpressure, pressure,el have approximately thesame samecompressibility compressibility factor, and and all deviate all deviate from from ideal-gas ideal-gas behavior behavior to about to about the the same same degree. degree. and all deviate from ideal-gas behavior to about the same degree. portamiento del gas ideal casi al mismo grado. and all deviate from ideal-gas behavior to about the same degree. Corresponding-states Corresponding-states correlations correlations ofofZof of based ononon this on this theorem theorem are are called called two-parameter two-parameter correcorreCorresponding-states correlations Zbased based this theorem are called two-parameter correCorresponding-states correlations ZZ this theorem are called two-parameter correLas correlaciones de estados correspondientes debased Ztwo están basadas en este teorema yPAlthough se. conocen como lations, lations, because because they they require require use use of the of the two reducing reducing parameters parameters T T and and P . Although these thesecorrec c c c lations, because they require use of the two reducing parameters T and P . Although these cTc and cPc . Although these lations, because they require use of the two reducing parameters laciones de dos parámetros, porque requieren del uso de los dos parámetros reducidos T , y P . Aunque c c correlations correlations are are very very nearly nearly exact exact for the for the simple simple fluids fluids (argon, (argon, krypton, krypton, and and xenon) xenon) systematic systematic estas correlations are very nearly exact for the simple fluids (argon, krypton, and xenon) systematic correlations are very nearly exact for the simple fluids (argon, krypton, and xenon) systematic correlaciones están cerca de ser exactas para fluidos simples (argón, kriptón y xenón) se observan desviaciodeviations deviations are observed are observed for more for more complex complex fluids. fluids. Appreciable Appreciable improvement improvement results results from from introintrodeviations are observed for more complex fluids. Appreciable improvement results from introdeviations are observed for more complex fluids. Appreciable improvement results from intrones sistemáticas para fluidos más complejos. Los resultados mejoran de manera apreciable a partir de la duction duction ofofaof of a third third corresponding-states corresponding-states parameter parameter (in addition (in addition totoTto to and PcP),cPP characteristic ofofof ofintrocTcand c and c),), duction athird corresponding-states parameter (in(in addition ),ccharacteristic characteristic duction athird corresponding-states parameter addition TTcand characteristic ducción de un tercer parámetro de estados correspondientes (además de T , y P ), característico c c molecular molecular structure; structure; the the most most popular popular such such parameter parameter isisthe the acentric acentric factor factor ω,ω,ω, introduced ω, introduced bybyby byde la molecular structure; the most popular such parameter the acentric factor introduced molecular structure; the most popular such parameter isis the acentric factor introduced estructura molecular; el más popular de estos parámetros es el factor acéntrico w, introducido por K. S. Pitzer 12 12 12 K.K.S. K. S. Pitzer andand and coworkers. coworkers.12 S.Pitzer Pitzer coworkers. K. S. Pitzer coworkers. 12 and y colaboradores. The The acentric acentric factor factor for for a apure a pure pure chemical chemical species species isisdefined defined with with reference reference totoits to vapor its vapor The acentric factor for chemical species defined with reference itsits vapor The acentric factor for apure chemical species isis defined with reference to vapor El factor acéntrico para una especie química pura estáofdefinido confluid respecto a su presión de vapor. pressure. pressure. Because Because the the logarithm logarithm of the of the vapor vapor pressure pressure a of pure a pure fluid is approximately is approximately linear linear pressure. Because the logarithm of the vapor pressure of a pure fluid is approximately linear pressure. Because thepresión logarithm of thedevapor pressure ofesa aproximadamente pure fluid is approximately linear con Puestoinque el logaritmo de la de vapor un fluido puro lineal en relación in the reciprocal reciprocal ofofabsolute of absolute temperature, temperature, inthe the reciprocal absolute temperature, in the of absolute temperature, el recíproco de reciprocal la temperatura absoluta, satsat d log dlog log Psaturado P sat dlog rPrrsat d dlog PrrP = S= S d(1/T d(1/T ) )==S S dd(1/T (d(1/T 1 / rTrr ))rr) satthe saturado sat where PrPsat Pisla the reduced reduced vapor vapor pressure, pressure, the reduced reduced temperature, temperature, and Sla the the slope slope ofofuna of grádondewhere Pwhere es presión de vapor reducida, TTrrTes temperatura reducida, yand Sand es pendiente de rthe the reduced vapor pressure, the reduced temperature, SisSS is the slope rwhere ris isis the vapor pressure, TTrisla isis the reduced temperature, and isis the slope of rPrrsatissat sat reduced saturado sat a plot of log of log P P vs. 1/T vs. 1/T . Note . Note that that “log” “log” denotes denotes a logarithm a logarithm to the to the base base 10. 10. fica dea log P en función de 1/T . Observe que “log” denota un logaritmo de base 10. sat r r aplot plot of log P vs. 1/T . Note that “log” denotes a logarithm to the base 10. r r rP r vs. 1/T r of log r r . Note that “log” denotes a logarithm to the base 10. a plot r IfIfthe the two-parameter two-parameter theorem theorem ofofcorresponding of corresponding states states were were generally generally valid, valid, the the slope slope Si el teorema detwo-parameter estados correspondientes con dos parámetros enwere general fuera válido, la pendiente S the two-parameter theorem corresponding states were generally valid, the slope IfIf the theorem of corresponding states generally valid, the slope would S would be the be the same same for for all pure all pure fluids. fluids. This This is observed is observed not not to be to true; be true; each each fluid fluid has has its its sería laSSmisma para todos los fluidos puros. Sin embargo, no se ha observado que esto sea cierto; cada fluido would be the same for all pure fluids. This is observed not to be true; each fluid has its S would be the same for all pure fluids. This is observed not to be true; each fluid has its own own characteristic characteristic value value ofofS, of which S, which could could ininprinciple in principle serve serve asasaas as a third third corresponding-states tiene su propio valor característico deS, S,which que en principio puede servir como elcorresponding-states tercer parámetro de estados own characteristic value S, which could principle serve athird corresponding-states own characteristic value of could in principle serve athird corresponding-states parameter. parameter. However, However, Pitzer Pitzer noted noted that that all vapor-pressure all vapor-pressure data data for for the the simple simple fluids fluids (Ar, (Ar, Kr, Kr, correspondientes. No obstante, Pitzer observó que todos los datos de presión de vapor para los fluidos simples parameter. However, Pitzer noted that all vapor-pressure data for the simple fluids (Ar, Kr, parameter. However, Pitzer noted that all vapor-pressure data for the simple fluids (Ar, Kr, satsat sat vs. saturado Xe) Xe) lie on lie the on the same same line line when when plotted plotted as log as log P P vs. 1/T 1/T and and that that the the line line passes passes through through sat (Ar, Kr, Xe) se encuentran sobre la misma línea cuando se grafica log P en función de 1/T , y que la línea r r Xe) lie on the same line when plotted as log P vs. 1/T and that the line passes through r r r r r rand that the line passes through Xe)satliesaton the same line when plotted as log rPr vs. 1/T saturado sat = −1.0 = −1.0 at T at = T 0.7. = 0.7. This This is illustrated is illustrated in Fig. in Fig. 3.12. 3.12. Data Data for for other other fluids fluids define define log log P P sat pasa por log P = –1.0 en T = 0.7. Esto se ilustra en la figura 3.12. Información para otros fluidos r r = −1.0 at T = 0.7. This is illustrated in Fig. 3.12. Data for other fluids define log P r r r log rPrr = −1.0 at rTr = 0.7. This is illustrated in Fig. 3.12. Data for other fluids definedefine other other lines lines whose whose locations locations can can bebefixed be fixed ininrelation in relation totopara the to the line line for the for the simple simple fluids fluids (SF) (SF) byla the by the otras líneas cuyas posiciones se fijan con respecto arelation la línea fluidos simples (FS) mediante diferencia: other lines whose locations can fixed the line for the simple fluids (SF) byby the other lines whose locations can be fixed in relation to the line for the simple fluids (SF) the difference: difference: difference: difference: saturado saturado sat sat sat loglog P –log loglog Psatrsat PrPsat P(SF) (SF) −− − PrP Psat sat(SF) rlog log log log (SF) − log rP r(SF) rP r r r The The acentric acentric factor factor isisdefined is defined as this difference difference evaluated evaluated at = 0.7: rTr= r0.7: The acentric defined asthis this difference evaluated atTat 0.7: El factor acéntrico sefactor define como laasdiferencia evaluada en Tr =at0.7: The acentric factor is defined as this difference evaluated TTr= = 0.7: satsat sat ) ωω≡ ≡ −1.0 −−log(P − log(P T)rT=0.7 Tr =0.7 ≡−1.0 −1.0 log(P r r )rsat ωω ≡ −1.0 − log(P )r T=0.7 =0.7 r r (3.48) (3.48)(3.48) (3.48) (3.48) Therefore can can bebe determined be determined for any for any fluid fluid from from TcTa,cTT P P ,de and a aTsingle measuremeasurePor loTherefore tanto, w seωωpuede determinar para cualquier fluido ,aPsingle yvapor-pressure devapor-pressure una sola medición de la precP, and csingle c, vapor-pressure Therefore can determined for any fluid from ,partir , cand measureTherefore ωω can be determined for any fluid from c , cPc , and a single vapor-pressure measure= 0.7. = 0.7. Values Values of ω of and ω and the the critical critical constants constants T , T P , , P and , and V for a for number a number of of se ment ment made made at T at T sión de vapor hecha a T = 0.7. Los valores para w y las constantes críticas T , P y V para varios fluidos r r c c c c c r c c c ment 0.7.Values Valuesofofωωand andthe thecritical criticalconstants constantsTcT,cP , cP,cand , andVcVcfor fora anumber numberofof mentmade madeatatTrTr==0.7. fluids fluids are are listed listed in App. in App. B. B. encuentran en el apéndice B. fluids fluidsare arelisted listedininApp. App.B.B. 12 La descripción completa seinS. encuentra en K. S. Pitzer, Thermodynamics, 3a. ed., apéndice 3, McGraw-Hill, 1212 12 Fully Fully described described ininK. K. S. Pitzer, Thermodynamics, Thermodynamics, 3d3ded., 3d App. ed., App. 3,3,McGraw-Hill, 3, McGraw-Hill, New New York, York, 1995. 1995. Nueva York. 12 Fully described K.K. S.Pitzer, Pitzer, Thermodynamics, ed., App. McGraw-Hill, New York, 1995. Fully described in S. Pitzer, Thermodynamics, 3d ed., App. 3, McGraw-Hill, New York, 1995. 03-SmithVanNess.indd 95 8/1/07 12:56:34 96 CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3.3.Volumetric Properties of Pure 96 log Prsat Figura 3.12: Dependencia Figure 3.12: Approximate aproximada con respecto a la temperature dependence of temperatura de la presión the reduced vaporde vapor reducida.pressure. �1 �2 log Prsaturado 1.0 0 1.0 1.2 0 1.2 1.4 1/Tr 1.4 1.6 1/Tr 1.6 1.8 1.8 2.0 2.0 1 Pendiente 2.3 Slope � �2.3 (Ar, Kr, Xe) (Ar, Kr, Xe) 2 1 1 Slope �Pendiente �3.2 3.2 1 1 1.43 T 1.430.7 � � (n-Octane) (n-octano) Tr 0.7 r La definición de w hace que su valor sea cero para el argón, kriptón y xenón, y la información experiThefactores definition ω makes its value krypton, and correlacionados xenon, and experimental mental produce de of compresibilidad parazero los for tresargon, fluidos que están por las mismas data yield factors for all three that es arelacorrelated by thedel same curvesteorema curvas cuando Z secompressibility representa como una función de Tr fluids y Pr. Ésta premisa básica siguiente when Z is representedcon as tres a function of Tr and Pr . This is the basic premise of the following de estados correspondientes parámetros: three-parameter theorem of corresponding states: Todos los fluidos que tienen el mismo valor w, cuando se les compara con la misma Tr y Pr Allmismo fluidsvalor having the same se value of ω ,del when compared atdethe same tienen el de Z, y todos desvían comportamiento gas idealTelr mismo and P , have about the same value of Z , and all deviate from idealr grado. gas behavior to about the same degree. VaporVapor y las & raíces de la ecuación cúbica Vapor-Like Roots ofde theestado Generic Cubicgenérica Equation of State may solve explicitly for para its three roots, the generic cubicdeequation of state, AunqueAlthough es posibleone resolver en forma explícita sus tres raíces, la ecuación estado cúbica genérica, 13 Convergence 13 Los problemas Eq. (3.42), practiceusualmente far more commonly by iterative procedures. ecuación (3.42), enislainpráctica se resuelvesolved mediante procedimientos iterativos. problems se areevitan most más likelyfácil avoided when the equation is rearranged to a forma form suited to the solude convergencia cuando la ecuación se reacomoda en una adecuada para encontrar for a particular root. largestesroot, i.e., vapor orde vapor-like volume, Eq. (3.42) una raíztion en particular. Para la raízFor másthe grande, decir, unavolumen vapor o de algo parecido, la is ecuación multiplied (V − b)/RT . It can se then be written: (3.42) se multiplicathrough por (V by – b)/RT. Ésta entonces puede escribir como: V = RT a(T ) V −b +b− P P (V + �b)(V + σ b) (3.49) (3.49) La solución para for V seVpuede encontrar con unaofrutina de un paquete software. Solution may be by trial,mediante iteration,ensayo, or withiteración the solveoroutine a software package.deAn initial estimate ideal-gas value RTRT/P. /P. For this este valuevalor is substituted onde V en Una estimación inicial for paraVVisesthe el valor del gas ideal Paraiteration, la iteración, se sustituye the rightde side of Eq. (3.49). resulting value of thelado leftizquierdo is then returned to the right al lado el lado derecho la ecuación (3.49).The El valor resultante deVV on en el se regresa después the process the change is suitably small. derechoside, y el and proceso continúacontinues hasta queuntil el cambio en Vin esVadecuadamente pequeño. Una ecuación para Z,forequivalente a lato(3.49) se obtiene a través la sustitución ZRT/P. Además, An equation Z equivalent Eq. (3.49) is obtained byde substituting V =VZ=RT /P. In la definición de dos adimensionales conduce a la simplificación. Así, addition, the cantidades definition of two dimensionless quantities leads to simplification. Thus, 13 Estos13 Such procedures are built into computerdentro software packages With these one procedimientos se encuentran integrados de los paquetesfor detechnical software calculations. de las computadoras parapackages cálculos técnicos. Con can solve routinely for V Vinmediante equationsuna such as (3.42) with little como thought to how is done. careful estos paquetes es posible calcular rutina en ecuaciones la as (3.42) conitmuy pocaHowever, labor mental de thought cómo fue hecho. should be tenerse given toespecial the question of whether the answers Sin embargo, debe cuidado con la pregunta de siare lasreasonable. respuestas en realidad son razonables. 03-SmithVanNess.indd 96 8/1/07 12:56:39 3.5. Cubic Equations of State 3.5. Cubic Cubiccúbicas Equations of State State 3.5. Equations of 3.5. Ecuaciones de estado 3.5. 3.5. Cubic Cubic Equations Equations of of State State 97 97 97 97 97 97 a(T P a(T))) bbbPP a(T ≡ ≡ (3.50) (3.51) )) bPP a(T β≡ ≡ bRT ≡ a(T (3.50) qqq ≡ (3.51) ββ (3.50) (3.51) RT ββ ≡ (3.50) (3.51) ≡ RT ≡ bbbRT (3.50) qq ≡ (3.51) RT RT RT bbRT RT RT With these substitutions Eq. (3.49) becomes: With these substitutions Eq. (3.49) becomes: becomes: With these substitutions (3.49) Mediante estas sustituciones, enEq. la ecuación (3.49) se obtiene: With With these these substitutions substitutions Eq. Eq. (3.49) (3.49) becomes: becomes: − Z− − ββ β ZZ (3.52) = + − qβ ZZ − − ββ+ σβ) (3.52) (3.52) Z= = 111 + + ββ β− − qβ qβ (Z + �β)(Z (3.52) ZZ (3.52) ZZ = (3.52) = 11 + + ββ − − qβ qβ(Z (Z + + �β)(Z �β)(Z + + σβ) σβ) (Z (Z + + �β)(Z �β)(Z + + σβ) σβ) Equations (3.50) and (3.51) in combination with Eqs. (3.45) and (3.46) yield: Con lasEquations ecuaciones (3.50) y (3.51) combinación con las ecuaciones (3.46) se obtiene: Equations (3.50) and (3.51)en in combination combination with with Eqs. (3.45) and and(3.45) (3.46)yyield: yield: (3.50) and (3.51) in Eqs. (3.45) (3.46) Equations Equations (3.50) (3.50) and and (3.51) (3.51) in in combination combination with with Eqs. Eqs. (3.45) (3.45) and and (3.46) (3.46) yield: yield: � α(T Prrr � α(T α(Trrr))) PP � = � (3.53) qq = = (3.54) � α(T P � α(Trr)) β= =� �P (3.53) = (3.54) ββ (3.53) q (3.54) r r � ββ = (3.53) (3.54) =� �TT (3.53) qq = = � (3.54) Trrr � TT Trrr TTrr � �TTrr Iterative solution of Eq. (3.52) starts with the value = on the right side. La solución iterativa de la (3.52) el valor =substituted 1 sustituido ladoside. derecho. El Iterative solution ofecuación Eq. (3.52) (3.52) startsinicia withcon the value value Z de = 1Z11 substituted substituted on en theelright right side. Iterative solution of Eq. starts with the ZZ = on the Iterative solution of Eq. (3.52) starts with the ZZ process = 11 substituted on the right side. Iterative solution of Eq. (3.52) starts with the value value =hasta substituted on the right side.final de The calculated value of Z is returned to the right side and the continues to convergence. valor calculado de Z se regresa al lado derecho y el proceso continúa la convergencia. El valor The calculated value of Z is returned to the right side and the process continues to convergence. The calculated value of Z is returned to the right side and the process continues to convergence. The of is returned to the side the process continues The calculated calculated value of ZZmediante isthe returned toZRT/P. the right right side and and theRT process continues to to convergence. convergence. final value of yields volume root through = /P. Z produce valor del value volumen V= Theelfinal final value of ZZ Z yields yields the volume volume root through through VV V= = ZZ Z RT RT/P. /P. The value of the root The The final final value value of of ZZ yields yields the the volume volume root root through through VV = = ZZRT RT/P. /P. Liquid & Liquid-Like Roots of the Generic Cubic Equation of State Liquid &raíces Liquid-Like Roots of of the the Genericcúbica Cubicgenérica Equation of of State State Liquid Liquid-Like Roots Generic Cubic Equation Líquido y las& de la ecuación de Liquid & Roots Generic Liquid & Liquid-Like Liquid-Like Roots of of the theestado Generic Cubic Cubic Equation Equation of of State State Equation (3.49) may be solved for the in the numerator of the final fraction to give: Equation (3.49) (3.49) may may be be solved solved for for the the VV V in in the the numerator numerator of of the the final final fraction fraction to to give: give: Equation Equation (3.49) may be for of the final fraction to give: Equation(3.49) (3.49)es may be solved solved for the the inenthe the numerator of the final fraction to give: � � Con la ecuación posible resolver paraVVVin elnumerator numerador de la fracción final obteniendo: �� RT + b P − V P �� � � � � RT + b P − V P RT + b P − V P = + (V + �b)(V + b) (3.55) bbPP − RT + +a(T − VV PP V= = bbb + + (V (V + + �b)(V �b)(V + + σσ σb) b) RT (3.55) VV (3.55) VV = (3.55) = bb + + (V (V + + �b)(V �b)(V + + σσb) b) (3.55) (3.55) a(T))) a(T a(T a(T)) This equation with starting value of = on the right side converges upon iteration to This equation equation with with aaa starting starting value value of of VV V = = bbb on on the the right right side side converges converges upon upon iteration iteration to to aaa This Esta ecuación con un valor inicial de value V = b of en el converge por iteración a una raíz de o This with starting = on side upon iteration to aa This equation equation with aaroot. starting value of V V lado = bb derecho, on the the right right side converges converges upon iteration to líquido liquid or liquid-like liquid or or liquid-like liquid-like root. root. liquid de algoliquid parecido. or liquid-like root. liquid or liquid-like root. An equation for equivalent to Eq. (3.55) is obtained when Eq. (3.52) is solved for the An equation equation for for ZZ Z equivalent equivalent to to Eq. Eq. (3.55) (3.55) is is obtained obtained when when Eq. Eq. (3.52) (3.52) is is solved solved for for the the An Una ecuación para Zfor quethe a lato se obtiene cuandowhen la ecuación (3.52) se resuelve para Z en An equation ZZequivale equivalent Eq. is Eq. is for Annumerator equation for equivalent to(3.55) Eq. (3.55) (3.55) is obtained obtained when Eq. (3.52) (3.52) is solved solved for the the Z in the of final fraction: in the the numerator numerator of of the the final final fraction: fraction: ZZ in � � el numerador denumerator la fracciónof ZZ in the in the the numerator offinal: the final final fraction: fraction: �1 + β − Z� � � � �11 + � + ββ − − ZZ � + σβ) (3.56) Z = β + (Z + �β)(Z 1 + β − Z 1 + β − Z + σβ) (3.56) Z = β + (Z + �β)(Z (3.56) Z = β + (Z + �β)(Z + σβ) qβ (3.56) ZZ = + σβ) σβ) (3.56) (3.56) = ββ + + (Z (Z + + �β)(Z �β)(Z + qβ qβ qβ qβ For iteration starting value of = is substituted on the right side. Once is known, the For iteration iteration aaa starting starting value value of of ZZ Z= = ββ β is is substituted substituted on on the the right right side. side. Once Once ZZ Z is is known, known, the the Para la For iteración, unis inicial de of Z en el on lado UnaOnce vez que se conoce, For aavalor starting value is the right ZZ is the For iteration iteration starting value of=ZZb= =seββsustituye is substituted substituted on thederecho. right side. side. Once isZknown, known, the la raíz volume root V = Z RT /P. volume root root is is VV = = ZZ RT RT/P. /P. volume del volumen es V = ZRT/P. volume root ZZRT /P. volume root is is VVof= =state RT /P. express Z as a function of Tr and Pr are said to be generalized, Equations which and PPrr are are said said to to be be generalized, generalized, Equations of of state state which which express express ZZ as as aa function function of of TTrr and Equations Las ecuaciones deof estado que expresan enaagases función de TTTrrryand Pr Any se que to deben ser generalizadas, PPrrdice are said Equations state express ZZZall as function of and are said to be be generalized, generalized, Equations of state which which expressato as function of because of their general applicability and liquids. equation of state can be because of of their their general general applicability applicability to to all all gases gases and and liquids. liquids. Any equation of of state state can can be be because Any equation debido because aput su campo de aplicación general para todos los gases y los líquidos. Es posible presentar cualquier of general applicability to gases liquids. Any of can because of their their general applicability to all allcorrelation gases and andfor liquids. Any equation equation of state state can be be into this form to provide a generalized the properties of fluids. This allows put into into this this form form to to provide provide aa generalized generalized correlation correlation for for the the properties properties of of fluids. fluids. This This allows allows put ecuación de estado en esta forma para proporcionar una correlación generalizada para las propiedades de los put to aa generalized correlation for of This allows put into into this this form form to provide provide generalized correlation for the the properties properties of fluids. fluids. Thissuch allows the estimation of property values from very limited information. Equations of state, as the estimation estimation of of property property values values from from very very limited limited information. information. Equations Equations of of state, state, such such as as the fluidos.the Esto permite el cálculo de valores de la propiedad a partir de información muy limitada. Las ecuacioof values limited information. Equations of such as the estimation estimation of property property values from from very very limitedwhich information. Equations of state, state, such as van der Waals and Redlich/Kwong equations, express as functions of and the van van der der Waals Waals and and Redlich/Kwong Redlich/Kwong equations, equations, which which express express ZZ Z as as functions functions of of TT Trr and and the nes de estado, la two-parameter de van Waals y la de Redlich/Kwong, expresan a Zfunctions sólo como the van der Waals and Redlich/Kwong equations, which express ZZ as of TTrrr and the vancomo der Waals andder Redlich/Kwong equations, whichque express as functions offunciones and de Tr only, yield corresponding states correlations. The Soave/Redlich/Kwong P r only, yield yield two-parameter two-parameter corresponding corresponding states states correlations. correlations. The The Soave/Redlich/Kwong Soave/Redlich/Kwong PPrr only, 14 15 yield corresponding states The Soave/Redlich/Kwong PPrr only, only, yield two-parameter two-parameter corresponding states correlations. The Soave/Redlich/Kwong y Pr, producen correlaciones de estado correspondientes decorrelations. dos15 parámetros. La ecuaciónfactor de Soave/Redlich/ 14 and 15 (SRK) equation the Peng/Robinson (PR) equation, in which the acentric enters 14 (SRK) equation and the Peng/Robinson (PR) equation, in which the acentric factor factor enters enters (SRK) equation and the Peng/Robinson (PR) equation, in which the acentric 14 15 14 and 15 in 14 15 en equation Peng/Robinson (PR) equation, which acentric factor enters (SRK) equation andr the the Peng/Robinson (PR) equation, in which the the acentric factor enters Kwong(SRK) (SRK) y la de Peng/Robinson (PR), las que el factor acéntrico se introduce a través de la funthrough function α(T ; ω) as an additional parameter, yield three-parameter correspondingthrough function function α(T α(Trr;; ω) ω) as as an an additional additional parameter, parameter, yield yield three-parameter three-parameter correspondingcorrespondingthrough through function α(T ; ω) as an additional parameter, yield three-parameter correspondingthrough function α(T ; ω) as an additional parameter, yield three-parameter correspondingción a(Tstates ; w) como un parámetro adicional, produce correlaciones de estados correspondientes con tres parár r correlations. The numerical assignments for parameters �, σ , �, and �, both for these r states correlations. correlations. The The numerical numerical assignments assignments for for parameters parameters �, �, σσ,, �, �, and and �, �, both both for for these these states correlations. The numerical assignments for �, ,, �, and �, both for states correlations. Thevan numerical assignments for parameters parameters �, σσpara �,are and �,ecuaciones both for these these metros.states Las asignaciones numéricas para los parámetros , s, W y Ψ, ambas y equations and for the der Waals and Redlich/Kwong equations, given in Table 3.1. equations and and for for the the van van der der Waals Waals and and Redlich/Kwong Redlich/Kwong equations, equations, are are given given in in Table Table 3.1. 3.1.para las equations equations and for the van der Waals and Redlich/Kwong equations, are given in 3.1. equations and for thegiven van derα(T Waals and Redlich/Kwong equations, are given in Table Table 3.1. ecuaciones de van der Waals y de Redlich/Kwong, se proporcionan en la tabla 3.1. También se dan expresioExpressions are also for ; ω) for the SRK and PR equations. r Expressions are are also also given given for for α(T α(Trr;; ω) ω) for for the the SRK SRK and and PR PR equations. equations. Expressions Expressions are also given for α(T ; ω) for the SRK and PR equations. Expressions are also given for α(T ; ω) for the SRK and PR equations. nes para a(Tr; w) para las ecuaciones SRKrr y PR. 14 Soave, Chem. Eng. Sci., vol. 27, pp. 1197–1203, 1972. 14 G. 14 G. Soave, Soave, Chem. Chem. Eng. Eng. Sci., Sci., vol. vol. 27, 27, pp. pp. 1197–1203, 1197–1203, 1972. 1972. G. 14 14G. Soave, Chem. Eng. Sci., vol. 27, pp. 1197–1203, 1972. G. Soave, Chem. Eng. Sci., vol. 27, pp. 1197–1203, 1972. 14 G. Soave, 15 Chem. Eng. Sci., vol. 27, pp. 1197-1203, 1972. Peng and D. B. Robinson, Ind. Eng. Chem. Fundam., vol. 15, pp. 59–64, 1976. 15 D.-Y. 15 D.-Y. Peng and D. B. Robinson, Ind. Eng. Chem. Fundam., vol. 15, 15, pp. pp. 59–64, 59–64, 1976. 1976. D.-Y. Peng and D. B. Robinson, Ind. Eng. Chem. Fundam., vol. 15 D. Y.15 15 Peng y D. B. and Robinson, Ind. Eng. Chem. Fundam., 15, pp.vol. 59-64, 1976. D.-Y. Peng Ind. Chem. Fundam., 15, 59–64, D.-Y. Peng and D. D. B. B. Robinson, Robinson, Ind. Eng. Eng. Chem. vol. Fundam., vol. 15, pp. pp. 59–64, 1976. 1976. 03-SmithVanNess.indd 97 8/1/07 12:56:53 98 98 98 98 98 98 CHAPTER CHAPTER 3. Volumetric 3. Volumetric Properties Properties of Pure of Pure Fluids Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CAPÍTULO 3. Propiedades volumétricas de fluidos puros Table Table 3.1:3.1: Parameter Parameter Assignments Assignments for for Equations Equations of State of State Table 3.1: Parameter Assignments forlas Equations of State TablaTable 3.1: Asignación de parámetros para ecuaciones de estado 3.1: Parameter Assignments for Equations of State Table 3.1:For Parameter Assignments for Equations For use use withwith Eqs. Eqs. (3.49) (3.49) through through (3.56) (3.56) of State For use with Eqs. (3.49) through (3.56) ParaFor su use usowith con las (3.49)(3.56) a (3.56) Eqs.ecuaciones (3.49) through through For use with Eqs. (3.49) (3.56) Eq.Eq. of de State ofestado State α(T α(T �Ω � � Ψ � Z c Z cZc r )r) r ) � � � sσ σ σ Ecuación a(T Eq. of State α(T � � Z r) Eq. of State α(T ) σ � � � Zccc r Eq. of State α(T ) σ � � � Z r vdWvdW (1873) 00 0 00 0 1/81/8 27/64 27/64 vdW (1873) (1873) 11 1 1/8 27/64 3/8 3/83/8 vdW (1873) 1−1/2 0 0 1/8 27/64 3/8 −1/2 –1/2 vdW (1873) 1 0 0 1/8 27/64 3/8 1 1 0 0 0.08664 0.08664 0.42748 0.42748 1/3 1/31/3 RK RK (1949) (1949) T T RK (1949) 10 00 0.08664 T vdW (1873) 1 1/8 27/64 3/8 r r −1/2 r 1 0 0.08664 0.42748 1/3 RK (1949) Tr−1/2 −1/2 † † 1 0 0.08664 0.42748 1/3 RK (1949) T SRK (1972) (1972)α aαSRK (Trr r ;(T ω) 0.086640.42748 0.42748 1/3 1/3 r;; ω) † 11 1√ 00√ 00 0√ 0.08664 0.08664 1/3 RK (1949) T SRKSRK (1972) 11√ 0.08664 0.42748 SRK SRK (1972) SRK αSRK (T(T 0.08664 0.42748 0.42748 1/3 1/3 r ;rω) ‡ω) √ √ †† ‡ 1 +11 (T ; ω) 1 0 0.08664 0.42748 1/3 SRK (1972) αα αPR † r PR PR (1976) (1976) α SRK (T ; (T ω) ; + 2 2 1 − 1 − 2 2 0.07780 0.07780 0.45724 0.45724 0.30740 0.30740 PR r r SRK (1972) (T ; ω) 0 0.08664 0.42748 1/3 ‡ w) √2 1 − √ √2 0.07780 0.45724 0.30740 PR (1976) αSRK 1 +√ PR (Trr; ω)‡ PR (1976) (1976) αPR (Trr ;; ω) ω)‡‡ + 22 11 − − 22 �0.07780 0.07780 0.45724 0.30740 PR�(T ��0.45724 �α � PR 11+ + 0.30740 2�� 2 1 √ 2 1 – √ 2 0.45724 0.30740 PR †(1976) a (T ; w) PR �� � r 1/2 1/2 † α (T ;(Tω); = 2 )ω0.07780 21� 2 α†SRK ω) = 1 + 1 (0.480 + (0.480 + 1.574 + 1.574 ω − ω 0.176 − 0.176 ω ) − 1 T − T �� � � 1/2 r r SRK r r �� 2 2 � � αSRK (Tr ; ω) = 1 + (0.480 + 1.574 ω −20.176 ω12/ 2) 12 − Tr1/2 1/2 2 †α (T ω) = + (0.480 (0.480 +ω1.574 1.574 ω− − 0.176 ω2 )))11 − −�TTr � † a †α ��2��2 SRK 1�= + + ω (T w)rr ;;=ω) +�(0�11.480 + 1.574 − 0.176 ω 0.176 ) (1 − Tω SRK r; (T r �r �� 1/2 1/2 2 2 ‡ α ‡SRK α (T ; (T ω) ; = ω) = 1 + 1 (0.37464 + (0.37464 + 1.54226 + 1.54226 ω − ω 0.26992 − 0.26992 ω ) ω 1 ) − 1 T − T � � ��2 1/2 PR PR r r r ‡ α (T ; ω) = � 1 + (0.37464 + 1.54226 ω − 0.26992 ω2 ) � 1 − T r �� 1/2 22 2 PR r r 2 ‡ 2 1 / 2 1/2 ‡ a ‡ (T αPR (T ω) = + (0.37464 (0.37464 +ω 1.54226 ω− −ω0.26992 0.26992 ω2))) 11 − − TTr 1= PR w)rr ;;=ω) + (011.37464 + 1.5422 − 0.26992 ) (1 − Trω + + 1.54226 ω PR α r; (T r Example Example 3.9 Ejemplo 3.9 3.9 Example 3.9 Example 3.9 Example 3.9 Given Given thatthat thethe vapor vapor pressure pressure of n-butane of n-butane at 350 at 350 K isK 9.4573 is 9.4573 bar,bar, findfind thethe molar molar Given that the vapor pressure of n-butane at 350 K is 9.4573 bar, find the molar Teniendo en cuenta que la presión de vapor para el n-butano a 350 K es 9.4573 bar, encuentre los Given that vapor pressureand ofand n-butane at 350 350 K K n-butane is 9.4573 9.4573 findconditions the molar volumes volumes of (a) ofthe (a) saturated-vapor saturated-vapor (b) (b) saturated-liquid saturated-liquid n-butane atbar, these at these conditions Given that vapor pressure of at is find the molar volumes of the (a) saturated-liquid n-butane at conditions volúmenes molares de saturated-vapor a) vapor saturadoand y n-butane b)(b) líquido saturado de n-butanobar, enthese estas condiciones, mevolumes ofthe (a) saturated-vapor and (b) saturated-liquid n-butane at these conditions asvolumes given as given byof by the Redlich/Kwong Redlich/Kwong equation. equation. (a) saturated-vapor and (b) saturated-liquid n-butane at these conditions asecuación given byde theRedlich/Kwong. Redlich/Kwong equation. diante la as given given by by the the Redlich/Kwong Redlich/Kwong equation. equation. as Solution Solution 3.9 Solución 3.9 3.9 Solution 3.9 Solution 3.9 Solution A partir deValues losofvalores de Pc para elfrom n-butano del apéndice Values Tof3.9 and Tc and PcTfor n-butane n-butane from App. App. B yield: B yield: B, se obtiene: cPcy for Values of cTc and Pc for n-butane from App. B yield: Values of T and P for n-butane from App. B yield: c c Values of Tc and Pc for n-butane from App. B yield: 9.4573 350350 9.4573 350 = 0.8233 9.4573= 0.2491 Pr = = 0.8233 andyand Pr =9.4573 = 0.2491 Tr = Tr = 350 = 0.8233 and Pr = = 0.2491 Tr =425.1 425.1 37.96 37.96= 350 9.4573 = = = 0.8233 and P 0.2491 T 425.1 37.96 and Prr = 37.96 = 0.2491 Trr = 425.1 = 0.8233 425.1 37.96 Parameter Parameter q is q given is given by Eq. by Eq. (3.54) (3.54) with with �, �, �, and �, and α(T α(T ) for ) for the the RK RK equation from from r (Tr) para laequation El parámetro q se qconoce por ecuación (3.54) W, α(T Ψr yr )a ecuación RK de la Parameter is given bylaEq. (3.54) with �, con �, and for the RK equation from Parameter q is given by Eq. (3.54) with �, �, and α(T ) for the RK equation from Table Table 3.1: 3.1: r Parameter q is given by Eq. (3.54) with �, �, and α(T ) for the RK equation from tabla 3.1: r Table 3.1: Table 3.1: 3.1: Table −1/2−1/2 �Tr�T � �−3/2−3/2 0.42748 0.42748 −1/2 r −3/2−3/2 �Tr−1/2 0.42748 −1/2= =� q =q =�T = 6.6048 Tr T (0.8233) (0.8233) −3/2 −3/2= 6.6048 r = =0.42748 � q = �T = = = 6.6048 T (0.8233)−3/2 rr�Tr −3/2 0.08664 �T � � 0.08664 r−3/2 � 0.42748 r −3/2 q = = = = 6.6048 T (0.8233) �T � 0.08664 r q = �Tr = � Tr = 0.08664 (0.8233) = 6.6048 �Eq. rr from Parameter Parameter β isβfound is �T found from Eq. (3.53): (3.53):0.08664 Parameter β is found afrom Eq. (3.53): El parámetro b se β determina partir de(3.53): la ecuación (3.53): Parameter β is found found from from Eq. (3.53): Parameter is Pr Eq. Pr (0.08664)(0.2491) (0.08664)(0.2491) P (0.08664)(0.2491) β =β�= � =(0.08664)(0.2491)= 0.026214 = 0.026214 Prrr= β = �TrP = = 0.026214 Tr= (0.08664)(0.2491) 0.8233 0.8233 β = � = 0.026214 T 0.8233 r β =�T = = 0.026214 0.8233 Trrwrite 0.8233 (a) (a) ForFor the the saturated saturated vapor, vapor, write the the RK RK form form of Eq. of Eq. (3.52) (3.52) which which results results upon upon (a) For the saturated vapor, write the RK form of Eq. (3.52) which results upon (a) For the saturated vapor, write the RK form of Eq. (3.52) which results upon substitution substitution of appropriate of appropriate values values for for � and � and σ from σ from Table Table 3.1: 3.1: a) Para (a) el vapor saturado, escribiendo la forma RKform laofecuación (3.52) lo que resulta por sustiFor the saturated vapor,values write the �RK Eq. (3.52) which results upon substitution of appropriate for and σdefrom Table 3.1: substitution of appropriate appropriate values for and 3.1: σ from from Table Table 3.1: 3.1: tución de valores apropiados para values y s defor la ��tabla substitution of and σ (Z − (Zβ) − β) (Z − β) Z =Z 1=+1β+−βqβ − qβ(Z − β) Z = 1 + β − qβZ (Z Z + (Z β) + β) = 11 + +β β− − qβ qβ Z(Z (Z−+β) β) ZZ = Z (Z + β) Z (Z + β) 03-SmithVanNess.indd 98 8/1/07 12:57:27 3.6. 3.6. Generalized Generalized Correlations Correlations for for Gases Gases 3.6. Correlaciones generalizadas para 3.6. Generalized Correlations forgases Gases 99 99 99 99 Iteration with an value Z= on Z Thus, Iteration with an initial initial value = 11 converges converges on Z= = 0.8305. 0.8305. Thus, La iteración con with un valor inicialvalue Z = 1ZZconverge en Z =on 0.8305. Así, Iteration an initial = 1 converges Z = 0.8305. Thus, Z v Z RT RT = (0.8305)(83.14)(350) (0.8305)(83.14)(350) = 2,555 cm33 mol −1 V V vv = = Z RT = (0.8305)(83.14)(350) = 2,555 cm mol−1 P 9.4573 V = P = = 2,555 cm3 mol−1 9.4573 P 9.4573 3 −1 −1.. An An experimental experimental value value is is 2,482 2,482 cm cm33 mol mol−1 An experimental value is 2,482 cm mol . 3 –1 Un valor experimental es 2 482 cm mol . (b) For apply Eq. in its RK (b)líquido For the the saturated saturated liquid, liquid, apply Eq. (3.56) (3.56) in en its su RK form: form: b) Para (b) el aplicaapply la ecuación (3.56) For thesaturado saturatedseliquid, Eq. (3.56) in its RKforma form:RK: � �1 + β − Z � � � 1 + β − Z� Z = β + Z (Z + β) 1 + β − Z Z = β + Z (Z + β) qβ Z = β + Z (Z + β) qβ qβ (1.026214 (1.026214 − −Z Z )) o or Z (1.026214 − Z ) or Z= = 0.026214 0.026214 + +Z Z (Z (Z + + 0.026214) 0.026214) (6.6048)(0.026214) or Z = 0.026214 + Z (Z + 0.026214) (6.6048)(0.026214) (6.6048)(0.026214) initial is = the right this equation. Iteration La etapaThe inicial es step la sustitución de Z of = bZ el on lado deof esta iteración conThe initial step is substitution substitution of Z en =β β on thederecho right side side of thisecuación. equation.La Iteration The initial step is substitution of Z = β on the right side of this equation. Iteration leads to convergence on the value Z = 0.04331. Whence, duce a laleads convergencia en el valor = 0.04331. De donde, to convergence on theZvalue Z = 0.04331. Whence, leads to convergence on the value Z = 0.04331. Whence, Z l Z RT RT = (0.04331)(83.14)(350) (0.04331)(83.14)(350) = 133.3 cm33 mol−1 V Vll = = Z RT = (0.04331)(83.14)(350) = 133.3 cm mol−1 9.4573 V = P = = 133.3 cm3 mol−1 P 9.4573 P 9.4573 3 −1 3 –1 An experimental value is 115.0 cm An experimental value is 115.0 cm mol−1.. Un valor experimental es 115.0 cm mol .33 mol An experimental value is 115.0 cm mol−1 . v l For For comparison, comparison, values values of of V V vv and and V Vll calculated calculated for for the the conditions conditions of of Ex. Ex. 3.9 3.9 by by all all four four For comparison, values of V and V calculated for the conditions of Ex. 3.9 by all four v l of the cubic equations of state considered here are summarized as follows: Por los valores de considered V y V calculados las condiciones del ejemplo 3.9 para las cuatro of comparación, the cubic equations of state here are con summarized as follows: of the cubic equations vof state considered here are summarized as follows: 3 −1 ecuaciones de estado cúbicasV se resumen de la siguiente forma: V ll /cm 33 mol −1 −1 V vv/cm /cm33 mol mol−1 V /cm mol−1 V /cm mol V l /cm3 mol−1 l RK 3 –1 Exp. Exp. V v/cm3RK mol–1 SRK Exp. vdW vdW RK SRK PR PR Exp. vdW vdWV /cm RK molSRK SRK PR PR Exp. vdW RK SRK PR Exp. vdW RK SRK PR Exp. PR Exp. 191.0 vdW 133.3 RK 127.8 SRK 112.6 PR 2,482 2,667 2,555 2,520 115.0 2,482vdW 2,667 RK 2,555 SRK 2,520 2,486 2,486 115.0 191.0 133.3 127.8 112.6 2,482 2,667 2,555 2,520 2,486 115.0 191.0 133.3 127.8 112.6 2 482 2 667 2 555 2the 520 2 486 115.0 191.0 133.3 127.8 112.6 The The Soave/Redlich/Kwong Soave/Redlich/Kwong and and the Peng/Robinson Peng/Robinson equations equations were were developed developed specifically specifically for for The Soave/Redlich/Kwong and the Peng/Robinson equations were developed specifically for vapor/liquid equilibrium calculations (Sec. 14.2). vapor/liquid equilibrium calculations (Sec. 14.2). vapor/liquid equilibrium calculations (Sec. 14.2). found Las ecuaciones de Soave/Redlich/Kwong de Peng/Robinson fueron forma such específica Roots of of most with aa software package as Roots of equations equations of state state yare are most easily easily found withdesarrolladas software en package such as para � � R R Roots ofvapor/líquido equations ofwhich state iteration are14.2). mostis easily foundpart with a software package such as cálculosMathcad de equilibrio (sección R R � � or Maple , in an integral of the equation-solving routine. Mathcad� or Maple� in which iteration is an integral part of the equation-solving routine. R or R ,, in Mathcad which iteration isand an integral part ofmás the fácil equation-solving routine. Las raícesvalues de lasMaple ecuaciones de estado se encuentran conparticular un paquete deof software Starting or bounds may be appropriate to root Starting values or bounds may be required, required, and must mustenbe beforma appropriate to the the particular root of R � Starting values or bounds may be required, and3.9 must be appropriate to the particular root of como Mathcad® o Maple®, en que la iteración es una parte integral de la rutina de solución de la ecuación. R � interest. A Mathcad program for solving Ex. is given in App. D.2. interest. A Mathcad� program for solving Ex. 3.9 is given in App. D.2. R program interest. A de Mathcad forosolving Ex. 3.9 given in App. Se puede requerir los valores iniciales de frontera másisapropiados paraD.2. la raíz que nos interese. Un programa Mathcad® para solucionar el ejemplo 3.9 está dado en el apéndice D.2. 3.6 3.6 GENERALIZED GENERALIZED CORRELATIONS CORRELATIONS FOR FOR GASES GASES 3.6 GENERALIZED CORRELATIONS FOR GASES CORRELACIONES GENERALIZADAS PARA GASES Generalized correlations find widespread use. Most popular are correlations of the kind de- 3.6 Generalized Generalized correlations correlations find find widespread widespread use. use. Most Most popular popular are are correlations correlations of of the the kind kind dedeGeneralized correlations find widespread use. Most popular are Z correlations of second the kindvirial developed by Pitzer and coworkers for the compressibility factor and for the veloped bygeneralizadas Pitzer and coworkers for the compressibility factor Z and for son the las second virial Las correlaciones tienen un uso muy extendido. Las más populares desarrolladas por 16 veloped by B. Pitzer and coworkers for the compressibility factor Z and for the second virial 16 coefficient 16 coefficient B.16 Pitzer ycoefficient colaboradores para el factor de compresibilidad Z y para el segundo coeficiente virial B. B. 16 16 16See See Pitzer, Pitzer, op. op. cit. cit. 16 See Pitzer, op. cit. Véase Pitzer, op. cit. 03-SmithVanNess.indd 99 8/1/07 12:57:46 100 CAPÍTULO 3. Volumetric Propiedades volumétricas de Fluids fluidos puros CHAPTER 3. Properties of Pure 100 1.2 1.2 4.0 4.0 1.0 1.0 0.8 0.8 Z 0 0.6 Z 0 0.6 0.4 0.4 Tr 0.7 Tr � 0.7 Gases Gases 0.9 0.9 1.5 1.5 1.2 1.2 1.0 1.0 Región de Two-phase dos fases region 0.7 0.7 C C 0.2 0.2 0 0 0.05 0.05 0.9 0.9 Líquidos comprimidos Compressed (Tr 1.0) liquids (Tr � 1.0) 0.1 0.1 0.2 0.2 0.5 0.5 Pr 1.0 1.0 2.0 2.0 5.0 5.0 10.0 10.0 Pr Figura 3.13: La correlación de Lee/Kesler para Z 0 = F 0 (Tr, Pr). Figure 3.13: The Lee/Kesler correlation for Z 0 = F 0 (Tr , Pr ). Correlaciones de Pitzer para el factor de compresibilidad Pitzer Correlations for the Compressibility Factor The correlation for la Z is: La correlación para Z toma forma: Z = Z 0 + ωZ 1 (3.57) (3.57) 0 y Z 1 son Z 0 and Z 1 arede functions ofr. both Tr and Whenesωel=caso 0, asdeislos thefluidos case for the simple funciones Tr y de P Cuando w =P0,r . como simples, el segundo donde Zwhere 0 becomes identical with Z . Thus a generalized corre0 fluids, the second term disappears, and Z término desaparece, y Z es idéntico a Z. Así, una correlación generalizada para Z como función de Tr y Pr, 0 = F 0(T , P ). forenZinformación as a functionpara of Treland Pr based on data just argon, krypton,laand xenonZprovides que estálation basada argón, el kriptón y elforxenón, proporciona relación r r 0 = F 0 (T , P ). By itself, this represents a two-parameter correspondingthe relationship Z r r Por sí misma, ésta representa una correlación de estados correspondientes con dos parámetros para Z. Ya que states correlation for Z . Because second term of relativamente Eq. (3.57) is apequeña relativelypara small correction el segundo término de la ecuación (3.57) the es una corrección esta correlación, su 0 0 to this correlation, its omission does not introduce large errors, and a correlation for Z be omisión no introduce grandes errores y es posible usar una correlación para Z y así obtenermay estimaciones used alone for quick but less accurate estimates of Z than are obtained from a three-parameter rápidas de Z, pero menos precisas que las que se consiguen a partir de una correlación de tres parámetros. Lacorrelation. ecuación (3.57) es una relación lineal simple entre Z y w para valores conocidos de Tr y de Pr. La and Equation (3.57) a simple linear and for given values of Tr de información experimental paraisZ, de fluidos querelation no son between simples, Z que sonω graficados en función w a Tr y . Experimental data for Z for nonsimple fluids plotted vs. ω at constant T and P P r r r do Pr constantes, producen líneas aproximadamente rectas y sus pendientes proporcionan valores para Z 1 a par1 1 = F 1provide approximately straight lines, and their Z slopes tir de losindeed cualesyield es posible construir la función generalizada (Tr, Pr).values for Z from which 1 1 function Z = del F (T bedesarrollada constructed.por Lee y Kesler17 es la más favorecida. r, P r ) canla Dethe lasgeneralized correlaciones disponibles tipo Pitzer, 17 has Of the Pitzer-type correlations available, the onededeveloped by de Lee and Kesler Aunque su desarrollo se fundamenta en una forma modificada la ecuación estado de Benedict/Webb/ 17 17 B. I. Lee and M. G. Kesler, AIChE J., vol. 21, pp. 510–527, 1975. B. I. Lee y M. G. Kesler, AIChE J., vol. 21, pp. 510-527, 1975. 03-SmithVanNess.indd 100 8/1/07 12:57:54 3.6. Generalized Generalized Correlations Correlations for for Gases Gases 101 3.6. 101 3.6. Correlations for Gases 101 101 101 3.6. Generalized Generalized Correlations forgases Gases 3.6. Correlaciones generalizadas para 3.6. Generalized Correlations for Gases 101 found greatest favor. Although its development is based on a modified form of the Bene0 1 found greatest favor. Although its development islos based on aamodified modified form of the BeneRubin,found ésta adquiere formaAlthough de las tablas que presentanis valores Z y Z form comoof funciones de T y de greatest its on ade 0Benefound greatestlafavor. favor. Although itsitdevelopment development onwhich modified ofthe theZBenedict/Webb/Rubin equation of state, state, takes the the form formisbased ofbased tables presentform values of and r 000and dict/Webb/Rubin equation of it takes of tables which present values of Z and Pr, quedict/Webb/Rubin se proporcionan en el apéndice E de las tablas E.1 a E.4. El uso de estas tablas con frecuencia requieequation of state, it takes the form of tables which present values of Z 1 as functions state, takes the form tables which present values ZBeneand found greatest favor. Although itsitdevelopment is of based a modified form of theUse Zdict/Webb/Rubin ofequation Tr and and Pof These are given in App. App. E as asonTables Tables E.1 through E.4.of of r .. These 111as as functions of T P are given in E E.1 through E.4. Use of Z 0 r r re de interpolación, la cual se trata al inicio del apéndice F. La naturaleza de la correlación se indica en Z functions of T and P . These are given in App. E as Tables E.1 through E.4. Use of functions Tr r andinterpolation, P These are given in App. Ethe as beginning Tables E.1ofthrough Useand of la fiZ astables r r .state, dict/Webb/Rubin equation of it takes theisform of at tables which present values of Z these often of requires which treated App. F.E.4. The nature 0 en función which these tables often requires interpolation, is treated at the beginning of App. F. The nature 1 gura 3.13, que es una gráfica de Z de P para seis isotermas. these tables often requires interpolation, which is treated at the beginning of App. F. The nature these tables oftenof isin treated at the beginning App. F.E.4. TheUse nature Z the ascorrelation functions Tr andinterpolation, Prby . These arewhich given App. EP asr for Tables E.1 of through of of isrequires indicated Fig. 3.13, 3.13, a rplot plot of Z0000vs. vs. six isotherms. isotherms. of the correlation is indicated by Fig. of P for six La correlación de Lee/Kesler proporciona resultados para gases que sonF.noThe polares o ligeraof the correlation is indicated by Fig. 3.13, aaaplot of ZZconfiables vs. P six isotherms. of theThe correlation is indicated by Fig. 3.13, plot of Z vs. P for six isotherms. rrrfor these tables often requires interpolation, which is treated at the beginning of App. nature Lee/Kesler correlation correlation provides provides reliable reliable results results for gases gases which which are are nonpolar nonpolar or or only only The Lee/Kesler for 0 mente polares; para éstos, los errores indicados no son mayores de 2 o 3%. Cuando se aplica a gases muy Lee/Kesler correlation results for which are ororonly of theThe correlation is indicated Fig. 3.13,reliable a plot ofresults vs. for indicated. six isotherms. The Lee/Kesler correlation provides reliable forPgases which arenonpolar nonpolar only rgases slightly polar; for these, these, errorsby ofprovides no more than 2 or or 3Z percent percent are When applied to slightly polar; for errors of no more than 2 3 are indicated. When applied to polaresslightly o asociados, se pueden esperar errores más grandes. polar; for errors ofofno 22oror 33percent are indicated. applied toto slightly polar; forthese, these, errors nomore morethan than percent are indicated. When applied The Lee/Kesler provides reliable results for be gases which areWhen nonpolar or only highly polar gases or tocorrelation gases that associate, larger errors can expected. highly polar gases or to gases that associate, larger can be expected. Los gases cuánticos (por ejemplo, hidrógeno, helio yerrors neón) nobe se ajustan al mismo comportamiento de highly polar gases or to gases that associate, larger errors can expected. highly polar gases or to gases that associate, larger errors can be expected. slightly polar; for these, errors of no more than 2 or 3 percent are indicated. When applied The quantum quantum gases gases (e.g., (e.g., hydrogen, hydrogen, helium, helium, and and neon) neon) do do not not conform conform to to the the same sameto The estados highly correspondientes como los fluidos normales. Algunas veces su análisis mediante las correlaciones The quantum gases (e.g., hydrogen, helium, and neon) not conform totothe polar gases or to gases that associate, largerTheir errors can bedo expected. The quantum gases (e.g., hydrogen, helium, andtreatment neon) do not conform thesame same corresponding-states behavior as do normal fluids. fluids. by the usual correlations correlations is corresponding-states behavior as do normal Their treatment by the usual is acostumbradas se adapta a través del uso denormal parámetros críticos eficaces que son dependientes desame lais temperacorresponding-states behavior as do fluids. Their treatment by the usual correlations 18 corresponding-states behavior as do normal fluids. Their treatment by the usual correlations is The quantum gases (e.g., hydrogen, helium, and neon) do not conform to the For sometimes accommodated by use of temperature-dependent effective critical parameters. 18 18 18 For sometimes accommodated by use of temperature-dependent effective critical parameters. 18 Para el hidrógeno, el gas cuántico más encontrado en los procesos químicos, las ecuaciones recomentura. sometimes For accommodated use effective critical parameters. For sometimes accommodated by use oftemperature-dependent temperature-dependent effective critical parameters. corresponding-states asof do normal fluids. Their treatment by the usual correlations is hydrogen, the quantumbehavior gasby most commonly found in in chemical processing, the recommended hydrogen, the quantum gas most commonly found chemical processing, the recommended 18 For dadas son: hydrogen, the quantum gas most commonly found in chemical processing, the recommended hydrogen, the quantum gas most commonly found in chemical processing, the recommended sometimes accommodated by use of temperature-dependent effective critical parameters. equations are: are: equations equations equations are: hydrogen,are: the quantum gas most commonly found in chemical processing, the recommended 43.6 43.6 (para el2H (for H H /K = = ) 2) (3.58) (3.58) Tc /K 43.6 43.6 equations are: (for (3.58) T 21.8 (for HH222))) (3.58) TTcc/K (3.58) (for 21.8 c /K== 1 + 43.6 21.8 21.8 ++ 2.016 T (for H2 ) (3.58) Tc /K =111+ 2.016 T 21.8TT 2.016 2.016 1+ 2.016 T 20.5 20.5 Pc /bar (for H H /bar = = ) ) (3.59) (3.59) 20.5 20.5 el2H P (3.59) (for c 44.2 (para PPc /bar (3.59) (for (3.59) (forHH222)))2 44.2 c /bar== 1 + 20.5 44.2 44.2 ++ 2.016 T Pc /bar =111+ ) (3.59) (for H 2 2.016 44.2TTT 2.016 2.016 1+ 2.016 51.5T −1 = 51.5 Vc /cm /cm3333mol mol−1 (3.60) (for H H2 )) 51.5 −1 = 51.5 V (3.60) (for 9.91 (para VVcc/cm ) )2) (3.60) (for elH22H mol−1== 1 − 9.91 (3.60) (3.60) (forH 9.91 c /cm mol 2 51.5 9.91 3 1 − −1 2.016 T 1 − Vc /cm mol = 1 −2.016 T (3.60) (for H2 ) 9.91TT 2.016 2.016 1Use − of these effective critical parameters for hywhere T is absolute temperature in kelvins. where isisabsolute absolute temperature ininkelvins. kelvins. Use of of these effective critical parameters for hy2.016 T effective where temperature in whereTTrequires Tis absolute temperature kelvins. ofthese these effectivecritical criticalparameters parametersfor forhyhydrogen the further further specification that Use ωUse = 0. 0. drogen requires the specification that ω = donde drogen Tdrogen es la requires temperatura absoluta en kelvins. El uso de parámetros paraforelhyhidrógeno the specification that ωω = thefurther further specification that =0.estos 0. these where Trequires is absolute temperature in kelvins. Use of effective críticos critical eficaces parameters requieredrogen la especificación de que w = 0. requires theadicional further specification that ω = 0. Pitzer Correlations Correlations for for the the Second Second Virial Virial Coefficient Coefficient Pitzer Pitzer PitzerCorrelations Correlationsfor forthe theSecond SecondVirial VirialCoefficient Coefficient Correlaciones de Pitzer para el compressibility-factor segundo coeficiente virial The tabular nature of the the generalized generalized compressibility-factor correlation is aa disadvantage, disadvantage, but but Pitzer Correlations for the Second Virial Coefficient The tabular nature of correlation is The tabular ofofthe compressibility-factor correlation isisaadisadvantage, but Thecomplexity tabularnature nature thegeneralized generalized compressibility-factor correlation disadvantage, but the of the the functions Z0000 and and Z1111 precludes precludes their their accurate accurate representation by simple simple Z representation by the complexity of functions Z and Z precludes their accurate representation by simple the complexity of the functions Z and Z precludes their accurate representation by simple the complexity of the functions Z The tabular nature of the generalized compressibility-factor correlation is a disadvantage, equations. However, we can can give give approximate analytical expression to these these functions forbut a pero la La naturaleza tabular de la correlación generalizada delanalytical factor deexpression compresibilidad es functions una desventaja, equations. However, we approximate to for 0approximate 1 precludes equations. However, we can give approximate analytical expression to these functions for equations. However, we can give analytical expression to these functions foraaa 0 1 and Z their accurate representation by simple the complexity of the functions Z limited range of pressures. The basis for this is Eq. (3.38), the simplest form of the virial complejidad derange las funciones Z y The Z impide su this representación exacta medianteform ecuaciones sencillas. Sin limited of pressures. basis for is Eq. (3.38), the simplest of the virial limited range ofofpressures. basis for this Eq. (3.38), the form ofofthe limited range pressures. The basis for thisisis Eq.de (3.38), thesimplest simplest form thevirial virial equations. However, we canThe give approximate analytical expression topara these for a equation: embargo, es posible dar una expresión analítica aproximada estas funciones unfunctions intervalo limitado de equation: equation: equation: limited range ofesto pressures. The basis for Bla this is Eq. (3.38), the simplest form of the virial Pforma P presiones. La base para es la ecuación (3.38), más simple de la ecuación virial: r B P P Z= = 11 + + BBPP = (3.61) =1+ + B̂ B̂ PP rr equation: (3.61) RT ==111+ Tr r ZZZ= (3.61) =11++ RT (3.61) +B̂B̂T r B P P RT TTr RT Z =1+ (3.61) (3.61) = 1 + B̂ r r RT Tr where B̂ B̂ is is a reduced reduced second second virial virial coefficient, coefficient, given given by: by: where isisaaareduced second virial coefficient, given reduced secondvirial virialreducido, coefficient, givenby: by: donde where B̂where es unB̂B̂segundo coeficiente conocido por: where B̂ is a reduced second virial coefficient, B given by: B P c PPcc (3.62) B̂ = = BBP (3.62) B̂ RTcc (3.62) B̂ (3.62) (3.62) B̂== RT c B P RT c RT (3.62) B̂ = c c RTc 18 J. M. Prausnitz, R. N. Lichtenthaler, and E. G. de Azevedo, Molecular Thermodynamics of Fluid-Phase Equilib18 J. M. Prausnitz, Prausnitz, R. R. N. N. Lichtenthaler, Lichtenthaler, and and E. E. G. G. de Azevedo, Azevedo, Molecular Molecular Thermodynamics Thermodynamics of Fluid-Phase EquilibEquilib18 18 J. M. Prausnitz, R. N. Lichtenthaler, andUpper E. G.de de Azevedo, Fluid-Phase Equilibria, 3dJ.M. ed., pp. 172–173, 172–173, Prentice Hall PTR, PTR, Saddle River, Molecular NJ, 1999. 1999. ThermodynamicsofofFluid-Phase ria, 3d ed., pp. Prentice Hall Upper Saddle River, NJ, J. ria, M. Prausnitz, R. N. Lichtenthaler y E.PTR, G. deUpper Azevedo, Molecular Thermodynamics of Fluid-Phase Equilibria, 3a. edición, pp. 3d ed., pp. 172–173, Prentice Hall Saddle River, NJ, 1999. 18 ria, 3d ed., pp. 172–173, Prentice Hall PTR, Upper Saddle River, NJ, 1999. J. M. Prausnitz, R. N. Lichtenthaler, and E. G. de Azevedo, Molecular Thermodynamics of Fluid-Phase Equilib172-173, Prentice-Hall PTR, Upper Saddle River, 1999. ria, 3d ed., pp. 172–173, Prentice Hall PTR, Upper Saddle River, NJ, 1999. 18 03-SmithVanNess.indd 101 8/1/07 12:58:17 102 CHAPTER 3. 3. Volumetric Volumetric Properties of Pure Pure Fluids CHAPTER Properties of CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3.3.Volumetric Properties of Pure Fluids 102 102 102 Thus, Pitzer and and coworkers coworkers proposed second correlation, correlation, which which yields values for B̂: B̂: Thus, Pitzer proposed second values for Así, Pitzer y colaboradores recomiendan unaaasegunda correlación, la cualyields produce valores para B̂: Thus, Pitzer and coworkers proposed a second correlation, which yields values for B̂: + ωB ωB111 = BB000 + B̂B̂ = B̂ = B 0 + ωB 1 (3.63) (3.63) (3.63) (3.63) Equations (3.61) and (3.63) (3.63) together become: (3.61) and together become: Al unir Equations las ecuaciones (3.61) y (3.63) se obtiene: Equations (3.61) and (3.63) together become: PPr PPr + ωB ωB111 Prrr = 11 + + BB000 Prrr + ZZ = Z = 1 + B 0 TTrrr + ωB 1 TTrrr Tr Tr Comparison of this this equation equation withse Eq. (3.57) provides provides the following following identifications: Comparison of with Eq. (3.57) the identifications: Al comparar esta ecuación con la (3.57) obtienen las identificaciones siguientes: Comparison of this equation with Eq. (3.57) provides the following identifications: PPr = 11 + + BB000 Prrr (3.64) (3.64) (3.64) ZZ0000 = (3.64) Z = 1 + B 0 TTrrr Tr PPr and = BB1111 Prrr and ZZ1111 = and Z = B TTrrr y Tr Second virial virial coefficients coefficients are are functions functions of of temperature temperature only, only, and and similarly similarly BB0000 and and BB1111 are are funcfuncSecond Second virial coefficients are functions of temperature only, and similarly B andthe B following are funcLos segundos viriales son funciones sólo de la temperatura, y de manera similar B0 y B1 son tions of ofcoeficientes reduced temperature temperature only. They are are reasonably reasonably well represented represented by tions reduced only. They well by the following tions of reduced temperature only. They are reasonably well represented by the following 19 funciones sólo de19 la temperatura reducida. Se representan bien mediante las ecuaciones siguientes:19 19 equations: equations: equations:19 0.422 0.422 = 0.083 0.083 − − 0.422 BB0000 = 1.6 1.6 B = 0.083 − TTrr1.6 Trr1.6 (3.65) (3.65) (3.65) 0.172 0.172 = 0.139 0.139 − − 0.172 BB1111 = 4.2 4.2 B = 0.139 − TTrr4.2 Trr4.2 (3.66) (3.66) (3.66) The simplest simplest form form of of the the virial virial equation equation has has validity validity only only at at low low to to moderate moderate pressures pressures The La forma más simple form de la of ecuación virial tiene validez sólo aonly presiones a moderadas, donde Z theThe virial equation has validity at lowdetobajas moderate pressures whereThe issimplest linear in in pressure. pressure. generalized virial-coefficient correlation is therefore therefore useful where ZZ is linear The generalized virial-coefficient correlation is useful es una función lineal de la presión. Por lo tanto, la correlación generalizada del coeficiente virial es útil sólo where Z is linear in pressure. The generalized virial-coefficient correlation is therefore useful 0 1 0 1 only where and ZZ 11 are are at at least least approximately approximately linear linear functions functions of of reduced reduced pressure. pressure. FigFigonly ZZ 00 and 0 y Zwhere 1 son al donde Zonly menos aproximadamente funciones lineales de la presión reducida. En la figura 3.14 se where Z and Z are at least approximately linear functions of reduced pressure. Fig0 0 ure 3.14 3.14 compares compares the the linear linear relation of of ZZ 00 to to PPrrr as as given given by by Eqs. Eqs. (3.64) (3.64) and and (3.65) (3.65) with with values values ure relation 0 en función comparaure la 3.14 relación lineal de Z de P como se conocen por las ecuaciones (3.64) y (3.65) con los compares the linear relation of Z to P as given by Eqs. (3.64) and (3.65) with values r 0 r 0 from the the Lee/Kesler Lee/Kesler compressibility-factor compressibility-factor correlation, correlation, Tables Tables E.1 E.1 and and E.3. E.3. The The two two corcorof ZZ 000from of valores of de ZZ from de la the correlación delcompressibility-factor factor de compresibilidad de Lee/Kesler, tablas E.1 y E.3. Las dos correlation, Tables E.1 and E.3. The cor-correlarelations differ Lee/Kesler by less less than than 2% 2% in in the the region region above above the dashed dashed line of of the figure. For two reduced relations differ by the line the figure. For reduced ciones difieren endiffer menos de 2%than en la región que está above arriba the de ladashed línea punteada defigure. la figura. Para temperaturelations by less 2% in the region line of the For reduced temperatures greater greater than than TTrrr ≈ ≈ 3, 3, there there appears appears to to be be no no limitation limitation on on the the pressure. pressure. For For lower lower temperatures ras reducidas mayoresgreater que Trthan ≈ 3,Tparece ser queappears no haytolímite enlimitation la presión. Para valores menores que Tr, el temperatures ≈ 3, there be no on the pressure. For lower r the allowable allowable pressure pressure range range decreases decreases with with decreasing decreasing temperature. temperature. A A point point is is values of of TTrrr the values intervalo de presión permisible disminuye conforme se reduce ladecreasing temperatura. Sin embargo, se alcanza un the allowable pressure range decreases with temperature. A point is values of T 20 r 20 20 reached, however, however, at at TTrrr ≈ ≈ 0.7 0.7 where where the the pressure pressure range range is is limited limited by by the the saturation saturation pressure. pressure. reached, 20 Esto 20 punto enreached, Tr ≈ 0.7, donde el intervalo de presión está limitado por la presión de saturation saturación.pressure. se indica however, at T ≈ 0.7 where the pressure range is limited by the 1 r 1 This is is indicated indicated by by the the left-most left-most segment segment of of the the dashed dashed line. line. The The minor minor contributions contributions of of ZZ 11 to to This por el segmento que estáby enthe el extremo izquierdo de the la línea. Seline. ignoran las contribuciones más de Thiscorrelations is indicated left-most segment of dashed The minor contributions of Zpequeñas to the are here neglected. In view of the uncertainty associated with any generalized the correlations are here neglected. In view of the uncertainty associated with any generalized Z 1 a lasthe correlaciones. En vista de la incertidumbre asociada con cualquier correlación generalizada, las descorrelations are here neglected. In view of the uncertainty associated with any generalized 0 0 correlation, deviations of of no no more more than than 2% 2% in in ZZ 0 are are not not significant. significant. correlation, deviations viaciones en Z 0 no mayores irrelevantes. correlation, deviationsdeof2% noson more than 2% in Z 0 are not significant. The relative relative simplicity simplicity of of the the generalized generalized second-virial-coefficient second-virial-coefficient correlation correlation does does much much The La simplicidad relativa de la correlación generalizada del coeficiente virial la hace muy recomendable. The relative the generalized second-virial-coefficient correlation does much to recommend recommend it. simplicity Moreover,oftemperatures temperatures and pressures of of many many chemical-processing chemical-processing opto it. Moreover, and pressures opPor otratoparte, las temperaturas y presiones de operación de varios procesos químicos se encuentranopdentro de recommend it. Moreover, temperatures and pressures of many chemical-processing erations lie lie within within the the region region where where itit does does not not deviate deviate by by aa significant significant amount amount from from the the erations la regiónerations donde no sewithin desvían enregion una cantidad significativa de la correlación del factor de compresibilidad. Al lie the where it does not deviate by a significant amount from the compressibility-factor correlation. correlation. Like Like the the parent parent correlation, correlation, itit is is most most accurate accurate for for nonpolar nonpolar compressibility-factor igual que la correlación generadora, ésta esLike másthe exacta para especies no polares, y menosfor para moléculas alcompressibility-factor correlation. parent correlation, it is most accurate nonpolar species and and least least accurate accurate for for highly highly polar polar and and associating associating molecules. molecules. species tamentespecies polaresand y asociadas. least accurate for highly polar and associating molecules. 19 19 19These These correlations first appeared in 1975 in the third edition of this book, attributed as personal communication Thesecorrelations correlationsfirst firstappeared appearedin in1975 1975in inthe thethird thirdedition editionof ofthis thisbook, book,attributed attributedas asaaapersonal personalcommunication communication 19 These correlations first appeared in 1975 in the third edition of this book, attributed as a personal communication to M. Abbott, who developed them. to M. M. who them. to M. M. M.Abbott, Abbott,aparecieron whodeveloped developed them. 19 Estas correlaciones por vez primera en 1975 en la tercera edición de este libro, como una comunicación personal de to M. M. Abbott, who developed them. M. M. Abbott, quien las desarrolló. 20 20 20Although Although the Lee/Kesler tables of Appendix list values for superheated vapor and subcooled liquid, they do not Althoughthe theLee/Kesler Lee/Keslertables tablesof ofAppendix AppendixEEElist listvalues valuesfor forsuperheated superheatedvapor vaporand andsubcooled subcooledliquid, liquid,they theydo donot not 20 Although 20 Aunque Lee/Kesler tables of Appendix E listvalores values para for superheated vapor and subcooled liquid, they do not lasvalues tablasthe de Lee/Kesler del apéndice E exhiben el vapor sobrecalentado y el líquido subenfriado, no proporprovide values at saturation conditions. provide at conditions. provide values atsaturation saturation conditions. provide at saturation conditions. cionan valores en values condiciones de saturación. 03-SmithVanNess.indd 102 8/1/07 12:58:34 3.6. Correlaciones generalizadas para 3.6. Generalized Generalized Correlations forgases Gases 3.6. Correlations for Gases 103 103 4.0 4.0 4.0 4.0 1.0 1.0 1.0 1.0 Z0 ZZ000 Z 2.4 2.4 2.4 2.4 1.8 1.8 1.8 1.8 0.9 0.9 0.9 0.9 0.8 0.8 0.8 0.8 103 T 0.8 TTrr � � 0.8 0.8 0.9 T rr � 0.8 0.9 0.9 0.9 1.0 1.0 1.0 1.0 1.1 1.1 1.1 1.1 1.5 1.5 1.5 1.5 1.3 1.3 1.3 1.3 0.7 0.7 0.70.0 0.7 0.0 0.0 0.0 0.5 0.5 0.5 0.5 1.0 1.0 1.0 1.0 1.5 1.5 1.5 1.5 Pr P Prrr P 2.0 2.0 2.0 2.0 2.5 2.5 2.5 2.5 0 correlación Figura 3.14: Comparación de correlaciones para for Z 0.ZLa del coeficiente virial is se represented representa por medio de líFigure 3.14: Comparison of correlations The virial-coefficient correlation by the Figure 3.14: 3.14: Comparison Comparison of of correlations correlations for for Z Z00... The The virial-coefficient virial-coefficient correlation correlation is is represented represented by by the the Figure neas rectas; la correlación de Lee/Kesler mediante los puntos. En la región por encima de la líneathe discontinua, las dos straight lines; the Lee/Kesler correlation, by the points. In the region above the dashed line two straight lines; lines; the the Lee/Kesler correlation, correlation, by by the the points. points. In In the the region region above above the the dashed dashed line line the the two two straight correlaciones difierendiffer por Lee/Kesler menos de 2%. correlations by less than 2%. correlations differ differ by by less less than than 2%. 2%. correlations Correlaciones parafor elthe tercer coeficiente virial Correlations for the Third Virial Coefficient Coefficient Correlations Third Virial Accurate precisa data for forpara third virial coefficients are viriales far less less es common than for second virial coefAccurate data third coefficients are far common second coefLa información losvirial terceros coeficientes bastantethan másfor escasa quevirial para los segundos ficients. Nevertheless, generalized correlations for third virial coefficients do appear in the ficients. Nevertheless, generalized for correlaciones third virial coefficients do appear the coeficientes viriales. No obstante, aparecencorrelations en la literatura generalizadas para losinterceros coliterature. literature. eficientes viriales. Equation (3.40) may be written: written: Equation (3.40) may be La ecuación (3.40) puede escribirse: Z= = 11 + + Bρ Bρ + + Cρ Cρ 222 Z (3.67) (3.67) (3.67) where ρes= =la1/V 1/V is molar molar density. Rewritten in reduced reduced form, reducida, this equation equation becomes: donde rwhere = 1/Vρ densidad molar. Al volverse a escribir en forma esta becomes: ecuación se convierte en: is density. Rewritten in form, this � � � Pr �222 Pr P P rr + Ĉ rr Z = 1 + B̂ (3.68) (3.68) Z = 1 + B̂ T Z + Ĉ T Z (3.68) Trrr Z Trrr Z donde el segundo coeficiente virialvirial reducido está definido por la by ecuación (3.62)and y elthe tercer coeficiente where the reduced reduced second coefficient B̂ is is defined defined Eq. (3.62), (3.62), reduced third virial where the second virial coefficient B̂ by Eq. and the reduced third reducido se define como: virial coefficient coefficient is is defined defined as: as: virial CP Pc222 C Ĉ ≡ Ĉ ≡ R 222Tcc222 R Tc cc A Pitzer-type Pitzer-type correlation forseĈ Ĉescribe: is written: written: Una correlación tipo correlation Pitzer para Ĉ A for is +ω ωC C 111 Ĉ = =C C 000 + Ĉ 03-SmithVanNess.indd 103 (3.69) (3.69) (3.69) 8/1/07 12:58:52 CHAPTER 3. Volumetric Properties of Pure Fluids 104 104 CHAPTER 3. Volumetric Properties of Pure Fluids CAPÍTULO 3. Propiedades volumétricas de21fluidos puros An expression for as a function of reduced temperature is given by Orbey and Vera: 0 An expression for C as a function of reduced temperature is given by Orbey and21Vera:21 0.02432 0.00313 Una expresión para C 0 como una función de temperatura reducida la dan Orbey y Vera: C 0 = 0.01407 + − (3.70) Tr T 10.5 0.02432 0.00313 C 0 = 0.01407 + − r10.5 (3.70) (3.70) Tr Tr 1 The expression for C given by Orbey and Vera is replaced here by one that is algebraically Aquí, la expresión para C 1 conocida por Orbey y Vera se reemplaza por una que en términos algebraicos es simpler, but essentially numerically: given by Orbey and Vera is replaced here by one that is algebraically The expression for C 1 equivalent más simple, pero numéricamente es equivalente: simpler, but essentially equivalent numerically: 0.05539 0.00242 C 1 = −0.02676 + − (3.71) (3.71) Tr2.7 Tr10.5 0.05539 0.00242 1 − (3.71) C = −0.02676 + 2.7 La ecuación (3.68) es cúbica en Z, y no es posibleTexpresarla la forma de la ecuación (3.57). Al esTen10.5 Equation (3.68) is cubic in Z , and cannot ber expressedr in the form of Eq. (3.57). With pecificarse Tr y Pr, la solución para Z es por iteración. Una aproximación inicial de Z = 1 en el lado derecho Tr andEquation Pr specified, solution for ZZ is by iteration. initial guess Z =of1 Eq. on the rightWith side (3.68) is cubic , and cannot beAn expressed in theofform (3.57). de la ecuación (3.68) por lo general nosinlleva a una convergencia rápida. of usually solution leads to rapid Tr Eq. and(3.68) Pr specified, for Z convergence. is by iteration. An initial guess of Z = 1 on the right side of Eq. (3.68) usually leads to rapid convergence. 104 C0 Condiciones de validez aproximada de laofecuación del gas ideal Conditions of Approximate Validity the Ideal-Gas Equation Conditions of arises Approximate Validity the Ideal-Gas The question often as to when the ideal-gasof equation may be used Equation as a reasonable ap- Con frecuencia surge la pregunta acerca de cuándo la ecuación del gas ideal puede utilizarse como una aproxitooften reality. Figure canthe serve as a guide. The question arises to3.15 when ideal-gas equation be used as a reasonable apmaciónproximation razonable a la realidad. Laasfigura 3.15 puede servir como unamay guía. proximation to reality. Figure 3.15 can serve as a guide. 10 10 10 Z 0 � 1.02 1 1 1 Figure 3.15: Region where Z 0 lies between 0.98 Region and 1.02, and the Figure 3.15: where Z 0 lies Figura 3.15: Región dondeisZa0 reasonable reside ideal-gas equation between 0.98 and 1.02, and the entre 0.98 y 1.02, y la ecuación del gas approximation. ideal-gas equation is a reasonable ideal es una aproximación razonable. approximation. Z 0 1.02 Z 0 � 1.02 Z 0 � 0.98 Pr 0.1 Pr Pr 0.1 0.1 Z 0 � 0.98 Z 0 0.98 0.01 0.01 0.001 0.001 0.01 0 0 0.001 1 0 1 1 2 Tr 2 Tr 3 2 Tr 3 4 3 4 4 21 H. Orbey and J. H. Vera, AIChE J., vol. 29, pp. 107–113, 1983. 21 H. Orbey and J. H. Vera, AIChE J., vol. 29, pp. 107–113, 1983. 21 H. Orbey y J. H. Vera, AIChE J., vol. 29, 107-113, 1983. 03-SmithVanNess.indd 104 8/1/07 12:59:01 3.6. Generalized Generalized Correlations Correlations for for Gases Gases 3.6. 3.6. Correlations Gases 3.6. Generalized Generalized Correlations for Gases 3.6. Correlaciones generalizadas parafor gases 105 105 105 105 105 Example 3.10 3.10 Example Example 3.10 Example 3.10 Ejemplo 3.10 Determine the the molar molar volume volume of of n-butane n-butane at at 510 510 K K and and 25 25 bar bar by by each each of of the the following: following: Determine Determine the molar volume of n-butane at 510 K and 25 bar by each of the following: Determine the molar volume of n-butane at 510 K and 25 bar by each of the following: (a) The The ideal-gas ideal-gas equation. equation. (a) (a) equation. (a) The The ideal-gas ideal-gas equation. La ecuación del gas ideal. (b) The The generalized generalized compressibility-factor compressibility-factor correlation. correlation. (b) (b) compressibility-factor correlation. (b) The The generalized generalized compressibility-factor correlation. La correlación generalizada del factor de compresibilidad. (c) Equation Equation (3.61), (3.61), with with the the generalized generalized correlation correlation for for B̂. B̂. (c) La ecuación (3.61)(3.61), con lawith correlación generalizada para B̂.for (c) the correlation (c) Equation Equation (3.61), with the generalized generalized correlation for B̂. B̂. (d) Equation Equation (3.68), (3.68), with with the the generalized generalized correlations correlations for for B̂ B̂ and and Ĉ. Ĉ. (d) La ecuación (3.68)(3.68), con laswith correlaciones generalizadas parafor B̂ yB̂ (d) the correlations and (d) Equation Equation (3.68), with the generalized generalized correlations for B̂Ĉ. and Ĉ. Ĉ. Determine the molar molar de volume of n-butane 510 K mediante: and 25 bar by each of the following: Determine el volumen n-butano a 510 K at y 25 bar a) b) c) d) Solution 3.10 Solución 3.10 3.10 Solution Solution Solution 3.10 3.10 a) Por la(a) ecuación del gas ideal, (a) By the the ideal-gas ideal-gas equation, By equation, (a) (a) By By the the ideal-gas ideal-gas equation, equation, RT (83.14)(510) −1 V= = RT mol−1 = (83.14)(510) = 1,696.1 1,696.1 cm cm33 mol RT (83.14)(510) RT (83.14)(510) V = = 33 −1 P 25 VV = = = P = mol−1 = = 1,696.1 1,696.1 cm cm mol 25 PP 25 25 (b)valores From the the values of and P Pen given in Table Table B.1 of App. App.B, B, b) De los de Tvalues conocidos la tabla B.1 del apéndice (b) From TTcc and in B.1 of B, cc given c y Pc of (b) (b) From From the the values values of of TTcc and and PPcc given given in in Table Table B.1 B.1 of of App. App. B, B, 510 25 510 25 = 510 = 25 = 1.200 1.200 Pr = = 0.659 0.659 510 = 25 = TTrr = P 425.1 37.96 TTrr = = PPrrr = = = 425.1 = 37.96 = 1.200 1.200 = 0.659 0.659 425.1 37.96 425.1 37.96 Interpolation in Tables E.1 and E.2 then provides: Interpolation in tablas TablesE.1 E.1yand then provides: La interpolación en las E.2E.2 proporciona: Interpolation in E.2 then Interpolation in Tables Tables E.1 E.1 and and E.2 then provides: provides: 0 1 = 0.865 Z = 0.038 0.038 0 =0.865 11 = ZZ 000Z= 0.865 ZZ 11 0.038 ZZ= = ZZ = = 0.865 0.865 = 0.038 0.038 Thus, by Eq. (3.57) with ω = 0.200, Thus, by Eq. (3.57)(3.57) with ωcon =w 0.200, Así, mediante la ecuación = 0.200, Thus, Thus, by by Eq. Eq. (3.57) (3.57) with with ω ω= = 0.200, 0.200, 0 1 + ωZ ωZ 1 = = 0.865 0.865 + + (0.200)(0.038) (0.200)(0.038) = = 0.873 0.873 = ZZ 0 + ZZ = ZZ = + ωZ ωZ11 = = 0.865 0.865 + + (0.200)(0.038) (0.200)(0.038) = = 0.873 0.873 = ZZ00 + (0.873)(83.14)(510) Z RT RT −1 (0.873)(83.14)(510) Z mol−1 = (0.873)(83.14)(510) = 1,480.7 1,480.7 cm cm33 mol and V= = ZZ RT (0.873)(83.14)(510) RT = = and V 33 −1 −1 P 25 mol = = 1,480.7 cm and V = P 25 mol = = 1,480.7 cm and V = y PP 25 25 If ZZ 11,, the the secondary secondary term, term, is is neglected, neglected, ZZ = = ZZ 00 = = 0.865. 0.865. This This two-parameter two-parameter If 1, Z = Z 0Z 3 con −1 If secondary term, = =cm This two-parameter If ZZ11,, the the secondary term, is is neglected, neglected, Z=1,467.1 = ZZ00 Así, = 0.865. 0.865. This two-parameter Si se desprecia el término secundario, Z 0.865. la correlación dethan estados co3 −1 mol , which is less less than corresponding-states correlation yields V = is corresponding-states correlation yields V = 1,467.1 cm 33 mol −1 −1,,, which 3cm –1, que mol which is corresponding-states correlation yields VV1==467.1 1,467.1 mol which is less less than than corresponding-states correlation yields 1,467.1 cm rrespondientes de dos parámetros se obtiene V = cm mol es 1% menor que el valor 1% lower than the value given by the three-parameter correlation. 1% lower than the value given by the three-parameter correlation. 1% lower than value given by the three-parameter correlation. 1% lower than the the value given by the three-parameter correlation. conocido por la correlación de tres parámetros. (c) Values Values of of B B00 and and B B11 are are given given by by Eqs. Eqs. (3.65) (3.65) and and (3.66): (3.66): (c) 00 and 0 1 (c) Values of B B (c) Values of B and B11 are are given given by Eqs. Eqs. (3.65) (3.65) and and (3.66): (3.66): c) Los valores de B y B se conocen por by las ecuaciones (3.65) y (3.66): 00 = −0.232 11 = 0.059 B B B = 0.059 B 00 =0 −0.232 11 1B= = BB B= ==−0.232 −0.232 = 0.059 0.059 –0.232 B B 0.059 Equations (3.63) (3.63) and and (3.61) (3.61) then then yield: yield: Equations Equations (3.61) yield: Equations (3.63) (3.61) then yield: De las ecuaciones (3.63) and yand (3.61) sethen obtiene: 00 + ωB11 = −0.232 + (0.200)(0.059) = −0.220 B̂ = B B̂ = B 00 + ωB 11 = −0.232 + (0.200)(0.059) = −0.220 B̂B̂ = + ωB ωB = = −0.232 −0.232 + + (0.200)(0.059) (0.200)(0.059) = = −0.220 −0.220 = BB + 0.659 0.659 = 0.879 0.879 = 11 + + (−0.220) (−0.220) 0.659 0.659 = ZZ = 1.200 = ZZ = = 0.879 0.879 = 11 + + (−0.220) (−0.220)1.200 1.200 1.200 3 mol−1 −1 , a value 3 from which V = 1,489.1 cm less than 1% higher higher than than that that given given from which V = 1,489.1 cm33 mol−1 value less than 1% −1,,, a from which VV == 1,489.1 cm mol aa value less than 1% higher than that given from which 1,489.1 cm mol value less than 1% higher than that given por la 3 –1 by the compressibility-factor correlation. de la cual V = 1 489.1 cmcorrelation. mol , un valor que es 1% mayor que el conocido by se theobtiene compressibility-factor by the compressibility-factor correlation. by the compressibility-factor correlation. correlación del factor de compresibilidad. 03-SmithVanNess.indd 105 8/1/07 12:59:21 106 CHAPTER 3. 3. CHAPTER CHAPTER CHAPTER 3. 3. CHAPTER 3. Volumetric Properties Properties of of Pure Pure Fluids Fluids Volumetric Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids Volumetric Properties of Pure CAPÍTULO 3. Propiedades volumétricas de Fluids fluidos puros 106 106 106 106 106 (d) Values Values of of C C000 and and C C111 are are given given by by Eqs. Eqs. (3.70) (3.70) and and (3.71): (3.71): (d) (d) Values of C and C are by Eqs. (3.70) (3.71): 0 0 0 1 (d) Values Values of yC CC and and C11 dados are given given by Eqs. (3.70) and and (3.71): d) Los valores de Cof están por by lasEqs. ecuaciones (3.70) y (3.71): (d) C are given (3.70) and (3.71): 000 = 0.0339 111 = 0.0067 C C C C1 11 = 0.0067 0= CCC = 0.0339 0==0.0339 0.0339 C= = = 0.0067 0.0067 0.0339 CC 0.0067 C0 = 0.0339 C = 0.0067 Equation (3.69) then yields: Equation (3.69) then yields: Equation (3.69) yields: Por lo tanto, la ecuación (3.69) nos da: Equation (3.69) then then yields: Equation (3.69) then yields: 0 00 + ω C111 = 0.0339 + (0.200)(0.0067) = 0.0352 Ĉ = C ωC = 0.0339 + (0.200)(0.0067) = 0.0352 Ĉ C 00 + ĈĈ = = =C C + +ω ωC C11 = = 0.0339 0.0339+ +(0.200)(0.0067) (0.200)(0.0067) = = 0.0352 0.0352 + ω C = 0.0339 + (0.200)(0.0067) = 0.0352 Ĉ = C With this value of Ĉ and the value of B̂ from part (c), Eq. (3.68) becomes, Ĉ and value of B̂ part (c), Eq. (3.68) becomes, Con esteWith valorthis de value Ĉ y elofvalor de the B̂ del inciso c),from la ecuación (3.68) se convierte en, With With this this value value of of Ĉ Ĉ and and the the value value of of B̂ B̂ from from part part (c), (c), Eq. Eq. (3.68) (3.68) becomes, becomes, With this value of Ĉ and the value of B̂ from part (c), Eq. (3.68) becomes, � � � � � � �� 0.659 �� �� 0.659 ��222 0.659 � 0.659 � � 0.659 � � 0.659 � Z = 1 + (−0.220) + (0.0352) 0.659 + 0.659 22 Z 11 + (−0.220) (0.0352) 0.659 0.659 ZZ = = + (−0.220) + (0.0352) 1.200Z 1.200Z = 1 + (−0.220) + (0.0352) 1.200Z 1.200Z Z = 1 + (−0.220) 1.200Z + (0.0352) 1.200Z 1.200Z 1.200Z 1.200Z 1.200Z 0.121 0.0106 0.121 0.0106 or Z = 11 − − 0.121 + 0.0106 0.121 0.0106 or Z 0.121 0.0106 or ZZ = = 11− + Z + Z22 or = − + Z o or Z =1− Z + Z ZZ222 ZZ Z −1 Whence, Z = 0.876 and V = 1,485.8 cm333 mol mol−1 −1 Whence, Z 0.876 and V 1,485.8 cm Whence, ZZ = = 0.876 and VV = = 1,485.8 cm 3 mol −1 3 Whence, = 0.876 and = 1,485.8 cm mol 3 mol -1−1 Whence, ZZ ==0.876 cm mol De donde, 0.876 and y VV ==1 1,485.8 485.8 cm The value value of of V V differs from from that that of of part part (c) (c) by by about about 0.2%. 0.2%. An An experimental experimental The The value of VV differs differs that of part (c) by about 0.2%. An experimental 3from −1 The value of differs from that of part (c) by about 0.2%. An experimental El valor de V difiere del que corresponde al inciso c) en aproximadamente Un valor value for V is 1,480.7 cm mol . Significantly, the results of parts (b), 0.2%. (c), and and 3 −1 The value of V differs from that of part (c) by about 0.2%. An experimental 3 mol −1 .. Significantly, value for V cm the results of parts (b), (c), value for VV is isis 1,480.7 1,480.7 cm Significantly, the results of parts (b), (c), and 33mol −1 –1 3 −1 value for 1,480.7 cm mol . Significantly, the results of parts (b), (c), and experimental para V es 1 480.7 cm mol . De manera significativa, los resultados de los incisos (d) are in excellent agreement. Mutual agreement at these conditions is suggested value for V is 1,480.7 cm mol . Significantly, the results of parts (b), (c), and (d) are in excellent agreement. Mutual agreement at these conditions is (d) are in excellent agreement. Mutual at conditions isissuggested suggested (d)seFig. are in excellent agreement. Mutual agreement agreement at these these conditions suggested b), c) y d) encuentran en excelente concordancia. La mutua correlación de estas condiciones se by 3.14. (d) are in excellent agreement. Mutual agreement at these conditions is suggested by Fig. 3.14. by Fig. 3.14. byla Fig. 3.14.3.14. sugiere en figura by Fig. 3.14. Example 3.11 3.11 Example Example Ejemplo 3.11 3.11 What pressure is generated when when 1(lb mol) mol) of methane methane is stored stored in aa volume volume of 2(ft) 2(ft)33 What What pressure pressure is is generated generated when 1(lb 1(lb mol) of of methane is is stored in in a volume of of 2(ft)3 What pressure is generated generated when 1(lb mol) mol) offollowing: methane is is stored stored in in aa volume volume of of 2(ft) 2(ft)33 at 122( calculations on each of theof ◦◦ F)? Base What pressure is when 1(lb methane ◦ F)? at 122(se Base calculations on each of the following: ¿Qué presión genera se almacena 1(lb de metano en un volumen de 2(pie) 3 a 122(°F)? at Base calculations on of the at 122( 122(◦◦F)? F)? Basecuando calculations on each each ofmol) the following: following: at 122( F)? Base calculations on each of the following: Apoye sus cálculos lo siguiente: (a) The en ideal-gas equation. (a) (a) The The ideal-gas ideal-gas equation. equation. (a) The The del ideal-gas equation. (a) ideal-gas equation. a) La ecuación gas ideal. (b) The Redlich/Kwong equation. (b) The Redlich/Kwong equation. (b) Redlich/Kwong equation. b) La ecuación Redlich/Kwong. (b) The The de Redlich/Kwong equation. (b) The Redlich/Kwong equation. (c) A generalized correlation. A correlation. (c) AA generalized generalized correlation. c) Una(c) correlación generalizada. (c) generalized correlation. (c) A generalized correlation. Solution 3.11 Solución 3.11 3.11 Solution Solution 3.11 Solution 3.11 3.11 Solution a) Por la(a) ecuación del gas ideal, By the the ideal-gas ideal-gas equation, (a) By equation, (a) By the ideal-gas equation, (a) By the ideal-gas equation, (a) By the ideal-gas equation, (0.7302)(122 + + 459.67) 459.67) RT (0.7302)(122 + RT = (0.7302)(122 = 212.4(atm) 212.4(atm) P = RT (0.7302)(122 +459.67) 459.67) = RT = P (0.7302)(122 + 459.67) RT = = 212.4(atm) PP = = V 2 = = 212.4(atm) = V 2 = 212.4(atm) P= V = 22 VV 2 (b) The The pressure as given given by the the Redlich/Kwong Redlich/Kwong equation is: (b) as by equation b) La presión sepressure conoce por la ecuación de Redlich/Kwong que es:is: (b) equation is: (b) The The pressure pressure as as given given by by the the Redlich/Kwong Redlich/Kwong equation is: (b) The pressure as given by the Redlich/Kwong equation is: a(T )) RT a(T )) RT − a(T (3.47) P= = RT a(T RT − (3.47) P a(T+) b) (3.47) PP = VRT − bb − V (V (V −V (3.47) =V + b) − (3.47) P= VV − − b V (V + b) − bb VV(V (V + + b) b) V− 03-SmithVanNess.indd 106 (3.47) 8/1/07 12:59:39 107 107 3.6. Generalized Correlations Gases 3.6. Correlaciones generalizadas parafor gases 107 3.6. Generalized Generalized Correlations Correlations for for Gases Gases 3.6. 107 107 –1/2 −1/2 3.6. 3.6. Generalized Generalized Correlations Correlations for for Gases Gases 107 Values a(T b come from (3.45) and (3.45) (3.46),ywhere Tr r) =inTr 107 Los valores de of a(T) y )b and se desarrollan de Eqs. las ecuaciones (3.46),α(T donde en r ) = a(T Eq. (3.45). Con Withlos values of Tde P from Table B.1 converted to (R) and (atm), −1/2 la ecuación valores T y P del apéndice B convertidos a (R) y (atm), c and c −1/2 in Values of of a(T a(T )) and and bb come come from fromc Eqs. Eqs.c (3.45) (3.45) and and (3.46), (3.46), where where α(T α(Tr )) = = TTr−1/2 Values in −1/2in Values of )))and bbbcome from Eqs. (3.45) and (3.46), where α(T TTTrr−1/2 rrrr)))= Values Values of ofa(T a(T a(T and and come come from from Eqs. Eqs. (3.45) (3.45) and and (3.46), (3.46), where where α(T α(T = = in in r r and P from Table B.1 converted to (R) and (atm), Eq. (3.45). With values of T c andTPc from 581.67 Eq. (3.45). With values of T Table B.1 converted to (R) and (atm), from converted Eq. = =B.1 1.695 and andPPPcccc= from fromTable Table Table B.1 B.1 converted convertedto to to(R) (R) (R)and and and(atm), (atm), (atm), Eq. Eq.(3.45). (3.45). (3.45).With With Withvalues values valuesof of ofTTTcccrcand Tc 343.1 T 581.67 581.67 Tr = = TTTT = = 581.67 = 1.695 1.695 581.67 581.67 T = TTTrr = = = 343.1 −1/2 2 (343.1) 2 = TTTccc(0.7302) = = 343.1 = =1.695 1.695 1.695 343.1 rr = (1.695) 6 TTcc 343.1 343.1 a = 0.42748 = 453.94(atm)(ft) (pie)6 −1/2 2 2 45.4 (1.695)−1/2 (0.7302)22 (343.1) (343.1)22 −1/2 (0.7302) (1.695) −1/2(0.7302) 22(343.1) a= = 0.42748 0.42748(1.695) = 453.94(atm)(ft) 453.94(atm)(ft)666 (1.695) (1.695)−1/2 (0.7302) (0.7302) (343.1) (343.1)22 = = (0.7302)(343.1) 66 aaaa= 0.42748 453.94(atm)(ft) 45.4 33 = = 0.42748 0.42748 = = 453.94(atm)(ft) 453.94(atm)(ft) 45.4 = 0.4781(ft) b = 0.08664 45.4 (pie) 45.4 45.4 45.4 (0.7302)(343.1) 3 (0.7302)(343.1) (0.7302)(343.1) = 0.4781(ft) 0.4781(ft) b= = 0.08664 0.08664 3333 (0.7302)(343.1) (0.7302)(343.1) Substitution of numerical values into 45.4 the Redlich/Kwong equation now yields: = = 0.4781(ft) bbbb= 0.08664 = = 0.4781(ft) 0.4781(ft) = = 0.08664 0.08664 45.4 45.4 Sustituyendo los valores numéricos en la ecuación de Redlich/Kwong, se obtiene: 45.4 45.4 Substitution of of numerical numerical values into into the the Redlich/Kwong Redlich/Kwong equation now now yields: yields: (0.7302)(581.67) 453.94 Substitution values equation Substitution now −the =equation 187.49(atm) Substitution SubstitutionPof of of=numerical numerical numericalvalues values valuesinto into into the theRedlich/Kwong Redlich/Kwong Redlich/Kwong equation equation now nowyields: yields: yields: 2 − 0.4781 (2)(2453.94 + 0.4781) (0.7302)(581.67) (0.7302)(581.67) 453.94 453.94 P= = (0.7302)(581.67) − = 187.49(atm) 187.49(atm) (0.7302)(581.67) (0.7302)(581.67) 453.94 453.94 P − = = − 2− − 0.4781 0.4781 (2)(2 + 0.4781) 0.4781) = (c) BecausePPPthe here is high, the generalized compressibility-factor cor= = pressure − −(2)(2 = =187.49(atm) 187.49(atm) 187.49(atm) (2)(2 + 2222− 0.4781 + 0.4781) − 0.4781 0.4781 (2)(2 (2)(2 + +of0.4781) 0.4781) relation is the proper − choice. In the absence a known value for Pr , an iterative (c) Because Because the the pressure pressure here here is is high, high, the the generalized generalized compressibility-factor compressibility-factor corcor(c) (c) Because here is high, the compressibility-factor corc) Ya que este caso lapressure presión alta, elección apropiada es la correlación del factor procedure isthe based on thees following equation: (c) (c)en Because Because the the pressure pressure here here is isla high, high, the thegeneralized generalized generalized compressibility-factor compressibility-factor corcor-de com, an iterative relation is the proper choice. In the absence of a known value for P r , an iterative relation isausencia the proper proper choice. In the absence absence of a known value for P r , an iterative relation is the choice. In the of a known value for P presibilidad. En de un valor conocido para P , el procedimiento iterativo se apoya an iterative iterative en la relation relation is is is the thebased proper proper choice. In In the the absence absence of ofr aa known known value value for for PPrrr,, an procedure onchoice. the following equation: Z RT Z (0.7302)(581.67) procedure is based based on on the following equation: procedure is the following equation: siguiente ecuación: =the = = 212.4 Z procedure procedure is is based basedPon on the following following equation: equation: V 2 Z RT Z (0.7302)(581.67) RT = ZZ(0.7302)(581.67) (0.7302)(581.67) = 212.4 Z P= = ZZZZRT RT RT = ZZ(0.7302)(581.67) (0.7302)(581.67) P = = 212.4 212.4 ZZ = V 2 becomes: = ,=this equation Because P = Pc P r= PPr = = = 45.4P = = 212.4 212.4 ZZ V VVV 2222 Pr = = 45.4P 45.4P , this this equation equation becomes: becomes: Because P P= =P Pc P 45.4P Z Because r rrr,, this equation becomes: Ya que Because P = PcPr PP =P = 45.4 esta ecuación Z = or Pr = = Pr Prrr,r = = =45.4P 45.4P 45.4P this thisserá: equation equation becomes: becomes: Because Because = = PP PccccPPP rr,,0.2138 212.4 0.2138 45.4Pr Z 45.4P Z= = 45.4P or Pr = = ZZZZ = 0.2138 0.2138 P Pr rrrr = 45.4P 45.4P Z or P r r Z = or P = = 0.2138 P 212.4 0.2138 One now assumes a starting value for Z , say gives Pr = 4.68, and ZZ = = 212.4 or orZ = 1. PPrrrThis = = 0.2138 = = 0.2138 0.2138 PPrrr o 212.4 0.2138 212.4 0.2138 0.2138 allows a new value 212.4 of Z to be calculated by Eq. (3.57) from values interpolated = 4.68, 4.68, and and One now now assumes assumes aa starting starting value value for for ZZ,, say say ZZ = = 1. 1. This This gives gives P Pr = One r = 4.68, One now assumes a starting value for Z , say Z = 1. This gives PPPrWith in Tables E.3 and E.4 at the reduced temperature of T = 1.695. this and new r = = 4.68, 4.68, and and One One now now assumes assumes a a starting starting value value for for Z Z , , say say Z Z = = 1. 1. This This gives gives r r allows a new value of Z to be calculated by Eq. (3.57) from values interpolated Se puede suponer un valor para Z, digamos Z =the 1. Ésta da Pvalues =continues 4.68, y permite rvalues allows new value of Zinicial Z of to P be calculated by and Eq. (3.57) from interpolated allows aaaanew of to be calculated by Eq. (3.57) from interpolated calculated, procedure until nocalcular value of Znew , a value new value r is allows allows new value value of of Z Z to to be be calculated calculated by by Eq. Eq. (3.57) (3.57) from from values values interpolated interpolated in Tables E.3 and E.4 at the reduced temperature of T = 1.695. With this new r con la ecuación un valor de Z astep partir de los valores interpolados lasfound tablas E.3 y in Tables(3.57) E.3 and E.4 atnuevo the reduced temperature ofThe = 1.695. With this new in E.3 and at the reduced temperature of TTTTrr = 1.695. new significant change occurs from one to the next. final valueWith of Zenthis so in inTables Tables Tables E.3 and andE.4 E.4 E.4 at atof the the reduced temperature temperature of of = 1.695. 1.695. With With this this new new rr = is calculated, calculated, and the procedure continues until no recienvalue of ZZE.3 , aareducida new value Prreduced E.4 a lavalue temperatura de T = 1.695. Con este valor nuevo de Z se calcula un valor r is and the procedure continues until no value of , new value of P calculated, and the continues until no of ZZat ,,,aaP new PPPrr is is 0.890 = value 4.14. of This may be confirmed byprocedure substitution into Eq. (3.57) rnew is is calculated, calculated, and and the the procedure procedure continues continues until until no noun paso value value of of Z a new value value of of r r significant change occurs from one step to the next. The final value of Z so found te de Prsignificant ,of y el procedimiento continúa queE.3 no ocurra cambio de significant change occurs fromhasta one step to the the next. The final final value of so found change occurs one step to next. The value ZZZZ so found Z 1 from Tables and E.4ningún interpolated atsignificativo Pof 4.14 and values for Z 0 and r = significant significant change change occurs occurs from from one one step step to to the the next. next. The The final final value value of of so so found found is 0.890 at P = 4.14. This may be confirmed by substitution into Eq. (3.57) r al siguiente. valor de=Z0.012, que se may encuentra es de 0.890 Pr = 4.14. Éste se(3.57) puede confirisTr0.890 0.890 at P PWith = 4.14. This may be be confirmed confirmed by para substitution into Eq. (3.57) rfinal is at = 4.14. This by substitution into Eq. =El1.695. ω 0=and is isla0.890 0.890 at at 4.14. 4.14. This This (3.57) may may be be confirmed confirmed by substitution substitution into into Eq. (3.57) (3.57) 0 y Zat 1 de rZ0en Z111 from from Tables E.3 and E.4 by interpolated P =Eq. 4.14 and of values forPPrrZ 0= r las mar porof sustitución la ecuación de los valores para Z tablas E.3 y E.4, of values for and Z Tables E.3 and E.4 interpolated at P = 4.14 and r ZZZ11 from Tables E.3 and E.4 interpolated at PPPr = 4.14 and values for ZZZ00 and and and from from Tables Tables E.3 E.3 and and E.4 E.4 interpolated interpolated at at = = 4.14 4.14 and and of of values values for for r r = 1.695. With ω = 0.012, T 0 1 r interpolados en P = 4.14 y T = 1.695. Con w = 0.012, = 1.695. With ω = 0.012, T r r + ωZ = 0.887 + (0.012)(0.258) = 0.890 Z = Z r TTTr = = =1.695. 1.695. 1.695.With With Withωωω===0.012, 0.012, 0.012, rr 0 1 + ωZ ωZ(0.890)(0.7302)(581.67) = 0.887 0.887 + + (0.012)(0.258) (0.012)(0.258) = = 0.890 0.890 =ZZZRT 00 + 11 = ZZ = = + +ωZ ωZ ωZ11= = =0.887 0.887 0.887+ + +(0.012)(0.258) (0.012)(0.258) (0.012)(0.258) = =0.890 0.890 0.890 = = ZZZ00+ PZZZ== = = 189.0(atm) V 2 Z RT (0.890)(0.7302)(581.67) RT = (0.890)(0.7302)(581.67) (0.890)(0.7302)(581.67) = 189.0(atm) ZZZZRT P= = RT RT factor (0.890)(0.7302)(581.67) P = (0.890)(0.7302)(581.67) = 189.0(atm) 189.0(atm) compressibilBecause the acentric is small, the P = = V 2 two- and three-parameter PP = = = V = = = = 189.0(atm) 189.0(atm) V 2222 Both the Redlich/Kwong ity-factor correlations are little different. equation and V V Because the acentric factor is small, the twoand three-parameter compressibilBecause the acentric factor is small, small, the twotwo- and and three-parameter compressibilthe acentric factor is the three-parameter compressibiltheBecause generalized compressibility-factor correlation give answers close to the exper-con dos Porque el factor acéntrico es pequeño, las correlaciones del factor de compresibilidad Because Because the the acentric acentric factor factor isdifferent. small, small, the theBoth twotwo-the and andRedlich/Kwong three-parameter three-parameter compressibilcompressibility-factor correlations are littleis equation and ity-factor correlations are little different. Both the Redlich/Kwong equation and ity-factor correlations are little different. Both the Redlich/Kwong equation and imental value of 185(atm). The ideal-gas equation yields a result that is high by y tres parámetros difieren un poco. Tanto la ecuación de Redlich/Kwong como la correlación ity-factor ity-factor correlations correlations are are little little different. different. Both Both the the Redlich/Kwong Redlich/Kwong equation equation and and the generalized compressibility-factor correlation give answers close to the experthe generalized compressibility-factor correlation respuestas give answers answers close to the the experexperthe generalized compressibility-factor correlation give close to 14.6%. generalizada del factor de compresibilidad proporcionan muy próximas al valor the the generalized generalized compressibility-factor correlation correlation give give answers answers close close to tois the thehigh experexperimental value of ofcompressibility-factor 185(atm). The The ideal-gas ideal-gas equation yields result that by expeimental value 185(atm). equation yields aa result result that is high high by value of 185(atm). The ideal-gas equation yields a that is by rimentalimental de 185(atm). La ecuación del gas ideal produce un resultado que es mayor en 14.6%. imental imental value value of of 185(atm). 185(atm). The The ideal-gas ideal-gas equation equation yields yields aa result result that that is is high high by by 14.6%. 14.6%. 14.6%. 14.6%. 14.6%. 03-SmithVanNess.indd 107 8/1/07 13:00:06 108 108 108 108 108 CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3.3.Volumetric Properties of Pure Example 3.12 Ejemplo 3.12 3.12 Example Example 3.12 3 vessel immersed A mass mass of g500 500 g of of gaseous gaseous ammonia is contained contained in aarecipiente 30,000-cm 3 sumergido en Una masa de 500 de g amoniaco gaseoso está contenida en in un de33 30 000 cm A of ammonia is 30,000-cm vessel immersed ◦ Example 3.12 A mass of 500 g of gaseous ammonia is contained in a 30,000-cm vessel immersed in a constant-temperature bath at 65 C. Calculate the pressure of the gas by: ◦ un baño a temperatura constante de 65 °C. Calcule la presión del gas mediante: in a constant-temperature bath at 65◦ C. Calculate the pressure of the gas by: 3 vessel in mass a constant-temperature bath at 65 C.is Calculate of the gas by: A of 500 g of gaseous ammonia containedthe in apressure 30,000-cm immersed (a) The ideal-gas equation; (b) A generalized correlation. ◦ a) in Laaecuación del gas ideal; b) unaat(b) correlación generalizada. (a) The ideal-gas equation; correlation. constant-temperature bath 65 AC.generalized Calculate the pressure of the gas by: (a) The ideal-gas equation; (b) A generalized correlation. (a) The ideal-gas equation; (b) A generalized correlation. Solución 3.12 3.12 Solution Solution 3.12 Solution 3.12 El volumen molar del amoniaco en el recipiente es: is: The molar molar volume volume of ammonia ammonia in the the vessel vessel The of in is: Solution 3.12 The molar volume oft ammonia in the vessel is: t V 30,000 V t t −1 V 30,000 V The molar volume oft ammonia vessel is:= 1,021.2 cm33 mol−1 = V t in =the30,000 =V = = = 1,021.2 cm3 mol−1 VV = n m/M 500/17.02 = 500/17.02 = 1,021.2 cm mol V = nt = m/M Vt 30,000 Vn m/M 500/17.02 = = = 1,021.2 cm3 mol−1 V gas = ideal, (a) By the the ideal-gas ideal-gas equation, a) Por la(a) ecuación del By nequation, m/M 500/17.02 (a) By the ideal-gas equation, (83.14)(65 + + 273.15) 273.15) RT RT (a) By the ideal-gas equation, = (83.14)(65 = 27.53 27.53 bar bar = = = PP = (83.14)(65 + 273.15) RT 1,021.2 1,021.2 = 27.53 bar P = VV = (83.14)(65 + 273.15) RT V 1,021.2 = = 27.53 bar = ≈ 27.53/112.8 27.53/112.8 0.244), the the genergener(b) Because Because the the P reduced pressure is low low (P (Pr ≈ == 00.244), (b) reduced pressure is V 1,021.2 r b) Ya que la presión reducida es menor (P ≈ 27.53/112.8 = 0.244), la correlación generalizada del 1the r ≈ 27.53/112.8 = 0.244), gener(b) Because the reduced pressure is low (P alized virial-coefficient correlation should suffice. Values of B and B are given 0 1 r 0 y BValues 1 se conocen alized virial-coefficient correlation shouldde suffice. of B 0 por and las B 1ecuaciones are given coeficiente virial debe ser suficiente. Los valores B (3.65) alized virial-coefficient correlation should Values of B and B the aregenergiven by Eqs. Eqs. (3.65) and (3.66), with TTr is = 338.15/405.7 = 0.834: 0.834: = 0.244), (b) Because theand reduced pressure low (Psuffice. r ≈ 27.53/112.8 (3.66), = r = 338.15/405.7 y (3.66),by Tr (3.65) = 338.15/405.7 = with 0.834: 0 and B 1 are given bycon Eqs. (3.65) and (3.66), with Tr =should 338.15/405.7 = 0.834: alized virial-coefficient correlation suffice. Values of B 0 = −0.482 B11 == −0.232 0= by Eqs. (3.65) and (3.66), with Tr = 338.15/405.7 0.834: BB00 B= −0.482 −0.232 –0.482 BB1 1= = –0.232 B = −0.482 B = −0.232 0 Substitution intoen Eq.la(3.63) (3.63) with(3.63) ω == 0.253 0.253 yields: Becuación = −0.482 B 1 = −0.232 Sustituyendo w = 0.253 se obtiene: Substitution into Eq. with ω yields: Substitution into Eq. (3.63) with ω = 0.253 yields: = −0.482 −0.482 + (0.253)(−0.232) = −0.541 −0.541 Substitution into Eq. with+ ω (0.253)(−0.232) = 0.253 yields: = B̂B̂ (3.63) = B̂ = −0.482 + (0.253)(−0.232) = −0.541 −(0.541)(83.14)(405.7) B̂RT RT B̂cc = −0.482 + (0.253)(−0.232) = −0.541 3 −1 = −161.8 −161.8 cm cm3 mol = −(0.541)(83.14)(405.7) = mol−1 = B̂ BB = B̂ RT c = −(0.541)(83.14)(405.7) P 112.8 3 −1 c 112.8 = −161.8 cm mol B = Pc = B̂ RT Pfor 112.8 c c P: −(0.541)(83.14)(405.7) Solve Eq. (3.38) = −161.8 cm3 mol−1 = = for P: Solve Eq.B(3.38) 112.8 c P: Solve Eq. (3.38)Pfor (83.14)(338.15) RT(3.38):(83.14)(338.15) Resolviendo P de lafor ecuación Solvepara Eq. (3.38) P: = (83.14)(338.15) = = 23.76 bar bar = RT = PP = RT − BB = 1,021.2 1,021.2 + + 161.8 161.8 = 23.76 23.76 bar P = VV − (83.14)(338.15) VRT −B 1,021.2 + 161.8 = 23.76 bar of pressure. The P =is not necessary, An iterative iterative solution solution because BB is is=independent An is Vnot−necessary, because B 1,021.2 + 161.8 independent of pressure. The An iterative solution is not necessary, because B is independent of pressure. The calculated P corresponds to a reduced pressure of P = 23.76/112.8 23.76/112.8 = 0.211. 0.211. r calculated P corresponds to a reduced pressure of Pr = = calculated P corresponds to a reduced pressure of P = 23.76/112.8 = 0.211. Reference to Fig. 3.14 confirms the suitability of the generalized virial-coefficient r An iterative solution is not necessary, because B is independent of pressure. The Referenceencontrar to Fig. 3.14 suitability of theBgeneralized virial-coefficient No es necesario unaconfirms solucióntheiterativa, porque es independiente de la presión. El Reference 3.14 confirms the suitability of of the virial-coefficient correlation. calculated PP Fig. corresponds to a reduced pressure 23.76/112.8 0.211. r r= correlation. valor calculado deto corresponde a una presión reducida dePgeneralized P = 23.76/112.8 ==0.211. La figura correlation. Experimental data indicate that the pressure is 23.82 bar at the given condiReference to Fig. 3.14 confirms the suitability of the generalized virial-coefficient 3.14 confirma que el uso de la indicate correlación coeficiente es apropiado. Experimental data thatgeneralizada the pressuredel is 23.82 bar atvirial the given condiExperimental data indica indicate that the pressure 23.82 barpara at the given condi- conocitions. Thus the ideal-gas ideal-gas equation yields an es answer highbar by about 15%, whereas correlation. Información experimental que layields presión deis23.82 las15%, condiciones tions. Thus the equation an answer high by about whereas tions. Thus the ideal-gas equation yields an answer high by about 15%, whereas the virial-coefficient correlation gives an answer in substantial agreement with exExperimental data indicate that the pressure is 23.82 bar at the given condidas. Dethe estevirial-coefficient modo, la ecuación del gas gives ideal an produce respuesta que es mayor enexcasi 15%, correlation answeruna in substantial agreement with the virial-coefficient correlation gives an answer in substantial agreement with experiment, even though ammonia is a polar molecule. tions. Thus the ideal-gas equation yields an answer high by about 15%, whereas mientrasperiment, que la correlación delammonia coeficiente even though is avirial polarproporciona molecule. una respuesta de conformidad conperiment, even though ammonia is el a polar molecule. the virial-coefficient correlation gives an answer inuna substantial agreement siderable con el experimento, aun cuando amoniaco es molécula polar. with experiment, even though ammonia is a polar molecule. 03-SmithVanNess.indd 108 8/1/07 13:00:23 3.7. Generalized Generalized Correlations Correlations for for Liquids Liquids 3.7. 3.7. Generalized Generalized Correlations for Liquids Liquids 3.7. Correlations 3.7. Correlaciones generalizadas para for líquidos 3.7 109 109 109 109 109 3.7 CORRELATIONS FOR 3.7 GENERALIZED GENERALIZED CORRELATIONSPARA FORLIQUIDS LIQUIDS CORRELACIONES GENERALIZADAS LÍQUIDOS Although the molar molar volumes oflíquidos liquids can can be calculated by means meanspor of medio generalized cubic equaequathe volumes liquids be calculated by of generalized cubic AunqueAlthough los volúmenes molares de losof son posibles de calcularse de ecuaciones cúbicas de Although the molar molar volumes of liquids can be calculated by means means of generalized cubic equaequaAlthough the volumes of liquids can be calculated by of generalized cubic tions of state, the results are often not of high accuracy. However, the Lee/Kesler correlation tions of state, the results are often not of high accuracy. However, the Lee/Kesler correlation estado, con frecuencia sonnot muy De cualquier modo, correlación de Lee/Kesler tions of state, state, los the resultados results are areno often not ofexactos. high accuracy. accuracy. However, the la Lee/Kesler correlation tions of the results often of high However, the Lee/Kesler correlation includes data para for subcooled subcooledsubenfriados, liquids, and and Fig. Fig.la3.13 3.13 illustrates illustrates curves for both both liquids liquidslíquidos and gases. gases. data for liquids, curves for and incluyeincludes información 3.13 ilustra curvas como para includes data forlíquidos subcooled liquids, and andyFig. Fig.figura 3.13 illustrates illustrates curves fortanto bothpara liquids and gases. gases. includes data for subcooled liquids, 3.13 curves for both liquids and Values for both phases are provided in Tables E.1 through E.4. Recall, however, that thisque esta Values for both phases are provided in Tables E.1 through E.4. Recall, however, that gases. Los valores ambas fases proporcionan en lasE.1 tablas E.1 aE.4. E.4. Recall, No obstante, recuerde Values forde both phases areseprovided provided in Tables Tables E.1 through E.4. Recall, however, thatthis this Values for both phases are in through however, that this correlation is most most suitable suitable for nonpolar and slightly slightly polar fluids. fluids. correlation is nonpolar and polar correlación es la más parafor fluidos no polares y ligeramente polares. correlation isadecuada most suitable suitable for nonpolar and slightly slightly polar fluids. fluids. correlation is most for nonpolar and polar Inestán addition, generalized equations are available available forlathe the estimation estimation of molar molar volumes volumes ofde líquiIn addition, generalized equations are for of of Además, disponibles ecuaciones generalizadas para de volúmenes molares In addition, generalized equations are available available forestimación the22estimation estimation of molar molar volumes volumes of In addition, generalized equations are for the of of 22 saturated liquids. The simplest equation, proposed by Rackett, is an example: 22 es un ejemplo: 22 saturated simplest proposed byby Rackett, is is anan example: dos saturados. Laliquids. ecuación más simple,equation, propuesta por Rackett, saturated liquids.The The simplest equation, proposed by Rackett,22 is an example: saturated liquids. The simplest equation, proposed Rackett, example: 2/7 sat (1−Trr ))2/72/7 =V V Z Z (1−T (3.72) V sat sat (1−Trr))2/7 = (3.72) V V saturado = ccV VccccZZc(1−T (3.72)(3.72) V sat = (3.72) V c An alternative form of this equation is sometimes useful: An alternative form ofof this equation is is sometimes useful: An alternative form of this equation sometimes useful: An alternative this equation sometimes En ocasiones es útil unaform forma alternativa de is esta ecuación:useful: P 2/7 ] rP [1+(1−T sat )2/7 ] ]] 2/7 rr ) ))2/7 Z = PrP Zrr cc[1+(1−T (3.73) sat [1+(1−T [1+(1−T Z sat = Z (3.73) rr =T Z (3.73)(3.73) ZZsat = Z (3.73) Z saturado Trr cc TTrr The only data required are the critical constants, given in Table B.1. Results are usually accuThe only data required are the critical constants, given inin Table B.1. Results are usually accuLa única información que se requiere son las constantes críticas, conocidas en la tabla B.1. Por lo regular los The only data required are the critical constants, given in Table B.1. Results are usually accuThe only data required are the critical constants, given Table B.1. Results are usually accurate to 1 or 2%. rate to 1 or 2%. resultadosrate tienen exactitud de 1 o 2%. rate to 11una or 2%. 2%. to or 23 developed a two-parameter corresponding-states Lydersen, Greenkorn, and Hougen23 23 desarrollaron 23 Lydersen, Greenkorn, and developed a aatwo-parameter corresponding-states Lydersen, Greenkorn y Hougen una correlación de estados correspondientes con dos Lydersen, Greenkorn, andHougen Hougen23 developed two-parameter corresponding-states Lydersen, Greenkorn, and Hougen developed two-parameter corresponding-states correlation for estimation of liquid volumes. It provides a correlation of reduced density ρrr as as reducorrelation for estimation of liquid volumes. It provides a correlation of reduced density ρ parámetros para la estimación del volumen líquidos. Ésta proporciona una of correlación de densidad correlation for estimation estimation of liquid liquidde volumes. It provides provides correlation of reduced reduced density as correlation for of volumes. It aa correlation density ρρrr as a function of reduced temperature and pressure. By definition, a function of reduced temperature and pressure. By definition, cida r r como una función de latemperature temperaturaand y lapressure. presión reducidas. Por definición, function of reduced reduced temperature and pressure. By definition, definition, aa function of By ρ Vc Vcc ρrr ≡ ≡ ρ ρρ= = VcV (3.74) ρ (3.74) (3.74)(3.74) ≡ρ =V ρρrr ≡ (3.74) ρcc = V ρ V ρcc V donde rwhere en el punto correlación generalizada se muestra en laby figura is the the density density at the the crítico. critical La point. The generalized generalized correlation is shown shown by Fig. 3.16. 3.16. Ésta es c es laρ where ρdensidad at critical point. correlation is Fig. 3.16. ccρ where ρis is the the density density at the the critical point.The The generalized generalized correlation is shown shown by Fig. 3.16. where at critical point. The correlation is by Fig. 3.16. cc is posible This usarla de manera directa con la ecuación (3.74) para determinar los volúmenes de los líquidos This figure may be used directly with Eq. (3.74) for determination of liquid volumes if the si se figure may be used directly with Eq. (3.74) for determination of liquid volumes if the This figure may be used directly with Eq. (3.74) for determination of liquid volumes if the This figure may be used directly with Eq. (3.74) for determination of liquid volumes if the conoce value el valor crítico.is mejor procedimiento consiste usar use un volumen líquido conocido value of del the volumen critical volume volume isUn known. A better better procedure procedure is to toenmake make use of aa single single known of the critical known. A is of known value of the critical volume is known. A better procedure is to make use of a single known value of the critical volume is known. A better procedure is to make use of a single known (estado liquid 1) y emplear identidad, liquid volumela(state (state 1) by by the the identity, identity, volume 1) liquid volume (state (state 1) by by the the identity, identity, liquid volume 1) ρr11 (3.75) =V V ρrρρ (3.75) V22 = rr11 (3.75) V = 11V Vρ (3.75) V22 = (3.75) V ρ11rr22 ρ ρrr22 = required required volume volume V where 2V= V where 2 required volume volume V22 == required where donde where V2 = volumen requerido V = = known known volume volume V11 == known volume V known volume conocido V1 =V11volumen ρ 11 ,, ρ ρrr22 = = reduced reduced densities densities read read from from Fig. Fig. 3.16 3.16 = reduced reduced densitiesleídas read from from Fig. 3.16 3.16 ,, ρρ densities read r r1,ρrrrρρ densidades reducidas de la Fig. figura 3.16 rr22 = rrr211= This method method gives gives good good results results and and requires requires only only experimental experimental data data that that are are usually usually available. available. This This method method gives gives good good results results and and requires requires only only experimental experimental data data that that are are usually usually available. available. This Este método da buenos resultados y sólo requiere de información experimental que usualmente está Figure 3.16 makes clear the increasing effects of both temperature and pressure on liquid den-disponiFigure 3.16 makes clear the increasing effects ofof both temperature and pressure onon liquid denFigure 3.16 makes clear the increasing effects of both temperature and pressure on liquid denFigure 3.16 makes clear the increasing effects both temperature and pressure liquid denble. La sity figura 3.16 muestra de forma clara los efectos al aumentar la temperatura y la presión sobre la densisity as the critical point is approached. asas the critical point is is approached. sity as the critical point is approached. sity the critical point approached. dad del líquido conforme se acerca al punto crítico. Correlations for the molar densities as functions of temperature are given for many pure Correlations forfor the molar densities asas functions ofof temperature are given forfor many pure Correlations for the molar densities as functions of temperature are given for many pure Correlations the molar densities functions temperature are given many pure 24 24 Las correlaciones para lascoworkers. densidades molares como funciones de la temperatura se conocen para varios liquids by Daubert Daubert and coworkers. 24 24 liquids by and liquids by by Daubert Daubert and and coworkers. coworkers. 24 liquids líquidos puros por Daubert y colaboradores. 22 H. G. Rackett, J. Chem. Eng. Data, vol. 15, pp. 514–517, 1970; see also C. F. Spencer and S. B. Adler, ibid., 22 H. G.G. Rackett, J. J. Chem. Eng. Data, vol. 15,15, pp.pp. 514–517, 1970; seesee also C.C. F. F. Spencer and S. S. B.B. Adler, ibid., 22 H. 22 G. Rackett, J. Chem. Chem. Eng. Data, vol. 15, pp. 514–517, 1970; see also C. F. Spencer and S. B. Adler, ibid., Rackett, Eng. Data, vol. 514–517, 1970; also Spencer and Adler, ibid., vol. 23,H.pp. pp. 82–89, 82–89, 1978, for aa review review of available available equations. vol. 23, 1978, for of equations. vol. 23, pp. 82–89, 1978, for a review of available equations. vol. 23, pp. 82–89, 1978, for a review of available equations. 22 H. G. Rackett, J. Chem. Eng. Data, vol. 15, pp. 514-517, 1970; véase también C. F. Spencer y S. B. Adler, ibid., vol. 23, pp. 82-89, 23 A. L. Lydersen, R. A. Greenkorn, and O. A. Hougen, “Generalized Thermodynamic Properties of Pure Fluids,” 23 A. L.L. Lydersen, R.R. A.A. Greenkorn, and O.O. A.A. Hougen, “Generalized Thermodynamic Properties of of Pure Fluids,” 23A. 23 A. L. Lydersen, R. A. Greenkorn, and O. A. Hougen, “Generalized Thermodynamic Properties of Pure Fluids,” Lydersen, Greenkorn, and Hougen, “Generalized Thermodynamic Properties Pure Fluids,” 1978, para una Wisconsin, revisión deEng. las ecuaciones disponibles. Univ. Expt. Sta. Rept. 4, 1955. 1955. Univ. Wisconsin, Eng. Expt. Sta. Rept. 4, Univ. Wisconsin, Eng. Expt. Sta. Rept. 4, 1955. Wisconsin, Eng. Expt.ySta. Rept. 4, 1955. 23 A. L. Univ. Lydersen, R. A. Greenkorn O. A. Hougen, “Generalized Thermodynamic Properties of Pure Fluids”, Univ. Wisconsin, Eng. 24 T. 4, 24 E. 1955. Daubert, R. R. P. P. Danner, Danner, H. H. M. M. Sibul, Sibul, and and C. C. C. C. Stebbins, Stebbins, Physical Physical and and Thermodynamic Thermodynamic Properties Properties of of Pure Pure Expt. Sta. Rept. T. E. Daubert, 24T. 24 T. E. Daubert, R. R. P. P. Danner, Danner, H. H. M. M. Sibul, Sibul, and and C. C. C. C. Stebbins, Stebbins, Physical Physical and and Thermodynamic Thermodynamic Properties Properties of of Pure Pure E. Daubert, Chemicals: Data Compilation, Taylor &yFrancis, Francis, Bristol, PA, extantand 1995. 24 T. Chemicals: Data Compilation, Taylor & Bristol, PA, extant 1995. E.Chemicals: Daubert, R. P. Danner, H. M. Sibul C. C. Stebbins, Physical Thermodynamic Properties of Pure Chemicals: Data Chemicals: Data Data Compilation, Compilation, Taylor Taylor & & Francis, Francis, Bristol, Bristol, PA, PA, extant extant 1995. 1995. Compilation, Taylor & Francis, Bristol, PA, existente 1995. 03-SmithVanNess.indd 109 8/1/07 13:00:32 110 CHAPTER 3.3.Volumetric Properties of Pure CAPÍTULO Propiedades volumétricas deFluids fluidos puros CHAPTER 3. Volumetric Properties of Pure Fluids 110 110 3.5 3.5 3.5 TrT�0.3 0.3 rr 3.0 3.0 3.0 2.5 2.5 2.5 �r r �r 2.0 2.0 2.0 Tr 0.95 Trr � 0.95 Tr � 0.95 1.0 0.97 0.97 0.99 0.97 0.99 0.99 1.5 1.5 1.5 1.0 1.0 0 0 1.0 0 Tr0.4 � 0.40.3 0.5 0.50.4 0.6 0.60.5 0.7 0.70.6 0.8 0.7 0.8 0.9 0.8 0.9 1.0 0.9 1.0 Líquido saturado Saturated Liquid Saturated Liquid 1 1 1 2 2 2 3 3 3 4 4 4 5 5 Pr Pr 5 Pr 6 6 6 7 7 7 8 8 8 9 9 9 10 10 10 Figura 3.16: Correlación de densidad generalizada para líquidos. Figure 3.16: Generalized density correlation for liquids. Figure 3.16: Generalized density correlation for liquids. Example Ejemplo 3.13 3.13 Example 3.13 For ammonia at 310 K, estimate the density of: Para el amoniaco a 310 K, calcule la densidad de: For ammonia at 310 K, estimate the density of: (a) The saturated liquid; (b) The liquid at 100 bar. a) El líquido saturado; b) El liquid; líquido (b) a 100 (a) The saturated Thebar. liquid at 100 bar. Solution Solución 3.13 3.13 Solution 3.13 (a) Apply Eq. (3.72) at the reduced temperature, Tr = 310/405.7 = 0.7641. With (3.72) atlathe reduced temperature, = 310/405.7 = 0.7641. With a) Aplique laApply ecuación temperatura reducida, = 0.7641. Con V Vc(a) = 72.47 Eq. and(3.72) Z c =a0.242 (from Table B.1), Tr T=r 310/405.7 C = 72.47 V = 72.47 and Z = 0.242 (from Table B.1), y ZC = 0.242 c (de la tabla B.1), c 2/7 2/7 V sat = Vc Z c(1−Tr ) 2/7= (72.47)(0.242)(0.2359) 2/7= 28.33 cm3 mol−1 saturado V V sat = Vc Z c(1−Tr ) = (72.47)(0.242)(0.2359) = 28.33 cm3 mol−1 For comparison, the experimental value is 29.14 cm3 mol−1 , a 2.7% difference. −1 , a 2.7% difference. For comparison, experimental is 29.14 cm3–1mol Para comparación, el valorthe experimental es value de 29.14 cm3 mol , y difiere en 2.7%. (b) The reduced conditions are: b) Las condiciones reducidas son: (b) The reduced conditions are: 100 Tr = 0.764 Pr = 100 = 0.887 Tr = 0.764 Pr =112.8 = 0.887 Sustituyendo el valorthe r rvalue, = 2.38ρ(de figura 3.16), VC 112.8 enand la ecuación (3.74), se gives: obtiene: Substituting 2.38 (from Fig.y3.16), Vc into Eq. (3.74) r =la Substituting the value, ρr = 2.38 (from Fig. 3.16), and Vc into Eq. (3.74) gives: Vc 72.47 V = Vc= 72.47= 30.45 cm3 mol−1 V =ρr =2.38 = 30.45 cm3 mol−1 ρr 2.38 03-SmithVanNess.indd 110 8/1/07 13:00:47 Problems 111 111 Problems Problems 111 Problemas 111 Problems 111 3 −1 In comparison with the experimental value of 28.6 cm mol , this result is higher 3 mol−1 , this result is higher In comparison comparison with the experimental value of 28.6 28.6 cm 3 mol –1,−1 3 mol by 6.5%. con En In comparación elthe valor experimental de of 28.6 cmcm este resultado eshigher mayor en 6.5%. with experimental value , this result 3 mol −1 In comparison with the experimental value of 28.6 cm resultisis higher 3 mol −1 for by 6.5%. 3 –1,,this saturated liquid a 310 If we startcon with the experimental value of 29.14 cm Si comenzamos el valor experimental de 29.14 cm mol para el líquido saturado by 6.5%. −1 for saturated liquid by 6.5%. mol If we start with the experimental experimental value ofthe 29.14 cm33 mol −1=for atwe 310 K, la Eq. (3.75) may be Para used. saturated liquid at T = 0.764, ρ K, se puede usar ecuación (3.75). elFor líquido saturado a T 0.764, r = 2.34 r r(de saturated liquid If start with the value of 29.14 cm 3 mol−1 r for saturated r1 liquid 1 =la figura If we start with the experimental value ofsaturated 29.14 cmliquid at 310 310 K, Eq. (3.75) may be used. For For theknown atEq. Tr (3.75) = 0.764, 0.764, ρr1 = = 2.34 Fig. 3.16). Substitution of intoat gives: 3.16). Con la(from sustitución de los valores conocidos envalues la ecuación (3.75) se obtiene: at K, Eq. (3.75) may be used. the saturated liquid T = ρ at 310 K, Eq. be used. of For the saturated liquid Trr =gives: 0.764, ρrr11 = 2.34 (from Fig.(3.75) 3.16).may Substitution known values into Eq.at (3.75) 2.34 (from Fig. 3.16). Substitution of known values into Eq. (3.75) gives: � � 2.34 (from Fig. 3.16). Substitution of known values into Eq. (3.75) gives: ρr1 2.34 � � � � V2 =ρV = 28.65 cm3 mol−1 = (29.14) 1 2.34 r � � 1 −1 ρ 2.34 ρ 2.38 r r V = 28.65 cm333 mol = V = (29.14) mol−1 1 2 1 ρr1 = (29.14) 2.34 = 28.65 cm VV22 = V 1 ρ 2.38 r = 28.65 cm mol−1 2 = V1ρr 2 = (29.14) 2.38 ρr22 agreement En esencia, resultado concuerda con el 2.38 valorthe experimental. This este result is in essential with experimental value. 1 interpolados This result is in indirecta essential agreement with thecorrelation experimental value. La aplicación de la Lee/Kesler con los valores de and Z 1 inDirect application ofcorrelación the Lee/Kesler with valuesdeofZ 0Z y0 Z This result is essential agreement with the experimental value. 0 and 1 inThis result is in essential agreement with the experimental value. 3 –1 3 −1 Z Direct application of the Lee/Kesler correlation with values of Z 1which las tablas E.1 application y E.2 conduce aE.1 un valor deleads 33.87tocm mol ,33.87 que tiene error importante, sin terpolated from Tables and E.2 a value ofvalues cmZ 0un mol , is and Z inDirect of the Lee/Kesler correlation with of 0 and Z 1 in3 of Directafrom application of the Lee/Kesler values Z −1 terpolated Tables E.1 and E.2owing leads to value ofwith 33.87 cm mol which is is 3 mol duda, debido la naturaleza altamente polar correlation del amoniaco. significantly in error, noand doubt toaathe highly polar cm nature of−1 ammonia. terpolated from Tables E.1 E.2 leads to value of 33.87 ,,,which 3 −1 terpolated from Tables E.1 andowing E.2 leads to highly a valuepolar of 33.87 cmofmol which is significantly in error, no doubt to the nature ammonia. significantly significantlyin inerror, error,no nodoubt doubtowing owingto tothe thehighly highlypolar polarnature natureof ofammonia. ammonia. PROBLEMAS PROBLEMS PROBLEMS PROBLEMS PROBLEMS 3.1. Exprese la expansión volumétrica y la and compresibilidad isotérmica como funciones de laof densidad 3.1. Express the volume expansivity the isothermal compressibility as functions –6 –1. ¿A ◦k −6 presión 3.1. Express the volume expansivity and the isothermal compressibility as functions of r y sus derivadas parciales. Para el agua a 50 °C y 1 bar, = 44.18 × 10 bar qué density ρ and its partial derivatives. For water at 50 C and 1 bar, κ = 44.18 × 10 3.1. the volume expansivity and the isothermal compressibility as functions of −6 3.1. Express Express−1 the volume expansivity andFor thewater isothermal as44.18 functions of ◦bar, density and its partial derivatives. at 50 50◦◦C Ccompressibility and 1 bar, = ×que 10−6 debe comprimirse el agua a 50must °C para que densidad cambie Suponga k−6esbyindepenbar ρρ .and To its what pressure water besucompressed at 50 C1%? toκκ change its× density density partial derivatives. For water at and 1 = 44.18 10 ◦ −1 ◦ density ρ and its partial derivatives. For water at 50 C and 1 bar, κ = 44.18 × 10 bar−1 To what pressure pressure must water be beofcompressed compressed at 50◦ C C to change change its density density by diente de P. 1%? Assume that κ ismust independent P. bar ... To bar−1Assume To what what pressure must water water be compressed atat 50 50◦ C to to change its its density by by 1%? that κ is independent of P. 1%? 1%? Assume Assumethat thatκκ isisindependent independentof of P. P. 3.2. Por general, elvolume coeficiente de expansión b y la compresibilidad isotérmica k depen3.2.loGenerally, expansivity β andvolumétrica isothermal compressibility κ depend on T and 3.2. Generally, volume expansivity β and isothermal compressibility κ depend on T and den de T y P. Demuestre que: P. Prove that: 3.2. volume compressibility κκ depend � isothermal �compressibility � � 3.2. Generally, Generally, volume expansivity expansivity ββ and and isothermal depend on on TT and and P. Prove that: that: ∂κ � � � �∂β � P. Prove � � � = − P. Prove that: ∂β ∂κ �∂β ∂�P � � =T− − ∂κ ∂β ∂κ ∂ T P = ∂ P ∂ T = − T ∂∂PP T ∂∂TT PP T P 3.3. The Tait equation for liquids is written for an isotherm as: 3.3. The Tait equation for liquids is written for an isotherm as: 3.3. Para una isoterma, la ecuación de Tait para líquidos se escribe 3.3. as:� como: � isotherm 3.3. The TheTait Taitequation equationfor forliquids liquidsisiswritten writtenfor foran an isotherm A� P as: � �� V =��V0 1 − AP PB + A P V = V 1 − 0 A P VV = B+ +P P = VV00 11− −B P hypothetical molar or specific volwhere V is molar or specific volume, V0Bis+the is the hypothetical molaranor orexpression specific volvolwhere V is molar or specific volume, V 0 is ume atel pressure, and A and B V are positive constants. Find for the the hypothetical molar where V isiszero molar specific volume, donde V es volumen o and específico yV0V elconstants. volumen molar o específico hipotético hypothetical molar or specific specific volwhere V molar or ormolar specific volume, 0 0isesthe ume at zero pressure, and A B are positive Find an expression for the a presión isothermal compressibility consistent with this equation. ume at zero pressure, and A and B are positive constants. Find an expression for the cero, que A y B and son constantes positivas. Encuentre una para laforcompresibilidad umemientras at zerocompressibility pressure, A and B are positive constants. Findexpresión an expression the isothermal consistent with this equation. equation. isothermal compressibility consistent with this isotérmica que sea consistente con esta ecuación. isothermal compressibility consistent with this equation. 3.4. For liquid water the isothermal compressibility is given by: 3.4. For liquid liquid water water the the isothermal isothermal compressibility compressibility is is given given by: by: 3.4. For 3.4. agua water líquidathe la isothermal compresibilidad isotérmicaisces conocida 3.4.Para Forelliquid compressibility given by: por: κ= c cc(P + b) V = κκκ = V (P (P + + b) =V V (P +b) b)only. If 1 kg of water is compressed where c and b are functions of temperature where c and b are functions of temperature only. If6011◦ C, kghow of water water iswork compressed muchis is required? isothermally and reversibly from 1 to 500 bar atIf where cyc band bbúnicamente are functions of temperature only. ◦ donde c son funciones de la temperatura. Sikg seof comprime kg de agua de manera where and are functions of temperature only. If 1how kg of water is1compressed compressed ◦ 3 −1 C, much work is required? isothermally and reversibly from 1 to 500 bar at 60 ◦ At 60 C, b = 2, 700 bar and c = 0.125 cm g . how much work isrequiere? required? isothermally and reversibly from 11 to 500 bar at 60 ◦C, ◦ 3 −1 isotérmica y reversible desde 1 hasta 500 bar a 60 °C, ¿cuánto trabajo se A 60 °C, b = C, how much work is required? isothermally and reversibly from to 500 bar at 60 At 60 60◦ C, C, bb = = 2, 2, 700 700 bar bar and and cc = = 0.125 cm cm3 g−1 . ◦ C, 3 gand –1. c = 0.125 2 At 700 bar y bc ==0.125 At 60 2, 700cm bar 0.125 cm3 gg−1.. 03-SmithVanNess.indd 111 8/1/07 13:01:01 112112 CAPÍTULO 3. Propiedades volumétricas de fluidos puros CHAPTER 3. Volumetric Properties of Pure Fluids 3.5. Calcule el trabajo reversible hecho al comprimir 1(pie)3 de mercurio a una temperatura constante de 3 of mercury 3.5.32(°F), Calculate theuna reversible work done in compressing 1(ft) atisotérmica a constantdel mercurio desde presión de 1(atm) hasta 3 000 (atm). La compresibilidad ◦ F) from 1(atm) to 3,000(atm). The isothermal compressibility temperature of 32( of a 32(°F) es: mercury at 32(◦ F) is: κ/(atm)−1 = 3.9 × 10−6 − 0.1 × 10−9 P(atm) 3.6. tetracloruro carbono líquido se asometen a un cambio de estado 3.6.Cinco Five kilogramos kilograms ofdeliquid carbon de tetrachloride undergo mechanically reversible, iso- isobárico, ◦ mecánicamente reversible a 1 bar, durante el cual la temperatura varía de 0 a 20 °C. Determine DV t, baric change of state at 1 bar during which the temperature changes from 0 C to 20◦ C. t t t , and t . The properties W,Determine Q, DH y �V DUt ,. W Suponga las �U siguientes propiedades tetracloruro de carbono líquido a 1 , Q, �Hque fordel liquid carbon tetrachloride –3 K–1, C = 0.84−3 ◦ −1–1, K–1, y r = bar y 0 °C son independientes de la temperatura: b = 1.2 × 10 P at 1 bar and 0 C may be assumed independent of temperature: β = 1.2 × 10 kJKkg –3 −1 K−1 , and ρ = 1,590 kg m−3 . 1C 590 kg m . P = 0.84 kJ kg 3.7. sustancia for para la que una constante, se an somete a un proceso isotérmico, mecánicamente 3.7.Una A substance which κ isk aes constant undergoes isothermal, mechanically reversible reversible del estado inicial (P , V ) al estado final (P , V ), donde V es el volumen 1 1 2 2 process from initial state (P1 , V1 ) to final state (P2 , V2 ), where V is molar volume.molar. the definition κ, show que that la thetrayectoria path of thedel process is described by: por: a) (a)A Starting partir dewith la definición de k of demuestre proceso está descrita VV ==A(T)exp(-k P) P) A(T ) exp(−κ b) (b)Determine una exacta which que proporcione el trabajo work isotérmico hecho Determine an expresión exact expression gives the isothermal done on 1 molsobre of 1 mol de esta sustancia con k constante. this constant-κ substance. 3.8. molmole de gas congas CP with = (7/2)R y C(7/2)R de Pexpands y T1 = V = (5/2)R 1 = 8 barfrom and se C Vexpande = (5/2)R P1600 = K a P2 = 1 3.8.Un One of ideal an ideal CP = bar por cada una de las trayectorias siguientes: 8 bar and T1 = 600 K to P2 = 1 bar by each of the following paths: Constant volume;b)(b) Constant temperature; Adiabatically. a)(a) Volumen constante; temperatura constante; c) (c) en forma adiabática. Assuming mechanical reversibility, , Q, , and �Hcada for proceso. each process. Suponiendo reversibilidad mecánica, calculate calcule W,WQ, DU�U y DH para Dibuje cada traSketch each path on a single P V diagram. yectoria en un solo diagrama PV. 3.9.Un Angas ideal gasinicialmente initially at 600 K and undergoes a four-step reversible 3.9. ideal, a 600 K y1010bar bar, se somete a un ciclomechanically mecánicamente reversible de cuacycle in a closed system. In step 12, pressure decreases isothermally to 3 bar; in step tro etapas en un sistema cerrado. En la etapa 12, la presión decrece isotérmicamente a 3 bar; en la 23, pressure decreases at constant volume to 2 bar; step volume decreases etapa 23, la presión disminuye a volumen constante a 2 in bar; en 34, la etapa 34, el volumenatdisminuye a constant pressure;y en andlainetapa step 41, 41,elthe gas returnsenadiabatically to itsainitial state.inicial. Take Considere: presión constante, gas regresa forma adiabática su estado = (7/2)R and C = (5/2)R. C P V CP = (7/2)R y CV = (5/2)R. (a) Sketch the cycle on a P V diagram. a) (b)Dibuje el ciclo en ununknown) diagrama PV. Determine (where both T and P for states 1, 2, 3, and 4. b) Determine (donde no se tienen ) tanto a T como a P para los estados 1, 2, 3 y 4. (c) Calculate Q, W , �U , and �H for each step of the cycle. c) Calcule Q, W, DU y DH para cada etapa del ciclo. 3.10. An ideal gas, C P = (5/2)R and C V = (3/2)R, is changed from tP = 1 3bar and 3.10. Un gas ideal,3con CP = (5/2)R y CV = (3/2)R, cambia de P = 1 bar y V1 = 12 m a P2 = 12 bar y V2t V1t = 12 m to P2 = 12 bar and V2t = 1 m3 by the following mechanically reversible 3 = 1 m mediante los procesos mecánicamente reversibles siguientes: processes: a) (a)Compresión Isothermalisotérmica. compression. b) (b)Compresión adiabática seguida porby enfriamiento a presiónpressure. constante. Adiabatic compression followed cooling at constant c) (c)Compresión adiabática seguida por enfriamiento a volumen constante. Adiabatic compression followed by cooling at constant volume. d) (d)Calentamiento a volumen constante seguido por enfriamiento a presión constante. Heating at constant volume followed by cooling at constant pressure. 03-SmithVanNess.indd 112 8/1/07 13:01:07 Problemas 113 Problems 113 113 Problems e) Enfriamiento a presión constante seguido por calentamiento a volumen constante. (e) Cooling at constant pressure followed by heating at constant volume. Problems (e) Cooling constant followed at constant t y DHpressure t para cada Calcule Q, W,atDU uno deby losheating procesos, y dibujevolume. las trayectorias de todos los prot , and �H t for each of these processes, and sketch the paths of Calculate Q, W , �U cesos en un solo diagrama PV. t , and �H t for each of these processes, and sketch the paths of Calculate Q, Won, �U all processes a single P V diagram. (e) Cooling at constant pressur all processes on a single P V diagram. 3.11. El gradiente térmico ambiental dT/dz caracteriza la variación local de la temperatura con la altura Calculate Q, W , �U t , and �H 3.11.enThe environmental lapseLarate dT /dz characterizes local variation de of acuerdo temperature la atmósfera terrestre. presión atmosférica varíathe con la elevación con la fórmula all processes on a single P V di 3.11. The lapse rate atmosphere. dT /dz characterizes the local variation temperature withenvironmental elevation in the earth’s Atmospheric pressure variesofwith elevation hidrostática. with elevation in the earth’s atmosphere. Atmospheric pressure varies with elevation according to the hydrostatic formula, according to the hydrostatic formula, 3.11. The environmental lapse rate d dP with elevation in the earth’s atm = −Mρg dP according to the hydrostatic for dz = −Mρg dz where M is molar mass, ρ is molar density, and g is the local acceleration of gravity. where M isthat mass, ρ is molar density, gT is related the oflocal gravity. donde esmolar lathe masa molar, rises densidad molar y glocal estolaacceleration de Asssume atmosphere anlaideal gas,and with Paceleración by the polytropic for-la gravedad. Asssume that the atmosphere is an ideal gas, with T related to P by the polytropic forSuponga que la atmósfera es un gas ideal, con T relacionada a P por la fórmula politrópica, ecuamula, Eq. (3.35c). Develop an expression for the environmental lapse rate in relation mula, Eq. (3.35c). Develop an expression for the environmental lapse rate in relation where M ción (3.35c). Desarrolle una expresión para el gradiente térmico ambiental en relación con ,isg,molar mass, ρ is m to M, g, R, and δ. Asssume that the atmosphere is RtoyM, d. g, R, and δ. mula, Eq. (3.35c). Develop an 3.12. An evacuated tank is filled with gas from a constant-pressure line. Develop an expres� to M, g, R, and δ. 3.12. Se llena con gas un tanque previamente vacío mediante una línea a presión constante. Desarrolle 3.12. An evacuated is filled withofgas a constant-pressure line. Develop anthe expresgas in sion relating tank the temperature thefrom gas in the tank to the temperature T of � una expresión que relacione la temperatura del gas en el tanque con la temperatura T ′ del gas en la the transfer gas in sion the temperature of the gasconstant in the tank the temperature T ofheat the relating line. Assume the gas is ideal with heattocapacities, and ignore línea. Suponga que el gas es ideal con capacidades caloríficas constantes e ignore la transferencia the line. Assume the gas is ideal with constant heat capacities, and ignore heat transfer An evacuated tank is filled with between the gas and the tank. Mass and energy balances for this problem are3.12. treated de entre el gas el tanque. Los and balances de balances masa y energía este problema consideran between the gas andythe tank. Mass energy for thispara problem are treatedse sion relating the temperature of incalor Ex. 2.13. en in el Ex.ejemplo 2.13. 2.13. the line. Assume the gas is idea between the gas and the tank. M 3.13. Show how Eqs. (3.36) and (3.37) reduce to the appropriate expressions for the four 3.13. Demuestre cómo las ecuaciones (3.36) y (3.37) se reducen a las expresiones apropiadas los 2.13. 3.13. Show how values Eqs. (3.36) and (3.37) reduce the appropriate expressions for the fourin Ex.para particular of δ listed following Eq.to(3.37). cuatro valores particulares de d enumerados después de la ecuación (3.37). particular values of δ listed following Eq. (3.37). 3.13. Show how Eqs. (3.36) and (3.3 3.14. A tank of 0.1-m3 volume contains air at3 25◦ C and 101.33 kPa. The tank is connected 3 volume 3.14. tanque contiene un volumen de 0.1 m 25de◦ Caire a 101.33 25 °C ykPa. 101.33 kPa. Elistanque estáparticular conectadovalues a of δ listed follo contains air at and The tank connected 3.14. Un Atotank of 0.1-m a compressed-air line which supplies air at the constant conditions of 45◦ C and ◦ una línea de aire comprimido que proporciona aire en condiciones constantes de 45 °C y 1 500 kPa. C and to1,500 a compressed-air line which supplies air at the constant conditions of 45 kPa. A valve in the line is cracked so that air flows slowly into the tank until Una válvula en valve la línea se que elso aire fluye bastante lentitud dentro del tanque 1,500 kPa. A theagrieta, linepressure. is así cracked air con flows slowly intoenough thehacia tank until 3.14. A tank of 0.1-m3 volume conta the pressure equalsinthe line If thethat process occurs slowly that the hasta que la presión se equilibra con la presión de la línea. Si el proceso ocurre muy lentamente, de the pressure equals the line pressure. If how the process occurs enough theto a compressed-air line which temperature in the tank remains at 25◦ C, much heat is lostslowly from the tank?that Assume ◦ tal manera que la temperatura se mantiene a 25 °C, ¿cuánto calor se pierde desde el tanque? Supontemperature in the tank remains at 25 C, how much heat is lost from the tank? Assume 1,500 kPa. A valve in the line air to be an ideal gas for which C P = (7/2)R and C V = (5/2)R. ga el an aireideal es un gas para que CP = (7/2)R CV(5/2)R. = (5/2)R. (7/2)R and C Vy = airque to be gas forideal which C Pel= the pressure equals the line pre in the tank remains 3.15. Gas at constant T and P is contained in a supply line connected through a valve to temperature a 3.15. aatTconstant y P constantes está contenido en una línea line de suministro conectada, mediante unatoválvula, air be an ideal gas for which 3.15. Gas Gas T and P is contained in a supply connected through a valve to a closed tank containing the same gas at a lower pressure. The valve is opened to allow aclosed un tanque cerrado que the contiene el mismo gas apressure. una presión menor. Laopened válvulatoseallow abre permitiendo tank containing same gas at a lower The valve is flow of gas into the tank, and then is shut again. que dentro tanque después se cierra una vez más. flowelofgas gasfluya into hacia the tank, anddel then is shuty again. 3.15. Gas at constant T and P is con (a) Develop a general equation relating n 1 and n 2 , the moles (or mass) of gas in the closed tank containing the same una ecuación general y nmoles moles (oUmasas) de gas en eloftanque a)(a) Desarrolle 1to 2, las na2 ,nthe (or mass) of gas in the Develop general equation n 1 and tank at athe beginning and relating endque of relacione the process, the properties and U , the flow gas into the tank, and th 1 2 altank principio y al final del proceso, con las propiedades U y U , la energía interna 1 end2ofUthe at the beginning the at process, to the properties U2 , the 1 and internal energy of theand gas end in theoftank the beginning and process, and del gas al principio yenthalpy al final proceso, H ′,supply laatentalpía del gas enthe la línea de suministro y(a) a Q,Develop el calor a general equation internal of del thethe gasgas in in theythe tank theline, beginning and end of the process,toand H � , theenergy of and to Q, heat transferred the � transferido a la sustancia en el tanque durante el proceso. Hmaterial , the enthalpy of the gas inthe theprocess. supply line, and to Q, the heat transferred to the tank at the beginning and in the tank during b) Reduzca la ecuación generalthe a su forma más simple para el caso especial de un gas ideal conenergy of the gas in material in the tank during process. (b) Reduce the general equation to its simplest form for the special case of an ideal internal capacidades caloríficas constantes. H � , the enthalpy of the gas (b) Reduce theconstant generalheat equation to its simplest form for the special case of an ideal gas with capacities. c) Además, reduzca la ecuación de b) para el caso n = 0. 1 material in the tank during with constant heat capacities. (c)gas Further reduce the equation of (b) for the case of n 1 = 0. d)(c) Después reduzcathe la equation ecuación of de(b) c) para el caso en que, también Q = 0. (b) Reduce the general equatio Further reduce for the case of n = 0. 1 (d) Further reduce the equation of (c) for the case in which, in addition, Q = 0. gas with constant heat capa (d) Further reduce the equation of (c) for the case in which, in addition, Q = 0. (c) Further reduce the equation (d) Further reduce the equation 03-SmithVanNess.indd 113 8/1/07 13:01:17 114 CAPÍTULO 3. Propiedades volumétricas de fluidos puros e) Considerando al nitrógeno como un gas ideal para el que CP = (7/2)R, aplique la ecuación apropiada para el que una alimentación estable de nitrógeno a 25 °C y 3 bar fluye hacia dentro de un tanque vacío de 4 m3 de volumen, y calcule las moles de nitrógeno que fluyen hacia dentro del tanque para igualar las presiones para dos casos: 1) Suponga que no fluye calor del gas al tanque o a través de las paredes del mismo. 2) El tanque pesa 400 kg, está perfectamente aislado, tiene una temperatura inicial de 25 °C, cuenta con calor específico de 0.46 kJ kg–1 K–1 y es calentado por el gas, por lo que siempre estará a la temperatura del gas en el tanque. 3.16. Desarrolle ecuaciones para determinar la temperatura final del gas que permanece en un tanque, después de que se extrajo gas desde una presión inicial P1 hasta una presión final P2. Las cantidades conocidas son la temperatura inicial, el volumen del tanque, la capacidad calorífica del gas, la capacidad calorífica total del contenido del tanque, P1 y P2. Suponga que el tanque siempre está a la temperatura del gas que permanece en éste y que está perfectamente aislado. 3.17. Un tanque rígido no conductor, con un volumen de 4 m3, se divide en dos partes desiguales separadas por una membrana delgada. Un lado de la membrana, que representa 1/3 del tanque, contiene gas nitrógeno a 6 bar y 100 °C, y el otro lado, que representa 2/3 del tanque, está vacío. La membrana se rompe y el gas llena el tanque. a) ¿Cuál es la temperatura final del gas? ¿Cuánto trabajo se hace? ¿El proceso es reversible? b) Describa un proceso reversible mediante el cual el gas pueda regresar a su estado inicial. ¿Cuánto trabajo se hace? Suponga que el nitrógeno es un gas ideal para el que CP = (7/2)R y CV = (5/2)R. 3.18. Un gas ideal, inicialmente a 30 °C y 100 kPa, experimenta los siguientes procesos cíclicos en un sistema cerrado: a) En un proceso mecánicamente reversible, primero hay una compresión adiabática a 500 kPa, a continuación un enfriamiento a presión constante de 500 kPa hasta 30 °C, y al final una expansión isotérmica hasta su estado original. b) El ciclo experimenta los mismos cambios de estado, pero cada etapa es irreversible con una eficiencia de 80% en comparación con la del correspondiente proceso mecánicamente reversible. Nota: la etapa inicial puede no ser más larga que el adiabático. Calcule Q, W, DU y DH para cada etapa del proceso y para todo el ciclo. Considere CP = (7/2)R y CV = (5/2)R. 3.19. Un metro cúbico de un gas ideal a 600 K y 1 000 kPa se expande hasta alcanzar cinco veces su volumen inicial de la siguiente manera: a) Mediante un proceso isotérmico mecánicamente reversible. b) Mediante un proceso adiabático mecánicamente reversible. 03-SmithVanNess.indd 114 8/1/07 13:01:17 Problemas 115 c) Mediante un proceso adiabático, irreversible, en el cual la expansión se hace contra una presión restringente de 100 kPa. Para cada caso calcule la temperatura y presión final y el trabajo realizado por el gas. CP = 21 J mol–1 K–1. 3.20. Un mol de aire, inicialmente a 150 °C y 8 bar, se somete a los cambios mecánicamente reversibles siguientes. Se expande isotérmicamente a una presión tal que cuando se enfría hasta 50 °C a volumen constante, su presión final es de 3 bar. Suponga que el aire es un gas ideal para el que CP = (7/2)R y CV = (5/2)R. Calcule W, Q, DU y DH. 3.21. Un gas ideal fluye en estado estacionario por un tubo horizontal. No se añade calor ni se hace trabajo de flecha. El área de la sección transversal del tubo cambia con la longitud, lo cual hace que cambie la velocidad. Deduzca una ecuación que relacione la temperatura con la velocidad del gas. Si por una sección del tubo pasa nitrógeno a 150 °C a una velocidad de 2.5 m s–1, ¿cuál es su temperatura en otra sección donde su velocidad es de 50 m s–1? Sea CP = (7/2)R. 3.22. Un mol de gas ideal, inicialmente a 30 °C y 1 bar, cambia a 130 °C y 10 bar mediante tres distintos procesos mecánicamente reversibles: a) El gas se calienta primero a volumen constante hasta que su temperatura es de 130 °C; a continuación, se comprime isotérmicamente hasta que su presión es de 10 bar. b) El gas se calienta primero a presión constante hasta que su temperatura es de 130 °C; a con­ tinuación, se comprime isotérmicamente hasta 10 bar. c) El gas se comprime primero isotérmicamente hasta 10 bar; a continuación, se calienta a presión constante hasta 130 °C. Calcule Q, W, DU y DH en cada caso. Considere CP = (7/2)R y CV = (5/2)R. En otro caso, considere CP = (5/2)R y CV = (3/2)R. 3.23. Un mol de gas ideal, inicialmente a 30 °C y 1 bar, experimenta los cambios mecánicamente reversibles siguientes. Se comprime de manera isotérmica hasta un punto tal que cuando se calienta a volumen constante hasta 120 °C, su presión final es de 12 bar. Calcule Q, W, DU y DH para el proceso. Tome CP = (7/2)R y CV = (5/2)R. 3.24. Un proceso consta de dos etapas: 1) un mol de aire a T = 800 K y P = 4 bar se enfría a volumen constante hasta T = 350 K. 2) Después el aire se calienta a presión constante hasta que su temperatura llega a 800 K. Si este proceso de dos etapas se reemplaza por una sola expansión isotérmica del aire desde 800 K y 4 bar hasta una presión final P, ¿cuál es el valor de P que hace que el trabajo de los dos procesos sea el mismo? Suponga reversibilidad mecánica y considere el aire como un gas ideal con CP = (7/2)R y CV = (5/2)R. 3.25. El siguiente es un esquema para encontrar el volumen interno VBt , de un cilindro de gas. El cilindro se llena con un gas a baja presión P1, y se conecta mediante una línea y válvula pequeñas a un tanque de referencia evacuado y de volumen conocido VAt . 03-SmithVanNess.indd 115 8/1/07 13:01:18 116 116 116 116 CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids CHAPTER 3. Volumetric Properties of Pure Fluids Propiedades de fluidos puros is opened, and gas flows through the CAPÍTULO line into the3.reference tank. volumétricas After the system is opened, and gas flows througha the line into the reference tank. After athe system returns to its initial temperature, sensitive pressure transducer provides value for t value returns toseits initial temperature, alathe sensitive pressure transducer provides for isválvula opened, and gas line into reference tank. After system pressure change �P inthrough thepor cylinder. Determine the cylinder volume VaBthe from Lathe abre y elflows gas fluye línea hacia elthe tanque de referencia. Después dethe que el sistema the pressure change �Pinicial, in the un cylinder. Determine the volume VaBt value fromunthe returns its initial temperature, atransductor sensitive pressure transducer provides for following regresa a to sudata: temperatura sensible acylinder la presión proporciona valor para el tV t from the following data: the pressure change �P in the cylinder. Determine the cylinder volume cambio de presión DP en el cilindro. Determine el volumen del cilindro V a partir de la informaB B • V At =data: 256 cmt3 . 3; b) AP/P = –0.0639. following ción siguiente: a) V = 256 cm t 3 1 • V A = 256 cmA . • �P/P 1 = −0.0639. t 3 V A = 256 cm . •• �P/P 1 = −0.0639. 3.26. Un cilindro horizontal, no conductor y cerrado, contiene un pistón flotante no conductor y sin fric•que �P/P 3.26.ción, A closed, nonconducting, horizontal cylinderA is fitted nonconducting, friction1 = divide al−0.0639. cilindro en dos secciones, y B. Laswith dos asecciones contienen masas iguales de 3.26.aire, A closed, nonconducting, horizontal fitted with frictionless, floating piston which divides thecylinder cylinder A 1(atm). and B. En Thelatwo sec- A se activa inicialmente en las mismas condiciones, T1is=into 300Sections K y Pa1 nonconducting, = sección less, floating whichhorizontal divides thecylinder Sections Aaumenta and TB. The two 3.26.untions Aelemento closed, nonconducting, isinto fitted with a nonconducting, frictioncontain equal masses ofeléctrico air, initially at the same conditions, 300 K secandTA en la sec1 = depiston calentamiento ycylinder la temperatura del aire lentamente: tions contain equal masses of air, initially the same conditions, TB. = 300two Ktemand adiabática less, which divides the cylinder into Sections A and The sec1and 1(atm).apiston An electrical heating element in Section A is activated, the air P 1 = ción A floating debido la transferencia de calor, y Tat en la sección B debido a la compresión B = 1(atm). An electrical heating element in Section A is activated, and the air temP tions contain equal masses of air, initially at the same conditions, T = 300 K and 1 peratures por slowly increase: T Alento in Section A because of heat and Section 1unTgas B inideal provocada el movimiento del pistón. Considere el transfer, aire como con CP = R, y peratures slowly increase: T in Section A because of heat transfer, and T in Section = 1(atm). An electrical heating element in Section A is activated, and the air temP A B B 1nbecause of adiabatic compression by the slowly moving piston. Treat air as an ideal sea el número de moles de aire contenidos en la sección A. Para el proceso descrito, evalúe uno A 7 Blos because of adiabatic compression by number the as ideal peratures T Ansiguientes: in Section A slowly because of heat transfer, and air TBA. in an Section with Cslowly R, and let the of moving moles ofpiston. air in Treat Section For the P = A be degas conjuntos de27increase: cantidades gas withasCdescribed, let n one of moving moles air in Treat Section Forideal the B because of adiabatic compression by the slowly piston. air A. as an P = A beofthe process evaluate thenumber following sets ofof quantities: 2 R, and 7 process as described, evaluate one of the following sets of quantities: gas with C = R, and let n be the number of moles of air in Section A. For the P A, si A 2 P(final) = 1.25(atm). a) (a)TAT, T,B Ty Q/n , and Q/n , if P(final) = 1.25(atm). A asB described, Aevaluate process one of the following sets of quantities: b) (a)TBT, AQ/n P(final), = 425 K. AB ,yand Q/n Asi, ifTAP(final) = 1.25(atm). Q/n if T A = 425 K. (b) TB ,, T A , and P(final), c) (b) T , Q/n y P(final), si T = 325 K.= ATB , T A , and BP(final) Q/n , and P(final), if T 425 K. K. (a) Q/n , if = 1.25(atm). A A B A , and P(final), A if TB = 325 (c) T AA, Q/n –1. d) (c) TAT , TB, Q/n y P(final), siP(final), Q/nA = if 3 kJ mol , and T = 325 425 (b) A P(final), if Q/n B A = 3 kJ K. mol−1 . (d) T AAB, TB , and A , T , and P(final), if Q/n = 3 kJ mol−1 . (d) T 325 K. (c) A Q/n B A , and P(final), if TB A −1 . constantes se somete a un proceso arbitrario 3.27. Un mol de un gas ideal con capacidades caloríficas P(final), if Q/n kJ mol (d) Tmole 3.27. One of, and an ideal gas with constant capacities undergoes an arbitrary mechanA , TB A = 3heat reversible. Muestre que: 3.27.mecánicamente One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechanically reversible process. Show that: ically reversible process. Show that: 3.27. One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechan1 ically reversible process. Show that: �U = 1 �(P V ) γ − 1 �(P V ) �U = 11 γ− �U = �(P V ) 3.28.Deduzca Derive an theelwork of mechanically reversible, isothermal γ −por 1 la compresión 3.28. unaequation ecuaciónfor para trabajo hecho isotérmica compression mecánicamente reversi3.28.ble Derive anofequation for an theuna work of mechanically reversible, isothermal of 1 mol a gas initial pressure P1 Pto1 ahasta final pressure P2final whenPcompression equation de 1 mol de gas,from desde presión inicial una presión cuando la ecuación de 2the of 1 mol a virial gas from an pressure Ptruncated to a final P2 when compression the equation 3.28.estado Derive equation for theinitial work of(3.11)] mechanically reversible, isothermal 1 truncada of state is the expansion [Eq. to:a:pressure es an laof expansión virial [ecuación (3.11)] of state thea virial expansion [Eq. (3.11)] Ptruncated to:pressure P2 when the equation 1 molisof gas from an initial pressure 1 to a final � Z = 1 + B P of state is the virial expansion [Eq. (3.11)] truncated to: Z = 1 + B� P How does the result compare with the Z corresponding = 1 + B � P equation for an ideal gas? How does the result compare with the corresponding equation for an ideal gas? se compara este resultado con la corresponding ecuación que corresponde ideal? How does theisresult compare with the equation foral angas ideal gas? 3.29.¿Cómo A certain gas described by the equation of state: 3.29. A certain gas is described by the equation of � state: � θ 3.29. gas segas describe mediante la ecuación de estado: �bstate: 3.29.Cierto A certain is described byPthe equation V = RT + of − θ �P P V = RT + �b − RT � P θ RT = RT of + T bonly. − For this P gas, determine expressions Here, b is a constant and θ is P aV function RT Here, is a constant and θ is a function T only. Forpressure this gas,coefficient determine (∂ expressions for thebisothermal compressibility κ andofthe thermal P/∂ T )V . for thebexpressions isothermal compressibility κ and the thermal pressure coefficient (∂ P/∂ T )V . Here, is a constant and θ is a function of T only. For this gas, determine expressions These should contain only T , P, θ , dθ/dT , and constants. Aquí, b es una constante y q es una función sólo de T. Para este gas, determine las expresiones para These expressions should contain only T ,the P, thermal θ , dθ/dTpressure , and constants. for the isothermal compressibility κ and coefficient (∂ P/∂ T )V .expresiones la compresibilidad isotérmica k y el coeficiente de presión térmica (∂P/∂T) . Estas V ◦ C the second These expressions should T and , P, third θ, dθ/dT and constants. 3.30.deben For methyl chloride atP,100 virial, coefficients are: contener sólo T, q,contain dq/dT yonly constantes. 3.30. For methyl chloride at 100◦ C the second and third virial coefficients are: 3 −1 ◦C B = −242.5 cmthe mol C =virial 25,200 cm6 mol−2 3.30.Para Forelmethyl at 100 second and ythird coefficients are: 3 el −1 −2 son: 3.30. clorurochloride deB metilo a 100 °C, segundo tercer coeficientes viriales = −242.5 cm mol C = 25,200 cm6 mol B = −242.5 cm3 3mol−1 C = 25,200 cm6 mol−2 B = –242.5 cm mol–1 C = 25 200 cm6 mol–2 03-SmithVanNess.indd 116 8/1/07 13:01:33 Problems 117 Problems 117 117 Problems Problemas 117 117 Problems Calculate the work of mechanically reversible, isothermal compression of 1 mol of Calculate the work of mechanically reversible, isothermal compression of 1 de mol of de metimethyl chloride 1 bar toisotérmica, 55 bar at mecánicamente 100◦ C. Base calculations the Calcule el trabajo defrom compresión reversible deon1of mol Calculate the work of mechanically reversible, compression 1 following molcloruro of ◦ C.isothermal Calculate the work of mechanically reversible, isothermal compression of 1 mol of methyl chloride from 1 bar to 55 bar at 100 Base calculations on the following ◦ C. Base forms1chloride of the virial lomethyl desde hasta 55from barequation: a1100 Apoye sus100 cálculos en las formas de la ecuación virial siguientes: bar °C. to 55 bar at calculations on the following methylofchloride 1 bar to 55 bar at 100◦ C. Base calculations on the following forms the virialfrom equation: forms of the virial equation: B C forms of the virial equation: Z = 1 +B +C 2 a) (a) (a) Z = 1 + BBV+ CC2V (a)(b) ZZ = 1 + V B+� PV+ � 2 (a) Z ==1 1++ V +V2 C P (b) ZB = 1 + BV�� P +VC2�� P22 C − B 2 b)(b) Z = 1 + B �P + C �P� 2 B � = B Z = 1 +and (b)where B P + C�CP=C − B22 2 � B B RT (RT B� = B and C = CC − where − )B22) B � = RT and donde y CC�� = where (RT B = = and where RT exactly the same result? (RT )22 Why don’t both equations give RT (RT ) Why don’t both equations give exactly the same result? Whyqué don’t both equations give exactly thecon same result? ¿Por no se obtiene el mismo resultado ambas ecuaciones? Why don’t both equations give exactly the same result? 3.31. Any equation of state valid for gases in the zero-pessure limit implies a full set of virial 3.31. Any equation ofShow state that validthe forsecond gases inand the zero-pessure limit implies a full setthe of generic virial coefficients. virial implied 3.31.Cualquier Any equation of state forque gases in thethird zero-pessure implies a fullby setcero, of virial 3.31. ecuación de valid estado, es válida para gasescoefficients enlimit el límite de presión implica un con3.31. coefficients. Any equation of state valid for gases in the zero-pessure limit implies a full set of virial Show that the second and third virial coefficients implied by the generic cubic equation of state, (3.42), are: coefficients. Show that theEq. second and third virial coefficients implied by thecoeficientes generic junto completo de coeficientes viriales. Demuestre que el segundo y tercero viriales coefficients. Show that Eq. the (3.42), second are: and third virial coefficients implied by the generic cubic equation of state, cubic equation of state, Eq. (3.42),cúbica are: genérica, ecuación (3.42), son: implícitos en la ecuación de estado cubic equation of state, Eq.a(T (3.42), (� + σ )ba(T ) ) are: C =2b2 +(� + σ )ba(T ) B = b −a(T ) ) RT )) C = b + (� + σσ)ba(T B = b − a(TRT )ba(T C = b22 + (� + RT B = b − a(T RT ) C =b + B = b − RT RT Specialize the result forRT B to the Redlich/Kwong equation RT of state, express it in reSpecialize the result for B to the Redlich/Kwong equation of state, express it in reduced form, and compare numerically with the generalized correlation Specialize the result for B toitthe Redlich/Kwong equation of state, express itforin Bre-for Specialize the result for B to the Redlich/Kwong equation of state, express in for reduced form, and compare it numerically with the generalized correlation forit B Adapte el resultado para B aitDiscuss lanumerically ecuación estado Redlich/Kwong, expréselo simple fluids, (3.65). whatde you find. duced form, andEq. compare with the de generalized correlation for B en forsu forma re­ duced form, and compare it numerically with the generalized correlation for B for simple fluids, Eq. (3.65). Discuss what you find. ducida compárelo numéricamente conyou la correlación generalizada para B para fluidos simples, simpleyfluids, Eq. (3.65). Discuss what find. simple fluids, Eq. (3.65). Discuss what you find. ecuación (3.65). Analice resultados. 3.32. Calculate Z and V forsus ethylene at 25◦ C and 12 bar by the following equations: 3.32. Calculate Z and V for ethylene at 25◦ C and 12 bar by the following equations: 3.32. Calculate Z and V for ethylene at 25◦◦C and 12 bar by the following equations: C and 12 bar thefollowing followingexperimental equations: values 3.32. Calculate Ztruncated and V for ethylene at 25[Eq. (a) The virial equation (3.40)] withbythe 3.32. Calcule Z truncated yV para elvirial etileno a 25 ºC[Eq. y 12(3.40)] bar mediante ecuaciones siguientes:values (a) The equation with thelas following experimental of truncated virial coefficients: (a) The virial equation [Eq. (3.40)] with the following experimental values (a) of The truncated virial equation [Eq. (3.40)] with the following experimental values virial coefficients: virial coefficients: a) Laof virial truncada [ecuación (3.40)], con los valores experimentales siguientes de los 3 6 ofecuación virial coefficients: mol−1 C = 7,200 cm mol−2 B = −140 cm 3 6 −1 −2 coeficientes viriales: C = 7,200 cm mol B = −140 cm mol C = 7,200 cm66 mol−2 B = −140 cm33 mol−1 −1 C = a7,200 cm mol−2 B virial = −140 cm3 mol (b) The truncated equation [Eq. (3.38)], with value of B –1 6 mol –2 from the generalized B = –140 cm mol C = 7 200 cm (b) The truncated virial equation [Eq. (3.38)], with a value of B from the generalized Pitzer correlation (3.63)]. (b) The truncated virial [Eq. equation [Eq. (3.38)], with a value of B from the generalized (b) Pitzer The truncated virial equation [Eq. (3.38)], with a value of B from the generalized correlation [Eq. (3.63)]. The correlation Redlich/Kwong equation. b) (c) LaPitzer ecuación virial truncada [ecuación (3.38)], con un valor de B obtenido de la correlación ge[Eq. (3.63)]. Pitzer correlation [Eq. (3.63)]. (c)neralizada The Redlich/Kwong equation.equation. de Pitzer [ecuación The Soave/Redlich/Kwong (c)(d) The Redlich/Kwong equation.(3.63)]. (c) The The Soave/Redlich/Kwong Redlich/Kwong equation. (d) equation. c)(d)(e) LaThe ecuación de Redlich/Kwong. The Peng/Robinson equation. Soave/Redlich/Kwong equation. (d) The Soave/Redlich/Kwong equation. (e) The Peng/Robinson equation. d)(e)LaThe ecuación de Soave/Redlich/Kwong. Peng/Robinson equation. Peng/Robinson equation. e) (e)Calculate LaThe ecuación de Peng/Robinson. 3.33. Z and V for ethane at 50◦ C and 15 bar by the following equations: 3.33. Calculate Z and V for ethane at 50◦◦ C and 15 bar by the following equations: 3.33. Calculate Z and V for ethane at 50 ◦C and 15 bar by the following equations: C 15 bar by the equations: 3.33.Calcule Calculate and ethane (a) The truncated virial with thefollowing following experimental values 3.33. Z y ZV paraVelfor etano aequation 50at°C50y [Eq. l5 and bar(3.40)] con las ecuaciones siguientes: (a) The truncated virial equation [Eq. (3.40)] with the following experimental values of truncated virial coefficients: (a) The virial equation [Eq. (3.40)] with the following experimental values (a) of The truncated virial equation [Eq. (3.40)] with the following experimental values virial coefficients: virial coefficients: a) Laof virial truncada [ecuación (3.40)], con los valores experimentales siguientes de los 3 6 ofecuación virial coefficients: mol−1 C = 9,650 cm mol−2 B = −156.7 cm 3 6 coeficientes viriales: −1 −2 C = 9,650 cm mol B = −156.7 cm mol C = 9,650 cm66 mol−2 B = −156.7 cm33 mol−1 −1 −2 C =a 9,650 cmB–2mol B= −156.7 cm3 mol –1 (b) The truncated virial equation [Eq. (3.38)], with value B = –156.7 cm mol C = 9 650 cm6 of mol from the generalized (b) The truncated virial equation [Eq. (3.38)], with a value of B from the generalized Pitzer correlation (3.63)]. (b) The truncated virial [Eq. equation [Eq. (3.38)], with a value of B from the generalized (b) The truncated virial equation [Eq. (3.38)], value ofde B Bfrom the generalized Pitzer correlation [Eq. (3.63)]. b) (c) LaPitzer ecuación virial truncada [ecuación (3.38)],with conaun valor obtenido a partir de la correlaThe correlation Redlich/Kwong equation. [Eq. (3.63)]. Pitzer correlation [Eq. (3.63)]. (c)(d) The Redlich/Kwong equation. ción generalizada de Pitzer [ecuación (3.63)]. The Soave/Redlich/Kwong (c) The Redlich/Kwong equation.equation. (c)LaThe The Redlich/Kwong equation. (d) Soave/Redlich/Kwong equation. c)(d) ecuación de Redlich/Kwong. (e)The The Peng/Robinson equation. Soave/Redlich/Kwong equation. (d) The Soave/Redlich/Kwong equation. Peng/Robinson equation. d) (e)LaThe ecuación de Soave/Redlich/Kwong. (e) The Peng/Robinson equation. Peng/Robinson equation. e) (e)LaThe ecuación de Peng/Robinson. 03-SmithVanNess.indd 117 8/1/07 13:01:48 CHAPTER CHAPTER 3. 3. Volumetric Volumetric Properties Properties of of Pure Pure Fluids Fluids 118118 118 CAPÍTULO 3. Propiedades volumétricas de fluidos puros 3.34. 3.34. Calculate Calculate Z and Z and V V forfor sulfur sulfur hexafluoride hexafluoride at 75 at ◦75 C◦and C and 1515 barbar byby thethe following following equaequa3.34. Calcule Z y V para el hexafluoruro de azufre a 75 °C y 15 bar mediante las ecuaciones siguientes: tions: tions: The The truncated truncated virial virial equation equation [Eq. [Eq. (3.40)] (3.40)] with with thethe following following experimental values values a)(a)(a) La ecuación virial truncada [ecuación (3.40)], con los valores experimental experimentales siguientes de los of of virial virial coefficients: coefficients: coeficientes viriales: 3 3 −1 −1 6 6 −2 −2 B == B–194 = −194 −194 cm mol mol C =C =15 = 15,300 15,300 cm6cm mol mol 3 cm –1 –2 B cm mol C 300 cm mol The The truncated truncated virial virial equation equation [Eq. [Eq. (3.38)], (3.38)], with with a value a value of of Bdefrom BBfrom thethe generalized generalized b)(b)(b) La ecuación virial truncada [ecuación (3.38)], con un valor obtenido de la correlación gePitzer Pitzer correlation correlation [Eq. [Eq. (3.63)]. (3.63)]. neralizada de Pitzer [ecuación (3.63)]. The The Redlich/Kwong Redlich/Kwong equation. equation. c)(c)(c) La ecuación de Redlich/Kwong. The The Soave/Redlich/Kwong Soave/Redlich/Kwong equation. equation. La ecuación de Soave/Redlich/Kwong. d)(d)(d) The The Peng/Robinson Peng/Robinson equation. equation. e)(e)(e) La ecuación de Peng/Robinson. 3 mol 3 mol −1 ,−1and ForFor sulfur sulfur hexafluoride, hexafluoride, Tc T= 318.7 318.7 K, K, Pc P= = 37.6 37.6 bar,bar, V V= = 198198 cmcm , and c = Para el hexafluoruro de azufre, Tc = 318.7 K, Pc c = 37.6 bar, cVc c= 198 cm3 mol–1, y w = 0.286. ω= ω= 0.286. 0.286. 3.35. Determine Z y V para el vapor a 250 °C 1 800 kPa a partir de lo siguiente: ◦ C◦y 3.35. 3.35. Determine Determine Z and Z and V for V for steam steam at 250 at 250 and C and 1,800 1,800 kPakPa byby thethe following: following: a)(a)(a) Mediante la ecuación virial truncada [ecuación (3.40)] con losexperimental valores experimentales The The truncated truncated virial virial equation equation [Eq. [Eq. (3.40)] (3.40)] with with thethe following following experimental values values siguientes de los coeficientes viriales: of of virial virial coefficients: coefficients: 3 3mol 6 6mol 3 –1−1 6 –2−2 −2 −1 = cm C 800 cm mol mol C =C =–5 = −5,800 −5,800 cmcm mol mol BB = B –152.5 = −152.5 −152.5 cmcm b)(b)(b) Con latruncated ecuación virial truncada [ecuación (3.38)], con unofvalor Bthe obtenido de la correlación The The truncated virial virial equation equation [Eq. [Eq. (3.38)], (3.38)], with with a value a value of B from B de from the generalized generalized generalizada de Pitzer [ecuación (3.63)]. Pitzer Pitzer correlation correlation [Eq. [Eq. (3.63)]. (3.63)]. c) Mediante las tablas de vapor (apéndice F). (c)(c) The The steam steam tables tables (App. (App. F).F). 3.36. Con respecto a las expansiones viriales, ecuaciones (3.11) y (3.12), demuestre que: 3.36. 3.36. With With respect respect to to thethe virial virial expansions, expansions, Eqs. Eqs. (3.11) (3.11) andand (3.12), (3.12), show show that: that: �� �� �� �� ∂ Z∂ Z ∂ Z∂ Z B � B=� = and B = B= and y ∂ P∂ PT,P=0 ∂ρ∂ρT,ρ=0 T,P=0 T,ρ=0 where where ρ1/V. ≡ 1/V 1/V . . donde rρ ≡≡ 3.37. 3.37. Equation Equation (3.12) when when truncated truncated to four toafour terms terms accurately accurately represents represents the volumetric volumetric data data 3.37. Cuando la(3.12) ecuación (3.12) se trunca cuatro términos, describe conthe exactitud la información volu◦C ◦with: for for methane methane gas gas at 0 at 0 C with: métrica para el gas metano a 0 °C con: 3 3 −1 −1 6 6 −2 −2 9 9 −3 −3 mol mol–1 CCC === 2,620 mol D =D =5= 5,000 5,000 cm9cm mol mol B= BB= −53.4 cmcm cm 3 mol 6mol –2 D –3 = −53.4 –53.4 2 2,620 620cm cmcm mol 000 cm mol Use Use these these data data to to prepare prepare plot a plot of of Z vs. Z gráfica vs. P for P for methane at 0at◦ C 0◦from C 0 to 0 to 200 200 bar.bar. a)(a)(a) Use esta información para apreparar una de methane Z en función defrom P para metano a 0 °C de 0 a (b)(b) To To what what pressures pressures do do Eqs. Eqs. (3.38) (3.38) and and (3.39) (3.39) provide provide good good approximations? approximations? 200 bar. b) ¿Para qué presiones las ecuaciones (3.38) y (3.39) proporcionan buenas aproximaciones? 3.38. 3.38. Calculate Calculate thethe molar molar volume volume of of saturated saturated liquid liquid andand thethe molar molar volume volume of of saturated saturated 3.38. Calcule elby volumen molar del líquido saturado y of delof volumen molar vapor saturado mediante la vapor vapor by the the Redlich/Kwong Redlich/Kwong equation equation forfor oneone the the following following anddel and compare compare results results ecuación de Redlich/Kwong para una de lascorrelations. condiciones y sustancias siguientes y compare los resulwith with values values found found byby suitable suitable generalized generalized correlations. tados con los valores encontrados mediante el empleo de las correlaciones generalizadas adecuadas. (a)(a) Propane Propane at 40 at ◦40 C◦where C where P sat P sat = 13.71 = 13.71 bar.bar. saturado a) Propano a 40 °C donde P = 13.71 bar. 03-SmithVanNess.indd 118 8/1/07 13:01:57 119 Problemas b) c) d) e) f) g) h) i) j) k) l) m) n) o) p) q) r) s) t) Propano a 50 °C donde P saturado = 17.16 bar. Propano a 60 °C donde P saturado = 21.22 bar. Propano a 70 °C donde P saturado = 25.94 bar. n-butano a 100 °C donde P saturado = 15.41 bar. n-butano a 110 °C donde P saturado = 18.66 bar. n-butano a 120 °C donde P saturado = 22.38 bar. n-butano a 130 °C donde P saturado = 26.59 bar. Isobutano a 90 °C donde P saturado = 16.54 bar. Isobutano a 100 °C donde P saturado = 20.03 bar. Isobutano a 110 °C donde P saturado = 24.01 bar. Isobutano a 120 °C donde P saturado = 28.53 bar. Cloro a 60 °C donde P saturado = 18.21 bar. Cloro a 70 °C donde P saturado = 22.49 bar. Cloro a 80 °C donde P saturado = 27.43 bar. Cloro a 90 °C donde P saturado = 33.08 bar. Bióxido de azufre a 80 °C donde P saturado = 18.66 bar. Dióxido de azufre a 90 °C donde P saturado = 23.31 bar. Dióxido de azufre a 100 °C donde P saturado = 28.74 bar. Dióxido de azufre a 110 °C donde P saturado = 35.01 bar. 3.39. Use la ecuación de Soave/Redlich/Kwong para calcular el volumen molar del líquido y del vapor saturados, para la sustancia y condiciones conocidas por uno de los incisos del problema 3.38, y compare sus resultados con los valores que se encuentran mediante las correlaciones generalizadas adecuadas. 3.40. Use la ecuación de Peng/Robinson para calcular los volúmenes molares del líquido y del vapor saturados para la sustancia y condiciones dadas por uno de los incisos del problema 3.38, y compare sus resultados con los valores que se encuentran mediante las correlaciones generalizadas adecuadas. 3.41. Calcule lo siguiente: a) El volumen ocupado por 18 kg de etileno a 55 °C y 35 bar. b) La masa de etileno contenida en un cilindro de 0.25 m3 a 50 °C y 115 bar. 3.42. El volumen molar de la fase vapor de un compuesto particular se reporta como 23 000 cm3 mol–1 a 300 K y 1 bar. Ninguna otra información está disponible. Sin que suponga comportamiento de un gas ideal determine una estimación razonable del volumen molar del vapor a 300 K y 5 bar. 3.43. Con una buena aproximación, ¿cuál es el volumen molar del vapor de etanol a 480 °C y 6 000 kPa? ¿Cómo se compara este resultado con el valor que corresponde a un gas ideal? 3.44. Se utiliza un recipiente de 0.35 m3 para almacenar propano líquido a su presión de vapor. Las consideraciones de seguridad dictan que a una temperatura de 320 K el líquido no debe ocupar más de 80% del volumen total del recipiente. Bajo estas condiciones determine las masas de vapor y de líquido dentro del recipiente. A 320 K la presión de vapor del propano es 16.0 bar. 03-SmithVanNess.indd 119 8/1/07 13:01:58 120 CAPÍTULO 3. Propiedades volumétricas de fluidos puros 3.45. Un tanque de 30 m3 contiene 14 m3 de n-butano líquido en equilibrio con su vapor a 25 °C. Estime la masa de vapor del n-butano contenida en el tanque. La presión de vapor del n-butano a la temperatura dada es 2.43 bar. 3.46. Calcule: a) La masa de etano contenida en un recipiente de 0.15 m3 a 60 °C y 14 000 kPa. b) La temperatura a la que 40 kg de etano, almacenados en un recipiente de 0.15 m3, ejercen una presión de 20 000 kPa. 3.47. ¿A qué presión debe llenarse un recipiente de 0.15 m3 a 25 °C para guardar 40 kg de etileno? 3.48. Si 15 kg de H2o se calientan a 400 °C en un recipiente de 0.4 m3, ¿que presión se genera? 3.49. A un recipiente de 0.35 m3 contiene vapor de etano a 25 °C y 2 200 kPa. Si el recipiente se calienta a 220 °C, ¿qué presión se genera dentro de él? 3.50. ¿Cuál es la presión en un recipiente de 0.5 m3 cuando se carga con 10 kg de dióxido de carbono a 30 °C? 3.51. A un recipiente rígido, lleno a la mitad de su volumen con nitrógeno líquido en su punto normal de ebullición, se le permite calentarse a 25 °C. ¿Qué presión se desarrolla? El volumen molar del nitrógeno líquido en su punto normal de ebullición es 34.7 cm3 mol–1. 3.52. El volumen específico del isobutano líquido a 300 K y 4 bar es 1.824 cm3 g–1. Calcule el volumen específico a 415 K y 75 bar. 3.53. La densidad del n-pentano líquido es 0.630 g cm–3 a 18 °C y 1 bar. Estime su densidad a 140 °C y 120 bar. 3.54. Calcule la densidad del etanol líquido a 180 °C y 200 bar. 3.55. Estime el cambio de volumen en la vaporización del amoniaco a 20 °C. A esta temperatura, la presión de vapor del amoniaco es 857 kPa. 3.56. La información PVT es posible adquirirse mediante el procedimiento siguiente. La masa m de una sustancia de masa molar M se introduce en un recipiente de volumen total V t conocido y térmicamente controlado. Al sistema se le permite alcanzar el equilibrio, y se miden la temperatura T y la presión P. a) Aproximadamente, ¿qué porcentajes de error se pueden permitir en las variables observadas (m, M, V t y P) si el error máximo permisible en el factor de compresibilidad Z es ±1%? b) Aproximadamente, ¿qué porcentajes de error se permiten en las variables observadas si el error máximo permisible en los valores calculados del segundo coeficiente virial B es ±1%? Suponga que Z 0.9 y que los valores de B se calculan mediante la ecuación (3.39). 03-SmithVanNess.indd 120 8/1/07 13:01:58 Problems Problems Problemas 121 121 121 3.57. gasdescribed descrito por de Redlich/Kwong y para temperatura mayor que Tc, desa3.57. For aa gas by the Redlich/Kwong equation for temperature greater than 3.57.Para Forun gas described byla theecuación Redlich/Kwong equation and and for aauna temperature greater than TTcc,, develop expressions two slopes, rrolle expresiones para lasfor dosthe pendientes límite, develop expressions for the two limiting limiting slopes, � � � � � � � � ∂∂ZZ ∂∂ZZ lim lim lím lím lim lim P→∞ P→0 P→∞ ∂∂ P P→0 ∂∂ P P TT P TT Observe quein ellimit límiteas 0,Vand → ∞, el límite conforme P→ Note the PP → that the limit as ∞, b. Note that that inen the limit asconforme → 0, 0, VVP → → ∞, ∞, and thatyin inque theen limit as PP → → ∞, VV → → b. ∞, V → b. metano 60(°F) yF)1(atm) es equivalente a 1(gal) de gasolina comoas 3.58. 3.58. 140(ft) of methane gas and is to of 3.58.SiIf If140(pie) 140(ft)333 de of gas methane gasa at at 60( 60(◦◦F) and 1(atm) 1(atm) is equivalent equivalent to 1(gal) 1(gal) of gasoline gasoline ascombustible para de automóvil, ¿cuál sería el volumen del tanque necesitato fuel an engine, what would be of tank hold fuelunfor formotor an automobile automobile engine, what would be the the volume volume of the theque tankserequired required topara holdmantener el methane 3,000(psia) and F) an equivalent to metano a 3at 000(psia) y 60(°F) en◦◦una equivalente a 10(gal) de of gasolina? F) in incantidad an amount amount equivalent to 10(gal) 10(gal) of gasoline? gasoline? methane at 3,000(psia) and 60( 60( 121 Problems 3.59. Determine aa good for factor hydrogen va3.59.Determine Determine good estimate for the the compressibility factor ZZ of of saturated saturated hydrogen va-de hidróge3.59. una buenaestimate estimación paracompressibility el factor de compresibilidad Z de vapor saturado at 25 and bar. For an ZZ = por atK 25yK K3.213 and 3.213 3.213 bar. For comparison, comparison, an experimental experimental value is 0.7757. = 0.7757. 0.7757. nopor a 25 bar. Por comparación, un valor experimentalvalue es Z is = 3.57. For a gas described by the Redlich/Kwong equation and for a temperature greater than 3.60. Boyle is temperature for 3.60.LaThe The BoyleTtemperature temperature is the the temperature for which: which: slopes, for limiting 3.60. temperatura de Boyleexpressions es aquella parathe la two que: c , develop � � � � � � � � ∂∂∂ZZZ ∂Z = lim = 00 lim lim lim lím P→0 PP TTT P→∞ ∂ P T P→0 ∂∂∂P P→0 Show that second virial coefficient is at Boyle temperature. a) (a) Muestre que el segundo coeficiente virial eszero ceroand la temperatura deas Boyle. (a) ShowNote that the the second virial coefficient is zero ata the the Boyle temperature. that in the limit as P→ 0, VBBB→ ∞, that in the limit P → ∞, V → b. la the correlación generalizada para (3.63), para estimar la temperatura b) (b) Use Use correlation for Eq. to the Boyle (b) Use the generalized generalized correlation forB,B, B,ecuación Eq. (3.63), (3.63), to estimate estimate the reduced reduced Boyle de Boyle 3.58. Ifpara 140(ft) of simples. methane reducida fluidos temperature for fluids. temperature for3simple simple fluids.gas at 60(◦ F) and 1(atm) is equivalent to 1(gal) of gasoline as fuel for an automobile engine, what would be the volume of the tank required to hold ◦ F) in an amount equivalent to 10(gal) of gasoline? methane 3,000(psia) andis 3.61. entregagas gas naturalat(suponga metano puro) a unato ciudad través de unat conducto con una relación 3.61. (assume pure delivered aa city pipeline aa volumetric 3.61.SeNatural Natural gas (assume pure methane) methane) is60( delivered to cityavia via pipeline at volumetric ◦◦F) promedio rate million standard cubic per Average delivery conditions volumétrica 150 millones piesfeet cúbicos estándar por día. Las condiciones de50( entrega rate of of 150 150de million standardde cubic feet per day. day. Average delivery conditions are are 50( F) 3.59. aDetermine: good estimate for the compressibility factor Z of saturated hydrogen vaand 300(psia). Determine: and 300(psia). Determine: son 50(°F) yDetermine 300(psia). por at 25 K and 3.213 bar. For comparison, an experimental value is Z = 0.7757. volumetric delivery rate cubic feet day. (a) The volumetric delivery rate in in actual actual cubic feet per per day.por día. a) (a) LaThe relación de entrega volumétrica en pies cúbicos reales (b) The molar delivery rate in kmol per hour. (b) The molar delivery rate in kmol per hour. Thede Boyle temperature the temperature b) La3.60. relación entrega molar eniskmol por hora. for which: −1.. gas at conditions m ss−1 (c) The gas velocity velocity atadelivery delivery conditions in m� c) (c) LaThe velocidad del gas las condiciones de in entrega en�m s–1. ∂Z = 0of lim The inside diameter The pipe pipe is is 24(in) 24(in) schedule-40 schedule-40 steel steel with with an an inside∂ P diameter of 22.624(in). 22.624(in). Standard Standard P→0 T interior de 22.624(in). Las condiciones Laconditions tubería es de acero de 24(in), cédula 40, con un diámetro ◦ ◦ F) and and 1(atm). 1(atm). conditions are are 60( 60( F) estándar son 60(°F) y 1 atm. (a) Show that the second virial coefficient B is zero at the Boyle temperature. 3.62. correlations use critical compressibility factor rather 3.62. Some Some corresponding-states corresponding-states correlations use the thefor critical compressibility factor ZZ cc,, rather (b) Use the generalized correlation B, Eq. (3.63), to estimate the reduced Boyle deω, estados correspondientes usan eltypes factor de compresibilidad crítica Zc, más 3.62. Algunas than acentric factor as a third parameter. The two of correlation (one based than the thecorrelaciones acentric factor ω, as a third parameter. The two types of correlation (one based temperature for simple fluids. que el factor acéntrico w, como un tercer parámetro. Las dos clases de correlación (uno on on TTcc,, PPcc,, and and ZZcc,, the the other other on on TTcc,, PPcc,, and and ω) ω) would would be be equivalent equivalent were were there there aa oneone- basado en ,to-one Pc y Zcorrespondence , el otro en T , P y w) serían equivalentes donde exista una correspondencia uno a uno Tcto-one c c and data B aa test between ZZcc methane) and ω. ω. The The data of of App. App. B allow allow test of ofatthis this between 3.61.correspondence Natural gas c(assume pure is delivered to a city via pipeline a volumetric y w. La información del apéndice B permite una prueba de esta correspondencia. Prepare entre Z c ◦ F) correspondence. Prepare aa plot of ω see how well with correspondence. Prepare plot of ZZccubic vs. feet ω to toper seeday. how well ZZcdelivery correlates with ω. ω.are 50( rate of 150 million standard Average conditions c vs. c correlates en función de w para ver qué tan bien Z se correlaciona con w. Desarrolle una una gráfica de Z c c Develop linear correlation (Z = a + bω) for nonpolar substances. Develop aaand linear correlation (Z = a + bω) for nonpolar substances. 300(psia). Determine: cc correlación lineal (Zc = a + bw) para sustancias no polares. The volumetric delivery rate in actual cubic volume) feet per day. 3.63. suggests that (paths of are 3.63. Figure Figure 3.3 3.3(a) suggests that the the isochores isochores (paths of constant constant volume) are approximately approximately figura 3.3 sugiere que las isocoras (trayectorias de volumen constante) sonisochores. líneas casi rectas en 3.63. Lastraight (b) on The delivery rate that in kmol per hour. models aa PPmolar TT diagram. Show the straight lines lines on diagram. Show that the following following models imply imply linear linear isochores. −1 un diagrama(c) PT.The Muestre que los siguientes modelos implican isocoras lineales. gas velocity at delivery conditions in m s . (a) (a) Constant-β, Constant-β, κκ equation equation for for liquids. liquids. (b) (b) Ideal-gas Ideal-gas equation. equation. (c) (c) Van Van der der Waals Waals The pipe is 24(in) schedule-40 steel with an inside of 22.624(in). Standard equation. equation. para líquidos k, –b constante. b) Ecuación dediameter gas ideal. c) Ecuación de van a) Ecuación conditions are 60(◦ F) and 1(atm). der Waals. 03-SmithVanNess.indd 121 3.62. Some corresponding-states correlations use the critical compressibility factor Z c , rather than the acentric factor ω, as a third parameter. The two types of correlation (one based on Tc , Pc , and Z c , the other on Tc , Pc , and ω) would be equivalent were there a oneto-one correspondence between Z c and ω. The data of App. B allow a test of this 10/1/07 correspondence. Prepare a plot of Z c vs. ω to see how well Z c correlates with ω. 14:08:10 122122 122 122 CAPÍTULO 3. Propiedades volumétricas de fluidos puros CHAPTER Volumetric Properties Fluids CHAPTER CHAPTER 3.3.3. Volumetric Volumetric Properties Properties ofofPure ofPure Pure Fluids Fluids 3.64. A continuación se muestra el árbol de decisión de la ecuación de estado. Para cada elemento discu3.64. Shown below Equation Decision Tree. For each item discuss the condi3.64. 3.64. Shown Shown below below isisthe isthe the Equation Equation ofofState ofState State Decision Decision Tree. Tree. For For each each item item discuss discuss the the condicondita tions las condiciones másits apropiadas para su uso. appropriate use. tions tions appropriate appropriate totoits to its use. use. �� � (a)(a) Ideal gas (a) Ideal Ideal gas gas ideal a) Gas Gas� Gas Gas Gas �� �� � b) virial Ecuación virial de dos términos (b) 2-term virial equation (b) (b) 2-term 2-term virial equation equation �� � c)equation Ecuación cúbica de estado (c)(c) Cubic equation (c) Cubic Cubic equation ofof state ofstate state d) Tablas de Lee/Kesler, apéndice E �� � (d) Lee/Kesler tables, Appendix (d) (d) Lee/Kesler Lee/Kesler tables, tables, Appendix Appendix EE E Gas Gas Gas ororor � ¿Gas o �� liquid? liquid? liquid? líquido? �� � (e)(e) Incompressible liquid (e) Incompressible Incompressible liquid liquid e) Líquido incompresible �� � (f) Rackett equation, Eq. (3.72) (f) (f) Rackett Rackett equation, equation, Eq. Eq. (3.72) (3.72) Liquid Liquid Liquid f) Ecuación de Rackett, ecuación (3.72) �� � Líquido �� � (g) Constant (g) (g) Constant Constant ββand βand and κκ κ g) b y k constantes �� � (h) Lydersen chart, Fig. 3.16 (h) (h) Lydersen Lydersen etetal. etal.al. chart, chart, Fig. Fig. 3.16 3.16 h) Diagrama de Lydersen, et al., figura 3.16 3.65. Un gas ideal, inicialmente a◦25 °C 1 bar, bar, se somete alfollowing siguiente proceso cíclicoininen ◦25 3.65. An ideal gas, initially C◦and undergoes the cyclic processes 3.65. 3.65. An An ideal ideal gas, gas, initially initially atat25 at25 C Cand and 1y1bar, 1bar, undergoes undergoes the the following following cyclic cyclic processes processes aina un a sistema cerrado: closed system: closed closed system: system: a)(a) En mecánicamente reversibles se comprime de manera to adiabática (a) reversible processes, compressed adiabatically to5to5bar, (a) InInprocesos mechanically Inmechanically mechanically reversible reversible processes, processes, ititprimero isitisfirst isfirst first compressed compressed adiabatically adiabatically 5bar, bar,a 5 bar, a ◦hasta ◦ C, continuación seataenfría a presión constante de 525 bar 25 °C yexpanded por últimoisotherseisotherexpande isotérC, and finally expanded isotherthen cooled pressure 25◦25 C, and and finally finally expanded then then cooled cooled at ataconstant aconstant constant pressure pressure ofof5of5bar 5bar bar toto to micamente a its suoriginal presión original. mally pressure. mally mally totoits toits original original pressure. pressure. b)(b) El ciclo es irreversible y cada etapa tiene una eficiencia de of 80% en comparación con el corres(b) The cycle and each step has anan efficiency ofof 80% compared with (b) The The cycle cycle isisirreversible isirreversible irreversible and and each each step step has has an efficiency efficiency 80% 80% compared compared with with pondiente proceso que es mecánicamente reversible. El ciclo completo consiste en una the corresponding mechanically reversible process. The cycle still consists the the corresponding corresponding mechanically mechanically reversible reversible process. process. The The cycle cycle still still consists consists ofofan ofan an etapa de compresión adiabática, una etapa de enfriamiento isobárico y una expansión isotérmica. adiabatic compression step, isobaric cooling step, and isothermal expansion. adiabatic adiabatic compression compression step, step, ananan isobaric isobaric cooling cooling step, step, and and ananan isothermal isothermal expansion. expansion. Calculate Q,Q, �U and �H for each step the process and for the cycle. Calculate Calculate Q, WW,W ,∆H �U , ,para and , and �H �H for for each each step step ofofof the process process and and for for the the cycle. cycle. Calcule Q, W, ∆U y,�U cada etapa del proceso y the para el ciclo. Considere que Take CTake = (7/2)R y CV PTake and CPC =(7/2)R (7/2)R and and CCVC =(5/2)R. (5/2)R. P= V= P=(7/2)R V=(5/2)R. =C(5/2)R. 3.66. Show that the density-series second virial coefficients can derived from isothermal 3.66. 3.66. Show Show that that the the density-series density-series second second virial virial coefficients coefficients can can be derived derived from from isothermal isothermala partir de 3.66. Demuestre que los segundos coeficientes viriales en serie debebe densidad pueden obtenerse volumetric data via the expression: volumetric volumetric data data via via the the expression: expression: información volumétrica isotérmica mediante la expresión: (Z(Z density) lim (Z −−1)/ρ −1)/ρ 1)/ρ ρρr(densidad (molar ρ(molar (molar density) density) ≡≡1/V ≡1/V 1/V BB= B== lim lim lím molar) 1/V) ρ→0 ρ→0 ρ→0 3.67. Haga uso de la ecuación del problema anterior yand ladata información deF.2 laF.2 tabla F.2 para obtener 3.67. Use the equation preceding problem and data from Table 3.67. 3.67. Use Use the the equation equation ofofthe ofthe the preceding preceding problem problem and data from from Table Table F.2 totoobtain toobtain obtain a avalue avalue value un valor de B para el agua a una de las siguientes temperaturas: water following temperatures: ofofof BBfor Bfor for water water atatone atone one ofofthe ofthe the following following temperatures: temperatures: ◦C ◦C ◦C ◦C ◦ C(b) ◦C ◦ Cb) ◦C ◦C a)(a) 300 °C 350 °C (b) 350 (c) 400 (a) 300 (b) 350 350 (c) (c) 400 400 (a) 300 300 03-SmithVanNess.indd 122 c) 400 °C 10/1/07 14:08:52 Problemas Problems Problems Problems 123 123 123 123 3.68. Obtenga los valores de W, ψ yand ZZcZcdados en la tabla 3.1 para: 3.68. 3.68. 3.68. Derive Derive Derive the the the values values values ofof of �, �, �, �, �, �, and and Zcgiven given ininin Table Table Table 3.1 3.1 3.1 for: for: for: cgiven a)(a) La ecuación de estado deequation Redlich/Kwong. (a) (a)The The The Redlich/Kwong Redlich/Kwong Redlich/Kwong equation equation ofofof state. state. state. b)(b) La ecuación de estado de Soave/Redlich/Kwong. (b) (b)The The The Soave/Redlich/Kwong Soave/Redlich/Kwong Soave/Redlich/Kwong equation equation equation ofofof state. state. state. c)(c) La ecuación de estado de Peng/Robinson. (c) (c)The The The Peng/Robinson Peng/Robinson Peng/Robinson equation equation equation ofofof state. state. state. 3.69. Suponga que se tienen disponibles datos de Z en función de Pr a Tr constante. Demuestre que el data data are are are available available available at constant constant constant TTr rT. .r Show .Show Show that that that the the the reduced reduced reduced densitydensitydensity3.69. 3.69. 3.69. Suppose Suppose Suppose ZZZvs. vs. vs.PPr P r data rvirial segundo coeficiente reducido enatat serie de densidad puede obtenerse a partir de tales datos series series series second second second virial virial virial coefficient coefficient coefficient can can can be be be derived derived derived from from from such such such data data data via via via the the the expression: expression: expression: mediante la expresión: B̂B̂B̂ ===lim lim lim (Z (Z (Z −−− 1)Z 1)Z 1)Z TTr rT/P /P lím r /P rr r PP r →0 rP→0 r →0 Sugerencia: fundamente el desarrolloon en la expansión virial en densidad completa, ecuación (3.12). Suggestion: Suggestion: Suggestion: Base Base Base the the the development development development on on the the the full full full virial virial virial expansion expansion expansion ininin density, density, density, Eq. Eq. Eq. (3.12) (3.12) (3.12) Utilice el resultado del problema anterior yand los datos de laTable tabla E.1 obtener un valor 3.70. 3.70. 3.70. 3.70. Use Use Use the the the result result result ofofof the the the preceding preceding preceding problem problem problem and and data data data from from from Table Table E.1 E.1 E.1 topara toto obtain obtain obtain aavalue avalue value ofofof de B̂ para fluidos simples con T = 1. Compare el resultado con el valor dado por la ecuación (3.65). r at B̂B̂B̂ for for for simple simple simple fluids fluids fluids atat TTr rT==r 1. =1.1. Compare Compare Compare the the the result result result with with with the the the value value value implied implied implied by byby Eq. Eq. Eq. (3.65). (3.65). (3.65). 3.71. La siguiente conversación fuewas escuchada en in los pasillos de una compañía de ingeniería. 3.71. 3.71. 3.71. The The The following following following conversation conversation conversation was was overheard overheard overheard inin the the the corridors corridors corridors ofofof aimportante alarge alarge large engineering engineering engineering firm. firm. firm. New New New engineer: engineer: engineer: ”Hi, ”Hi, ”Hi, boss. boss. boss. Why Why Why the the the big big big smile?” smile?” smile?” Ingeniero novato: “Hola, jefe. ¿Por qué tan contento? Old-timer: Old-timer: Old-timer: ”I ”I ”I finally finally finally won won won a a wager a wager wager with with with Harry Harry Harry Carey, Carey, Carey, from from from Research. Research. Research.He He He bet bet bet me me me that that that Me aposVeterano: “Por fin le gané una apuesta a Uriel Ortega, del departamento de investigación. I I couldn’t I couldn’t couldn’t come come come up up up with with with a a quick a quick quick but but but accurate accurate accurate estimate estimate estimate for for for the the the molar molar molar volume volume volume of of of ararar- del argón tó que no se me podría ocurrir una estimación rápida pero precisa para el volumen molar ◦◦CC ◦C gon gon gon at at at 30 30 30 and and and 300 300 300 bar. bar. bar. Nothing Nothing Nothing to to to it; it; it; I I used I used used the the the ideal-gas ideal-gas ideal-gas equation, equation, equation, and and and got got got about about about a 30 °C33y3 300 bar. Nada más fácil: utilicé la ecuación del gas ideal y obtuve aproximadamente 83 −1 −1 −1 83 83 cm cm cm–1 mol mol . .Harry .negó Harry Harry shook shook shook his his his head, head, head, but but but paid paid paid up. up. up. What What What do dodo you you you think think think about about that?” that?” that?” 383 cm mol .mol Uriel con la cabeza, pero pagó. ¿Qué piensas acerca deabout esto? New New New engineer engineer engineer (consulting (consulting (consulting his his his thermo thermo thermo text): text): text): ”I”I”I think think think you you you must must must be bebe living living living right.” right.” right.” Ingeniero novato (consultando su texto de termodinámica): “Pienso que debe de estar en lo correcto”. Argon Argon Argon atatat the the the stated stated stated conditions conditions conditions isisis not not not an anan ideal ideal ideal gas. gas. gas.Demonstrate Demonstrate Demonstrate numerically numerically numerically why why why the the the Elold-timer argón a won las condiciones old-timer old-timer won won his his his wager. wager. wager.establecidas no es un gas ideal. Demuestre numéricamente por qué el veterano ganó la apuesta. 3.72. 3.72. 3.72. Five Five Fivemol mol molofofof calcium calcium calcium carbide carbide are are are combined combinedwith with with10 1010mol mol molofofde ofwater water waterinen ininaaun closed, aclosed, closed,rigid, rigid, rigid, 3.72. Cinco moles de carburocarbide de calcio secombined combinan con 10 moles agua recipiente cerrado, rígi33internal 3internal 3 high-pressure high-pressure high-pressure vessel vessel vessel of of of 1800 1800 1800 cm cm cm internal empty empty empty volume. volume. volume. Acetylene Acetylene Acetylene gas gas gas is is is produced produced produced do, de alta presión y con un volumen vacío interno de 1 800 cm . Se produce gas de acetileno meby byby the the the reaction: reaction: diante lareaction: reacción: (s) ++ 2H 2H 2H → → CC2C (g) + Ca(OH) Ca(OH) Ca(OH) (s) CaC CaC CaC CaC ++ O(l) → H (g) +++ 22(s) 2(s) 22O(l) 2 O(l) 22H 2 (g) 22(s) 2 (s) 2(s) 2H 22(g) ElThe recipiente contiene un embalaje con una porosidad de 40% para evitar ladecomposidescomposición exploThe The vessel vessel vessel contains contains contains packing packing packing with with with aaporosity aporosity porosity ofofof 40% 40% 40% tototo prevent prevent prevent explosive explosive explosive decomposidecomposi◦de ◦CC ◦C siva del acetileno. Las condiciones iniciales son 25 °C 1bar, bar, ythe la reacción llega atoto completarse. and and and 11ybar, 1bar, and and and the the reaction reaction reaction goes goes goes to tion tion tion ofofof the the the acetylene. acetylene. acetylene. Initial Initial Initial conditions conditions conditions are are are 25 2525 La reacción esThe exotérmica, pero debido a but la transferencia detransfer, calor, lathe temperatura final es de sólo completion. completion. completion. The The reaction reaction reaction isis is exothermic, exothermic, exothermic, but but owing owing owing tototo heat heat heat transfer, transfer, the the final final final temperatemperatempera◦◦C. ◦la 125 °C. Determine presión final en el recipiente. ture ture ture isisis only only only 125 125 125 C. C. Determine Determine Determine the the the final final final pressure pressure pressure ininin the the the vessel. vessel. vessel. –1 ◦◦C, ◦ C, 333mol 3mol −1 −1 −1 Nota: aAt 125 °C elC, volumen molar delof Ca(OH) de 33.0 cm mol .. .Ignore los efectos de cualquier the the the molar molar molar volume volume volume ofof Ca(OH) Ca(OH) Ca(OH) 33.0 33.0 33.0 cm cm cm mol Ignore .Ignore Ignore the the the effects effects effects Note: Note: Note: AtAt 125 125 125 2 es 22is 2isis gas (por ejemplo, aire) que de manera inicial haya estado presente en dicho recipiente. ofofof any any any gases gases gases (e.g., (e.g., (e.g., air) air) air) initially initially initially present present present ininin the the the vessel. vessel. vessel. ◦◦CC ◦C 3.73. Se requiere almacenar 3535,000 000 kg deofofpropano, que es recibido como un gas a 1(atm). 10 °C y 1(atm). Se 3.73. 3.73. 3.73. Storage Storage Storage isisis required required required for for for 35,000 35,000 kg kgkg of propane, propane, propane, received received received asasas aagas agas gas atatat 10 10 10 and and and 1(atm). 1(atm). proponen dos opciones: Two Two Two proposals proposals proposals have have have been been been made: made: made: ◦◦CC ◦ and (a) (a) Store Store Store ititas itasas aacomo gas agas gas atatun at 10 1010 and 1(atm). 1(atm). a)(a) Almacenarlo gasC a and 101(atm). °C y 1(atm). ◦◦CC ◦C and and 6.294(atm). 6.294(atm). 6.294(atm). For For For this this thiseste modo (b) (b) (b) Store Store Store it it as it as as a a liquid a liquid liquid in in in equilibrium equilibrium equilibrium with with with its itsits vapor vapor vapor at 10 1010 b) Almacenarlo como un líquido en equilibrio con suatat vapor aand 10 °C y 6.294(atm). Para mode mode mode of of of storage, storage, storage, 90% 90% 90% of of of the the the tank tank tank volume volume volume is is is occupied occupied occupied by by by liquid. liquid. liquid. de almacenamiento, 90% del volumen del tanque es ocupado por líquido. 03-SmithVanNess.indd 123 10/1/07 14:09:43 124 124124 CAPÍTULO 3. Propiedades volumétricas de fluidos puros CHAPTER Volumetric Properties of Pure Fluids CHAPTER 3. 3. Volumetric Properties of Pure Fluids Compare las dostwo opciones, discutiendo lospros puntos acons favorofy each. en contraquantitative de cada una.where Trate de ofrecer Compare proposals, discussing Compare thethe two proposals, discussing pros andand cons of each. BeBe quantitative where argumentos cuantitativos siempre que sea posible. possible. possible. 3.74. La definición del factor de compresibilidad Z, ecuación (3.10), puede escribirse de la forma más 3.74. The definition compressibility factor , Eq. (3.10), may written more 3.74. The definition of of compressibility factor Z , ZEq. (3.10), may be be written in in thethe more intuitiva: intuitive form: intuitive form: V V Z≡ Z≡ V (gas (idealgas) ideal) V (idealgas) where both volumes are at the same T and P. Recall ideal aunmodel sub-es una suswhere both volumes are at the same T and P. Recall that ideal gasgas isque aismodel donde ambos volúmenes se encuentran a las mismas T that y an P. an Recuerde gassubideal stance comprising particles with no intermolecular forces. Use the intuitive definition stance comprising particles with no intermolecular forces. Use the intuitive definition tancia modelo que tiene partículas sin fuerzas intermoleculares. Utilice la definición intuitiva de Z of Z argue to argue that: of Z to that: para sostener que: attractions promote values Z 1. < 1. (a)(a) Intermolecular promote values of of Zvalores < a) LasIntermolecular atraccionesattractions intermoleculares promueven de Z < 1. repulsions promote values Z 1. > 1.de Z > 1. (b)(b) Intermolecular promote values of of Zvalores > b) LasIntermolecular repulsionesrepulsions intermoleculares promueven Aequilibrio balance of repulsions implies Z =1.(Note 1.(Nótese (Note gas es un caso (c)(c) A of attractions andand repulsions implies thatthat Z= 1. thatthat an an ideal gas c) Unbalance deattractions atracciones y repulsiones implica que Z= que unideal gas ideal a special case there or repulsions.) is aisspecial case forfor which areare no no attractions or repulsions.) especial para el que nowhich haythere atracciones ni attractions repulsiones). 3.75. Escriba forma general ecuación estado 3.75. Write the general form of una an equation ofdestate 3.75. Write thela general form of de an equation of state as:as: como: =+11 Z +rep ZZrep ZZatr(T, (T, (ρ) −attr (T, ZZ 1= + rep(r) attr (ρ) − –Z ρ)r)ρ) Z= where Z (r) represents from representsla contriburep (ρ) attr ZZrep represents contributions from repulsions, andand ρ) represents where donde representa la contributions contribución de lasrepulsions, repulsiones yZ attr ZZatr(T, (T,(T, r)ρ) representa rep(ρ) contributions from attractions. What repulsive and attractive contributions contributions from attractions. areare thethe repulsive and attractive contributions ción de las atracciones. ¿CuálesWhat son las contribuciones repulsivas y de atracción detolatoecuación de the Waals equation of state? the vanvan derder Waals of state? estado de van der equation Waals? 3.76. below four proposed modifications Waals equation of state. 3.76. A Given continuación se proponen cuatro modificaciones avan la van ecuación deequation estado deofvan der Waals. ¿Son 3.76. Given below areare four proposed modifications of of thethe derder Waals state. Are any of these modifications reasonable? Explain carefully; statements such as, “It afirmaciorazonables algunas de estas modificaciones? Explique de manera cuidadosa poras,qué Are any of these modifications reasonable? Explain carefully; statements such “It las isn’t cubic in volume.” do not qualify. nes del tipoin“no es cúbica en el volumen”, no son válidas. isn’t cubic volume.” do not qualify. RTRT a a P= (a)(a) −− P= V b− b V V V− RTRT a a P= (b)(b) P= −2 − 2 (V − b) V V (V − b) RTRT a a P= (c)(c) P= − −2 2 V (V − b) V V V (V − b) RTRT a a P= −− 2 (d)(d) P= V V V 2V 3.77. With reference to Pb. 2.43, assume to be an ideal develop expression 3.77. With reference Pb. 2.43, assume air air toque be an ideal andand develop an an expression 3.77. Con respecto altoproblema 2.43, suponga el aire esgas, ungas, gas ideal y desarrolle una expresión en la giving the household air temperature as a function of time. giving the household air temperature as a function of time. que proporcione la temperatura del aire doméstico como una función del tiempo. 3.78. A garden hose water valve shut closed insethe sun, of 3.78. AUna garden hose with thethe water valve shut andand thethe nozzle closed sitssits in the sun, fullfull ofexpuesta 3.78. manguera dewith jardín con la válvula del agua y lanozzle boquilla cerradas encuentra a los ◦ C and 6 bar. After some time the temperature ◦10 liquid water. Initially, the water is at C and 6 bar. After some time the temperature liquid water. Initially, the water is at 10 rayos del sol y llena de agua líquida. Al principio, el agua tiene una temperatura de 10 °C y 6 bar. ◦ C. Owing to the increase in temperature and pressure and the of the water rises to ◦40 of the water totiempo 40 C. la Owing to the increase temperature pressure and the Después de rises cierto temperatura del aguainasciende a 40 and °C. Obedeciendo al incremento de elasticity of the hose, the internal of the hose increases 0.35%. Estimate elasticity of the hose, theasí internal of the hose increases by 0.35%. Estimate temperatura y presión, como adiameter ladiameter elasticidad de la manguera, el by diámetro interno de ésta se inthe final pressure of the water in the hose. the final pressure of the water in the hose. crementa 0.35%. Calcule la presión final del agua en la manguera. −1 −1 Data: β(ave) = 250 × −6 10−6−1 K−1 ; κ(ave) = 45 × −6 10−6 bar = 250 × 10 = 45 × ×10 Data: Datos:β(ave) b(prom) = 250 × 10–6KK–1; ;κ(ave) k(prom) = 45 10–6bar bar–1 03-SmithVanNess.indd 124 8/1/07 13:02:44 Capítulo 4 Efectos térmicos La transferencia de calor es una de las operaciones más comunes en la industria química. Considere, por ejemplo, la fabricación del etilenglicol (un agente anticongelante) mediante la oxidación de etileno a óxido de etileno y de su hidratación posterior a glicol. La reacción de oxidación catalítica resulta más efectiva cuando se realiza a temperaturas cercanas a 250 °C. Por lo tanto, los reactivos etileno y aire se calientan a esta temperatura antes de que entren al reactor. Para diseñar el precalentador es necesario saber cuánto calor se transfiere. Las reacciones de combustión del etileno con el oxígeno en la cama catalítica tienden a incrementar la temperatura. Sin embargo, si se elimina el calor del reactor, la temperatura no aumenta más de 250 °C. Las temperaturas superiores promueven la formación de CO2, que es un producto no deseado. Para el diseño del reactor se requiere conocer la rapidez de transferencia de calor, y ésta depende de los efectos térmicos asociados con las reacciones químicas. El producto óxido de etileno se hidrata a glicol por la absorción en agua. El calor se desprende debido no sólo al cambio de fase y al proceso de disolución, sino también por una reacción de hidratación entre el óxido de etileno disuelto y el agua. Al final, el glicol se recupera del agua a través de la destilación, un proceso de vaporización y condensación que origina la separación de una solución en sus componentes. Todos los efectos térmicos importantes se ilustran por medio del proceso, relativamente simple, de la fabricación de una sustancia química. A diferencia de los efectos del calor sensible, caracterizados por los cambios de temperatura, los efectos térmicos de una reacción química, la transición de fase, así como la formación y la separación de soluciones se determinan a partir de medidas experimentales hechas a temperatura constante. En este capítulo, la termodinámica se aplica a la evaluación de la mayoría de los efectos térmicos que acompañan a las operaciones físicas y químicas. No obstante, los efectos térmicos de los procesos de mezclado, que dependen de las propiedades termodinámicas de las mezclas, se tratan en el capítulo 12. 4.1 EFECTOS DEL CALOR SENSIBLE La transferencia de calor a un sistema en el que no hay transiciones de fase, ni reacciones químicas o algún cambio en la composición, provoca que cambie la temperatura del sistema. Nuestro propósito es desarrollar relaciones entre la cantidad de calor transferida y el cambio de temperatura resultante. Cuando el sistema es una sustancia homogénea de composición constante, la regla de la fase indica que al fijar los valores de dos propiedades intensivas se establece su estado. Por lo tanto, la energía interna molar o específica de una sustancia se puede expresar como una función de las otras dos variables de estado. Cuando 125 04-SmithVanNess.indd 125 8/1/07 13:07:19 126 CAPÍTULO 4. Efectos CHAPTER 4. Heat Heat Effectstérmicos 126 126 CHAPTER 4. 4. Effects CHAPTER Heat Effects 126 126 CHAPTER 4. Heat Effects éstas seother seleccionan en forma arbitraria como temperatura y volumen molar and o específico, U = U(T, V). state variables. When these are arbitrarily selected as temperature molar or or specific specific other state variables. When these are arbitrarily selected as temperature and molar De donde, other state When these are arbitrarily selected as temperature and molar or specific volume, U variables. =U U (T, (T, V V ). ). Whence, other state variables. When these are arbitrarily selected as temperature and molar or specific volume, U = Whence, volume, U = U (T, V ). Whence, � � � � volume, U = U (T, V ). Whence, � � � � ∂U � ∂U � � ∂U � ∂U dU = dT + V � dT + � ∂U � dd V dU = = � ∂U ∂T T V dT ∂V V T dV dU + ∂∂U ∂U ∂ dU = ∂ T VVV dT + ∂ V TTT d V ∂T V ∂V T As aa result result of Eq. (2.16)(2.16), this becomes: becomes: Como resultado de la ecuación ésta será: As of Eq. (2.16) this As a result of Eq. (2.16) this becomes: � � As a result of Eq. (2.16) this becomes: � � ∂U � � ∂U dT + + � ∂U � dd V V dU = =C C V dT dU V dT + ∂V V T dV dU =C ∂U V V ∂ T dU = C V dT + ∂ V TT d V ∂V T The final term may be set equal to zero in in two two circumstances: circumstances: The final final term term may may be be set set equal equal to to zero zero The in two circumstances: Existe laThe posibilidad quebeelset término seainigual cero bajo dos circunstancias: final termdemay equal final to zero two acircumstances: • For For any any constant-volume constant-volume process, process, regardless regardless of of substance. substance. • • For anyproceso constant-volume regardless of substance. • Para cualquier a volumenprocess, constante sin importar la sustancia. • For any constant-volume process, regardless of substance. • Whenever the internal energy is independent of volume, regardless of of the the process. process. This This Whenever the internal internal energy is independent independent of volume, volume,sinregardless regardless • Siempre que la energía interna energy sea independiente del volumen, considerar proceso. Esto es verda•• Whenever the is of ofelthe process. This is exactly true for ideal gases and incompressible fluids and approximately true for low• is Whenever the internal energy is independent of volume, regardless of the process. This is exactly true for ideal gases and incompressible fluids and approximately true for lowdero para gases ideales y fluidos incompresibles, y aproximadamente cierto para gases a baja exactlygases. true for ideal gases and incompressible fluids and approximately true for low-presión. pressure is exactly true for ideal gases and incompressible fluids and approximately true for lowpressure gases. pressure gases. pressure gases. En cualquier caso, dU = =C In either either case, dU CVV dT dT In case, dU =C C dT V dT In either case, dU = V V In either case, dU = � C T V dT � T22 � T y C V dT dT (4.1) (4.1) and �U = = � T22 C (4.1) and �U V dT (4.1) and �U = TT1T2 C V V 1 (4.1) and �U = TT11 C V dT T1 For a mechanically reversible constant-volume process, Q = �U , and Eq. (2.19) may be escriPara unFor proceso a volumen constante reversible, ∆U,�U y la and ecuación (2.19) may se puede mechanically reversiblemecánicamente constant-volume process, QQ Q= = = Eq. (2.19) (2.19) be For aa mechanically reversible constant-volume process, �U ,, and Eq. may be written for a unit mass or a mole: bir parawritten unaamol For mechanically reversible constant-volume process, Q = �U , and Eq. (2.19) may be foro aamasa unit unitaria: mass or aa mole: mole: written for unit mass or � T2 written for a unit mass or a mole: � T � TT22 C V dT Q = �U = Q= = �U �U = = �T T22 C C V dT dT Q 1 V V Q = �U = TTT111 C V dT T1 Similarly, the the molar molar or or specific specific enthalpy enthalpy may may be expressed expressed as as a function function of of temperature temperature Similarly, be De manera similar, la entalpía molar o específica se expresa en función de la temperatura y de la preSimilarly, the H molar or(T, specific enthalpy may be expressed as aa function of temperature and pressure. Then = H P), and Similarly, the molar or specific enthalpy may be expressed as a function of temperature and pressure. Then H = H (T, P), and sión. En tal caso, H = H(T, P), y and pressure. Then H = H (T, P), and � � � � and pressure. Then H = H (T, P), and � � � � ∂H H� ∂H H� � � ∂ ∂ dT + + � ∂ H � dd P P dH H= = � ∂ H � dT T P dT + ∂H P T dP dd H = ∂∂ T H ∂ P P T d H = ∂ T PP dT + ∂ P TT d P ∂T P ∂P T As a result of Eq. (2.20) this becomes: As a result of Eq. (2.20) this becomes: As a result of Eq. (2.20) this becomes: Como resultado deof la Eq. ecuación ésta será: � � As a result (2.20)(2.20), this becomes: � � ∂H H� � ∂ dT + + � ∂ H � dd P P dH H= =C C P dT P dT + P T dP dd H =C ∂∂ H P P P T d H = C P dT + ∂ P TT d P ∂P T Again, two circumstances allow the final final term term to to be be set set equal equal to to zero: zero: Again, two two circumstances circumstances allow allow the the Again, final term to be set equal to zero: twocircunstancias circumstancespermiten allow theque final term to befinal set equal to zero: Una vezAgain, más, dos término seasubstance. igual a cero: • For For any constant-pressure constant-pressure process, process,elregardless regardless of the the • any of substance. • For anyproceso constant-pressure process, regardless of the substance. • Para cualquier a presión constante, sin importar la sustancia. • For any constant-pressure process, regardless of the substance. • Whenever the enthalpy of the substance is independent of pressure, pressure, regardless regardless of of the the • Whenever the enthalpy of the substance is independent of Whenever the enthalpy oftrue thefor substance is independent of pressure, regardless the Esto que la entalpía de la sustancia sea independiente de la presión, sin considerar el of proceso. • Cada•vez process. This is exactly ideal gases and approximately true for low-pressure • process. WheneverThis the is enthalpy thefor substance is independent of pressure, of the process. This exactlyoftrue true for ideal gases gases and approximately approximately true regardless for low-pressure low-pressure exactly ideal and for es precisamente cierto is para gases ideales, y aproximadamente válido paratrue gases a baja presión. gases. process. This is exactly true for ideal gases and approximately true for low-pressure gases. gases. gases. 04-SmithVanNess.indd 126 8/1/07 13:07:21 4.1. Sensible Heat Effects 4.1. Sensible Heat Effects 4.1. Sensible Sensible Heat Effects 4.1. 4.1. Sensible Heat Effects Effects 4.1. Efectos del calorHeat sensible In either case, In either case, Ineither either case, En cualquier caso, In In either case, case, y and and and and and 127 127 127 127 127 d H = C P dT H = C dT H= = CPPPPdT dT dH =C C dT ddddH H = C dT � T2P �� �H = �� TTTT2T2222C P dT CC dT �H = CPPPPdT dT �H= = T1 C dT �H �H = T TTTT 11111 P 127 (4.2) (4.2) (4.2) (4.2) (4.2) (4.2) Q ∆H = �H mechanically reversible,reversibles constant-pressure, closed-system processes Por otraMoreover, parte, Q = parafor procesos mecánicamente de sistema cerrado, a presión constante Moreover, Q = �H for mechanically reversible, constant-pressure, closed-system processes Moreover, Qand =�H �Hthe fortransfer mechanically reversible, constant-pressure, closed-system processes Moreover, Moreover, Q Q = = �H for for mechanically mechanically reversible, reversible, constant-pressure, constant-pressure, closed-system closed-system processes processes and [Eq. (2.23)] for of heat in steady-flow exchangers where �E [ecuación (2.23)] y para la transferencia de calor en intercambiadores de flujo estableP donde�E ∆EKP are y ∆EK son and are [Eq. (2.23)] and for the transfer of heat in steady-flow exchangers where �E and�E �EKKKKare are [Eq.(2.23)] (2.23)] andfor for the transfer ofheat heat insteady-flow steady-flow exchangerswhere where�E �EPPPPand �E [Eq. [Eq. (2.23)] and and for the transfer transfer of of heat in in steady-flow exchangers where �E =the 0 [Eq. (2.33)]. In cualquier either case, negligible P and�E K are insignificantes y Wand 0 s[ecuación (2.33)]. En caso, exchangers s =W = 0 [Eq. (2.33)]. In either case, negligible and W = 0 [Eq. (2.33)]. In either case, negligible and W s [Eq.(2.33)]. (2.33)].InIneither eithercase, case, negligible negligibleand andWWssss==00[Eq. � ��� TTT222 � (4.3) (4.3) Q = �H = TTT222 C P dT CC dT (4.3) Q = �H = CPPPPdT dT (4.3) Q= =�H �H= = T1 C dT (4.3) (4.3) Q Q = �H = P T TTTT 11111 La aplicación más común de esta application ecuación enofingeniería es enislatotransferencia de calor en flujo estable. The common engineering this equation steady-flow heat transfer. The common engineering application of this equation isis to steady-flow heat transfer. Thecommon commonengineering engineeringapplication applicationof ofthis thisequation equationis isto tosteady-flow steady-flowheat heattransfer. transfer. The The common engineering application of this equation to steady-flow heat transfer. Temperature Dependence of the Heat Capacity Dependencia de laDependence temperaturaof de la capacidad calorífica Temperature Dependence of the Heat Capacity Temperature Dependence ofthe the HeatCapacity Capacity Temperature Heat Evaluation of the integral in Eq. (4.3) requires knowledge of the temperature dependence of Evaluation of the integral in Eq. (4.3) requires knowledge of the temperature dependence of Evaluation of theintegral integral inEq. Eq.given (4.3) requires knowledge ofthe thethe temperature dependence of Evaluation Evaluation of the the integral in in Eq. (4.3) (4.3) requires requires knowledge knowledge of of the temperature temperature dependence dependence of of La evaluación laof integral deislausually ecuación (4.3) requiere el conocimiento de la dependencia de la temperatuthe heatdecapacity. This by an empirical equation; two simplest expressions the heat capacity. This is usually given by an empirical equation; the two simplest expressions the heat capacity. This is usually given by an empirical equation; the two simplest expressions the the heat heat capacity. capacity. This This is is usually usually given given by by an an empirical empirical equation; equation; the the two two simplest simplest expressions expressions ra de la of capacidad practicalcalorífica. value are:Ésta, por lo general, se proporciona por una ecuación empírica; las dos expresioof practical value are: of practical valueare: are: of practical practical value are: nes másof simples devalue valor C P práctico son: CP −2 CC CC and CPPPP = α + βT + γ T 222 CPPPP = a + bT + cT −2 C C −2 2 2 −2 and = α + βT + γ T = + bT + cT RP = RP = and =ααα++ +βT βT++ +γγγTTT2 =aaaa+ +bT bT+ +cT cT−2 −2 y and and = βT = + bT + cT RRR RRR R R where α, β, and γ and a, b, and c are constants characteristic of the particular substance. With where α, β, and γ and and a, b, and are constants characteristic of the particular substance. With where α,así β,and and and a,cb, b, and areconstants constants characteristic ofthe theparticular particular substance. With del donde a, b exception y α, g, como a, blast ya, son constantes características laof sustancia particular. Con excepción where where α, β, β, and and a, b, and and ccccare are constants characteristic characteristic of the particular substance. substance. With With the ofγγγthe term, these equations are of thedesame form. We therefore combine the exception of the last term, these equations are of the same form. We therefore combine the exception of the last term, these equations are of the same form. We therefore combine último the término, estas ecuaciones tienen la misma forma. Por lo tanto, las combinamos para proporcionar una the exception exception of of the the last last term, term, these these equations equations are are of of the the same same form. form. We We therefore therefore combine combine them to provide a single expression: them to provide single expression: themto toprovide provideaaaasingle singleexpression: expression: sola expresión: them them to provide single expression: CP −2 CC (4.4) CPPPP = A + BT + C T 222+ DT −2 C −2 −2 = + BT + CC + DT (4.4) RP = =AAAA+ +BT BT+ +C CTTTT222+ +DT DT−2 (4.4) (4.4) −2 = + BT + + DT (4.4) (4.4) R R RR where either C or D is zero, depending on the substance considered. Because the ratio C P /R Donde where ya seaeither C o DC esor cero dependiendo de laon sustancia considerada. Puesto que la the relación where either CC or D isis zero, depending on the substance considered. Because the ratio CC /Rno tiene where either C or Dis iszero, zero, depending on thesubstance substance considered. Because theratio ratioC CPPP/R P/R /R either or D D zero, depending on the the substance considered. considered. Because Because the ratio C iswhere dimensionless, the units ofdepending C PP/R P are governed by the choice of R. dimensiones, las unidades de C dependen de la elección de R. are governed by the choice of R. is dimensionless, the units of C are governed by the choice of R. is dimensionless, the units of C P P P aregoverned governed by bythe thechoice choice ofofR. R. isisdimensionless, dimensionless, the units units CCPgases As shown inthe Chap. 6,ofof for the ideal-gas heat capacity, rather than the actual heat caPPare Como se mostrará en el capítulo 6, para los gases seheat usa capacity, la capacidad calorífica del gasheat ideal, en lugar As shown inin Chap. 6, for gases the ideal-gas heat capacity, rather than the actual caAs shown Chap. 6,for forgases gases the ideal-gas heat capacity, rather than theactual actual heat caAs shown Chap. Chap. 6, 6, for gases the the ideal-gas ideal-gas heat capacity, rather rather than than the the actual heat cacapacityAs is shown used ininin the evaluation of such thermodynamic properties as the enthalpy. Theheat reason de la capacidad calorífica real en la evaluación de las propiedades termodinámicas como la entalpía. La razón pacity is used in the evaluation of such thermodynamic properties as the enthalpy. The reason pacity is used in the evaluation of such thermodynamic properties as the enthalpy. The reason pacity isused usedininthe theevaluation evaluationofofevaluation such suchthermodynamic thermodynamic properties propertiesas asthe theenthalpy. enthalpy. The reason ispacity thatisthermodynamic-property is most conveniently accomplished inThe tworeason steps: es que is la evaluación termodinámica de la propiedad se consigue en forma más conveniente siguiendo is that thermodynamic-property evaluation is most conveniently accomplished in two steps: is that thermodynamic-property evaluation is most conveniently accomplished in two steps: isthat that thermodynamic-property thermodynamic-property evaluation evaluationisisideal-gas most mostconveniently conveniently accomplished accomplished inintwo twosteps: steps: dos first, calculation of values for a hypothetical state wherein ideal-gas heat capacities pasos: primero, se calculan los valores un hipotético estado delwherein gas ideal, en donde se usan las capacidafirst, calculation of values for aaapara hypothetical ideal-gas state wherein ideal-gas heat capacities first, calculation of values for hypothetical ideal-gas state wherein ideal-gas heat capacities first, first, calculation calculation of of values values for for a hypothetical hypothetical ideal-gas ideal-gas state state wherein ideal-gas ideal-gas heat heat capacities capacities are used; second, correction of the ideal-gas-state values to the real-gas values. A real gas des caloríficas delsecond, gas ideal; segundo,of hace una corrección de los del estado de A gas ideal para que are used; second, correction the ideal-gas-state values to the real-gas values. A real gas areused; used; second, correction ofsethe the ideal-gas-state values tovalores thewhen real-gas values. A real gas are are used; second, correction correction of the ideal-gas-state ideal-gas-state values to to the the real-gas real-gas values. values. A real gas gas becomes ideal in the limit as ofP → 0; if it were to values remain ideal compressed toreal a finite reflejenbecomes los valores delin gas real. Unas gas será en to el límite conforme P→ 0; si continuara siendo ideal becomes ideal in the limit as PPPreal → 0; ifififideal itititwere were to remain ideal when compressed to aaafinite finite becomes ideal in the limit as → 0; were to remain ideal when compressed to finite becomes ideal ideal in the the limit limit as P → → 0; 0; if it were to remain remain ideal ideal when when compressed compressed to to a finite pressure, it would exist in a hypothetical ideal-gas state. In such states gases have propercuandopressure, se comprime a presiones finitas, existiría en un estado de gas ideal hipotético. Los gases en sus estapressure, itititwould would exist in aaahypothetical hypothetical ideal-gas state. In such states gases have properpressure, would exist in hypothetical ideal-gas state. In such states gases have properpressure, it would exist exist in in a hypothetical ideal-gas ideal-gas state. state. In In such such states states gases gases have have properproperties that reflect their molecular structure just as do real gases. Ideal-gas-state heat capacities dos de ties gas tienen propiedades reflejan just su estructura molecular al igual que en loscapacities gases reales. Por ties that reflect their molecular structure just as do real gases. Ideal-gas-state heat capacities tiesideal thatreflect reflecttheir their molecular structure just asdo doreal real gases.Ideal-gas-state Ideal-gas-state heat capacities ig molecular ig que ties that that reflect molecular structure structure as as do real gases. Ideal-gas-state heat heat capacities (designated by Ctheir thereforejust different for gases. different although functions of ig ig ig ig )gases; ig and C ig ig) are P V ig ig ig lo tanto, las capacidades térmicas del gas ideal (denotadas por C y C son diferentes para gases distintos; ig ig (designated by C and C ) are therefore different for different gases; although functions of and C ) are therefore different for different gases; although functions of (designated by C P V and and CCVVVVV))are aretherefore therefore different differentfor fordifferent differentgases; gases;although althoughfunctions functionsof of (designated (designatedby by CCPPPPPare temperature, they independent of pressure. aunquetemperature, son funciones de la temperatura, son independientes de la presión. temperature, they are independent of pressure. temperature, they are independent of pressure. temperature, they they are arecapacities independent independent ofofpressure. pressure. Ideal-gas heat increase smoothly with increasing temperature toward an upper Las capacidades caloríficas delincrease gas ideal aumentan deincreasing manera uniforme conforme se la Ideal-gas heat capacities increase smoothly with increasing temperature toward an upper Ideal-gas heatcapacities capacities increase smoothly with increasing temperature toward anincrementa upper Ideal-gas heat capacities increase smoothly smoothly with with increasing temperature temperature toward toward an an upper upper limit, Ideal-gas which is heat reached when all translational, rotational, and vibrational modes of molecular temperatura hacia un límite superior, el cual se alcanza cuando se excitan completamente todos los modos de ig limit, which is reached when all translational, rotational, and vibrational modes of molecular limit, which is reached when all translational, rotational, and vibrational modes of molecular limit, limit,which which isisreached reached when alltranslational, translational, rotational, rotational, and and vibrational modes modes of molecular molecular motion are fully excitedwhen [seeall Eq. (16.18)]. The influence ofvibrational temperature on Cof for argon, ig ig P ig movimiento molecular traslación, rotación y vibración [véase of la ecuación (16.18)]. En laargon, figura 4.1 se ig motion are fully excited [see Eq. (16.18)]. The influence of temperature on CC for argon, motion are fullyde excited [see Eq.(16.18)]. (16.18)]. Theinfluence influence oftemperature temperature onC Cig for argon, motion motion are are fully fully excited excited [see [see Eq. Eq. (16.18)]. The The influence of temperature on on for for argon, P P nitrogen, water, and carbon dioxide is illustrated in Fig. 4.1. Temperature dependence is exPPP ig muestranitrogen, la influencia de la temperatura en C para el argón, el nitrógeno, el agua y el dióxido de La nitrogen, water, and carbon dioxide is illustrated in Fig. 4.1. Temperature dependence is exnitrogen, water, and carbon dioxide is illustrated in Fig. 4.1. Temperature dependence iscarbono. exP nitrogen, water, water,and andby carbon carbon dioxide dioxide isisillustrated illustrated ininhere Fig. Fig.written: 4.1. 4.1. Temperature Temperaturedependence dependenceisis exexpressed analytically equations such as Eq. (4.4), dependencia de la temperatura se expresa en forma analítica mediante ecuaciones como la (4.4), que aquí se pressed analytically by equations such as Eq. (4.4), here written: pressed analytically by equations such as Eq. (4.4), here written: pressed pressedanalytically analyticallyby byequations equationssuch suchasasEq. Eq.(4.4), (4.4),here herewritten: written: ig escribe como: C ig igig −2 CC CPPigPig = A + BT + C T 222+ DT −2 C −2 222 + PPP = −2 DT A + BT + C T R + DT = A + BT + C T −2 DT −2 ==AA++BT BT++CCTT ++DT RRRR 04-SmithVanNess.indd 127 8/1/07 13:07:25 128 CAPÍTULO 4. Efectos térmicos CHAPTER 4. Heat Effects 128 Los valores de los parámetros se dan en la tabla C.1 del apéndice C para varios gases orgánicos e inorgánicos 1 comunes. En laofliteratura es posible ecuaciones exactas más complejas. Values the parameters areencontrar given in Table C.1 ofmás App. C for pero a number of common organic and inorganic gases. More accurate but more complex equations are found in the literature.1 7 CO 2 7 CO 2 6 6 H 2O H 2O 5 5 Figura 4.1: Capacidades caloríficas de 4.1: nitrógeno, Ideal-gas heat gas idealFigure del argón, agua y capacities of bióxido de carbono. argon, nitrogen, water, and carbon dioxide. CPig R CPig N2 4 R N2 4 3 3 Ar Ar 2 2 500 500 1 000 T/K 1500 1000 T/K 1 500 2 000 2000 Como resultado deof la Eq. ecuación lasideal-gas dos capacidades caloríficas del gas ideal están relacionadas: As a result (3.19),(3.19), the two heat capacities are related: ig ig CV C = P −1 R R ig (4.5) (4.5) ig The temperature dependence of C V /R follows from the temperature dependence of C P /R. ig La dependencia de la temperatura de CVig/R es consecuencia de la dependencia de la temperatura de CP /R. ig ig ig The de effects of temperature C Vobtienen are determined experiment, most often Los efectos la temperatura sobreon CPigCoP Cor se de manerabyexperimental; con frecuencia, se V from spectroscopic data and knowledge of molecular structure through calculations based on calculan mediante métodos de la mecánica estadística a partir de datos espectroscópicos y del conocimiento statisticalmolecular. mechanics. Where data areexperimental not available,semethods estimation are de la estructura Cuando noexperimental se tiene información empleanofmétodos de estimación, 2 2 employed, as described by Reid, Prausnitz, and Poling. como los descritos por Reid, Prausnitz, Poling y O’Connell. ideal-gas heat capacities exactly forpara real gases gases reales only atsólo zeroapressure, AunqueAlthough las capacidades caloríficas del gasare ideal son correct correctas presión cero, la the departure of real gases from ideality is seldom significant at pressures below severaldebars, desviación de la idealidad de los gases reales rara vez es significativa en presiones por debajo varios bar, ig ig ig here Cy C are usually good approximations to their true heat capacities. y en esteand caso CPig por lo general son buenas aproximaciones de sus capacidades térmicas reales. P Cand V V 1 Véase F. 1 See A. Aly y L. L. and Lee,L. Fluid Phase Equilibria, vol. 6, pp. 169-179, y su bibliografía; véase también T.see E. Daubert, R. P. F. A. Aly L. Lee, Fluid Phase Equilibria, vol. 6, pp.1981, 169–179, 1981, and its bibliography; also Danner, H. M. y C. R. C. Stebbins, Thermodynamic Properties of Pureand Chemicals: Data Compilation, & Francis, T. E. Sibul Daubert, P. Danner,Physical H. M. and Sibul, and C. C. Stebbins, Physical Thermodynamic Properties Taylor of Pure Chemicals: Bristol, PA, existente Data 1995.Compilation, Taylor & Francis, Bristol, PA, extant 1995. 2 J. M. Prausnitz, B. E. Poling y J. P. O’Connell, The Properties of Gases and Liquids, 5a. edición, capítulo 3, McGraw-Hill, Nueva 2 York, 2001. J. M. Prausnitz, B. E. Poling, and J. P. O’Connell, The Properties of Gases and Liquids, 5th ed., chap. 3, McGrawHill, New York, 2001. 04-SmithVanNess.indd 128 8/1/07 13:07:27 4.1. Sensible Heat Effects 4.1. Sensible Heat Effects 4.1. Sensible Heat Effects Example 4.1 4.1. Efectos del calor sensible 129 129 129 Example 4.1 The parameters listed in Table C.1 require use of Kelvin temperatures in Eq. (4.4). Example 4.1 The parameters listed Table C.1 require use of Kelvin temperatures in Eq. (4.4). Equations sameinform may also be developed for use with temperatures in ◦◦C, Ejemplo 4.1◦◦of the ◦ 129 C, Equations thebut same mayC.1 also be developed for use with temperatures (R), parameters and (ofF), the form parameter values are different. The molar heatincapacity of The listed in Table require use of Kelvin temperatures Eq.in(4.4). ◦ F), but theen Los parámetros lastate tabla requieren del usoof de temperaturas Kelvin en ecuación (R), and enumerados (in values different. The molar heat capacity methane isC.1 given asare a function temperature in kelvins by: C, Equations ofthe theideal-gas same parameter form may also be developed for use with temperatures inla◦ of (4.4). De esta forma, pueden desarrollar ecuaciones para con temperaturas methane in thetambién ideal-gas state is given as aare function of temperature kelvins by: of en °C, (R), and (◦ F), but these parameter values different. Theusarse molar in heat capacity ig ig (R) y (°F), aunque los valores de as losaparámetros. La capacidadincalorífica molar del meC PP state methane inson the diferentes ideal-gas is given function of temperature kelvins by: −3 −6 22 −3 −6 ig =dada 1.702 + 9.081 × 10 T − 2.164 × 10 T tano en estado de gas ideal está como una función de la temperatura en kelvins por: C P R −3 −6 2 ig = 1.702 + 9.081 × 10 T − 2.164 × 10 T CRP −3 −6 T 2 ig ig = 1.702 9.081Table × 10 C.1. T −Develop 2.164 × 10 where the parameterR values are+from an equation for C PP /R for ig ◦ where the parameter temperatures in ◦ C. values are from Table C.1. Develop an equation for C Pig/R for ◦ C. donde los valores de los parámetros se toman de la tabla C.1. Desarrolle una ecuación para Cig /R con tempetemperatures in where the parameter values are from Table C.1. Develop an equation for C PP /R for raturas en °C. temperatures in ◦ C. Solution 4.1 Solution 4.1 Solución 4.1 The relation between the two temperature scales is: Solution 4.1 La relación entre las dos escalas de latemperature temperaturascales es: is: The relation between the two T K = t ◦◦C scales + 273.15 The relation between the two temperature is: = tt ◦°C TTKK= C ++273.15 273.15 Therefore, functionde of t,t, T K = t ◦ C + 273.15 Por lo tanto, como as unaa función Therefore, as a function of t, ig CPPig as a function of t, −3 Therefore, −6 (t + 273.15)22 ig = 1.702 + 9.081 × 10−3 (t + 273.15) − 2.164 × 10−6 CR 2 P −3 −6 ig = 1.702 + 9.081 × 10 (t + 273.15) − 2.164 × 10 (t + 273.15) CRP −3 ig ig = 1.702 − 2.164 × 10−6 (t + 273.15)2 C PP+ 9.081 × 10 (t + 273.15) R −3 −6 t22 −3 ig = 4.021 + 7.899 × 10 or t − 2.164 × 10−6 o CR P −3 −6 2 or ig = 4.021 + 7.899 × 10 t − 2.164 × 10 t CRP = 4.021 + 7.899 × 10−3 t − 2.164 × 10−6 t 2 or R Gas mixtures of constant composition behave exactly as do pure gases. In an ideal-gas Las mezclas de gasesofdeconstant composición constante seanother, comportan como losideal-gas gases puros. En Gasthe mixtures exactlyand asexactamente do pure In an mixture molecules have no composition influence onbehave one each gasgases. exists independent of una mezcla de gases ideales las moléculas carecen de alguna influencia entre sí, y cada gas existe de manera mixture themixtures molecules no influence onaone another, andaseach gasmole-fraction-weighted exists independent of the others. The ideal-gas heat capacity of mixture is therefore Gas of have constant composition behave exactly do the pure gases. In an ideal-gas independiente de los otros. Debidoheat a eso, la capacidad calorífica del gases gas ideal de una mezcla es la suma de las the others. The ideal-gas capacity of a mixture is therefore the mole-fraction-weighted sum of the heat capacities of the individual gases. Thus for A, B, and C the molar heat mixture the molecules have no influence on one another, and each gas exists independent of capacidades caloríficas ponderadas con la fracción mol de los gases individuales. De este modo, para los sum of theofheat capacities of thecapacity individual gases. Thus gases A, and C the molar heat capacity aThe mixture in the ideal-gas state the others. ideal-gas heat of is: a mixture is for therefore theB,mole-fraction-weighted gases A,capacity B y C la capacidad calorífica molar de una mezcla en el estado de gas ideal es: of aheat mixture in theofideal-gas state is:gases. Thus for gases A, B, and C the molar heat sum of the capacities the individual ig ig ig ig ig ig ig ig = y AACis: (4.6) capacity of a mixture in the C ideal-gas state BC P CC P Pmixture PAA + y B PBB + yC PCC P P mixture ig ig ig ig C Pmixture = y A C PA + y B C PB + yC C PC (4.6) (4.6) mezcla ig ig ig ig ig ig ig ig ig ig C Pmixture = yAC y B C PB + (4.6) C C PCA, B, and C in the ideal-gas where C PPAA , C PPBB , and C PPCC are the molar heat capacities ofypure PA + ig ig ig ig ig ig donde Cwhere , C y C son las capacidades caloríficas molares de A, B y C puras en estado de gas ideal y yA, C , C , and C are the molar heat capacities of pure A, B, and C in the ideal-gas state, and y , y , and y are mole fractions. A B C A B C PB PC PA PBPA PC igy las igfracciones yB y yCstate, representan molares. and ,Cygases, yCC ig are mole fractions. A B ,, and As the heat capacities of solids and liquids are found by experiment. Paramwhere C Pwith , and are the molar heat capacities of pure A, B, and C in the ideal-gas PB PC A Alstate, igualfor que conthe los gases, lasfractions. capacidades caloríficas sólidos y líquidos seParamdeterminan de As with capacities and liquids arelos found bygiven experiment. eters the dependence of of C PPsolids as expressed by de Eq. (4.4) are for a few solids and yocurre , gases, y B , and yCheat are mole Atemperature maneraeters experimental. Los parámetros para de liquids la temperatura de Cgiven como expresan for temperature ofdependencia CofPC.solids asCorrelations expressed by are for amany few solids Pexperiment. and liquids in Tables C.2 and C.3 oflaApp. forEq. the heat capacities ofse solidsmedianAsthe with gases, the dependence heat capacities and are(4.4) found by Paramte la ecuación están dados paraC.3 algunos y líquidos en las33(4.4) tablascapacities C.2 y C.3 apéndice and Tables C.2 ofGreen App. Correlations for the heat ofdel many solids andliquids liquids given by and Perry and and by Daubertby et Eq. al. eters for(4.4) theinare temperature dependence ofsólidos CC. expressed are given for a few solids C. Las P as 3 correlaciones para are lasTables capacidades deand diversos sólidoset líquidos están dadas por Perry y Green, and given by Perry andofGreen by Daubert and liquids liquids in C.2 andcaloríficas C.3 App. C. Correlations foryal. the heat capacities of many solids 3 33R. Daubert así como por ygiven colaboradores. H. Perry D. Green, Perry’s Chemical Handbook, and liquids areand by Perry and GreenEngineers’ and by Daubert et 7th al.3ed., Sec. 2, McGraw-Hill, New York, 3 R. T. 1997; Daubert op. cit. H.E.Perry and etD.al., Green, Perry’s Chemical Engineers’ Handbook, 7th ed., Sec. 2, McGraw-Hill, New York, 1997; E. Daubert et al., op. cit. 3 R.T.H. Perry and D. Green, Perry’s Chemical Engineers’ Handbook, 7th ed., Sec. 2, McGraw-Hill, New York, 3 R. H. 1997; T. yE.D.Daubert al., op.Chemical cit. Perry Green,etPerry’s Engineer’s Handbook, 7a. edición, sección 2, McGraw-Hill, Nueva York, 1997; T. E. Daubert et al., op. cit. 04-SmithVanNess.indd 129 8/1/07 13:07:30 130 130 130 130 130 130 130 130 CHAPTER 4. Heat Effects CHAPTER 4. 4. Heat Effects CHAPTER Effects CAPÍTULO 4. Heat Efectos térmicos CHAPTER 4.4. Heat Effects CHAPTER Heat Effects CHAPTER 4. 4. Heat Heat Effects Effects CHAPTER Evaluation the Sensible-Heat Integral Evaluation of the Sensible-Heat Integral Evaluación de laof integral del calor sensible Evaluation of the Sensible-Heat Integral Evaluation of the Sensible-Heat Integral � � Evaluation of the Sensible-Heat Integral � Evaluation of the Sensible-Heat Integral Evaluation of the Sensible-Heat Integral Evaluation of the integral isisaccomplished by substitution for CCPPCas aafunction of TT,,T , ��CCPPCdT Evaluation ofof thethe integral dT accomplished byby substitution forfor as function ofof Evaluation integral is accomplished substitution a function P dT P as � Cse La evaluación deby laofformal integral ∫ CP �dT consigue mediante labysustitución para CCPresult función de Evaluation the integral dT istemperature accomplished substitution for asasaisis function ofofT, TT,seguida Pcomo followed integration. For limits of TT00Tand TTfor the conveniently Evaluation of theintegral integral CPPPPdT dTFor is accomplished bysubstitution substitution forthe C function followed byof formal integration. For temperature limits of and result conveniently followed by formal integration. temperature limits of Tthe is conveniently Evaluation of the integral dT accomplished by substitution for as aaafunction function of TTen Evaluation the CC isis accomplished by CCresult as of ,, , forma 0yand PPPresult por la integración formal. Para los límites de las temperaturas de T T, el resultado se expresa followed by formal integration. For temperature limits of T and T the is conveniently 0 0 expressed as: followed by formal integration. For temperature limits of T and T the result is conveniently expressed as: 0 expressed as: followed by formal formal integration. integration. For For temperature temperature limits limits of of TT00 and and TT the the result result isis conveniently conveniently followed by conveniente como: expressed as: expressed as: expressed as: �� � �� � expressed �� T�T as: BB B22 22 CC C33 33 DD D − 1 �1� ��ττ − � CTCPPC P τ 1− 2 − 2 1) 3 − 3 1) − + + + = AT (4.7) �ττ−−11� � dTdT == ATAT (4.7) (τ0 (τ −1) 1)1) ++ (τ − 1)1) ++ (τ − 1)1) + +DD� BBTT002T(τ CCTT003T(τ ���TTTTCCP dT 00(τ − (τ − (τ − (4.7) (4.7) 2 3 0 0 RRPPPRdT 2 3 T τ C τ − 1 B C D 2 2 3 3 τ − 1 2 3 T τ = AT T T (4.7) (τ − 1) + (τ − 1) + (τ − 1) + B C D 0 TT00 TC 2 3 T τ 0 0 (τ − 1) + 2 (τ 3 (τ 22 − 1) + 33 − 1) + 0 0 0 0 2 3 dT = AT T T (4.7) 0 R dT (τ − 1) + (τ − 1) + (τ − 1) + dT = AT T T (4.7) 0 (τ T T (4.7) 0 2 3 T τ (τ − 1) + − 1) + (τ − 1) + = AT 0 0 0 T0T 0 RR 222 0 333 00 τττ TTT 000 TT00 0 R TT T ττ ≡ donde where where ≡ where τ ≡T where ττ≡≡TTT0T 0T where where τ ≡ TT0 0 where τ≡ 0 is straightforward. T T 00�H Given and TT,,Tthe of QQ Q or �H isisTthe calDados T y T,Tel de Qcalculation ocalculation ∆H es directo. Esor menos directo el cálculoLess deLess T,direct dados ythe Q ocal∆H. En Given T00Tcálculo and the calculation ofof or �H is is straightforward. Less direct cal0 Given 0the , the straightforward. direct is 0 and Given T and T , the calculation of Q or �H isisstraightforward. Less direct is the cal0 and Q or �H . Here, an iteration scheme may be useful. Factoring culation of TTTT ,,T given T Given and T , the calculation of Q or �H straightforward. Less direct is the caleste caso, un Given planteamiento iterativo es de utilidad. Al factorizar (τ – 1) de cada término del lado derecho de culation ofof given and Q or �H . Here, an iteration scheme may be useful. Factoring 0 0 and Q or �H . Here, an iteration scheme may be useful. Factoring culation T , given T 0 Given and T , the calculation of Q or �H is straightforward. Less direct is the caland TT, the 0 calculation of Q or �H is straightforward. Less direct is the calculation ofof TTeach ,00,given and QQ oror�H . . Here, anan iteration scheme may bebeuseful. Factoring 0 on (τ − 1) from term the right-hand side of Eq. (4.7) gives: culation given and �H Here, iteration scheme may useful. Factoring la ecuación (τ −(4.7) 1)1) from term on the right-hand side of Eq. (4.7) gives: 0 (τ − from each term on the right-hand side of Eq. (4.7) gives: culation ofse , given given and Q or �H . Here, an iteration scheme may be useful. Factoring culation of TTobtiene: ,each TTT and Q or �H . Here, an iteration scheme may be useful. Factoring 00 (τ(τ−−1)1)from each term on the right-hand side ofofEq. (4.7) gives: eachterm termon theright-hand right-handside sideof Eq.(4.7) (4.7)gives: gives: (τ − − 1) 1) from from each term the right-hand side of Eq. Eq. (4.7) gives: �� �on (τ �from �on�the � T�T each T C B C DD D� � B B22 2 C C33 322 2 �� ��T CPPC P − + + + ττ + + = ��AT (τ (τ −1) 1)1) + (τ +1) 1)1) ++ (τ + 1)1) + +DD���(τ dTdT == ATAT BBTT002T(τ CCTT003T(τ �� TTTCCP dT 00 + − + τ 1) + 0+ 2 2+ + 0 (τ 0 (τ RRPPPRdT 2 3 TD B C C 2 3 B C 2 3 Tτ00T0(τ(τ−−1)1) + (τ + 1) + (τ + τ + 1) + = AT T T TT00 TC 2 3 0 2 + τ + 1) +ττD 22 (τ + 1) + 33 (τ 0 0 2 + dT = AT T T 0 R dT 0 + 22TT000(τ (τ + + 1) 1) + + 33TT000(τ (τ + + ττ + + 1) 1) + + ττTT0 (τ dT = = AT AT00 + (τ − − 1) 1) T0 RR 22 33 ττTT000 TT0T0 0 R TT − T − T 0 T −0T Because ττ − Because − 1= = TT−−TT0 0 Because τ 1− 1= T T − T00TT00 τ − 1 = T − Ya que Because Because Because −−111= == TT0 0 Because τττ− this may be written: TT000 this may bebe written: this may written: this may be written: this may be written: ésta se puede escribir como: �� � �� � this may may this be �be � Twritten: �written: �� �� CC C22 22 DD D� BB B ��TT CTCPPC P �(T � � 2 2 + + + 1) + − = (τ0 (τ +1) 1)1) ++ (τ +τ+ τ+ + 1)1) + +DD (T(T −T T00)T) ) dTdT = =AA+ ++ CCTT002T(τ BBTT00(τ �� TTTCCP dT + (τ τ + − A T 2 2 0 2 2 RRPPPRdT 2 3 C C D B 2 2 τ T C B 2 3 (τ + 1) + (τ + τ + 1) + −−TT0 ) )0 = A + T T D TT00 TC τ T 2 3 0 τ T 22 (τ 22 + τ + 1) + 002 0 (T 0 (τ + 1) + (T dT = A + T T 0 R dT 0 (τ + + 1) 1) + + 33TT000(τ (τ + + ττ + + 1) 1) + + τ T0222 (T (T − − TT00)0) dT = = AA + + 22TT00(τ T0 RR 22 33 τττTTT TT0T0 0 R 000 We identify the quantity in square brackets as �C /R,/R, where �C isis defined as aa mean WeWe identify thethe quantity in in square brackets asas �C�C where �C�C defined asas mean PP��HP PP��HP identify quantity square brackets where is defined a mean H�/R, H�H H where We identify the quantity in square brackets as �C � /R, �C � is defined as aamean P P H H heat capacity: We identify the quantity in square brackets as �C � /R, where �C � is defined as mean Identificamos la cantidad entre los rectangulares ⟨C ⟩ se define como una heat capacity: P P⟩ H /R, P H heat capacity: We identify the quantity quantity inparéntesis square brackets brackets as �C �CPPP�como �HHH/R, /R,⟨C where �CPdonde � is defined as a mean � is defined as a mean H We identify the in square as where �C P HH heat capacity: heat capacity: capacidad heatcalorífica capacity:media: heat capacity: �C CC C22 22 DD D BB B �C�C PP��HP H�H 2 + 2 ττ + + + 1) + (4.8) = AA+ �C (τ0 (τ +1) 1)1) ++ (τ + + 1) + (4.8) == ++ CCTT002T(τ DD BBTT00(τ + (τ + τ + 1) + (4.8) A T PP�H 2 22 2 � �C 3C �CRPRP��H �C D 20 222+ τ + 1) + ττT D 2 T2T0 (τ(τ++1)1)++C 3 T3T022(τ (4.8) T RHH ==AA++2BB τ T 0 (τ + τ + 1) + (4.8) (4.8) 0 2 0(τ + + 1) 1) + + 33TT000(τ (τ + + ττ + + 1) 1) + + τ T02220 (4.8) = AA + + 22TT00(τ RR = (4.8) RR 22 33 τττTTT 000 Equation (4.2) may therefore be written: Equation (4.2) may therefore bebe written: Equation (4.2) may therefore written: Equation (4.2) may therefore bebewritten: (4.2)may may therefore written: Equation (4.2) may therefore be written: written: (4.2) therefore be DebidoEquation aEquation eso, la ecuación (4.2) se escribe como: (T (T − (4.9) �H = −TT00)T) 0 ) (4.9) �H = �C �C�C PP��HP (4.9) �H = H�(T H −− (T TT0 ) ) (4.9) �H ==�C PP�H � (T − (4.9) (4.9) �H �C 0 (T − − TT00)) (4.9) �H = = �C �CPP��HHH(T (4.9) �H The angular brackets enclosing CCPPCidentify itit as aa mean value; subscript “H ”” denotes aa mean The angular brackets enclosing identify as mean value; subscript “H“H denotes mean The angular brackets enclosing it as a mean value; subscript ” denotes a mean P identify The angular brackets enclosing C identify it as a mean value; subscript “H ” denotes asimilar mean P value specific to enthalpy calculations, and distinguishes this mean heat capacity from a similar The angular brackets enclosing C identify it as a mean value; subscript “H ” denotes a mean “H” value specific to to enthalpy calculations, and distinguishes this mean heat capacity from a P value specific enthalpy calculations, and distinguishes this mean heat capacity from a similar Los paréntesis angulares que contienen se identifican como su valor medio; en tanto, el subíndice The angular brackets enclosing identify it as a mean value; subscript “H ” denotes a mean The angular brackets enclosing CCC identify it as a mean value; subscript “H ” denotes a mean P PP value specific to enthalpy calculations, and distinguishes this mean heat capacity from a similar quantity introduced in the next chapter. value specific to enthalpy calculations, and distinguishes this mean heat capacity from a similar quantity introduced in in thethe next chapter. quantity introduced next chapter. denota value un valor medio para cálculos dedistinguishes la entalpía ythis distingue esta capacity capacidad calorífica media de value specific toespecífico enthalpy calculations, and distinguishes this mean heat heat capacity from similar specific to enthalpy calculations, and mean from aa similar quantity introduced inin the next chapter. Solution of Eq. for TT gives: quantity introduced the next chapter. Solution ofof Eq. (4.9) for Solution Eq. (4.9) for Tgives: gives: una cantidad similar que será introducida en el siguiente capítulo. quantity introduced in(4.9) the next chapter. quantity introduced in the next chapter. Solution ofofEq. (4.9) for TTgives: Solution Eq.(4.9) (4.9) forT gives: La solución de laof (4.9) T da: Solution ofecuación Eq. (4.9) for Tpara gives: Solution Eq. for gives: �H �H �H+ T TT = (4.10) ++ T00T0 (4.10) �H T= = (4.10) �H �H �C TT==�C (4.10) �H PP��HP �C �H++TT0 0 H (4.10) (4.10) = �C + T (4.10) + T (4.10) � TT = 0 P 0 �CPP��H�H �C �C HH) allows evaluation of �C � by Eq. (4.8). AA starting value for TT (and hence for ττ = TTP/T evaluation ofof �C�C Eq. (4.8). starting value forfor hence forfor /T PP �HP 00 ) 0allows ) allows evaluation � byby Eq. (4.8). A starting value T(and (and hence τ= = T /T allows evaluation ofof�C �HH� H by Eq. (4.8). A starting value for T (and hence for τ = T /T P 0 )a Substitution of this value into Eq. (4.10) provides new value of T from which to reevaluate ) allows evaluation �C by Eq. (4.8). (4.8). A starting value for T (and hence for τ = T /T Substitution of this value into Eq. (4.10) provides a new value of T from which to reevaluate P 0) allows Un valor para Tof(yfor por tanto de τ Eq. = T/T evaluación deof⟨CTPof ⟩of mediante la ecuación Substitution this (4.10) a new evaluation value from which reevaluate allows evaluation �C �HHHby byto Eq. (4.8). Ainicial starting value for (andinto hence for =provides /T00la A starting value TTvalue (and hence for = TT/T 0ττ) permite H �C ) � Eq. (4.8). P P Substitution ofofthis value into Eq. (4.10) provides a new value of TTfrom which totoreevaluate �C continues to convergence on aun value of Tde Substitution this valueinto into Eq. (4.10) provides new value fromwhich which reevaluate �C �HPH.�.HIteration Iteration continues to(4.10) convergence onon afinal final value ofof T.of .Tof PP�este Al sustituir valor la ecuación se obtiene valor T.TT a partir del cualto vuelve a evaluar �C . Iteration continues to convergence anuevo final value Substitution ofen this value into Eq. (4.10) provides new value of from which tosereevaluate reevaluate Substitution of this value Eq. (4.10) provides aaanew value from �C � . Iteration continues to convergence on a final value of T . PPH �C � . Iteration continues to convergence on a final value of T . ⟨CP⟩ H. �C La continúa hastato la convergence convergenciaon para un valor �CPPiteración Iteration continues to convergence on final valuefinal of TTde ��HHH.. Iteration continues aa final value of .. T. 04-SmithVanNess.indd 130 8/1/07 13:07:37 4.1. 4.1. Sensible Sensible Heat Heat Effects Effects 4.1. Efectos del calorHeat sensible 4.1. Sensible Effects 4.1. Sensible Sensible Heat Heat Effects Effects 4.1. 131 131 131 131 131 131 Example 4.2 Ejemplo 4.2 4.2 Example Example 4.2 required Example Calculate heat Calculate the the 4.2 heat required to to raise raise the the temperature temperature of of 1 1 mol mol of of methane methane from from 260 260 ◦◦Cnecesario Calculate the heat para required to raise the temperature of 1de mol of methane Calculeto calor aumentar la de 1 mol metano de 260from amay 600260 °C en un a process at a pressure sufficiently low that methane be toel600 600 C in in a steady-flow steady-flow process attemperatura a pressure sufficiently low that methane may be Calculate the heat required required to raise raise the temperature of 11 mol mol of methane from 260 Calculate the heat to the temperature of of methane from 260 ◦C to 600 in a steady-flow process at a pressure sufficiently low that methane may be un gas procesoconsidered de flujo estable a una presión lo suficientemente baja para que el metano se considere ◦ ◦ an ideal gas. considered ideal gas. process to 600 C C in in aan a steady-flow steady-flow process at at aa pressure pressure sufficiently sufficiently low low that that methane methane may may be be to 600 ideal. considered an ideal gas. considered an an ideal ideal gas. gas. considered Solution 4.2 Solution 4.2 Solución 4.2 Solution 4.2 Solution 4.2 Solution 4.2 Equation in with Eq. (4.7) the result. ParamLa ecuación (4.3) (4.3) en combinación con la (4.7) proporciona el resultado requerido. parámetros Equation (4.3) in combination combination with Eq. (4.7) provides provides the required required result.Los Paramig igEquation (4.3) combination with Eq.temperatures (4.7) provides the required result. Paramig /Rin eters for C come from Table C.1; are: para CPEquation /R se obtienen de la tabla C.1; las temperaturas son: Equation (4.3) income combination withC.1; Eq.temperatures (4.7) provides provides the required required result. result. ParamParam(4.3) combination with Eq. (4.7) the eters for C /Rin from Table are: P Pig /R eters for Cig come from Table C.1; temperatures are: ig eters for for CCPPP /R /R come come from from Table Table C.1; C.1; temperatures temperatures are: are: eters 873.15 873.15 1.6377 T T ττ = 873.15 = T00 = = 533.15 533.15 K K T= = 873.15 873.15 K K = 533.15 = 1.6377 873.15 = 1.6377 T0 = 533.15 K T = 873.15 K τ = 873.15 533.15 = 533.15 533.15 K K = 873.15 873.15 K K = 533.15 = = 1.6377 1.6377 TT00 = TT = ττ = �� 873.15 ig 533.15 533.15 ig 873.15 C � 873.15 C PPig dT Whence, Q Whence, Q= = �H �H = =R R�� 873.15 ig 873.15 C Pig dT CR Whence, Q = �H = R 533.15 R PP dT 533.15 C De donde, Whence, Q= = �H �H = = RR 533.15 dT Whence, Q R dT R 533.15 R 533.15 �� −3 −3 9.081 � 9.081 × × 10 10−3 2 22 T Q T02(τ (τ − − 1) 1) + + 9.081 2× 10−3 (τ − − 1) 1) Q= = (8.314) (8.314)��1.702 1.702 T T00(τ 9.081 × × 10−3 T02022 (τ222 − 1) Q = (8.314) 1.702 T0 (τ − 1) + 9.081 2 10 T (τ − − 1) 1) + + (τ − 1) � Q= = (8.314) (8.314) 1.702 1.702TT00(τ T (τ − 1) Q 2 00 � 22 −6 −6 � 2.164 × 10 2.164 × 10 3 3 3 3 � −6 T − (τ − − 1) 1)� = = 19,778 19,778 JJ T0 (τ − 2.164 3× 10−6 2.164 × × 10−6 T03033 (τ333 − 1) = 19,778 J − 2.164 3 10 (τ − − 1) 1) = = 19,778 19,778 JJ − TT00 (τ − 3 33 Uso de las funciones definidas Use Use of of Defined Defined Functions Functions Use of Defined Functions �� Use of Defined Functions Use of Defined Functions La integral a menudo en los cálculos termodinámicos. Por lo As tanto, por razones de conveThe integral often in calculations. aa matter of P/R)dT � (C P /R) The∫(C integral (C /R) dT dTaparece often appears appears in thermodynamic thermodynamic calculations. As matter of conveconveP �� (Cde The integral /R) dT often appears inofthermodynamic calculations. As a matter of conveniencia,The el lado derecho la define ecuación se define como la función ICPH(T0,T;A,B,C,D) y convese escribe una nience, we the right side Eq. (4.7) as function, ICPH(T0,T;A,B,C,D), nience, we therefore therefore define the(4.7) right side Eq. (4.7) as the the function, ICPH(T0,T;A,B,C,D), The integral (CPPP/R) /R) dT often often appears inof thermodynamic calculations. As aa matter matter of of conveintegral (C dT appears in thermodynamic calculations. As nience, we therefore define the right side of Eq. (4.7) as the function, ICPH(T0,T;A,B,C,D), rutina de computadora para su evaluación. En tal caso la ecuación (4.7) será entonces: and write a computer routine for its evaluation. Equation (4.7) then becomes: and write a computer routine for its evaluation. Equation (4.7) then becomes: nience, we we therefore therefore define define the the right right side side of of Eq. Eq. (4.7) (4.7) as as the the function, function, ICPH(T0,T;A,B,C,D), ICPH(T0,T;A,B,C,D), nience, and write a computer routine for its evaluation. Equation (4.7) then becomes: and write write aa computer computer routine routine��for for its evaluation. evaluation. Equation Equation (4.7) (4.7) then then becomes: becomes: and TT its CP � C ICPH(T0,T;A,B,C,D) �� TTT C PP dT dT ≡ ≡ ICPH(T0,T;A,B,C,D) ICPH(T0,T;A,B,C,D) CR TT0 C RPP dT ≡ ICPH(T0,T;A,B,C,D) 0 dT ≡ ≡ ICPH(T0,T;A,B,C,D) ICPH(T0,T;A,B,C,D) T0 R dT R TT00 R The function name is ICPH, and the quantities in are the and El nombre la función es ICPH, y lasand cantidades entre paréntesis son las T0 y T,T Thedefunction name is ICPH, the quantities in parentheses parentheses arevariables the variables variables T00seguidas and T T ,, por los The function name is ICPH, and the quantities in parentheses are the variables T and T , 0 followed by parameters A, B, C, and D. When these quantities are assigned numerical values, parámetros B, by C yparameters D. Cuando estas se les valores numéricos, la notación representa followed A,a B, C, and When these quantities are assigned numerical values, The A, function name ICPH, andcantidades theD. quantities inasignan parentheses are the variables variables and The function name isis ICPH, and the quantities in parentheses are the TT00 and TT,, followed by parameters A, B, C, and D. When these quantities are assigned numerical values, the notation represents aaA, value for the integral. Thus, for evaluation of in un valorfollowed para la integral. Así, para la de Q en el ejemplo the notation represents value forand theD. integral. Thus, for the the 4.2: evaluation of Q Qnumerical in Ex. Ex. 4.2: 4.2:values, followed by parameters parameters A, B,evaluación C, and D. When these quantities are assigned assigned numerical values, by B, C, When these quantities are the notation represents a value for the integral. Thus, for the evaluation of Q in Ex. 4.2: the notation notation represents represents aa value value for for the the integral. integral. Thus, Thus, for for the the evaluation evaluation of of Q Q in in Ex. Ex. 4.2: 4.2: the Q 8.314 ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) 19,778 Q = 8.314 ×× ×ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0)== =19 19,778 Q= = 8.314 778 J JJ Q = 8.314 × ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) = 19,778 J Q= = 8.314 8.314 × × ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) ICPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) = = 19,778 19,778 JJ Q Representative computer programs for evaluation of the integral are given in D. Representative computer programs for evaluation of the integral are given in App. App. D.integral. En el apéndice D se dan programas representativos de computadora para are la evaluación de laD. Representative computer programs for evaluation of the integral given in as App. For added flexibility the programs also evaluate the dimensionless quantity �C � /R given P For added flexibility the programs also evaluate the dimensionless quantity �C � /R as given H Representative computer programs for evaluation of the integral are given in App. D.expresa Representative computer programs for evaluation of the integral are given in App. D. P H Para mayor flexibilidad, los programas también evalúanthe la cantidad adimensional ⟨C se ForEq. added flexibility the programs also evaluate dimensionless quantity �CPP⟩ �HH/R /Rcomo as given by (4.8). The right-hand side of this equation is another function, MCPH(T0,T;A,B,C,D). by Eq. (4.8). The right-hand side of this equation is another function, MCPH(T0,T;A,B,C,D). For added flexibility the programs also evaluate the dimensionless quantity �C � /R as given For added flexibility the programs also evaluate the dimensionless quantity �C � /R as given P P mediante ecuación (4.8). El lado side derecho de equation esta ecuación es otra función, MCPH(T0,T;A,B,C,D). Con HH bylaEq. (4.8). The right-hand of this is another function, MCPH(T0,T;A,B,C,D). With definition, Eq. becomes: With this definition, Eq. (4.8) (4.8) becomes: by Eq.this (4.8). The right-hand right-hand side of this this equation equation isis another another function, function, MCPH(T0,T;A,B,C,D). MCPH(T0,T;A,B,C,D). by Eq. (4.8). The side of esta definición, ecuación (4.8) será: With thisladefinition, Eq. (4.8) becomes: With this this definition, definition, Eq. Eq. (4.8) (4.8) becomes: becomes: With �C �C PP ��H H = MCPH(T0,T;A,B,C,D) �CRP �H = MCPH(T0,T;A,B,C,D) �CPR �C MCPH(T0,T;A,B,C,D) P��HH = MCPH(T0,T;A,B,C,D) = MCPH(T0,T;A,B,C,D) MCPH(T0,T;A,B,C,D) R = RR 04-SmithVanNess.indd 131 8/1/07 13:07:42 CHAPTER 4. Heat Effects 132 CHAPTER 4. Heat Effects 132 CHAPTER 4. Heat Effectstérmicos 132 132 CAPÍTULO 4. Efectos A specific numerical value of this function is: A specific numerical value of this function is: A numérico specific numerical value of this function Un valor específico de esta función es: is: MCPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) = 6.9965 MCPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) = 6.9965 6.9965 representingMCPH(533.15,873.15;1.702,9.081E-3,-2.164E-6,0.0) �C P � /R for methane in the calculation of Ex. 4.2. By Eq.=(4.9), representing �C P �HH /R for methane in the calculation of Ex. 4.2. By Eq. (4.9), representando a ⟨CP⟩�C el metano eninelthe cálculo del ejemplo la ecuación (4.9), representing /R for methane calculation of Ex.4.2. 4.2.Mediante By Eq. (4.9), H /R P �Hpara �H = (8.314)(6.9965)(873.15 − 533.15) = 19,778 J �H = (8.314)(6.9965)(873.15 − 533.15) = 19,778 J ∆H== (8.314)(6.9965)(873.15 (8.314)(6.9965)(873.15 –−533.15) 778 J J �H 533.15)==1919,778 Example 4.3 Example 4.3 Ejemplo 4.3 What is the final temperature when heat in the amount of 0.4 × 1066(Btu) is added to Example 4.3 What is the final temperature when heat in the amount of 0.4 × 10 (Btu) is added to ◦ 25(lblamol) of ammonia in a steady-flow process at 1(atm)? ¿Cuál será temperatura final initially cuandoat se500( agrega una × 106(Btu) a 25(lb mol) ◦F) 6 (Btu) What theoffinal temperature when heat amountdeofcalor 0.4 ×de100.4 is added to 25(lb is mol) ammonia initially at 500( F) in in the a cantidad steady-flow process at 1(atm)? de amoniaco inicialmente a 500(°F) en un proceso de flujo estable a 1(atm)? ◦ 25(lb mol) of ammonia initially at 500( F) in a steady-flow process at 1(atm)? Solution 4.3 Solución 4.3 Solution 4.3 4.3 Si ∆HSolution esIfel�H cambio de la entalpía parafor 1(lb mol), Q= y , and is the enthalpy change 1(lb mol), Q n=∆H n �H If �H is the enthalpy change for 1(lb mol), Q = n �H , and If �H is the enthalpy change for × 1(lb Q = n �H , and 6 Q 0.4 10mol), �H = Q = 0.4 × 106 = 16,000(Btu)(lb mol)−1 25 6 = 16,000(Btu)(lb mol)−1 �H = n = Qn 0.4 × 2510 �H = = = 16,000(Btu)(lb mol)−1 n requires 25 temperatures in kelvins; therefore, conversion The heat-capacity equation La ecuación para la capacidad calorífica requiere que lasintemperaturas estén en kelvins; por lo The requires temperatures kelvins; therefore, conversion −1 is equivalent of allheat-capacity units to the equation SI system is indicated. Because 1 J mol to –1 es equivalente −11 is tanto, seThe necesita convertir todas lasrequires unidades al sistema SI.kelvins; Puesto que J mol of all units to the SI system is indicated. Because 1 J mol equivalent to heat-capacity equation temperatures in therefore, conversion −1 , divide the preceding result by 0.4299: 0.4299(Btu)(lb mol) –1, el resultado a 0.4299(Btu)(lb mol) se divide entreby0.4299: , divideanterior preceding result 0.4299(Btu)(lb mol) of all units to the SI−1system isthe indicated. Because 10.4299: J mol−1 is equivalent to −1 , divide the preceding result by 0.4299: 0.4299(Btu)(lb mol)�H 37,218 mol–1−1 ∆H= = 16,000/0.4299 16 000/0.4299 == 218 JJJmol �H = 16,000/0.4299 =3737,218 mol−1 −1 �H = 16,000/0.4299 500 + 459.67= 37,218 J mol With T0 = 500 + 459.67 = 533.15 K 1.8 With T0 = = 533.15 K Con 500 +1.8 459.67 With T0 = = 533.15 K 1.8 then for any value of T : then for any value of T : A continuación, paravalue cualquier valor de T: then for any �C P �H of T : �C P �H = MCPH(533.15,T;3.578,3.020E-3,0.0,-0.186E+5) R �C PR�H = MCPH(533.15,T;3.578,3.020E-3,0.0,-0.186E+5) = MCPH(533.15,T;3.578,3.020E-3,0.0,-0.186E+5) R Iteration between this equation and Eq. (4.10) starts with a value T ≥ T0 , and Iteration between thisvalue: equation and Eq. (4.10) starts with a value T ≥ T0 , and converges on the final convergesbetween on the final Iteration this value: equation and Eq. (4.10) starts with a value T ≥ T0 , and La iteración entre esta ecuación y la (4.10) comienza con un valor T ≥ T0, y converge al valor converges on the final T =value: 1,250 K or 1,790(◦◦F) final: T = 1,250 K or 1,790( F) ◦ T = 1,250 K or 1,790( T = 1 250 K o 1 790 (°F) F) 04-SmithVanNess.indd 132 8/1/07 13:07:46 4.2. Latent of Pure Substances 4.2. Calores latentesHeats de sustancias puras 4.2 133 133 4.2 LATENT HEATS OF SUBSTANCES CALORES LATENTES DE PURE SUSTANCIAS PURAS Cuando When una sustancia pura se convierte al estado líquido desdeorelvaporized estado sólido seliquid vaporiza a partir de un a pure substance is liquefied from the solid state from othe at constant estado líquido a presión constante, no hay cambio en lathe temperatura; de cualquier modo, of el proceso pressure, no change in temperature occurs;alguno however, process requires the transfer a requierefinite la transferencia finita de calorheat a laeffects sustancia. Estos the efectos llamados amount of de heatuna to cantidad the substance. These are called latenttérmicos heat of son fusion calor latente de fusión y calor latente de vaporización. De manera similar, existen calores de transición and the latent heat of vaporization. Similarly, there are heats of transition accompanying the que acompañan el cambio de una sustancia desolid un estado a otro;forpor ejemplo,the el heat calorabsorbed absorbidowhen cuando los change of a substance from one state sólido to another; example, cristalesrhombic rómbicoscrystalline de azufre cambian a una estructura monoclínica a 95 °C es de 360isJ por sulfur changes to the monoclinic structure at y951◦bar C and 1 bar 360cada J forátomogramo. each gram-atom. La principal estos of procesos es processes la coexistencia dos fases. De acuerdo con la regla de The característica characteristic de feature all these is thede coexistence of two phases. Acfase, un cording sistema to dethe dosphase fases rule, que está formadosystem por unaconsisting sola especie univariante, estado intensivo se a two-phase of aessingle species yis su univariant, and determina la especificación de una sola intensiva. calor latenteproperty. que acompaña un camitspor intensive state is determined bypropiedad the specification of Así, just el one intensive Thus athe bio de fase es heat una accompanying función sólo dea la temperatura, está relacionada con otras deltosistema latent phase change isque a function of temperature only,propiedades and is related medianteother una system ecuación termodinámica exacta: properties by an exact thermodynamic equation: �H = T �V sat d P saturado dT (4.11) (4.11) where for a pure species at temperature T , donde para una especie pura a temperatura T, �H = latent heat ∆H = calor latente �V = volume change accompanying the phase change ∆V = cambio de volumen que acompaña el cambio de fase sat P = saturation pressure Psaturado = presión de saturación The derivation of this equation, known as the Clapeyron equation, is given in Chap. 6. satcapítulo La deducción de estaEq. ecuación, ecuación de Clapeyron, se da denP el 6. slope of /dT is the When (4.11) isconocida applied como to the vaporization of a pure liquid, Cuando se aplica la ecuación (4.11) a curve la vaporización de un líquido puro, d�V Psaturado /dTdifference es la pendiente the vapor pressure-vs.-temperature at the temperature of interest, is the de la curva de presión vapor en la temperatura a la liquid, temperatura de interés, ∆V esheat la diferencia between molarde volumes of función saturateddevapor and saturated and �H is the latent of entre losvaporization. volúmenes molares de vapor saturado líquido saturado, y ∆H es el calor de vaporización. Thus values of �H may ybedecalculated from vapor-pressure andlatente volumetric data. Así, los valores de ∆Hheats se pueden calcular a partir de la presión de vapor y de información volumétrica. Latent may also be measured calorimetrically. Experimental values are available 4 Correlations Los también miden de manera calorimétrica. dispone de valores experimentales at calores selectedlatentes temperatures forsemany substances. forSethe latent heats of many com4 Las correlaciones para para muchas sustancias a diferentes temperaturas. los calores latentes data aredenotmuchos pounds as a function of temperature are given by Daubert et al.5 Nevertheless, 5 Sin embargo, no siempre compuestos en función deatlathe temperatura están porand Daubert y colaboradores. always available temperature of dados interest, in many cases the data necessary for aphay información a laare temperatura de interés, y enevent muchos casos tampoco se conoce información plication disponible of Eq. (4.11) also not known. In this approximate methods are used for necesariaestimates para aplicar ecuación En esta asituación se usanBecause métodos aproximados para estimar el of thelaheat effect (4.11). accompanying phase change. heats of vaporization are efecto térmico que most acompaña un cambio fase. Debido a que losthey calores vaporización los más imby far the important from adepractical point of view, havedereceived most son attention. 6 Alternative portantesOne desde un puntois de han recibido mayor known atención. Un procedimiento de uso común es procedure usevista of apráctico, group-contribution method, as UNIVAP. meth6 el método la contribución de grupo, conocido como UNIVAP. Los métodos alternativos sirven para uno odsdeserve one of two purposes: de dos propósitos: • Prediction of the heat of vaporization at the normal boiling point, i.e., at a pressure of • Predicción del caloratmosphere, de vaporización en as el 101,325 punto dePa. ebullición normal, es decir, a una presión de 1 at1 standard defined mósfera estándar, definida como 101 325 Pa. 4 V. Majer and V. Svoboda, IUPAC Chemical Data Series No. 32, Blackwell, Oxford, 1985; R. H. Perry and D. Green, op. cit., Sec. 2. 4 5 Daubert et al., op. cit. V. Majer yT.V.E.Svoboda, IUPAC Chemical Data Series No. 32, Blackwell, Oxford, 1985; R. H. Perry y D. Green, op. cit., sección 2. 5 T. E. Daubert et al., op. cit. 6 M. Klüppel, S. Schulz, and P. Ulbig, Fluid Phase Equilibria, vol. 102, pp. 1–15, 1994. 6 M. Klüppel, S. Schultz y P. Ulbig, Fluid Phase Equilibria, vol. 102, pp. 1-15, 1994. 04-SmithVanNess.indd 133 8/1/07 13:07:47 CHAPTER 4. 4. Heat Heat Effects Effects 134 CHAPTER 134 CHAPTER 4. Heat Effects 134 134 CHAPTER 4. 4. Heat Effects CAPÍTULO Efectos térmicos CHAPTER 4. Heat Effects 134 Estimation of of the the heat heat of of vaporization vaporization at at any any temperature temperature from from the the known known value value at at aa •• Estimation •• Estimation of the heat of vaporization at any temperature from the known value at • Estimación del calor de vaporización a cualquier temperatura a partir del valor conocido a una Estimation of the heat of vaporization at any temperature from the known value at aatempesingle temperature. single temperature. single temperature. • Estimation of the heat of vaporization at any temperature from the known value at a ratura particular. single temperature. single temperature. Rough estimates estimates of of latent latent heats heats of of vaporization vaporization for for pure pure liquids liquids at at their their normal normal boiling boiling Rough Rough estimates of heats of for their boiling Estimaciones burdas de los calores latentes de vaporización paraliquids líquidosat en sus puntos Rough estimates of latent latent heats of vaporization vaporization for pure pure liquids atpuros their normal normal boilingde ebupoints are are given given by Trouton’s Trouton’s rule: points by rule: points are given by Trouton’s rule: estimates of regla latent heats of �H vaporization for pure liquids at their normal boiling llición normal están dadas la de Trouton: pointsRough are given bypor Trouton’s rule: �Hnn �H ∼ 10 10 points are given by Trouton’s rule: n∼ �H 10 RTnnn ∼ RT ∼ 10 �H RT RTnn ∼ 10 is the the absolute absolute temperature temperature of of the theRT normal boiling point. point. The The units units of of �H �Hnn,, R, R, and and TTnn where TTnn is normal boiling where n the absolute temperature of point. The units of �H and where T is n ,, R, absolute temperature of the the normal normal boiling boiling The �H and TTnn where must beTnchosen chosen so that �H �H /RTnn is is dimensionless. dimensionless. Datingpoint. from 1884, 1884,units thisof rule still provides must be that Dating from this rule still provides n is the so n R, nn/RT must be chosen so that �H /RT is dimensionless. Dating from 1884, this rule still provides is the absolute temperature of the normal boiling point. The units of �H , R, and where T n n ncheck n n, Rprovides n deben donde Taamust es labetemperatura absoluta del punto de ebullición normal. Las unidades ∆H y TnTse chosen sowhether that �Hvalues is dimensionless. from thisde rule still nsimple simple check on on whether values calculated by other otherDating methods are1884, reasonable. Representative by methods are reasonable. Representative n /RT ncalculated a simple check on whether values calculated by other methods are reasonable. Representative must be chosen so that �H /RT is dimensionless. Dating from 1884, this rule still provides n n elegir de tal forma que ∆H /RT no tenga dimensiones. Desde 1884, esta regla proporciona una comprobaa simple check on whether values calculated by other methods are reasonable. Representative n n experimental values for this ratio are Ar, 8.0; N , 8.7; O , 9.1; HCl, 10.4; C H , 10.5; H S, experimental values for this ratio are Ar, 8.0; N22, 8.7; O22, 9.1; HCl, 10.4; C66H66, 10.5; H22S, values for this ratio are Ar, O ,, 9.1; HCl, 10.4; C S, aexperimental simple on whether values calculated byN methods reasonable. Representative 2 ,, 8.7; ción simple de sicheck los valores calculados por otros Valores experimental values this ratioson arerazonables Ar, 8.0; 8.0; Nother O22métodos. 9.1;are HCl, 10.4; experimentales C66H H66,, 10.5; 10.5; H H22represenS, O, 13.1. for 10.6; and H 13.1. 10.6; and H 2 8.7; 22O, O, 13.1. 10.6; and H experimental values for this ratio are Ar, 8.0; N , 8.7; O , 9.1; HCl, 10.4; C H , 10.5; H S, 2 2 10.4; C26H6, 10.5; H2S, 10.6, 6 y6 H2O, 2 7 7 tativos de estaOf relación son Ar, 8.0; N , 8.7; O , 9.1; HCl, 13.1. 10.6; and H O, 13.1. 2 2 Of the the2same same nature, nature, but but not not quite quite so so simple, simple, is is the the equation equation proposed proposed by by Riedel: Riedel: 7 7 Of same nature, but so is proposed by O, 13.1. 10.6; and H2naturaleza, 7 De la misma pero nonot tanquite simple, es la ecuación propuesta por Riedel: Of the the same nature, but not quite so simple, simple, is the the equation equation proposed by Riedel: Riedel: 7 Of the same nature, but not quite so simple, is the equation proposed by Riedel: �Hnn − 1.013) 1.013) 1.092(ln PPcc − �H 1.092(ln �H 1.013) PPc − = 1.092(ln (4.12) (4.12) n= 1.092(ln �H − 1.013) n c = (4.12) RT 0.930 − T RT 0.930 − T n r n r nn = (4.12) (4.12) �H 1.092(ln Pc−−T1.013) RT 0.930 n r RTn = 0.930 − Trnn (4.12) n where PPcc is is the the critical critical pressure pressureRT innbars bars and and 0.930 is − theTrreduced reduced temperature at at TTnn.. Equation Equation where in TTrrnn is the temperature P is the critical pressure in bars and T is at donde Pwhere es la presión crítica en bar y T es la temperatura reducida a Tntemperature . Resulta sorprendente que, para ser c r c r n n where is is the critical pressure bars and Trn expression; is the the reduced reduced temperature at TTnn.5.5 Equation Equation (4.12) isPcsurprisingly surprisingly accurate forinan an empirical expression; errors rarely exceed exceed percent. (4.12) accurate for empirical errors rarely percent. (4.12) is surprisingly accurate for an empirical expression; errors rarely exceed 5 percent. where P is the critical pressure in bars and T is the reduced temperature at T . Equation una expresión empírica, la ecuación (4.12) es exacta; los errores rara vez exceden a 5%. Aplicándola c r n n (4.12) isto accurate for an empirical expression; errors rarely exceed 5 percent.al agua Applied tosurprisingly water itit gives: gives: Applied water Applied to water it gives: (4.12) is surprisingly accurate for an empirical expression; errors rarely exceed 5 percent. se obtiene: Applied to water it gives: Applied to water it gives:�H 1.092(ln 220.55 220.55 − − 1.013) 1.013) �Hnn 1.092(ln 220.55 − 1.013) �H = 1.092(ln = 13.56 13.56 n= 1.092(ln 220.55 − 1.013) = �H n = = 13.56 RTnn = 1.092(ln 0.930 − 0.577 0.577 RT 0.930 − �H 220.55 − 1.013) = 13.56 RT 0.930 − 0.577 n RTn = 0.930 − 0.577 = 13.56 0.930 − 0.577= −1 =n(13.56)(8.314)(373.15) (13.56)(8.314)(373.15) = 42,065 42,065 JJ mol mol−1 Whence, �HnnRT = Whence, �H De donde, −1 = (13.56)(8.314)(373.15) = 42,065 J mol Whence, �H −1 n (13.56)(8.314)(373.15) = 42,065 J mol Whence, �Hn =−1 −1 −1 −1 −1 This corresponds to 2,334 J g ; the steam-table value of 2,257 J g is loweren by3.4%. 3.4 percent. percent. –1 –1 This corresponds to 2,334 J g ; the steam-table value of 2,257 J g is lower by 3.4 = (13.56)(8.314)(373.15) = 42,065 mol Whence, �H n −1 −1 Esto corresponde a 2 334 to J g2,334 ; el Jvalor lasteam-table tabla de vapor deof 2 257 J gJ g −1 es is menor This corresponds gg−1;;de the value 2,257 lower by 3.4 percent. This corresponds to 2,334 J the steam-table value of 2,257 J g is lower by 3.4 percent. Estimates of the latent heat of vaporization of a pure liquid at any temperature from the a partir Estimates of the latent heat of vaporization of a pure liquid at any temperature from the −1 −1 Las estimaciones del calor latente de vaporización de aunpure líquido puro aiscualquier temperatura Estimates of the latent heat of vaporization of liquid at any temperature from the This corresponds to 2,334 J g ; the steam-table value of 2,257 J g lower by 3.4 percent. Estimates of the latent heat of vaporization of pure liquid at any temperature from the conoknown value at a single temperature may be based on a known experimental value or on a value known value at a single temperature may be based on a known experimental value or on a value de un valor conocido a auna temperatura particular, se pueden hacer con base en temperature un value valor or experimental known value at single temperature may be based on a known experimental on a value Estimates of the latent heat of vaporization of a pure liquid at any from the 88 has known value at a single temperature may be based on a known experimental value or on a value 8 estimated by Eq. (4.12). The method proposed by Watson has found wide acceptance: estimated by Eq. (4.12). The method proposed by Watson found wide acceptance: 8 cido o sobre unvalue valor estimado por la ecuación (4.12). El método propuesto por Watson goza una amplia estimated by Eq. (4.12). The proposed by Watson found acceptance: known a single may be based a known experimental or on adevalue 8 has estimated byat Eq. (4.12).temperature The method method proposed byon Watson has found wide widevalue acceptance: aceptación: 8 has found wide acceptance: �0.38 � by Watson � � estimated by Eq. (4.12). The method proposed 0.38 � − TTrr22 � �H22 �H �0.38 �111− − T �H 0.38 = (4.13) = (4.13) r 2 2 −TTTrrr2�0.38 �H112 = � 111− (4.13) �H − �H 11 = (4.13) (4.13) 1 − T �H �H �H211 = 11 − − TTrrr211 (4.13) �H1 its − Tis This equation equation is is simple simple and and fairly fairly accurate; accurate; its1use use isr1illustrated illustrated in in the the following following example. example. This This is and fairly use in following Esta ecuación es simple ysimple bastante su uso seits ilustra el siguiente ejemplo. This equation equation is simple andexacta; fairly accurate; accurate; its use is isenillustrated illustrated in the the following example. example. This equation is simple and fairly accurate; its use is illustrated in the following example. 134 Example 4.4 Example Ejemplo 4.4 4.4 Example 4.4 −1,, estimate Given that that the the latent latent heat heat of of vaporization vaporization of of water water at at 100 100◦◦C C is is 2,257 2,257 JJ gg−1 estimate Given Example Given that the4.4 latent ◦◦heat of vaporization of water at 100◦◦C is 2,257 J g−1 −1, estimate –1 Dado que el calor latente de vaporización del aguaof a water 100 °Cates100 2 ◦257 , calcule calor latente a Given that theat latent C isJ g2,257 J g−1 el , estimate the latent heat at 300 C. of vaporization the latent heat 300 C. ◦heat the at thatheat the latent ◦C. 300 °C.Given the latent latent heat at 300 300◦heat C. of vaporization of water at 100 C is 2,257 J g , estimate the latent heat at 300 C. 77L Riedel, Chem. Chem. Ing. Ing. Tech., Tech., vol. vol. 26, 26, pp. pp. 679–683, 679–683, 1954. 1954. Riedel, 7L Riedel, Chem. Ing. Tech., vol. 26, pp. 679–683, 1954. 7L L Riedel, Chem. Ing. Tech., vol. 26, pp. 679–683, 1954. 878K. K. Riedel, M. Watson, Watson, Ind. Eng. Chem., vol. 35, 679–683, pp. 398–406, 398–406, 1943. Chem. Ing. Tech., vol.vol. 26, pp. 1954. M. Ind. Eng. Chem., 35, pp. 1943. 8L 7 L. Riedel, M. Watson, Ind. Eng. Chem., vol. 35, pp. 398–406, 1943. Chem. Ing. Tech., vol. 26, pp. 679-683, 1954. 8K. K. M. Watson, Ind. Eng. Chem., vol. 35, pp. 398–406, 1943. 8 K. M. Watson, 8 K. M. Watson, Ind. Eng.Ind. Chem., 35, pp. 1943. 1943. Eng.vol. Chem., vol.398-406, 35, pp. 398–406, 04-SmithVanNess.indd 134 8/1/07 13:07:52 4.3. Standard Heat of Reaction 4.3. Calor estándar de reacción 135 135 Solución 4.4 Solution 4.4 calor latente a 100 °C = 2◦ 257 J g–1 �H = latent heat at 100 C = 2,257 J g−1 calor1 latente a 300 °C ◦ �H2 = latent heat at 300 C 373.15/647.1 = 0.577 Tr1 = 373.15/647.1 = 0.577 573.15/647.1 = 0.886 Tr2 = 573.15/647.1 = 0.886 En seguida, ecuación Thenpor by la Eq. (4.13), (4.13), Sean ∆H1 = Let ∆H2 = Tr1 = Tr2 = �H2 = (2, 257) � 1 − 0.886 1 − 0.577 �0.38 = (2, 257)(0.270)0.38 = 1,371 J g−1 value given en in las the tablas steam de tables is 1,406 J g−1 El valorThe proporcionado vapor es 1 406 J g.–1. 4.3 4.3 STANDARD HEAT OF REACTION CALOR ESTÁNDAR DE REACCIÓN Heat térmicos effects discussed so hasta far have been physical processes. Chemical also are Los efectos analizados ahora hanforsido sólo para procesos físicos. reactions Las reacciones químicas accompanied either by the transfer of heat or by temperature changes during the course of también se acompañan, ya sea por una transferencia de calor o por variaciones de temperatura durante el reaction—in some cases both. These effects are manifestations of the differences in de molectranscurso de una reacción, y enby algunos casos por ambas. Estos efectos son manifestaciones las diferenstructure, molecular and therefore in energy, of the products and reactants. For example, cias en ular la estructura y, en consecuencia, en la energía de los productos y dethelosreactants reactivos. Por in a combustion reaction possess greater energy on account of their structure than do the prodejemplo, los reactivos en una reacción de combustión poseen una energía mayor que los productos a causa de ucts, and this energía energy must be transferred the surroundings as heato or produce obtenidos products a una su estructura, y esta debeeither ser transferida a lostoalrededores como calor productos at an elevada. elevated temperature. temperatura Each vast number of possible may be outmaneras, in many y cada Cada una de of lasthe muchas reacciones químicaschemical se puedereactions llevar a cabo de carried diferentes different ways, and each reaction carried out in a particular way is accompanied by a particular reacción que se realiza en un procedimiento particular se acompaña por un efecto térmico propio. La tabuheat effect. Tabulation of all possible forreacciones all possiblerealizables reactions is WeDebido lación de todos los efectos térmicos posibles heat para effects todas las es impossible. poco práctica. therefore calculate the heat effects for reactions carried out in diverse ways from data a eso, calculamos los efectos térmicos para las reacciones que se realizan de diversas maneras, afor partir de reactions out inque a standard way.deThis reduces the required data to minimum. información paracarried reacciones se realizan modo estándar. Esto reduce al amínimo la información reThe heat associated with a specific chemical reaction depends on the temperatures of querida. both the reactants and products. A consistent (standard) basis for treatment of reaction heat El calor asociado con una reacción química determinada depende de la temperatura de los reactivos y effects results when the products of reaction and the reactants are all at the same temperature. de los productos. Una base consistente (estándar) para el tratamiento de los efectos térmicos de una reacción Consider the flow-calorimeter method for measurement of heats of combustion of fuel se obtiene cuando los productos de una reacción y los reactivos, todos, están a la misma temperatura. gases. The fuel is mixed with air at ambient temperature and the mixture flows into a combusConsidere el método del calorímetro de flujo para medir los calores de combustión de los gases comtion chamber where reaction occurs. The combustion products enter a water-jacketed section bustibles. El combustible se mezcla con aire a temperatura ambiente y la mezcla fluye hacia una cámara de in which they are cooled to the temperature of the reactants. Because no shaft work is produced combustión donde ocurre la reacción. Los productos de la combustión entran a una sección de enfriamiento by the process, and the calorimeter is built so that changes in potential and kinetic energy are con agua, en la que se enfrían a la temperatura de los reactivos. Puesto que no se produce trabajo de flecha negligible, the overall energy balance, Eq. (2.32), reduces to debido al proceso, y el calorímetro está construido de tal forma que son insignificantes los cambios en las energías potencial y cinética, el balance de energíaQtotal, la ecuación (2.32), se reduce a = �H = ∆H Thus the heat flowing from the calorimeter Q and absorbed by the water is equal in magnitude to the enthalpy change caused by the combustion reaction. The enthalpy change of reaction �H Así, la transmisión del calor desde el calorímetro y que es absorbido por el agua es idéntico en magnitud al is called the heat of reaction. cambio de la entalpía causado por una reacción de combustión. El cambio de la entalpía de reacción ∆H se conoce como el calor de reacción. 04-SmithVanNess.indd 135 8/1/07 13:07:53 136 136 CHAPTER 4. Heat Effects CHAPTER 4. Heat Effects 136 CAPÍTULO 4. Efectos térmicos For purposes of data tabulation with respect to the reaction, For purposes of data tabulation with respect to the reaction, Con el propósito de tabular la información con→ respecto la reacción, a A + bB l L + ma M a A + bB → l L + m M aA + bB → lL + mM the standard heat of reaction is defined as the enthalpy change when a moles of A and b moles the standard heat of reaction is defined as theTenthalpy change when of a moles of and bofmoles of B in their standard se states at como temperature react to form l moles L and mAA moles M in el calorof estándar de standard reacción define el cambio de entalpía cuando aofmoles dem y b moles B in their states at temperature T react to form l moles L and moles of MdeinB en sus their standard states at the same temperature T . estadostheir estándar a temperatura T same reaccionan para formar standard states at the temperature T . l moles de L y m moles de M en sus estados estándar a la misma temperatura T. A standard state is a particular state of a species at temperature T A standard state conditions is a particular state of acomposition, species at temperature T and at specified of pressure, and physical Un estado estándar es un conditions estado particular de una especie a temperatura T y en condicioand at specified of pressure, composition, and physical condition as, e.g., gas, liquid, or solid. nes determinadas de presión, composición y condición física, por ejemplo, gas, líquido o condition as, e.g., gas, liquid, or solid. sólido. A standard-state pressure of 1 standard atmosphere (101,325 Pa) was in use for many standard-state pressure of standard atmosphere Pa) wasisin1 use 5 Pa), years,Aand older data tabulations are1 for this pressure. The (101,325 present standard bar for (105many Durante muchos años se utilizó como presión de estadoThe estándar a 1standard atmósfera estándar (101 325 Pa) years, and older data tabulations are for this pressure. present is 1 bar (10 Pa), but for purposes of this chapter, the difference is of negligible consequence. With5 respect to y las antiguas tabulaciones de datos son para esta presión. Ahora el estándar es 1 bar (10 Pa); de cualquier but for purposes this chapter, the difference is of negligible consequence. With For respect to composition, the of standard states used in this chapter are states of the pure species. gases, modo, composition, para los finesthe de standard este capítulo, la diferencia tiene consecuencias insignificantes. Con respecto a la states used in this chapter are states of the pure species. For gases, the physical state is the ideal-gas state and for liquids and solids, the real state at the standardcomposición, en estestate capítulo los estados estándar queliquids se usanand sonsolids, los estados destate las especies puras. Para los the ideal-gas and for real at the standardstatephysical pressure andisatthethe system state temperature. In summary, thethe standard states used in this gases, el estado físico es el estado de gas ideal; y para líquidos y sólidos, el estado real a la presión del estado state pressure chapter are: and at the system temperature. In summary, the standard states used in this estándarchapter y a la temperatura del sistema. En resumen, en este capítulo los estados estándar usados son: are: • Gases: The pure substance in the ideal-gas state at 1 bar. • Gases: sustancia pura ensubstance el estado de gasideal-gas ideal a 1 state bar. • Gases: The pure in the at 1 bar. • Liquids and líquido solids: oThe real puro pure real liquid solid at 1 bar. • Líquidos y sólidos: sólido a 1orbar. • Liquids and solids: The real pure liquid or solid at 1 bar. Property values in theen standard state are denoted by the symbol. For example, Los valores de una propiedad el estado estándar se indican condegree el símbolo de grado. Por ejemplo, CP° values in estado the state are denoted by theestándar degree C P◦◦ isProperty thecalorífica standard-state heatstandard capacity. Because the elstandard state forsymbol. gaseslos is For the example, ideal-gas es la capacidad en el estándar. Puesto que estado para gases es el estado de C Ppara is the standard-state heat capacity. the standard state for isal the ideal-gas ◦ for gas ideal, gases CP°isesidentical idéntica a CPigC , yigla,Because información deTable la tabla C.1 se gases aplica estado estándar gases with and the data of C.1 apply to the standard state para state, Clos ig P P for gases is identical with C P , estándar and of Table C.1 apply to the standard state es la state, C P◦ las los gases. condiciones para estado son fijas excepto la temperatura, que siempre forTodas gases. All conditions for aun standard statethe aredata fixed except temperature, which is always for All conditions for aStandard-state standard stateproperties are fixedson except temperature, is always del sistema. Debido a eso, las system. propiedades del estado estándar funciones sólo de lawhich temperatura. El estado the gases. temperature of the are therefore functions of temperature the temperature of the system. Standard-state properties are therefore functions of temperature estándaronly. elegido los gases hipotético, ya que 1 bar los gases reales ideales.gases Sin embargo, Thepara standard statees chosen for gases is aahypothetical one, for atno1 son bar actual are not estos only. state chosen for gases hypothetical one, at 1un barpoco actual gases arelas not gases rara vezThe se standard desvíanthey mucho de lodeviate ideal, ymuch enislaafrom mayoría de los casos son diferentes entalpías ideal. However, seldom ideality, and informost instances enthalpies for ideal. However, they seldom deviate much from ideality, and in most instances enthalpies for para el estado del gas realata11bar barand y elthe estado del gas ideal. the real-gas state ideal-gas state are little different. the real-gas state 1 bar the ideal-gas different. Cuando se proporciona un and calor reacción paraare unalittle reacción particular, éstethe sestoichiometric ajusta para los coefiWhen a heatatof reaction isde given for astate particular reaction, it applies for When a heat of reaction is given for a particular reaction, it appliesse for the stoichiometric cientes coefficients estequiométricos en la ecuación. Si cada coeficiente estequiométrico duplica, as written. If each stoichiometric coefficient is doubled, the heat of también reaction lo is hace el asexample, written. If each stoichiometric coefficient the heat of reaction is calor decoefficients reacción.For Por ejemplo,the la ammonia reacción de síntesis reaction de amoniaco sedoubled, puede como: doubled. synthesis mayisbe written:escribir doubled. For example, the ammonia synthesis reaction may be written: 1 ◦ = −46,110 J N + 332 H2 → NH3 �H298 12 2 ◦ = −46,110 J N + H → NH �H 2 2 3 298 2 2 ◦ = −92,220 J �H298 or N2 + 3H2 → 2NH3 ◦ = −92,220 J �H298 or N2 + 3H2 → 2NH3 o ◦ indicates that the heat of reaction is the standard value for a temperature The symbol �H298 ◦ indicates that the heat of reaction is the standard value for a temperature The symbol �H ◦ 298 of 298.15 (25 C).que el calor de reacción es el valor estándar para una temperatura de 298.15 K El símbolo ∆H° Kindica of 298.15298K (25◦ C). (25 °C). 4.4 STANDARD HEAT OF FORMATION STANDARD HEAT FORMATION 4.4 4.4 CALOR ESTÁNDAR DEOF FORMACIÓN Tabulation of data for just the standard heats of reaction for all of the vast number of possible Tabulation ofimpractical. data for just the heats of reaction all the vast number ofposibles possible La tabulación de is información para losstandard calores the estándar de reacción deof todas las reacciones reactions Fortunately, standard heat for of any reaction can be calculated ifes poco reactions is impractical. Fortunately, the standard heat of any reaction can be calculated if práctica. Por fortuna, el calor estándar de cualquier reacción se puede calcular si se conocen los calores estándar de formación de los compuestos que participan en una reacción. Una reacción de formación se define 04-SmithVanNess.indd 136 8/1/07 13:07:55 4.4. Standard Heat of Formation 4.4. Standard Heat of Formation 137 137 4.4. Calor estándar de formación 137 the standard heats of formation of the compounds taking part in the reaction are known. A the standard heats of formation of thereaction compounds takingaspart in the which reaction are known. A formation is defined a reaction forms a single compound from its constituent formation reaction iscomo defined as a reaction which forms a single compound from its constituent una reacción donde sólo se forma un compuesto a partir de sus elementos que lo constituyen. Por ejem1 elements. For1 example, the reaction C + 2 O2 + 2H2 → CH3 OH is the formation reaction for elements. For example, reaction OH is the formation reaction for plo,the la reacción C + O + 2H → CH OH es la reacción de formación para el metanol. La reacción 2 2 H O + 3SO → H SO is not a formation reaction, because it forms H2O + 2 reaction methanol. The 2 3 2 4 methanol. The reaction O + SO → H SO is not formation because it forms SO3H→ H SO no es una reacción de formación, porque nocompounds. forma ácidoFormation sulfúrico areactions partir deare los unelementos 2 sulfuric 2 from 4 2 43 acid not the aelements butreaction, from other sulfuric acid not from thedeelements but from other compounds. Formation reactions are unsino otros compuestos. Se sobreentiende que las reacciones de formación dan como resultado 1 mol del derstood to produce 1 mol of product; the heat of formation is therefore based on 1 mol of the derstood to produce 1compuesto; mol ofcompound product; heat of formation is therefore based on 1 mol of the por lothe tanto, el calor de formación se refiere a 1 mol del compuesto formado. formed. compound formed. Los calores cualquier temperatura a partir la información de ifla the capacidad Heatsdeofreacción reactiona at any temperature cansebecalculan calculated fromdeheat-capacity data Heats of reaction at any temperature can be calculated from heat-capacity data if the calorífica si se conoce el valor para una temperatura; en consecuencia, la tabulación de los datos se value for one temperature is known; the tabulation of data can therefore be reduced to thereduce a value for one temperature iscompilation known; of datade thereforeat be reduced totemperatura. the la recopilación dethe losoftabulation calores estándar formación para una sola La usual elección usualfor para esta standard heats ofcan formation a single temperature. The choice compilation of standard heats of formation at a single temperature. The usual choice for temperatura es de 298.15 K o 25 °C. El calor estándar de formación de un compuesto a esta temperatura se ◦ this temperature is 298.15 K or 25 C. The standard heat of formation of a compound at this ° this temperature is 298.15 Ktemperature or por 25◦ el C. símbolo The standard heat of formation of a compound at this representa ∆H . El símbolo de grado indica que éste es el valor estándar, el subíndice f ◦ f is represented by the symbol �H f298 . The degree symbol denotes the standard ◦ . 298 temperature is represented the symbol �H The degree symbol denotes the standard muestraby que se trata de un calor de formación, y el 298 es la temperatura absoluta aproximada en kelvins. Las value, subscript f fidentifies a heat of formation, and the 298 is the approximate absolute 298 value, subscript f identifies aestos heatvalores of formation, and theofcomunes 298 the tablas de para sustancias se approximate encuentran enabsolute los manuales, las recopilaciones temperature in kelvins. Tables theseisvalues for common substances maypero be found in standard más 9 Una lista de valores abreviada 9se da en temperature in kelvins. Tables of these values for common substances may be found in standard extensas están disponibles en trabajos especializados de referencia. handbooks, but the most extensive compilations available are in specialized reference works. 9 handbooks, but the most extensive available are ininspecialized reference la tabla C.4abridged delcompilations apéndice An list ofC.values is given Table C.4 of App. C.works. An abridged list of values is givenWhen in C.4 of químicas App. C. se Cuando lasTable ecuaciones estándar de reaction reacciónmay también se chemical equations arecombinan combinedpor byadición, addition,los thecalores standard heats of When chemicalpueden equations are combined by addition, the standard heats of reaction may sumar para dar el calor estándar de una reacción resultante. Esto es posible porque la entalpía also be added to give the standard heat of the resulting reaction. This is possible because en- es una also be added to givefunción the standard heat thefunction, resultingand reaction. This is ende estado, yofsus cambios para ciertos estados inicialinitial ybecause finaland son independientes de la trayectoria. En thalpy is a state its changes forpossible given final states are independent of thalpy is a state function, and its changes for given initial and final states are independent of particular, siempre es posible combinar las ecuaciones de formación y los calores estándar de formación para path. In particular, formation equations and standard heats of formation may always be compath. In particular, formation equations and standard heats(que of formation may comproducirbined cualquier ecuación no sea enitself sí misma unabeecuación de formación), y ésta se acompato produce anydeseada desired equation (not aalways formation equation) and its accompanying bined to produce anyñadesired equation (not itself a formation equation) and its accompanying de sustandard calor estándar de reacción. Las ecuaciones escritas para este fin con frecuencia incluyen heat of reaction. Equations written for this purpose often include an indication ofuna the indicastandard heat of reaction. Equations written for this purpose often include an indication of the ción del estado físico de cada reactivo y producto, es decir, las letras g, l o s se ponen entre paréntesis physical state of each reactant and product, i.e., the letter g, l, or s is placed in parenthesesdespués physical state of each product, i.e., the letter g,unl, gas, or s líquido is placed in parentheses dereactant la fórmula química para indicar si es quizá innecesario, ya que una afterand the chemical formula to show whether it isoasólido. gas, a Esto liquid, or aparezca solid. This might seem after the chemical formula show whether it is a gas, a liquid, or a solid. This might seem especieto química pura a una temperatura particular y a 1 bar por lo general puede existir sólo en un estado fíunnecessary, because a pure chemical species at a particular temperature and 1 bar can usually unnecessary, becausesico. a pure chemical species at a particular temperature and 1 bar can usually Sinexist embargo, con frecuencia se suponen estados ficticios por conveniencia. only in one physical state. However, fictitious states are often assumed for convenience. exist only in one physical state. However, fictitious are often assumed convenience. ◦ C.CO(g) Considere la reacción de states desplazamiento a “gas defor agua” CO2(g) + H2(g) H2O(g) a 25 °C. Consider the reaction CO 25→ This +water-gas2 (g) + H2 (g)◦ → CO(g) + H 2 O(g) at Consider the reaction CO (g) + H (g) → CO(g) + H O(g) at 25 C. This water-gasEsta reacción se encuentra usualmente en la industria química, aunque se realice únicamente temperaturas 2 2 2 shift reaction is commonly encountered in the chemical industry, though it takes place aonly at shift reaction is commonly encountered in the chemical industry, though it takes place only at ◦ ◦ mayores de 25 °C. De cualquier modo, la información que se emplea es a 25 °C, y el paso inicial enany cualquier temperatures well above 25 C. However, the data used are for 25 C, and the initial step in ◦ C, and the initial step in any temperatures well above 25◦calculation C. data used arethis for 25 cálculo de However, los efectos térmicos relativos a esta reacción es para el heat calorofestándar la ◦reacción a ofthe heat effects for reaction is evaluation theevaluar standard reactionde at 25 C. ◦ C. calculation of heat effects this reaction is evaluation the standard heat of reaction at 25 25 °C.for Las reacciones de formación pertinentes y sus calores de formación de la tabla C.4 son: The pertinent formation reactions and their heats of formation from Table C.4 are: The pertinent formation reactions and their heats of formation from Table C.4 are: CO2 (g): C(s) + O2 (g) → CO2 (g) �H ◦f298 = −393,509 J CO2 (g): C(s) + O2 (g) → CO2 (g) �H ◦f298 = −393,509 J que hydrogen el hidrógenoisesanunelement elemento �H ◦f = 0 H2 (g): Puesto Because 298 H2 (g): Because hydrogen is an element �H ◦f298 = 0 1 ◦ CO(g): C(s) + 2 O2◦(g) → CO(g) �H f298 = −110,525 J CO(g): C(s) + 12 O2 (g) → CO(g) �H f298 = −110,525 J 1 H2 O(g): H2 (g) + 2 O◦2 (g) → H2 O(g) �H ◦f298 = −241,818 J H2 O(g): H2 (g) + 12 O2 (g) → H2 O(g) �H f298 = −241,818 J Because the reaction is actually carried out entirely in the gas phase at high temperature, convista de carried que la reacción se lleva cabophase por completotemperature, en fase gaseosa altas temperaturas, la conveBecause the reactionEn is actually out entirely in standard thea gas con-a muy venience dictates that the statesatofhigh all ◦products and reactants at 25◦ C be taken as the dicta que los of estados estándarand de reactants todos los at productos reactivos a 25 °C se consideren como el estado venience dictates thatniencia the standard states all products 25 C beytaken as the de gas ideal a 1 bar, aun cuando en estas condiciones el agua en realidad no exista como un gas. 9 For example, see TRC Thermodynamic Tables—Hydrocarbons and TRC Thermodynamic Tables—Non9 For example, see TRC Thermodynamic Tables—Hydrocarbons and TRC Thermodynamic Tables—Nonhydrocarbons, serial publications of the Thermodynamics Research Center, Texas A & M Univ. System, College hydrocarbons, serial publications of the Thermodynamics Texas A & M Univ.Properties,” System, College Station, Texas; “The NBSResearch Tables ofCenter, Chemical Thermodynamic J. Physical and Chemical Reference Data, Station, Texas; “The NBS Tables ofvol. Chemical Thermodynamic Properties,” J. Physical and op. Chemical Reference Data, 11, supp. 2, 1982. See also, T. E. Daubert et al., cit. Where data are unavailable, estimates based only on 9 Por ejemplo, véase Thermodynamic Tables—Hydrocarbons y TRC Thermodynamic Tables—Non-hydrocarbons, publicaciones vol. 11, supp. 2, 1982. See also, T.molecular E. Daubert et al., may op. cit. Wherebydata unavailable, estimates based on Fluid Phase Equilibria, vol. 103, structure be found theare methods of L. Constantinou and only R. Gani, seriales Center, A & Fluid M Univ. System, Collagevol. Station, molecular structure may be founddel by the methods of L.Research Constantinou andTexas R. Gani, Phase Equilibria, 103, Texas; “The NBS Tables of Chemical Therpp.Thermodynamics 11–22, 1995. modynamic Properties”, J. Physical and Chemical Reference Data, vol. 11, supp. 2, 1982. Véase también T. E. Daubert et al., op. cit. pp. 11–22, 1995. Cuando no hay datos disponibles las estimaciones se basan únicamente en la estructura molecular y se pueden encontrar con los métodos de L. Constantinou y R. Gani, Fluid Phase Equilibria, vol. 103, pp. 11-22, 1995. 04-SmithVanNess.indd 137 9/1/07 12:49:05 Leyen 138 CHAPTER 4. Heat Effects CHAPTER 4. 4. Heat Effects CAPÍTULO Efectos térmicos 138 138 ideal-gas state at 1 bar, even though water cannot actually exist as a gas at these conditions. Escribir lasstate reacciones deeven formación, tal forma su suma deseada, requiere ideal-gas at 1 bar, though de water cannotque actually existproduzca as a gas la at reacción these conditions. Writing the formation reactions so that their sum yields the desired reaction, requires that Writing the formation so that their sum yields desired reaction, requires tiene that signo que la reacción de formación para elreactions CO2 se escriba a la inversa; en the tal caso el calor de reacción the formation reaction for CO2 be written in reverse; the heat of reaction is then of opposite the formation reaction for CO be written in reverse; the heat of reaction is then of opposite contrario a su calor estándar de formación: 2 sign to its standard heat of formation: sign to its standard heat of formation: ◦ = 393,509 J �H298 CO2 (g) → C(s) + O2 (g) ◦ = 393,509 J �H298 CO2 (g) → C(s) + O2 (g) 1 ◦ = −110,525 J C(s) + 21O2 (g) → CO(g) �H298 ◦ = −110,525 J C(s) + 2 O2 (g) → CO(g) �H298 1 ◦ H2 (g) + 21O2 (g) → H2 O(g) �H298 J ◦ == −241,818 H2 (g) + 2 O2 (g) → H2 O(g) �H298 −241,818 J ◦ = 41,166 J CO2 (g) + H2 (g) → CO(g) + H2 O(g) �H298 ◦ = 41,166 J CO2 (g) + H2 (g) → CO(g) + H2 O(g) �H298 The meaning of this result is that the enthalpy of 1 mol of CO mol1 of H2de O is greater than que la ElThe significado resultado es the queenthalpy la entalpía 1 mol deplus CO 1más mol O es mayor meaningde of este this result is that of 1demol of CO plus 1 mol of H2 O H is2greater than the enthalpy of 1 mol of CO2 plus 1 mol of H2 by 41,166 J when each product and reactant is entalpíathe de 1enthalpy mol de CO más of 1 mol H2 en 41 166 cadaJ producto y reactivo es considerado of 12 mol CO plus 1 mol of HJ2cuando by 41,166 when each product and reactant is como 2de ◦ taken as the pure gas at 25 ◦C in its ideal-gas state at 1 bar. taken as °C the en pure gas at 25 in ideal its ideal-gas el gas puro a 25 el estado delCgas a 1 bar.state at 1 bar. In this example the standard heat of formation of H2 O is available for its hypothetical OH is2O available for itsestándar hypothetical this example the standard of formation of H de En este In ejemplo, se dispone del calor heat estándar de formación en su estado de gas ideal ◦ ideal-gas standard state at 25 ◦C. One might expect the value2 of the heat of formation of water ideal-gas standard state at 25 que C. One might expect the value of the heat of se formation ofsuwater hipotético a 25 °C. Se podría esperar el valor del calor de formación del agua liste para real ◦ for to be listed for its actual state as a liquid at 1 bar and 25 ◦C. As a matter of fact, values estado C. As aestán matter of fact, values for ya que tolíquido be listed for its actual state as a los liquid at 1 para bar and 25 estados como un a 1 bar y 25 °C. De hecho, valores ambos dados en la tabla C.4, both states are given in Table C.4 because they are both frequently used. This is true for many both states are given C.4 because they are both que frequently used. This is true for líquidos many a 25 ◦C se usancompounds con frecuencia. Esto in es Table cierto muchos compuestos normalmente como that normally existpara as liquids at 25 and 1 bar. Cases do arise,existen however, in which that normally exist as liquids atvalor 25◦ Csólo andestá 1 bar. Cases doelarise, however, incomo which °C y a 1acompounds bar. Sin embargo, hay casos en los que un dado para estado estándar value is given only for the standard state as a liquid or as an ideal gas when what is needed isun líquia valuecuando is given only the standard as a liquid or as an ideal when what is ejemplo needed is do o gasthe ideal que for se necesita elstate otro queexample, éste gas fuera el caso other value.loSuppose this wereesthe casevalor. for theSuponga preceding with only del the standardanterior the other value. Suppose this were the case for the preceding example, with only the standard y que sólo estándar de formación del H2then O líquido. debemos una ecuación heatseofconociera formationelofcalor liquid H O available. We would includeAhora an equation forincluir the physical heat of formation of liquid H22 O available. We would then include an equation for the physical para el change cambio that físico que transforma agua su estado estándar estado transforms water el from itsdesde standard state as a liquidcomo into un its líquido standarda su state as anestándar change that transforms water from its standard state as a liquid into its standard state as an ideal The change forpara this physical process is es theladifference heats de of formacomo un gas gas. ideal. El enthalpy cambio de entalpía este proceso físico diferenciabetween entre losthe calores ideal gas. The enthalpy change for this physical process is the difference between the heats of of water in its two standard states: ción delformation agua en sus dos estados estándar: formation of water in its two standard states: −241,818 − (−285,830) = 44,012 J –241 818 − – (–285 830) ==4444,012 012 J J −241,818 (−285,830) This is approximately the latent heat of vaporization of water at 25◦◦C. The sequence of steps C. secuencia The sequence of steps This is approximately the latent of vaporization water at °C. 25 La Éste es is aproximadamente el calor latenteheat de vaporización delof agua a 25 de etapas es ahora: now: is now: ◦ CO2 (g) → C(s) + O2 (g) �H298 J ◦ == 393,509 CO2 (g) → C(s) + O2 (g) �H298 393,509 J ◦ = −110,525 J C(s) + 121O2 (g) → CO(g) �H298 ◦ = −110,525 J C(s) + 2 O2 (g) → CO(g) �H298 1 ◦ = −285,830 J H2 (g) + 21O2 (g) → H2 O(l) �H298 ◦ = −285,830 J H2 (g) + 2 O2 (g) → H2 O(l) �H298 ◦ H2 O(l) → H2 O(g) �H298 J ◦ == 44,012 H2 O(l) → H2 O(g) �H298 44,012 J ◦ = 41,166 J CO2 (g) + H2 (g) → CO(g) + H2 O(g) �H298 ◦ = 41,166 J CO2 (g) + H2 (g) → CO(g) + H2 O(g) �H298 This result is of course in agreement with the original answer. This result is of course in agreement the original answer. Por supuesto, este resultado concuerda con lawith respuesta original. Example Example 4.5 4.5 Ejemplo 4.5the standard heat at 25◦◦C for the following reaction: Calculate Calculate the standard heat at 25 C for the following reaction: Calcule el calor estándar a 25 4HCl(g) °C para la 2H2 O(g) + 2Cl2 (g) +reacción O2 (g) →siguiente: 4HCl(g) + O2 (g) → 2H2 O(g) + 2Cl2 (g) 4HCl(g) + O2(g) → 2H2O(g) + 2Cl2(g) 04-SmithVanNess.indd 138 8/1/07 13:08:00 4.5. Standard Heat of Combustion 4.5. Calor estándar de combustión 139 139 Solución 4.5 Solution 4.5 Los calores estándar de formación a 298.15 K de la tabla C.4 son: Standard heats of formation at 298.15 K from Table C.4 are: HCl(g): –92 307 J H2O(g): –241 818 J HCl(g): −92,307 J H22O(g): −241,818 J La combinación siguiente da el resultado deseado: The following combination gives the desired result: 4HCl(g) → 2H22(g) + 2Cl22(g) 2H22(g) + O22(g) → 2H22O(g) 4HCl(g) + O22(g) → 2H22O(g) + 2Cl22(g) ◦◦ = (4)(92,307) �H298 298 ◦◦ = (2)(−241,818) �H298 298 ◦◦ = −114,408 J �H298 298 This result is four times the standard heat of combustion of 4HCl(g) (see below). Este resultado es cuatro veces el calor estándar de combustión de 4HCl(g) (véase más adelante). 4.5 CALOR ESTÁNDAR DEOF COMBUSTIÓN 4.5 STANDARD HEAT COMBUSTION Sólo muy pocas reacciones de formación se pueden llevar a cabo en las condiciones de interés y, por lo tanto, Only a few reactions can actuallysebe carried out at de theforma conditions of interest, andde reacla información paraformation estas reacciones normalmente debe determinar indirecta. Una clase therefore thesefácil reactions usually determinedesindirectly. One of reaction ción que conducedata de for manera y pormust sí misma al be experimento la reacción de kind combustión, y muchos readily itself surgen to experiment is the combustion and many standard heats of Una calores that estándar de lends formación de los calores estándar dereaction, combustión, medidos por calorimetría. formation come from standard of combustion, combustion reacción de combustión se define comoheats una reacción entre unmeasured elementocalorimetrically. o compuesto y elAoxígeno para formar reaction is defineddeascombustión. a reaction between an elementorgánicos or compound oxygen to form productos determinados Para compuestos que and están formados sólospecified de carbono, hiFor organic compounds madeyup of carbon, oxygen drógenocombustion y oxígeno,products. los productos son dióxido de carbono agua, pero elhydrogen, estado deland agua puedeonly, ser vapor o are carbon state ofquemada. the water may be either vapor or líquido.the Laproducts información siempredioxide se basaand en 1water, mol debut la the sustancia liquid. Data are always based onde 1 mol of the substance burned. Una reacción como la formación n-butano: A reaction such as the formation of n-butane: 4C(s) + 5H2(g) → C4H10(g) 4C(s) + 5H22(g) → C44H10 10(g) no puede realizarse en la práctica. De cualquier modo, esta ecuación se obtiene a partir de la combinación de is not feasible in practice. However, this equation results from combination of the following las reacciones de combustión siguientes: combustion reactions: 4C(s) + 4O22(g) → 4CO22(g) 5H22(g) + 2 121 O22(g) → 5H22O(l) 2 11 4CO22(g) + 5H22O(l) → C44H10 10(g) + 6 22 O22(g) 4C(s) + 5H22(g) → C44H10 10(g) ◦◦ = (4)(−393,509) �H298 298 ◦◦ = (5)(−285,830) �H298 298 ◦◦ = 2,877,396 �H298 298 ◦◦ = −125,790 J �H298 298 This result is the standard heat of formation of n-butane listed in Table C.4. Este resultado es el calor estándar de formación del n-butano proporcionado en la tabla C.4. 04-SmithVanNess.indd 139 8/1/07 13:08:03 140 CHAPTER 4. Heat Effects CHAPTER 4. Effects CHAPTER 4. Heat Heat Effects CAPÍTULO 4. Efectos CHAPTER 4. Heat Effectstérmicos 140 140 140 140 ◦ ◦◦ 4.6 TEMPERATURE DEPENDENCE OF � H 4.6 TEMPERATURE DEPENDENCE OF � H 4.6 TEMPERATURE DEPENDENCE OF � H 4.6 DEPENDENCIA CON LA TEMPERATURA H ◦∆H ° 4.6 TEMPERATURE DEPENDENCE OF �DE In the the foregoing sections, standard heats of of reaction reaction are are discussed for for aa reference reference temperature In sections, standard heats discussed temperature En las secciones anteriores, los calores estándar se analizaron temperatura de referencia In theforegoing foregoing sections, standard heats de of reacción reaction are discussed para for a una reference temperature In the foregoing sections, standard heatstheofcalculation reaction areofdiscussed for a reference temperature of 298.15 K. In this section we treat standard heats of reaction at other other of K. this calculation of heats reaction at de 298.15 K. En esta sección se tratawe el treat cálculo calor estándar de reacción temperaturas, of 298.15 298.15 K. In In this section section we treat the thedel calculation of standard standard heats aof ofotras reaction at other pero a of 298.15 K. from In this section we treat the at calculation of standard heats of reaction at other temperatures knowledge of the value the reference temperature. temperatures from reference partir del conocimiento delknowledge valor a la of temperatura de referencia. temperatures from knowledge ofthe thevalue valueat atthe the referencetemperature. temperature. temperatures fromchemical knowledge of the may valuebeatwritten: the reference temperature. The general general reaction The be La reacción químicachemical general reaction se puedemay escribir: The general chemical reaction may bewritten: written: The general chemical reaction may be written: |ν |A + |ν |A + → |ν |A + |ν |A + |ν |ν11|A |A11 + +|ν |ν22|A |A22 + +········· → → |ν |ν33|A |A33 + +|ν |ν44|A |A44 + +········· |ν111|A111 + |ν222|A222 + · · · → |ν333|A333 + |ν444|A444 + · · · i | is aa stoichiometric where |ν coefficient and stands for chemical formula. The species on where |ν coefficient and stands for chemical formula. The donde |ν estequiométrico y Ai indica fórmula especies enspecies la parteon izquierda where |νcoeficiente is a stoichiometric stoichiometric coefficient and AAAiiuna stands foraaaquímica. chemicalLas formula. The species on iii||is i| es un where |νare a stoichiometric coefficient and Aiii stands for aconvention chemical formula. The species on i | is i is as follows: the left reactants; those on the right, products. The sign for ν the are those on the The convention ννiii isde es la siguiente: son reactivos, que las de la sonproducts. productos. Lasign convención delfor signo νifollows: isas as follows: theleft leftmientras arereactants; reactants; those onderecha theright, right, products. The sign convention for the left are reactants; those on the right, products. The sign convention for νi is as follows: (+) for products and negative (−) forreactivos reactants positive products and negative (−) for positive positiva(+) (+)for para productos y negativa (–) para (+) for products and negative (−) forreactants reactants positive positive (+) for products and negative (−) for reactants Theνννijunto withcon theirsus accompanying signsnúmeros are called called stoichiometric numbers. numbers. For example, example, when The signs stoichiometric For Los signos se llaman estequiométricos. Por ejemplo, cuando lawhen reacción de The iνiii with with their their accompanying accompanying signs are are called stoichiometric numbers. For example, when Theammonia νi with their accompanying signs are called stoichiometric numbers. For example, when the synthesis reaction is written: reaction síntesisthe delammonia amoniacosynthesis se escribe como: isiswritten: the ammonia synthesis reaction written: the ammonia synthesis reaction is written: N + 3H → 2NH N +3H 3H22 → → 2NH NN22 + + 3H 2NH33 N2222+ 3H2222 → 2NH333 N22 = H22 = NH33 = then = −1 = −3 =2 NH then νννNN νννHH νννNH en tal caso then −1 −3 N22 = −1 H22 = −3 NH33 = 22 then νN νH νNH 22 = −1 22 = −3 33 = 2 This sign convention allows the definition of standard heat of reaction to be expressed Esta convención de signo permite definición calor estándar reacción expresado en forma This allows the definition of aaastandard heat reaction to expressed Thissign signconvention convention allowsla the definitionde ofun standard heatof ofde reaction tobe be expressed This sign convention allows the definition of a standard heat of reaction to be expressed mathematically by the simple equation: matemática por la ecuación: mathematically mathematicallyby bythe thesimple simpleequation: equation: mathematically by the simple equation: ◦◦ ◦◦ ≡ (4.14) H (4.14) �H νννiiH (4.14) �H ≡ Hi◦i◦ (4.14) �H◦◦ ≡ (4.14) �H ◦ ≡ iii νiii Hiii◦ i ii donde H °i es la entalpía de las especies i en su estado estándar, y la sumatoria se hace sobre todos los produc◦◦◦ is the enthalpy ◦ of species i in its standard state and the summation is over all products where H i isisentalpía the species ii in state is where theenthalpy enthalpy ofestado species inits itsstandard standard stateand andthe thesummation summation isover allproducts products whereHHiiLa tos y reactivos. en elof estándar de un compuesto químico es igual aover su all calor de formación i◦ is the enthalpy of species i in its standard state and compound the summation is over all products where H and reactants. The standard-state enthalpy of a chemical is equal to its heat of i and reactants. The standard-state enthalpy of a chemical compound is equal to its heat and reactants. The standard-state enthalpy of a chemical compound is equal to its heat of más lasand entalpías en el The estado estándar de sus elementos que lo constituyen. Siislas entalpías en el of estado esreactants. standard-state enthalpy of a chemical compound equal to its heat of formation plus the standard-state enthalpies of its constituent elements. If the standard-state formation plus the standard-state enthalpies of its constituent elements. If the standard-state formation plus the standard-state enthalpies of its constituent elements. If the standard-state tándar de todos losplus elementos de manera arbitraria seofhacen iguales a cero como la base de cálculo, entonces formation the standard-state enthalpies its constituent elements. If the standard-state enthalpies of all elements are arbitrarily set equal to zero as the basis of calculation, then the enthalpies of elements are arbitrarily set to as of then enthalpies of all allestándar elementsde are arbitrarily set equal equal to zero zero as the the basis basis of calculation, calculation, then the the ◦ ° = ∆H °f la entalpía en el estado cada compuesto es igual su calor formación. En este caso, H ◦◦◦ = enthalpies of all elements are arbitrarily setisequal to aof zero as thedebasis of calculation, then the ◦ ◦◦f◦ i standard-state enthalpy of each compound its heat formation. In this event, H �H i i standard-state enthalpy of each compound is its heat of formation. In this event, H = �H i f standard-state enthalpy of each compound is its heat of formation. In this event, H = �H i ◦ y la ecuación (4.14) serán: standard-state enthalpy of each compound is its heat of formation. In this event, Hiii = �Hf◦fifii and Eq. (4.14) becomes: i and andEq. Eq.(4.14) (4.14)becomes: becomes: ◦ and Eq. (4.14) becomes: ◦◦◦ = ν �H (4.15) �H ◦◦f◦ ◦ i i f νν �H (4.15) �H = �H i (4.15) (4.15) �H = (4.15) �H ◦ = iii νiii �Hf◦fifii i i where the summation summation over all products and reactants. Esto Thisformaliza formalizes the procedure procedure descrito dedonde la sumatoria se hace sobre todosall losproducts productos yii reactants. reactivos. el the procedimiento en where the isis and where the summation is over over all products and reactants. This This formalizes formalizes the procedure dedewhere the summation is over all products and reactants. This formalizes the procedure de-estándar scribed in the preceding section for calculation of standard heats of other reactions from stanla sección anterior, para el cálculo de los calores estándar de otras reacciones a partir de los calores scribed scribedin inthe thepreceding precedingsection sectionfor forcalculation calculationof ofstandard standardheats heatsof ofother otherreactions reactionsfrom fromstanstanscribed inse the preceding section calculation dard heats ofaplica formation. Appliedfor to the the reaction,of standard heats of other reactions from stande formación. Si a la reacción, dard of Applied dardheats heats offormation. formation. Appliedto to thereaction, reaction, dard heats of formation. Applied to the reaction, (g) → 2H O(g) + 2Cl (g) 4HCl(g) + O 4HCl(g) (g)→ → 2Cl 22(g) 22(g) 2(g) 2(g) 2H O(g) 2Cl 4HCl(g) + → 2H 2H222O(g) O(g)++ + 2Cl (g) 4HCl(g) ++O OO 4HCl(g) + O222(g) → 2H222O(g) + 2Cl222(g) Eq. (4.15) is written: La ecuación (4.15)is escribe como: Eq. written: Eq.(4.15) (4.15) isse written: ◦◦ ◦◦ = 2�H◦◦◦◦ Eq. (4.15) is written: �H − 4�H HCl �H �H◦◦◦ = = 2�H 2�Hff◦ffHHHH22OOOO − −4�H 4�H◦ff◦f◦fHCl HCl HCl �H = 2�H fH22O − 4�H fHCl H22 O HCl With data from Table C.4 for 298.15 K, this becomes: With Withdata datafrom fromTable TableC.4 C.4for for298.15 298.15K, K,this thisbecomes: becomes: With data from Table 298.15 K, this becomes: Con la información de la tablaC.4 C.4forpara 298.15 K, ésta será: ◦ ◦ ◦298 ◦ = �H = (2)(−241,818) − (4)(−92,307) = −114,408 �H (2)(−241,818) − (4)(−92,307) 298 �H298 = (2)(−241,818) − (4)(−92,307) = −114,408 −114,408JJJ ◦ = (2)(−241,818) − (4)(−92,307) = 298 �H298 = −114,408 J in agreement agreement with the the result of of Ex. 4.5. 4.5. in in agreementwith with theresult result ofEx. Ex. 4.5. in agreement the result Ex. 4.5.4.5. en concordancia con with el resultado delofejemplo � � � � � � � � 04-SmithVanNess.indd 140 8/1/07 13:08:08 ◦ ◦ 4.6. Temperature Dependence of �H 141 �H 4.6. Temperature Dependence of 141 �H◦◦◦◦◦ 4.6. Temperature Temperature Dependenceof of�H 141 141 4.6. Dependence 141 �H 4.6. Temperature Dependence of 141 4.6. Dependencia con la temperatura de ∆H° �H 4.6. Temperature Dependence of 141 ◦ ◦ ◦ �H �H◦ 4.6. 4.6.Temperature Temperature Dependence Dependence ofof of 141 141 �H 4.6. Temperature Dependence 141 �H 4.6. Temperature Dependence of 141 For standard reactions, products and reactants are always atstandard-state the standard-state pressure of For standard reactions, products and reactants are always atatthe pressure of For standardestándar, reactions, products andyreactants reactants are alwaysestán theastandard-state standard-state pressure of Para lasFor reacciones los productos reactivosare siempre la presión delpressure estado estándar de standard reactions, products always at of For standard reactions, products and reactants are always atthe the standard-state pressure of 1 bar. Standard-state enthalpies areand therefore functions of temperature only, and by (2.21), Eq. (2.21), For standard reactions, products and reactants are always at the standard-state pressure of 1 bar. Standard-state enthalpies are therefore functions of temperature only, and by Eq. For For standard standard reactions, reactions, products products and and reactants reactants are are always always at at the the standard-state standard-state pressure pressure of of 1 bar. Standard-state enthalpies are therefore functions of temperature only, and by Eq. (2.21), For standard reactions, products and reactants are always at the standard-state pressure of 1 bar. Debido a eso las entalpías del estado estándar son funciones sólo de la temperatura, y por la ecuación For standard reactions, products and reactants areof always at the standard-state pressure of 111 bar. enthalpies are functions temperature only, bar. Standard-state Standard-state enthalpies are therefore therefore functions of temperature only, and and by by Eq. Eq. (2.21), (2.21), bar. Standard-state enthalpies are therefore functions of temperature only, and by Eq. (2.21), ◦ ◦ ◦ofof 1bar. bar. Standard-state Standard-state enthalpies enthalpies are are therefore therefore functions of temperature temperature only, only, and and byby by Eq. Eq. (2.21), (2.21), Standard-state enthalpies are therefore functions temperature only, and Eq. (2.21), (2.21),111bar. = C dT H bar. Standard-state enthalpies are therefore functions of temperature only, and by Eq. (2.21), ◦ = C dT ddHHi◦◦◦◦d◦functions i P =CC CP◦◦◦◦Pi dT dTi dddH = dT Hiii◦i = P◦Pii◦ii dT H ◦ i◦◦◦= C◦P P◦dT = = C C dT d H d H = C dT d H i =C dT dproduct H i iii product Pior where subscript i identifies a particular or reactant. Multiplying byand νi and summing PPP where subscript identifies particular by summing iii reactant. wheresubscript subscriptiiii identifies identifiesaaaaparticular particularproduct productor or reactant. Multiplying Multiplyingby byννννiiiii and and summing where reactant. summing where subscript identifies particular product or reactant. Multiplying by and summing over all products and reactants gives:product or reactant. Multiplying where subscript i identifies a particular Multiplying by ν and summing over all products and reactants gives: where subscript subscript i iiidentifies iand identifies a aaparticular aparticular particular product product oror or reactant. reactant. Multiplying byby by and summing summing over all products and reactants gives: where subscript identifies product reactant. Multiplying and summing dondewhere el subíndice i identifica un producto o reactivo particular. Al Multiplying multiplicar por ννiiννyνiiand sobre todos los where subscript identifies particular product or reactant. Multiplying by and summing over all products and reactants gives: over all products reactants gives: iisumar over all products and reactants gives: ◦ ◦ over over all all products products and and reactants reactants gives: gives: over all products and reactants gives: productos y reactivos se obtiene: over all products and reactants gives:ν d H ◦ ◦ ν d H = ν C dT ii◦◦◦◦ = i ν CCP◦◦◦◦i dT Pi H = dT i dT iCPP νννi idddH νννi C H = dT ii◦i◦i◦= i dT P◦◦P i νiiiii d H i νiiiiiC = i i ◦ ◦ ◦ i ◦ ◦ i P di idH = iiiiνiννC νiiC dT dT i dT iiiiνiννν iC ddH H = dT iH i= PCPP ii= i bei placed i ii Because νai constant, is a constant, it may inside thei Pidifferential: i iiplaced i i the Because ν is it may be inside ii i differential: i Becauseνννiiiiisis isaaaconstant, constant,itititmay maybe beplaced placedinside insidethe thedifferential: differential: Because Because constant, may be placed inside the differential: Because ννiisisis aaconstant, itmay may be placed inside the differential: ◦se ◦ inside ◦ Because constant, it it may be be placed placed inside the the differential: Because aaconstant, constant, it may be placed inside the differential: PuestoBecause que νi esνiννuna constante, puede colocar dentro de la diferencial: iiiis Because isad(ν constant, it may be placed inside the differential: ◦ ◦ d(ν◦◦◦◦i)H=i ) = νi C ◦ν◦◦◦i C or differential: Hi◦ = νi C ◦ν◦◦◦◦i C dT or dd d ννi HHi◦ν◦◦◦◦i = dT Pi dT Pi dT iH d(ν Hi◦i)))= = νiiiiC CP◦Pi dT dT or = νiiiiC CP◦Pi dT dT iH iC iH iC d(ν ν or d ν = ν d(ν H = ν dT or d ν H = ν dT i ◦ i i i i i i i i i P i P i i i P i P i d(νi H or H◦Hii◦◦= ◦ ii◦◦◦) = i i νi C◦ P ◦= i i νi C◦ P i◦i dT i◦i dT P◦◦idT P◦◦idT oor d(ν = iii νiννC νiiC dT oror or ddddd iiiiiνiνννH νiiiiH = iii νiννC νiiC dT d(ν i dT i dT iiiid(ν iH iC iC iiiH � d(ν H )◦)= = H = iH PC i iii = PC i)ii)= � iPPP iPPP iii dT iii dT ◦ . The standard i i standard i by i ◦ ◦ � The term ν H is the heat of reaction, defined Eq. (4.14) as �H � i� i ii i i ii iEq. (4.14) ias i �H ◦. The i ii i νi H i The term is the standard heat of reaction, defined by standard ◦ i i i i νννiH � Theterm term Hi◦◦◦◦i isis isthe thestandard standardheat heatof ofreaction, reaction,defined definedby byEq. Eq.(4.14) (4.14)as as�H �H◦◦◦◦...The Thestandard standard The The term H the standard heat of reaction, defined by Eq. (4.14) as �H The standard i i i i � � � i i i i i heat-capacity change of reaction is defined similarly: i◦◦is the � ° The term ν H standard heat of reaction, defined by Eq. (4.14) as �H . The standard El término Σ ν H es el calor estándar de reacción, definido por la ecuación (4.14) como ∆H°. El cambio en heat-capacity change of reaction is defined similarly: ◦ ◦ ◦ ◦ i i i i i iνH ◦ is ◦.The iis The The term term ν ν H the is the standard standard heat heat of of reaction, reaction, defined defined by by Eq. Eq. (4.14) (4.14) as as �H �H . . The standard standard heat-capacity change of reaction is defined similarly: The term H the standard heat of reaction, defined by Eq. (4.14) as �H The standard i i The term ν H is the standard heat of reaction, defined by Eq. (4.14) as �H . The standard heat-capacity heat-capacity change of reaction reaction is is defined defined similarly: similarly: i ii change i i iii of heat-capacity change of reaction is defined similarly: la capacidad calorífica estándar de reacción se define de manera similar: ◦ heat-capacity heat-capacity change change ofof of reaction reaction isis defined defined similarly: ◦ heat-capacity change reaction defined similarly: heat-capacity change of reaction isis defined similarly: ◦similarly: �C (4.16) �C ≡≡P ≡ ννi CCP◦ν◦◦◦◦i C Pi (4.16) �CP◦◦◦◦P◦≡ (4.16) i i �C ν C (4.16) �C ≡ ν C (4.16) P ◦ i i iiC◦P i P P P P P i �C ≡ ν (4.16) i i ◦ i ◦ P◦◦◦ ≡ iν C i CP◦◦i �C �C (4.16) (4.16) (4.16) �C (4.16) iC �C ≡ iiii iνννiiC (4.16) PiPPP P PP≡ P≡ i i i As a result of these definitions, i ii i As result of these definitions, Asaaaaresult result of these definitions,la ecuación anterior As these definitions, As result of these definitions, Como resultado deof estas definiciones, será: As a result of these definitions, As As a aaresult aresult result ofof of these these definitions, definitions, As these definitions, ◦ = �C As result of these definitions, ◦ dT◦ dT ◦ d �H (4.17) d �H ==�C (4.17) P �H◦◦◦◦◦= �CP◦◦◦◦P◦dT dT (4.17) (4.17) dddd�H �C (4.17) �H = �C dT (4.17) P P P �H �C (4.17) ◦ P◦dT ◦ ◦◦= ◦= ◦◦dT ddd�H d�H �H = �C �C dT (4.17) (4.17) = �C (4.17) �H = �C dT (4.17) P of PPPdT This is fundamental the fundamental equation relating heats reaction to temperature. This is the equation relating heats of reaction to temperature. Ésta esThis la ecuación fundamental que relaciona a los calores de reacción con la temperatura. This is the fundamental equation relating heats of reaction to temperature. fundamental equation relating heats This is is the theUpon fundamental equation relating heats of of reaction reaction to to temperature. temperature. integration Eq. (4.17) becomes: This is the fundamental equation relating heats of reaction to temperature. Upon integration Eq. (4.17) becomes: Al integrar lafundamental ecuación (4.17) se obtiene: This This isis is the the fundamental fundamental equation equation relating relating heats heats ofof of reaction reaction toto to temperature. temperature. Upon integration Eq. (4.17) becomes: This the equation relating heats reaction temperature. This isUpon the fundamental equation relating heats of reaction to temperature. integration Eq. (4.17) becomes: Upon integration Eq. (4.17) becomes: Upon integration Eq. (4.17) becomes: �� T � T �C ◦ Upon Upon integration integration Eq. Eq. (4.17) (4.17) becomes: becomes: ◦ Upon integration Eq. (4.17) becomes: Upon integration Eq. (4.17) becomes: ��� TTTT �C ◦+ ◦ = �H ◦◦◦◦ P dT P ◦ ◦ �C R (4.18) �H �C �C ==�H ++0R� (4.18) ◦PdT �H P◦P �C �H0◦◦◦◦0◦+ R���T TTT�C (4.18) (4.18) �H◦◦◦◦◦= RdT ◦P T �C �H R (4.18) �H = �H + R dT (4.18) �H T ◦◦dT 0R P �C T 0 0 0 0 �C �H RR TTTT0 RRPRPPdT (4.18) �H PdT ◦ 0◦◦◦+ ◦ ◦◦= ◦= = �H �H + RR dT (4.18) (4.18) �H �H = �H (4.18) �H = �H + R T0000 RRR dT dT (4.18) �H 0 00+ 0+ ◦los ◦ are◦ heats ◦son °0�H R and �H are heats of reaction at temperature T and at reference temperature T0 where donde ∆H° y ∆H calores de reacción a temperatura T y a una temperatura de referencia T0T, 0respectiT T R T 0 0 0 and �H of reaction at temperature T and at reference temperature where �H ◦ ◦ T 0 heats of reaction at temperature 0 ◦ ◦ ◦ ◦ ◦ ◦ 0 and �H are T and at reference temperature where �H heats at TTTcapacity and at reference temperature TTTTestá where and �H �H are heats of of reaction reaction at temperature temperature and atcada reference temperature where �H ◦ and 0000 dada 00◦0◦0◦are If the temperature dependence of the heat of each product and reactant vamente. Sirespectively. la�H dependencia de la temperatura de la capacidad calorífica de producto y reactivo and �H are heats of reaction at temperature and at reference temperature where �H respectively. If the temperature dependence of the heat capacity of each product and reactant ◦ ◦ ◦ ◦ 0 0◦ are and �H �H are heats heats of of reaction reaction atat at temperature temperature TTTTand and at at reference reference temperature temperature T0TTT000 where where �H �H◦and respectively. Ifthe the temperature dependence of theheat heatcapacity capacity of each product andreactant reactant and �H heats of reaction temperature and at reference temperature where �H and �H heats of reaction at temperature and at reference where �H respectively. If dependence the of each product and respectively. If the dependence of the heat capacity of each product and reactant 0temperature 0 are 00are is given by Eq. (4.4), then the integral is of given by thecapacity analog of Eq. (4.7) (τTtemperature ≡ por la ecuación (4.4), en taltemperature caso la integral está dada por el análogo de la ecuación (4.7) (τT0 ):/T ≡reactant respectively. If the temperature dependence of the heat of each product and 0T):/ T0): is given by Eq. (4.4), then the integral isisgiven by the analog of Eq. (4.7) (τ ≡ /T respectively. respectively. If If the the temperature temperature dependence dependence of of the the heat heat capacity capacity of of each each product product and and reactant reactant is given by Eq. (4.4), then the integral given by the analog of Eq. (4.7) (τ ≡ T /T ): respectively. If the temperature dependence of the heat capacity of each product and reactant respectively. If(4.4), the temperature dependence ofby thethe heat capacity of(4.7) each(τ product is given by then is analog of ≡ is given by Eq. Eq. (4.4), then the the integral integral is given given by the analog of Eq. Eq. (4.7) (τ ≡ TTT/T /T ): reactant 0000): �and � � by is by Eq. (4.4), then the integral is given by the analog of Eq. (4.7) (τ ≡ /T 0 ):� � �given ◦ (4.4), Tby isis given given by Eq. Eq. (4.4), (4.4), then then the the integral integral isis is given given by by the the analog analog ofof of Eq. Eq. (4.7) (4.7) (τ (τ≡≡ ≡ T�TτT /T T/T /T ): ◦ Eq. then the integral given by the analog Eq. (4.7) (τ T �C �1 0 ): �C 0τ010): − �B �C �D isis given by Eq. (4.4), then the integral is given by the analog of Eq. (4.7) (τ ≡ /T ):� � � � ����given ◦ − �B �C �D T 2 2 3 3 P ◦ ◦ TTT �C ◦ 2 2 3 3 P �C τ − 1 �B �C �D � � (τ − 1) + (τ − 1) + (τ − 1) + dT = (�A)T T T (4.19) (4.19) �CPPP◦PdT = (�A)T0 (τ − 0 1) + �B − 11� 1� �B TT02222(τ �C TT03333(τ �D�� −−1) ++�C −−1) ++�D (4.19) �0τττ − ����T TTT �C � (τ− −1) 1)+ + (τ222022− 1)+ (τ333033− 1)+ =(�A)T (�A)T0000(τ (4.19) − �B �C �D RdT = 20020(τ 30030(τ T τ�� (4.19) ◦ P◦◦◦dT T T 1) 1) (τ − 1) + (τ − 1) + (τ − 1) + dT = (�A)T T T (4.19) T R 2 3 T τ 0 �C 0 �C τ − τ − 1 1 �B �B �C �C �D �D T0T�C 0 τ − 1 �B �C �D 0 �C τ − 1 �B �C �D R 2 3 T τ (τ − 1) + (τ − 1) + (τ − 1) + dT = (�A)T T T (4.19) 2 2 2 2 3 3 3 3 P P 2 2 3 3 P 0 0 T R 2 3 T τ R 2 3 T τ 2 2 3 3 P TTT0000 (τ −− 1) −1) 1) ++ + 2 T0TTT00(τ (τ−− 1) −1) 1) ++ + 3 T0TTT00(τ −1) 1) +++ T0000 dT== = (�A)T (�A)T (4.19) (4.19) (τ (τ(τ−−1) dT (�A)T (4.19) 0 (τ 000(τ (τ − 1) + (τ − 1) + dT = (�A)T (4.19) RRdT T0 RR 2222 00�A 3333A 00(τ − 1) +T0T T000 τ ττττ T0TTT R T 0 00where ν by definition, ≡ i i ν AAi where by definition, �A ≡≡ where bydefinition, definition, �A≡ donde por definición where by �A Aiiii where by definition, �A ≡ i ννννiiiiiiA A where by definition, �A ≡ iA i i i i ν A where where by by definition, definition, �A �A ≡ ≡ where by definition, �A ≡ i iννν i i i AAiii where by definition, �A ≡ i i �D. with analogous definitions for �B, �C, and with analogous definitions for �B, �C, and �D. i i i with analogous definitions for �B, �C, and �D. i con definiciones análogas para ∆B, ∆C y ∆D. with definitions for �B, �C, and with analogous analogous definitions for �B, �C, and �D. �D. a mean heat capacity change of reaction is deAn alternative formulation results when with analogous definitions for �B, �C, and �D. An alternative formulation results when a mean heat capacity change of reaction isisdewith with analogous analogous definitions definitions for for �B, �B, �C, �C, and and �D. �D. Analternative alternative formulation results when meanheat heat capacity change ofreaction reaction de-de reacwith analogous definitions for �B, �C, and �D. Una formulación alternativa se obtiene cuando se define un cambio enchange la capacidad calorífica with analogous definitions for �B, �C, and An formulation results when aaaamean capacity of is An alternative formulation results when mean heat capacity change of reaction is dedefined in analogy to Eq. (4.8): results when�D. An alternative formulation mean heat capacity change of reaction is defined in analogy to Eq. (4.8): An An alternative alternative formulation formulation results results when when a mean a mean heat heat capacity capacity change change of of reaction reaction is is dedefined in analogy to Eq. (4.8): An alternative formulation results when a mean heat capacity change of reaction is deción media analogía con la ecuación An alternative formulation results when a mean heat capacity change of reaction is defined in analogy to (4.8): fineden in analogy to Eq. Eq. (4.8): (4.8): fined in analogy to Eq. (4.8): ◦(4.8): ◦ fined fined in in analogy analogy to to Eq. Eq. (4.8): � ��C fined in analogy to Eq. (4.8): �B �C �D fined in analogy to P◦Eq. (4.8): ��C �B �C �D H ◦◦◦����H P ��C �B �C �D = �A + T0 (τT+ + T 22222(τT2220222+ (τ 2τ + ++1)�D + (4.20) ��C ��C 0 (τ1)++1)�C �B �C �D H = �A ++�B ++τ1) (4.20) ◦P�H P H H P◦P 0 (τ ��C = �A T T (τ + 1) + (τ + τ 1) + (4.20) (4.20) �B �C �D 2τ T 2 R 2 3 ◦ ◦ 0 = �A + T T (τ + 1) + + τ + 1) + (4.20) = �A + T T (τ + 1) + (τ + τ + 1) + (4.20) 0 ◦ 2 2 H P R 2 3 000(τ + 1) +�C τ T 2 � � ��C ��C � �B �B �C �D �D ��C 0 0 �B �C �D 0 0 2 2 � ��C �B �C �D 2 R 2 3 = �A + T T (τ + τ + 1) + (4.20) 0 20222 2 222 PRPPH P HH T20 0 (τ R = �A �A ++ + 222T0TTT T(τ(τ(τ+++ (τ + 1)1) 1) ++ + 333T0TT τ τ+ τ++ 1)1) 1) +++τττTTT (4.20) (4.20) �A (4.20) 000(τ = �A + (τ++ + 1) + (4.20) RR H== 200022 RR 3333 000(τ + τ + 1) +τ ττTττ0T Equation (4.18) becomes:2222 R then TT00020 Equation (4.18) then becomes: Equation (4.18) then becomes: Por lo tanto la ecuación (4.18) será: Equation (4.18) becomes: Equation (4.18) then then becomes: Equation (4.18) then becomes: ◦ � (T − T ) Equation Equation (4.18) (4.18) then then becomes: becomes: Equation (4.18) then becomes: ◦ + ◦��C ◦ (T ◦ = ◦�H �H = �H + ��C (4.21) Equation (4.18) then becomes: �H (4.21) H− P(T �H◦◦◦◦◦= =�H �H0◦◦◦◦0◦+ +0��C ��CP◦◦◦◦P◦����HH(T −TTTT00000)))) 0 (4.21) (4.21) �H − (4.21) �H = �H + ��C (T − (4.21) H P H P 0 0 H P 0 �H = �H + ��C � (T − T ) (4.21) ◦ ◦ ◦ ◦ ◦ ◦◦= ◦ = P 0◦◦ + �H �H �H + ��C ��C �◦◦H �(T (T −−− T0TT )00)) (4.21) (4.21) �H = �H ��C �H (T (4.21) �H =�H �H ��C (4.21) PP 0 00+ 0+ HH(T − T00 ) PP�H � � �� � �� � � � � � � �� � � � �� � �� � � � � � � �� � � � � � � � � � � � �� � �� � � �� � � �� � � �� � � �� � 04-SmithVanNess.indd 141 8/1/07 13:08:23 142 142 142 142 142 142 CHAPTER CHAPTER 4. 4. Heat Heat Effects Effects CHAPTER 4.Heat Heat Effects CHAPTER 4. CAPÍTULO 4. Effects Efectos CHAPTER 4. Heat Effectstérmicos The The integral integral of of Eq. Eq. (4.19) (4.19) is is of of the the same same form form as as that that of of Eq. Eq. (4.7), (4.7), and and in in analogous analogous The integral of Eq. (4.19) is of same form as that of Eq. (4.7), and in analogous Lafashion integral de la ecuación (4.19) tiene lathe misma forma que laof dada por la ecuación (4.7), y de manera The integral of Eq. (4.19) is of the same form as that Eq. (4.7), and in analogous may be set equal to a function: fashion may be set of equal to(4.19) a function: The integral Eq. is of the same form as that of Eq. (4.7), and in analogous fashion may be set equal to a function: análoga puede asignársele una función: fashion may bebe setset equal a function: ��to Tto ◦◦ fashion may equal a function: � TT �C �C ◦ dT = IDCPH(T0,T;DA,DB,DC,DD) �� P ◦ P T �C �C dT = IDCPH(T0,T;DA,DB,DC,DD) P ◦ dT T �C P R IDCPH(T0,T;DA,DB,DC,DD) TT00 RdT IDCPH(T0,T;DA,DB,DC,DD) P == R dT = IDCPH(T0,T;DA,DB,DC,DD) T T0 0 R R ◦◦ and of A, where “D” denotes “�”. analogy requires simple replacement of C �C where “D” “∆”. denotes “�”.T0The The analogy requires simple replacement of CoPP yby by �C and of∆A, A, PP ◦ and dondewhere “D” denota La“�”. analogía requiere la requires sustitución simple de CP por ∆C de�C A,◦ etc., por where “D” denotes “�”. The analogy simple replacement of C by of A, etc. El P P “D” denotes The analogy requires simple replacement of C by �C and of A, P ◦ and etc. by �A, etc. The same computer program serves for evaluation of either integral. The only PC by P etc. by �A, etc. The same computer program serves for evaluation of either integral. The only where “D” denotes “�”. The analogy requires simple replacement of �C of A, P La P The mismo programa deetc. computadora sirve para evaluar cualquiera de las integrales. única diferencia etc. by �A, The same computer program serves for evaluation of either integral. only es el etc. byby �A, etc. same computer serves forfor evaluation of of either integral. The only difference is in the function name. difference isetc. inThe the function name. program etc. �A, The same computer program serves evaluation either integral. The only nombre de la función. difference is in the function name. difference is as in in the function name. Just function MCPH is defined to represent �C so MDCPH by analPP��H /R, Just as function MCPH is MCPH definedse to define represent �Crepresentar so function function MDCPH byMDCPH analdifference is the function name. H /R, so Del mismo modo que laMCPH función para ⟨C /R,MDCPH la función por Just as function is to represent �C function by analP⟩ H MDCPH ◦◦defined P �/R, Just as function MCPH is is defined tothus, represent �C�C �H soso function byby analH /R, ogy is defined to represent ��C � /R; P o ogy is defined to represent ��C � /R; thus, Just as function MCPH defined to represent � /R, function MDCPH analH P ◦ �⟩H /R; P P H analogía se define para representar ⟨∆C / R; así, ◦ ogy is defined to represent ��C thus, ogy is is defined to to represent ��C HH thus, P◦P /R; P ◦◦��H ogy defined represent ��C ��C P �H /R; thus, ��C � H P ◦ H P ◦ �P �H = ��C = MDCPH(T0,T;DA,DB,DC,DD) MDCPH(T0,T;DA,DB,DC,DD) ��C ◦ PR MDCPH(T0,T;DA,DB,DC,DD) ��C RH MDCPH(T0,T;DA,DB,DC,DD) P �H= = R = MDCPH(T0,T;DA,DB,DC,DD) RR Ejemplo 4.6 Example 4.6de la reacción de síntesis del metanol a 800 °C: Calcule el calor estándar Example 4.6 Example 4.6 ◦ Example 4.6 heat Calculate Calculate the the standard standard heat of of the the methanol-synthesis methanol-synthesis reaction reaction at at 800 800◦◦C: C: ◦ C: Calculate the standard heat of the methanol-synthesis reaction at 800 CO(g) + 2H2(g) → CH3OH(g) Calculate the standard heat of of the methanol-synthesis reaction at at 800 ◦ C: Calculate the standard heat the methanol-synthesis reaction 800C: (g) → CH OH(g) CO(g) + 2H CO(g) + 2H22 (g) → CH33 OH(g) (g) → CH OH(g) CO(g) + 2H 2 → CH3 OH(g) 3 CO(g) 2H2H 2 (g) Solution 4.6 Solución 4.6 (g) → CH OH(g) CO(g)+ + Solution 4.6 4.6 2 3 Solution Solution 4.6 Solution Apply Eq. (4.15) to reaction for temperature K and Aplicando la ecuación (4.15) a esta reacción, para la temperatura de TTreferencia T0 = 00 = Apply Eq.4.6 (4.15) to this this reaction for reference reference temperature = 298.15 298.15 K298.15 and K, y Apply Eq. (4.15) to this reaction for reference temperature T= 298.15 and 0 = (4.15) to to this reaction temperature T0T 298.15 KK and with heat-of-formation data from Table C.4: con laApply información de calor de formación dereference lareference tabla with Eq. heat-of-formation data fromfor Table C.4: C.4: Apply Eq. (4.15) this reaction for temperature 0 = 298.15 K and with heat-of-formation data from Table C.4: with heat-of-formation data from Table C.4: ◦◦ ◦◦ = with heat-of-formation data from Table C.4:(−110,525) = −90,135 J �H = �H �H = −200,660 −200,660 − − (−110,525) = −90,135 J �H 00◦ = 298 ◦ = 298 ◦ ◦ = �H −200,660 − (−110,525) = −90,135 �H −200,660 −− (−110,525) == −90,135 J JJ �H 0◦ �H298298 ◦= = 0 = = �H −200,660 (−110,525) −90,135 �H La evaluación de los parámetros en in la ecuación se on basa entaken la información 0 parameters 298 Evaluation of (4.19) is data from C.1: Evaluation of the the parameters in Eq. Eq. (4.19)(4.19) is based based on data taken from Table Tabletomada C.1: de la Evaluation of the parameters in Eq. (4.19) is based on data taken from Table C.1: Evaluation of the parameters in Eq. (4.19) is based on data taken from Table C.1: tabla C.1: 3 6 −5 Evaluation of theiiparameters is based10 on6 C data 3 B −5from ννii in Eq. A 10 10 D A (4.19) 10 10 C taken 10 D Table C.1: 3B 6C 3 B 6C −5−5 A 1010 B 1010 10 D i ii νi ννi A A 10 D 3 6 −5 10 B 10 C 100.000 D CH 11i 2.211 CH33OH OH 2.211 12.216 12.216 −3.450 −3.450 0.000 CH OH 1 2.211 12.216 −3.450 0.000 3 CH OH 1 2.211 12.216 −3.450 0.000 CO −1 0.557 0.000 3 CO −1 3.376 12.216 0.557 −3.450 0.000 −0.031 −0.031 CH 1 3.376 2.211 0.000 CO3 OH −1−2 −1 3.376 0.557 0.557 0.000 0.000 −0.031 −0.031 CO 3.376 H 3.249 0.422 0.000 0.083 2 H −2 3.249 0.422 0.000 0.083 CO −1 3.376 0.557 −0.031 2 H2 −2 3.249 3.249 0.422 0.422 0.000 0.000 0.083 0.083 H2H −2−2 3.249 0.422 0.000 0.083 2 From From its its definition, definition, From its definition, From itsits definition, A partir de su definición, From definition, �A �A = = (1)(2.211) (1)(2.211) + + (−1)(3.376) (−1)(3.376) + + (−2)(3.249) (−2)(3.249) = = −7.663 −7.663 �A = (1)(2.211) + (−1)(3.376) + (−2)(3.249) = −7.663 �A = (1)(2.211) ++ (−1)(3.376) ++ (−2)(3.249) = −7.663 ∆A = (1)(2.211) +(−1)(3.376) (–1)(3.376) (–2)(3.249) == –7.663 �A = (1)(2.211) + (−2)(3.249) −7.663 Similarly, Similarly, Similarly, Similarly, De manera similar, 55 Similarly, −3 −6 −3 −6 �C �D �B �C = = −3.450 −3.450 × × 10 10 �D = = −0.135 −0.135 × × 10 10 �B = = 10.815 10.815 × × 10 10 −3 −6 5 5 −3 −6 �C = −3.450 × 10 �D = −0.135 × 10 �B = 10.815 × 10 3 –6 5 �C == −3.450 ××× 10 = −0.135 10105 �B =∆B 10.815 ×× 1010 −3 = 10.815 × 10 ∆C = –3.450 1010−6 �D ∆D = –0.135 ××10× �C −3.450 �D = −0.135 �B = 10.815 The The value value of of the the integral integral of of Eq. Eq. (4.19) (4.19) for for TT = = 1,073.15 1,073.15 K K is is represented represented by: by: The value of the integral of Eq. (4.19) for T = 1,073.15 K is represented by: The of of thethe integral of of Eq.Eq. (4.19) forfor TTT=== 1,073.15 K is represented by:by: El valor devalue lavalue integral de la ecuación para 1 073.15 KK seis representa por: The integral (4.19) 1,073.15 represented IDCPH(298.15,1073.15;-7.663,10.815E-3,-3.450E-6,-0.135E+5) IDCPH(298.15,1073.15;-7.663,10.815E-3,-3.450E-6,-0.135E+5) = = −1,615.5 −1,615.5 K K IDCPH(298.15,1073.15;-7.663,10.815E-3,-3.450E-6,-0.135E+5) = −1,615.5 K IDCPH(298.15,1073.15;-7.663,10.815E-3,-3.450E-6,-0.135E+5) = −1,615.5 KK IDCPH(298.15,1073.15;-7.663,10.815E-3,-3.450E-6,-0.135E+5) = −1,615.5 Then Then by by Eq. Eq. (4.18), (4.18), Then by Eq. (4.18), (4.18), En este caso, por la ecuación Then by Eq. (4.18), Then by Eq. (4.18), ◦◦ = −90,135 + 8.314(−1,615.5) = −103,566 J �H �H = −90,135 + 8.314(−1,615.5) = −103,566 J ◦ = ◦∆H° �H + 8.314(−1,615.5) −103,566 = −90,135 –90 135 8.314 (–1 615.5)=== –103 566 JJ J �H = −90,135 ++ 8.314(−1,615.5) −103,566 �H ◦ = −90,135 + 8.314(−1,615.5) = −103,566 J 04-SmithVanNess.indd 142 8/1/07 13:08:31 4.7. Heat Effects of Industrial Reactions 4.7. Efectos térmicos de las reacciones industriales 143 143 4.7 HEATTÉRMICOS EFFECTS OF REACTIONS EFECTOS DEINDUSTRIAL LAS REACCIONES INDUSTRIALES 4.7 En las secciones anteriores se analizó el calor de reacción. reacciones industriales raras The preceding sections have dealt withestándar the standard heat of Las reaction. Industrial reactions areveces se llevan ararely cabo carried bajo condiciones estado estándar. Además,Furthermore, en las reacciones reales los reactivos pueden no out under de standard-state conditions. in actual reactions the reacestar presentes ennot proporciones estequiométricas, la reacción no llevarámay a cabo completo, y la tempetants may be present in stoichiometric proportions, thesereaction notpor go to completion, ratura final puede diferir de la inicial. Por otra parte, es probable que se presenten inertes y ocurran and the final temperature may differ from the initial temperature. Moreover,especies inert species may de manera simultánea No obstante, los cálculos de los efectos térmicos de las reacciones be present, and varias severalreacciones. reactions may occur simultaneously. Nevertheless, calculations of the reales están los principios considerados, y se ilustran de mejorconsidered forma mediante un best ejemplo. heat basados effects ofenactual reactionsyaare based on the principles already and are illustrated by example. Ejemplo 4.7 Example 4.7 ¿Cuál es la temperatura máxima que se puede alcanzar por combustión de metano con un exceso de maximum temperature can be reached by the combustion of methane aire deWhat 20%?isElthe metano y el aire entran en that un quemador a 25 °C. with 20% excess air? Both the methane and the air enter the burner at 25◦ C. Solución 4.7 Solution 4.7 La reacción es CH4 + 2O2 → CO2 + 2H2O(g) para la cual, The reaction is CH4 + 2 O2 → CO2 + 2H2 O(g) for which, ◦ = −393,509 + (2)(−241,818) − (−74,520) = −802,625 J �H298 H �H � 0 0 Because the maximum attainable temperature (called the theoretical flame temDado que se buscaislasought, temperatura (llamada teórica de flama), se perature) assumemáxima that thealcanzable combustion reactiontemperatura goes to completion adisupone abatically que la reacción de combustión realiza hasta su terminación (Q = 0). Si los (Q = 0). If the kinetic-seand potential-energy changes adiabática are negligible and cambiosifen energías potencial y cinética son despreciables, y si W = 0, el balance energético s = 0, the overall energy balance for the process reduces to �H = 0. For Wlas s total para el proceso se reduce aof∆H 0. Con el propósito calcular lapath temperatura purposes of calculation the=final temperature, anydeconvenient between final the es posible utilizar estadoisinicial y final. Ladiagram. trayectoria elegiinitialcualquier and final trayectoria states may conveniente be used. Theentre path el chosen indicated in the da se indica en el siguiente diagrama. Reactants at 1 bar and 25�C Reactivos a 1 bar 1y mol 25 °CCH4 2.41 mol mol O CH 24 9.03 2.4 mol mol N O22 9.03 mol N2 04-SmithVanNess.indd 143 �H�P H P Products at 1 bar and T�K Productos a 1 bar 1ymol T/K CO2 21 mol mol H CO 2O 2 0.42 mol mol O H22O 9.03 0.4 mol mol N O22 9.03 mol N2 �H�298 H298 8/1/07 13:08:34 144 CHAPTER 4. Heat Effects CHAPTER 4. Heat Effects CHAPTER 4. Effects CHAPTER CHAPTER 4. Heat Heat Heat Effects Effects CAPÍTULO 4. Efectos térmicos CHAPTER CHAPTER 4. 4. Heat Heat Effects Effects 144 144 144 144144 144 144 When one mole of methane burned is the basis for all calculations, the followCuandoWhen un When mol deone metano quemado seburned toma como base para todos los cálculos, el aire que When one mole of methane burned the basis for all calculations, the followone mole of burned isis for the When one mole mole ofmethane methane of methane burned isthe the is basis the basis basis forall for allcalculations, all calculations, calculations, thefollowthe followfollowWhen one mole of burned is basis for all the When onecantidades mole of methane methane burned is the the basis for all calculations, calculations, the followfollowing quantities of oxygen and nitrogen are supplied by the entering air: entra proporciona las siguientes de oxígeno y nitrógeno: ing quantities of oxygen and nitrogen are supplied by the entering air: ing of and nitrogen are by air: ingquantities ing quantities quantities ofoxygen oxygen of oxygen andand nitrogen nitrogen aresupplied are supplied supplied bythe by theentering the entering entering air:air: ing ing quantities quantities of of oxygen oxygen and and nitrogen nitrogen are are supplied supplied by by the the entering entering air: air: Moles O required 2.0 Moles deMoles O2Moles requeridas = 2.0 Moles O required 2.0 O ====2.0 Moles O2222required O required 2.0 = 2.0 2 required Moles O ==(0.2)(2.0) 2.0 Moles O required 2.0 = 0.4 22 required Moles excess O = 0.4 Moles enMoles exceso de O = (0.2)(2.0) 2 2 Moles excess O = (0.2)(2.0) 0.4 ====0.4 excess OO222=O=(0.2)(2.0) (0.2)(2.0) = (0.2)(2.0) 0.4 = 0.4 Moles Moles excess excess 2 Moles excess O (0.2)(2.0) ===0.4 == (0.2)(2.0) 0.4 Moles excess O= 22 = Moles N entering (2.4)(79/21) 9.03 Moles que entran de N (2.4)(79/21) 9.03 2 2 Moles N entering = (2.4)(79/21) 9.03 Moles NN222entering ==(2.4)(79/21) ====9.03 Moles Moles N entering entering (2.4)(79/21) = (2.4)(79/21) 9.03 = 9.03 2 Moles Moles N N22 entering entering == (2.4)(79/21) (2.4)(79/21) == 9.03 9.03 Los gases que salen del quemador contienen 1 mol de CO , 2 moles deO(g), H0.4 O(g), 0.4 de O2, , 2 mol H O(g), mol O ,,and and The gases leaving the burner contain 1 mol CO 22mol 20.4 2,2, ,22 HH222O(g), mol O and The gases leaving the burner contain mol CO H mol O The gases leaving the contain 111mol CO ,mol 2 mol H O(g), 0.4 0.4 mol mol O222,moles and TheThe gases gases leaving leaving theburner the burner burner contain contain mol 1 mol CO22CO 2mol 2O(g), 2 0.4 2,,O 2 , and , 2 mol H O(g), 0.4 mol O and The leaving the burner contain 1 mol CO , 2 mol H O(g), 0.4 mol O , and The gases gases leaving the burner contain 1 mol CO 2 2 2 2 2 2 y 9.03 moles de N . Dado que el cambio de entalpía debe ser independiente de la trayectoria, 9.03 mol N . Because the enthalpy change must be independent of path, 2 2..N 9.03 mol NN Because the enthalpy change must be independent of path, 9.03 mol N the change must be of 9.03 9.03 molmol .Because Because theenthalpy the enthalpy enthalpy change change must must beindependent be independent independent ofpath, path, of path, 2 . Because 9.03 the must 9.03 mol mol N N22222.. Because Because the enthalpy enthalpy change change must be be independent independent of of path, path, ◦ ◦ ◦ ◦◦ = ◦ �H ◦◦◦ + ◦+ �H + �H = �H = (A) �H �H = (A) �H = (A) 298 �H�H +�H �H + �H = �H = �H =00000= 0 (A)(A) ◦◦298298 ◦P= PP◦P 298 P�H �H + �H = �H = (A) �H298 (A) 298 298 + �HPP = �H = 0 where all enthalpies are on the basis of 1 mol CH burned. The enthalpy change 44burned. where all enthalpies are on the basis of mol CH burned. The enthalpy change where all enthalpies of CH The enthalpy change where where all all enthalpies enthalpies areon are onthe on thebasis the basis basis of111of 1mol mol 1 mol CH burned. burned. TheThe enthalpy enthalpy change 44CH 4quemado. donde todas las entalpías se are toman con base en CH cambio enchange la entalpía where all enthalpies are on the of mol CH The enthalpy change where all enthalpies areare onheated the basis basis of 1298.15 1mol molde CH burned. The El enthalpy change 44 burned. of the products as they from K to T4Ttois: is: of the products as they are heated from 298.15 K to T is: of the products as they are heated from 298.15 K to T of the of the products products as they as they are are heated heated from from 298.15 298.15 K to K is: T is: de los productos a medida que son calentados de 298.15 K a T es: of the products as they are heated from 298.15 K to T is: of the products as they are heated from 298.15 K to T is: ◦◦� ◦(T − 298.15) ◦◦◦◦ = ◦�C �H �C (B) �H �C (T − 298.15) (B) �H == ��H (B) �H �H = �C =◦◦P�C �H(T (T �− (T −298.15) 298.15) − 298.15) (B)(B) ◦P ◦ P P P P �H (B) �HPP = = �C �CPPPP◦◦P�H �HHHP(T (TH − − 298.15) 298.15) (B) ◦ ◦ ◦ ◦ ◦ where we define �C � as the mean heat capacity for the total product stream: where we define �C as the mean heat capacity for the total product stream: where we �C ��H heat capacity for product stream: where where wedefine we define define �CP�C �HHPas �as the as mean the mean mean heat heat capacity capacity forthe for thetotal the total total product product stream: stream: Hthe o where we as the heat the product stream: where we define define �C �CPPPP◦◦P�H �la as the mean mean heat capacity capacity for thelatotal total product stream: H donde ⟨C calorífica mediafor para corriente total de productos: H capacidad P⟩ H se define como ◦◦� ◦ ≡ ◦◦ � ◦ �C �C �C ≡ �C �C ��H ��H �C◦◦P�C �H ≡ �≡ ≡ nnnni�C �C n ◦◦P�C �H � �C �CPPPP◦◦P�H �HHHP≡ ≡H ii nniiiii�C �CPiPPP◦i◦Piiiii�H �HHHPi H (A) (B) � � �� � � ii i ii i The simplest procedure here is to sum the mean-heat-capacity equations for En esteThe caso, el simplest procedimiento más simple es lasmean-heat-capacity ecuaciones de laequations capacidad calorífica The simplest procedure here to sum the mean-heat-capacity equations for simplest procedure here isis the for TheThe simplest procedure procedure here here isto to issum sum to sumar sum themean-heat-capacity the mean-heat-capacity equations equations for for The procedure here is equations for The simplest simplest procedure here is to to sum sum the the mean-heat-capacity mean-heat-capacity equations for the products, each multiplied by its appropriate mole number. Because C = 0 for media para los productos, cada una multiplicada por el número denumber. moles apropiado. Puesto que C the products, each multiplied by its appropriate mole number. Because == for the products, each multiplied by its appropriate mole number. Because CCC= 000= for the the products, products, each each multiplied multiplied by by its its appropriate appropriate mole mole number. Because Because C for 0 for the products, each multiplied by its appropriate mole the products, each(Table multiplied by its(4.8) appropriate mole number. number. Because Because C C= = 00 for for each product gas C.1), Eq. yields: = 0 paraeach cada producto gaseoso (tabla C.1), la(4.8) ecuación (4.8) produce: each product gas (Table C.1), Eq. (4.8) yields: product gas (Table C.1), Eq. (4.8) yields: each each product product gas gas (Table (Table C.1), C.1), Eq. Eq. (4.8) yields: yields: each (4.8) each product product gas gas (Table (Table C.1), C.1), Eq. Eq. (4.8) yields: yields: � � ���� � ���� � � � � � �� �� � � � n B n D � � � � i i i i i i n B n D nii B Bi(τ + 1) + ii innnii D D nii i � Di ini ii T iBB iD ii inn ◦◦◦�◦�� ◦= ◦◦◦◦��� ◦= = n �C = R n A + �C i i i 0 n B n D i i i i T = n �C = R n A + (τ + 1) + �C T n �C R n A + (τ + 1) + �C H H P P i i i i T T � � = = n �C n �C � � = = R R n A n A + + (τ (τ + 1) + + 1) + �C �C i i i 0 i i �C �HHHHP= =H ii nniiii�C �CPiPPP◦i◦Piiiii�H �HHHHPi = =H R R ii nniiiiA Aiiii + +i i2222 2 TT0000(τ (τ0+ + 1) 1) + + τττiτTTTT2022τ222T 2 �CPPPP◦◦P�H i i i i i i 22 ττ TT00000 0 ii ii Data from Table C.1 are combined as follows: Data from Table C.1 are combined as follows: Data from Table C.1 are combined as Data Data from from Table Table C.1 C.1 are are combined combined asfollows: follows: as siguiente follows: manera: La información de la tabla C.1 se combina de la Data Data from from Table Table C.1 C.1 are are combined combined as as follows: follows: = = (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) = 43.471 = = (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) = 43.471 AAAA= = = A = nnnniAAAAniiii= A=(1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) = (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) =43.471 43.471 = 43.471 A A= = ii nniiiiiA Aii = =i (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) (1)(5.457)+(2)(3.470)+(0.4)(3.639)+(9.03)(3.280) = = 43.471 43.471 � � �� � � � � �� � � � � �� � ii i ii i −3 5. 5 −3 −3 −3 = 9.502 × 10 and D = D = −0.645 × 10 Similarly, = = 9.502 × 10 and D = D = −0.645 × 10 .. . Similarly, = De la misma manera, × D × Similarly, BBBB= B =9.502 9.502 = 9.502 ×10 10 ×−3 10and andyand D= = D = nnnniD D ni = D =−0.645 −0.645 = −0.645 ×10 10 ×55.510 Similarly, Similarly, = B = nnnniBBBBiniiii= −3 and Similarly, Bii = =i 9.502 9.502 × × 10 10−3 and D D= = iiii nniiiiiiD Diiiii = =i −0.645 −0.645 × × 10 1055.. Similarly, B B= = iiii nniiiiiiB ii ii o ◦◦◦�◦�� /R /R is therefore represented by: For the product stream �C Por lo tanto, para laproduct corriente de productos ⟨C ⟩ H / Rrepresented se representa por: is therefore represented by: For the product stream �C by: For the product stream �C P�C �H◦/R /R �His /R istherefore therefore isPtherefore represented represented by: by: ForFor the the product stream stream �C is represented For �HHHHP/R /R is therefore therefore represented by: by: For the the product product stream stream �C �CPPPP◦◦P�H MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) MCPH(298.15,T;43.471,9.502E-3,0.0,-0.645E+5) Equations (A) and (B) are combined and solved for Al combinarse las ecuaciones (A) yare (B) y resolviendo para T, seTfor Equations (A) and (B) combined and solved for TTobtiene: Equations (A) and (B) are combined and solved for Equations Equations (A)(A) andand (B)(B) are are combined combined andand solved solved for T:::: T : Equations Equations (A) (A) and and (B) (B) are are combined combined and and solved solved for for TT :: ◦ ◦◦◦ ◦ �H �H �H 298 �H�H ◦◦298298 298 298 = 298.15 − �H �H = 298.15 − TTTT= 298.15 − ◦ = T 298.15 = 298.15 − − 298 298 ◦ ◦ ◦ �C TT = = 298.15 298.15 − − �C �C ����HHHP◦ �H �CP�C �C �CPPPP◦◦P�H �H o Ya que las capacidades caloríficas medias dependen deTT,, H primero se evalúa ⟩ H an para un valor ◦◦◦�◦��⟨C P ◦for Because the mean heat capacities depend on first evaluate �C for asBecause the mean heat capacities depend on first evaluate �C for an asBecause the mean heat capacities depend on TT ,, ,first evaluate �C H P Because Because the the mean mean heat heat capacities capacities depend depend on on T T first , first evaluate evaluate �C �C � � for for anasan as-as◦ ◦ H P PP�H HPnos H an the mean heat capacities depend on T , first evaluate �C for an asBecause the mean heat capacities depend on T , first evaluate �C � for an assupuestoBecause de T > 298.15, y se sustituye el resultado en la ecuación anterior. Esto dará un nuevo PP H sumed value of Tof > 298.15, and substitute the result in the preceding equation. H equation. sumed value of > 298.15, and substitute the result in the preceding equation. sumed value of and substitute the result in sumed sumed value value ofTT T> > T 298.15, > 298.15, 298.15, andand substitute substitute the the result result inthe the in preceding the preceding preceding equation. equation. o> value of T 298.15, and substitute the result in the preceding equation. ◦ sumed value of T > 298.15, and substitute the result in the preceding equation. valor desumed T para el cual ⟨C ⟩ se evalúa nuevamente. El procedimiento continúa hasta lograr el H Pvalue ◦��H◦is This yields new value of T for which �C is reevaluated. The procedure This yields new value of for which �C reevaluated. The procedure This yields aaaanew of �C The procedure This This yields yields new a new value value ofTT of T for for T which for which which �CP◦P◦P◦◦�C �is is reevaluated. is reevaluated. TheThe procedure procedure HHP is Hreevaluated. P���H yields a new value of T for which �C reevaluated. The procedure This yields a new value of T for which �C � is reevaluated. The procedure límite enThis el valor final, PP H continues to convergence on the final value, H continues to convergence on the final value, continues to on value, continues continues toconvergence convergence to convergence onthe on thefinal the final final value, value, continues continues to to convergence convergence on on the the final final value, value, ◦◦C ◦ T ==2 2,066 066 KK oor 11,793 793 ◦◦°C == 2,066 KK K or or 1,793 TTTT= K CCC C T2,066 2,066 = 2,066 or or 1,793 1,793 1,793 TT = or 1,793 = 2,066 2,066 K K or 1,793◦◦C C � � �� � 04-SmithVanNess.indd 144 � � �� � 8/1/07 13:08:48 4.7. 4.7. Heat Heat Effects Effects of of Industrial Industrial Reactions Reactions 4.7. Heat Effects of Industrial Reactions 4.7. Efectos térmicos de las reacciones industriales 4.7. Heat Effects of Industrial Reactions 145 145 145 145 145 Example Ejemplo 4.8 4.8 Example 4.8 Example 4.8 One method for the “synthesis gas” of H Un método la fabricación del “gas deof (principalmente una mezcla de CO H 2the ) es la reOne para method for the manufacture manufacture ofsíntesis” “synthesis gas” (a (a mixture mixture of CO CO and and H22))y is is the Example 4.8 One method for the manufacture of “synthesis gas” (a mixture CO and H2pressure: ) is the catalytic reforming of CH with steam at high temperature andof atmospheric formación catalítica del CHof temperatura y a presión atmosférica:pressure: catalytic reforming CH44vapor with asteam at high elevada temperature and atmospheric 4 con One method for of theCH manufacture ofat“synthesis gas” (a mixture of CO and H2 ) is the catalytic reforming high temperature and atmospheric pressure: 4 with steam CH (g) + H O(g) → CO(g) + 3H (g) catalytic reforming of CH with steam at high temperature and atmospheric pressure: 4 (g) 2 (g) 4CH (g) + H2 O(g) CO(g) + 3H2(g) CH 4 4 + H22O(g) → CO(g) + 3H 2 CH4 (g) + H2 O(g) → CO(g) + 3H2 (g) The only reaction to be is the water-gas-shift (g)considered + Huna + 3H2 (g) La única reacción adicional queCH ocurre con extensión apreciable es reaction: la reacción de desplazamiento 4be 2 O(g) The only other other reaction to considered is→ theCO(g) water-gas-shift reaction: The other reaction to be considered is the water-gas-shift reaction: de gas deonly agua: + → CO + H2 (g) reaction: The only other reaction CO(g) to be considered water-gas-shift 2 (g) CO(g) +H H22O(g) O(g) is →the CO 2 (g) + H2 (g) CO(g) H2O(g) CO +H H22(g) (g) CO(g) + H+2 O(g) →→CO 2(g)+ 2 (g) IfIf the reactants are supplied in the ratio, 2 mol steam to 11 mol ifif heat CO(g) + H O(g) → CO (g) + H 4 ,, and 2steam to2 (g) the reactants are supplied in the2 ratio, 2 molde mol CH CH heat is is 4CHand Si losIf reactivos se suministran en proporción de 22 moles vapor por 1 mol de si se proporciona 4y the reactants are supplied in the ratio, mol steam to 1 mol CH , and if heat is 4 supplied to the reactor so that the products reach a temperature of 1,300 K, the CH 4 supplied to the reactor so that the products reach a temperature of 1,300 K, the CH 4 calorsupplied alis que productos alcancen 1 300 K de to temperatura, CH convierte por 4Assuming Ifreactor, the reactants are supplied in the ratio, reach 2 mol steam 1 mol CH4el ,CO. and ifseheat to de themodo reactor solosthat temperature 1,300 K, the CH converted and the product 17.4 mol-% 4 is is completely completely converted andthe theproducts product stream streamacontains contains 17.4 of mol-% CO. Assuming completo yreactants la salida de productos contiene 17.4% mol-% de Suponiendo que los supplied to converted the reactor so that the products reach a CO. temperature of 1,300 K, reactivos the CH4 se cais the completely and the product stream contains 17.4 mol-% CO. Assuming to be preheated to 600 K, calculate the heat requirement for the reactor. the reactants to be preheated to 600 K, calculate the heat requirement for the reactor. lentaron previamente a 600 K, calcule el requerimiento de calor el reactor. completely converted andtothe product stream 17.4 mol-% theisreactants to be preheated 600 K, calculate thecontains heat para requirement forCO. the Assuming reactor. the reactants to be preheated to 600 K, calculate the heat requirement for the reactor. Solution 4.8 Solución 4.8 Solution 4.8 Solution 4.8 ◦ las Los calores estándar reacción a 25 °C 25 para dos reacciones se calculan a partir de la inforThe heats Solution 4.8de The standard standard heats of of reaction reaction at at ◦25◦C C for for the the two two reactions reactions are are calculated calculated from from mación de la tabla C.4: The standard heats of reaction at 25 C for the two reactions are calculated from the the data data of of Table Table C.4: C.4: ◦ theThe datastandard of Tableheats C.4: of reaction at 25 C for the two reactions are calculated from ◦ = 205,813 J (g) H �H the dataCH of Table ◦ 298 CH44 (g) + +C.4: H22O(g) O(g) → → CO(g) CO(g) + + 3H 3H22(g) (g) �H 298 = 205,813 J ◦ CH4 (g) + H2 O(g) → CO(g) + 3H2 (g) �H298 = 205,813 J ◦◦ = −41,166 CH H O(g) → CO(g) + 3H 205,813 JJ �H CO(g) ◦ 4 (g) + 298 298 CO22(g) (g) + +H H22(g) (g) �H CO(g) + H22 O(g) → CO 298 = −41,166 J ◦ �H298 = −41,166 J CO(g) + H2 O(g) → CO2 (g) + H2 (g) These two reactions be added give aaHthird reaction: ◦ O(g) COato (g) −41,166 J CO(g) + Hmay These two reactions be→ added to give third 2may 2fin 2 (g) reaction: Estas dos reacciones pueden ser sumadas de+obtener una�H tercera= reacción: These two reactions may be added to give a third reaction: 298 ◦ + 2H2may O(g)be→ COto +a4H �H TheseCH two4 (g) reactions added give third ◦ = 2 (g) 2 (g)reaction: 298 �H = 164,647 164,647 JJ CH 4 (g) + 2H2 O(g) → CO2 (g) + 4H2 (g) ◦ 298 �H298 = 164,647 J CH4 (g) + 2H2 O(g) → CO2 (g) + 4H2 (g) ◦ The Any the reactions constitutes an third reaction (g) +three 2H2 O(g) → CO2 (g)un+conjunto 4Hindependent �Hset. 164,647 J reacción no CH 4las 2 (g) independiente. Cualquier parpair de of tres reacciones constituye La tercera 298 = Any pair of the three reactions constitutes an independent set. The third reaction Any pair of the three reactions constitutes an independent set. The third reaction is not independent; it is obtained by combination of the other two. The reactions es independiente, debido a it que se obtienebydecombination la combinación las otras este caso, las is not independent; is obtained of thedeother two. dos. The En reactions Anymás pair of the three reactions constitutes an independent third reaction is most not independent; it work is obtained by combination of the otherset. two.The The reactions convenient to here are third: reacciones convenientes parawith trabajar son: most convenient to work with here are the the first first and and third: is not independent; it iswith obtained by the combination of the other two. The reactions most convenient to work here are first and third: ◦ = CH → CO(g) + �H 205,813 J (A) ◦ 2 O(g) 2 (g) 298 CH44(g) (g) + + Hwork →here CO(g) + 3H 3H �H (A) most convenient toH with are the first and third: 2 O(g) 2 (g) 298 = 205,813 J ◦ CH4 (g) + H2 O(g) → CO(g) + 3H2 (g) �H298◦ = 205,813 J (A) ◦ CH (g) + 2H O(g) → CO (g) + 4H (g) �H = 164,647 J (B) ◦ = 205,813 4 (g) CH H222O(g) → CO(g) �H298 CH CO22 (g)++3H 4H22(g) 164,647 J (A) (B) 4 4 (g) ++2H 2 (g) 298 ◦ 298 �H298 = 164,647 J (B) CH4 (g) + 2H2 O(g) → CO2 (g) + 4H2 (g) First determine the fraction of CH converted by each of these reactions. As ◦ 4 First determine of CO CH24(g) converted by each of298 these reactions. CH4 (g) +the 2H2fraction O(g) → + 4H2 (g) �H = 164,647 J As (B)aa First determine the fraction of CH converted by each of these reactions. As basis for calculations, let 1 mol CH and 2 mol steam be fed to the reactor. If 4 4 basis for calculations, let 1 mol CH4 and por 2 mol steam be fed toreacciones. the reactor.aComo If base Primero determine la fracción de CH convertido cada una de estas 4CH determine thebyfraction ofthen CH converted by each of these reactions. As a basis for calculations, let 1 mol 2 mol steam be fed to the reactor. If xFirst mol CH reacts Eq. (A), 1 − x mol reacts by Eq. (B). On this basis the 4 and 4 4 x mol CH reacts by Eq. (A), then 1 − x mol reacts by Eq. (B). On this basis the 4 para los cálculos sethe supondrá que al reactor se le alimenta con 1 be mol de CH 2 moles de vapor. 4 ybasis basis for4 of calculations, let 1then mol CH 2reacts mol by steam fed to the reactor. If x mol CH reacts by Eq. (A), 1 − mol Eq. (B). On this the products reaction are: 4x and products the reactionpor are:la ecuación (A), entonces 1 – x mol reacciona en la ecuación Si x moles de CH CHof reaccionan x mol by Eq. then 1 − x mol reacts by Eq. (B). On this basis the products of the reaction are:(A), 44 reacts (B). Sobre esta base losreaction productos de la reacción son: CO: xx products of the are: CO: CO: x : 3x + 4(1 H 2 3x + 4(1 − − x) x) = = 44 − − xx H2 : CO: :CO2 :: 3x1x1+− 4(1 − x) = 4 − x H2CO x −x :2 1 − 3xx +x 4(1 − x) x) = 4 −x x COH O: 22 − − x− − 2(1 2(1 − − x) = =x H222:O: 1− x 2(1 − x) = x O: 2 : 2 − x− H2CO Total: 525 mol − x products − 2(1 − x) = x H 2 O: Total: mol products Total: 5 mol products productos mol Total: 5 mol products 04-SmithVanNess.indd 145 8/1/07 13:08:56 CHAPTER 4. Heat Effects CAPÍTULO 4. Efectos CHAPTER Effects CHAPTER4.4. Heat Heat Effectstérmicos 146 146 146 146 La fracción mol fraction de CO en corriente de productos dondex xx== Themole mole fraction ofla CO in theproduct product streames isx/5 x/5= =0.174, 0.174;de whence = 0.870. 0.870. Así, de The ofof CO stream isis x/5 whence The mole fraction COininthe the product streamde x/5=en =0.174; 0.174; whence x y=0.870. 0.870.moles en acuerdo con la base elegida, reaccionan 0.870 moles CH la ecuación (A) 0.130 Thus,ononthe the basischosen, chosen,0.870 0.870 molCH CH4reacts reactsbybyEq. and 0.130mol mol reacts 4Eq. ( A)and Thus, (A) Thus, onlado, thebasis basis chosen,de 0.870mol mol CH444en reacts by Eq.( (A) A)productos and0.130 0.130es: molreacts reacts la (B). by Por otro la cantidad especie laspecies corriente de byEq. Eq. (B).Furthermore, Furthermore, thecada amounts ofthe thespecies inthe the productstream streamare: are: (B). the amounts of in product by Eq. (B). Furthermore, the amounts of the species in the product stream are: Moles CO==x x= =0.87 0.87 Moles de CO Moles MolesCO CO==xx==0.87 0.87 = = 3.13 Moles H Moles de H 44–4−x−x=x=3.13 22 2= = Moles H 3.13 Moles H22 = 4 − x =3.13 Moles CO = 1 − x = 0.13 Moles de CO22 2==11–−x x= =0.13 Moles MolesCO CO22 = 1 − x =0.13 0.13 Moles 0.87 Moles de HH = =x x= =0.87 2O 2O Moles 2O==xx==0.87 MolesHH22O 0.87 We now a path,for for purposes calculation, to objeto proceed at a 600 Ahora,We para finesdevise de cálculo, diseñamos unaoftrayectoria con defrom ir de reactants los reactivos Wenow nowdevise deviseaapath, path, forpurposes purposesofofcalculation, calculation,totoproceed proceedfrom fromreactants reactantsatat 600 K to products at 1,300 K. Because data are available for the standard heats of K a los600 productos a 1 300 K. Debido a que se dispone de la información del calor estándar de atat1,300 600KKtotoproducts products 1,300K. K.Because Becausedata dataare areavailable availablefor forthe thestandard standardheats heatsofof ◦ ◦ C, reaction at25 themost most convenient path theque oneincluya whichincludes includes thereactions reactions ◦ C,the reacción a 25 °C, la25 trayectoria más conveniente esisis una las reacciones a 25 °C (298.15 reaction at convenient path the one which the ◦ reaction the most convenient path is the one which includes the reactions ◦ at 25 C, K). ◦muestra atse 25 This is shownschematically in theaccompanying accompanying diagram. ◦ C (298.15 K). Esto de forma esquemática enschematically el siguiente diagrama. La línea punteada representa atat 25 25◦CC(298.15 (298.15K). K).This Thisisisshown shown schematicallyininthe the accompanyingdiagram. diagram. The dashed line represents the actual path for which the enthalpy change �H . la trayectoria real para la cual el cambio de entalpía es ∆H. the Dado que el cambio enis�H la entalpía es The dashed line represents the actual path for which enthalpy change is The dashed line represents the actual path for which the enthalpy change is �H. . Because this enthalpy change is independent of path, independiente de laenthalpy trayectoria: Because this change is independent of path, Because this enthalpy change is independent of path, ◦ �H== =�H �H◦◦R◦◦++ + �H �H◦298 �H◦◦P◦◦ �H ◦◦ ++�H �H �H RR �H PP 298 298 + �H R P 298 H �H �H �H �H HPP �H� �H� � PP �H �H� P Reactivos at a 11bar Reactants bar Reactants Reactantsatat at111bar bar Reactants y 600 600 KK bar and 600K and and 600 K and 600 1mol mol CHK4 111 44 molCH CH mol CH 4 2 mol HO O 222mol 2 22O molHH H O mol 2 Productos a 1 bar Products at Products Productsatat at111bar bar Products y 1 300 KKbar and 1,300 and and1,300 1,300KK K and 1,300 0.87 mol CO 0.87 0.87mol molCO CO 0.87 mol 3.13mol mol CO H2 3.13 22 3.13 molHH H 3.13 mol 2 2 0.13 mol CO 0.13 mol CO 22 0.13 mol mol CO CO 0.13 2 0.87 mol H O 2O 0.87 2O 0.87mol molHH H 0.87 mol 22O HRR �H� �H� RR �H� �H� R � H �H � 298 �H 298 �H �� �H 298 298 ◦ Forthe thecalculation calculationofof of�H �H◦298 reactions( A) A)and and(B) (B)must mustboth bothbebe betaken takeninto into For ◦◦ , ,,reactions the calculation �H reactions (( A) and (B) must both taken into 298 Paraaccount. losFor cálculos de ∆H° es necesario tomar en cuenta las reacciones (A) y (B). Dado que 298 298mol CH 298 reacts by (A) and 0.13 mol reacts by (B), Because 0.87 by and 0.13 reacts by (B), account. Because 0.87 4 44reacts account.(A) Because 0.87mol molCH CH by4(A) (A) andmoles 0.13mol mol en la ecuación reaccionan 0.87 moles de CH y 0.13 en lareacts (B), by (B), 4 reacts ◦ �H◦298 (0.87)(205,813)++(0.13)(164,647) (0.13)(164,647)==200,460 200,460J J �H ◦◦ ==(0.87)(205,813) �H298 298 = (0.87)(205,813) + (0.13)(164,647) = 200,460 J 298 Theenthalpy enthalpychange changeofofthe the reactantsfor for coolingfrom from600 600 Ktoto298.15 298.15KKis:is: The El cambio entalpíachange de los of reactivos al ser enfriados 600 600 K aK298.15 K es:K is: Thede enthalpy thereactants reactants forcooling coolingdefrom K to 298.15 � � �� �� ◦ (298.15−− −600) 600) = �H◦◦R◦◦== ◦� i �C◦P nnni �C �H (298.15 600) �H RR Pi◦�iH�H (298.15 i �C R � � � i ii i i H Pii H P ◦◦ Pi H /Rare: are: where the values �C oof�C /R the ◦◦� ��son: H P donde where los valores de ⟨Cof ⟩ �C /PiR where thevalues values Pof H /R are: PiiH i H CH : MCPH(298.15,600;1.702,9.081E-3,-2.164E-6,0.0) MCPH(298.15,600;1.702,9.081E-3,-2.164E-6,0.0)== =5.3272 5.3272 CH 4 :44: MCPH(298.15,600;1.702,9.081E-3,-2.164E-6,0.0) CH 5.3272 4 H O: MCPH(298.15,600;3.470,1.450E-3,0.0,0.121E+5) = 4.1888 2 HH2 O: MCPH(298.15,600;3.470,1.450E-3,0.0,0.121E+5) MCPH(298.15,600;3.470,1.450E-3,0.0,0.121E+5)==4.1888 4.1888 22 O: 04-SmithVanNess.indd 146 8/1/07 13:09:04 4.7. Heat Heat Effects Effects of Industrial Industrial Reactions Reactions 4.7. 4.7. Efectos térmicos de of las reacciones industriales 147 147 147 Whence, Whence, De donde, �H◦R◦◦R◦ = = (8.314)[(1)(5.3272) (8.314)[(1)(5.3272) + + (2)(4.1888)](298.15 (2)(4.1888)](298.15 − − 600) 600) = = −34,390 −34,390 JJ �H RR El cambio entalpíachange de los of productos a medida queare son calentados de 298.15 a 1 300 Thede enthalpy the products products as they they heated from 298.15 298.15 to 1,300 1,300 K is isK se calThe enthalpy change of the as are heated from to K cula de calculated manera similar: similarly: calculated similarly: � � � � �H◦P◦◦P◦ = = �H PP �C ◦P◦P◦◦��HH �nniiii�C � PP H H iiii i iii (1,300 − − 298.15) 298.15) (1,300 /Rovalues are: are: where �C �C ◦◦◦◦��HH/R where iH donde los valoresPPPPiiide ⟩ / R son: H ⟨CPvalues i H CO: CO: H2222:: H CO2222:: CO H2222O: O: H MCPH(298.15,1300;3.376,0.557E-3,0.0,-0.031E+5) == 3.8131 3.8131 MCPH(298.15,1300;3.376,0.557E-3,0.0,-0.031E+5) MCPH(298.15,1300;3.249,0.422E-3,0.0,0.083E+5) == 3.6076 3.6076 MCPH(298.15,1300;3.249,0.422E-3,0.0,0.083E+5) MCPH(298.15,1300;5.457,1.045E-3,0.0,-1.157E+5) == 5.9935 5.9935 MCPH(298.15,1300;5.457,1.045E-3,0.0,-1.157E+5) MCPH(298.15,1300;3.470,1.450E-3,0.0,0.121E+5) == 4.6599 4.6599 MCPH(298.15,1300;3.470,1.450E-3,0.0,0.121E+5) Whence, Whence, De donde, �H◦P◦◦P◦ = = (8.314)[(0.87)(3.8131) (8.314)[(0.87)(3.8131) + + (3.13)(3.6076) (3.13)(3.6076) �H PP + (0.13)(5.9935) + (0.87)(4.6599)] × (1,300 (1,300 − − 298.15) 298.15) + (0.13)(5.9935) + (0.87)(4.6599)] × = 161,940 161,940 JJ = Therefore, Therefore, Por lo tanto, �H ==−34,390 −34,390 200,460 +161 161,940 =328 328,010 ∆H= –34 390 + ++200 460 ++ 940 == 010 J JJ �H 200,460 161,940 328,010 2 se 22suponen insignificantes. El proceso del tipo de estableflow paraforelwhich cual WW y ∆u 22 /2 are presumed s,ss,∆z Thees process is one oneflujo of steady steady , �z, and/2�u The process is of flow for which W ss �z, and �u /2 are presumed Así, negligible. Thus, negligible. Thus, QQ= ==�H �H =328 328,010 Q 328,010 ∆H == 010 JJJ This result result isbasado on the theen basis of mol CH CH4444 fed fed1to tomol thede reactor. Este resultado estáis unaof alimentación de CH4 al reactor. This on basis 11 mol the reactor. Example 4.9 Ejemplo 4.9 4.9 Example A boiler boiler is is fired fired with with aa high-grade high-grade fuel fuel oil oil (consisting (consisting only only of of hydrocarbons) hydrocarbons) having having aa A −1 Una caldera se enciende con aceite combustible gran poder calorífico sólo as en hidro−1 at −1 standard heat of of combustion combustion of −43,515 −43,515de 25◦◦◦◦C C with CO CO(que (g) consiste and H O(l) standard heat of JJ gg−1 at 25 2222(g) and H2222O(l) as –1with carburos); tiene un calor estándar de combustión de –43 515 J g a 25 °C con CO (g) y H O(l) 2 2 products. The The temperature temperature of of the the fuel fuel and and air air entering entering the the combustion combustion chamber chamber is is como products. ◦ ◦ los productos. La temperatura del combustible y del aire que entran a la cámara de combustión ◦ ◦ ◦ ◦ ◦ ◦ 25 C. C. The The air air is is assumed assumed dry. dry. The The flue flue gases gases leave leave at at 300 300 C, C, and and their their average average es de 25 25 °C. Se supone que el aire es seco. Los gases de combustión salen a 300 °C y su análisis promedio analysis (on (on aa dry dry basis) basis) is is 11.2% 11.2% CO CO2222,, 0.4% 0.4% CO, CO, 6.2% 6.2% O O2222,, and and 82.2% 82.2% N N2222.. Calculate Calculate analysis (sobre una base seca) es de 11.2% de CO , 0.4% de CO, 6.2% de O y 82.2% de N . Calcule la 2 2 2 the fraction fraction of of the the heat heat of of combustion combustion of of the the oil oil that that is is transferred transferred as as heat heat to to the the boiler. boiler.fracción the del calor de combustión del aceite que se transfiere como calor a la caldera. 04-SmithVanNess.indd 147 8/1/07 13:09:11 148 CHAPTER 4. Heat Effects CHAPTER 4. 4. Heat Effects CAPÍTULO Efectos térmicos 148 148 Solución 4.9 Solution 4.9 Se toman como base Solution 4.9100 moles de los gases de combustión secos, consistentes en: Take as a basis 100 mol dry flue gases, consisting of: 11.2 moles Take as a basis 100 mol dry CO flue2 gases, consisting of: COCO2 0.4mol moles 11.2 11.2 mol CO O2 CO 2 6.2mol moles 0.4 CO 0.4 mol N2 O2 82.2 moles 6.2 mol O2 6.2 mol Total 100.0 moles N2 82.2 mol 82.2 mol N2 Totalel vapor 100.0Hmol Este análisis, sobre base seca, no considera O presente en los gases de combustión. La 2mol Total 100.0 por la reacción de combustión se origina de O unvapor balance de oxígeno. El cantidadThis de Hanalysis, 2O formada on a dry basis, does not take into account the H present in This analysis, on a dry basis, does not takeeninto account the H22 Ode vapor present inrestante O2 proporcionado por el aire representa 21mol-% mol de la corriente aire. El 79% the flue gases. The amount of H2 O formed by the combustion reaction is found the flue gases. The of H2 O formed by the combustion Así, reaction is found es N2, elfrom cual permanece sinamount cambio el proceso por cada 100 moles an oxygen balance. Thedurante O2 supplied in thedeaircombustión. represents 21 mol-% of the from an oxygen balance. The O supplied in the air represents 21 mol-% of the por el 2 de gasesairsecos de combustión se presentan 82.2 moles de N , los cuales son suministrados 2 stream. The remaining 79% is N2 , which goes through the combustion process air The remaining 79% is N2 , which goes through the combustion process aire, y elunchanged. O2stream. que acompaña 2 es: N appearing in 100 mol dry flue gases is supplied Thus theeste 82.2Nmol unchanged. Thus the 82.2 mol N22 appearing in 100 mol dry flue gases is supplied with the air,Moles and thedeOO2 2accompanying N2=is:(82.2)(21/79) = 21.85 que entran en this el aire with the air, and the O2 accompanying this N2 is: Moles O entering in air = (82.2)(21/79) = 21.85 Sin embargo, Moles O22 entering in air = (82.2)(21/79) = 21.85 However, Moles de O2 en los gases secos de combustión = 11.2 + 0.4/2 + 6.2 = 17.60 However, O2 ines theeldry flue gases = 11.2 + 20.4/2 + 6.2 = 17.60 La diferencia entreMoles estas cifras número moles de O que reaccionan para formar H2O. Por Moles O2 in the dry flue de gases = 11.2 + 0.4/2 + 6.2 = 17.60 lo tanto,The sobre la base de 100 moles de gases de combustión secos, difference between these figures is the moles of O that react to form H O. The difference between these figures is the moles of O22 that react to form H22 O. Therefore on the basisdeofH100 mol dry flue gases,– 17.60)(2) = 8.50 Moles = (21.85 Therefore on the basis of 100 mol dry flue gases, 2O formadas formed = (21.85 − de 17.60)(2) = 8.50 = 8.50 Moles deMoles H2 enH el2 O combustible = moles agua formadas Moles H O formed = (21.85 − 17.60)(2) = 8.50 2 H2 in the fuel = moles water formed = 8.50 La cantidad de C en elMoles combustible está dada por unof de carbono: Moles H2 in the fuel = moles ofbalance water formed = 8.50 The amount of C in the fuel is given by a carbon balance: of C indethe fuel is given by a carbon balance: MolesThe de Camount en los gases combustión = moles de C en el combustible = 11.2 + 0.4 = 11.60 Moles C in flue gases = moles C in fuel = 11.2 + 0.4 = 11.60 Moles C in flue gases = moles C in fuel = 11.2 + 0.4 = 11.60 Estas cantidades de C y H2 juntas dan: These amounts of C and H2 together give: These amounts of C and H2 together give: MasaMass de combustible quemado = (8.50)(2) + (11.6)(12) = 156.2 g of fuel burned = (8.50)(2) + (11.6)(12) = 156.2 g Mass of fuel burned = (8.50)(2) + (11.6)(12) = 156.2 g H2O(l) Si esta cantidad de combustible es quemada por completo a CO 2(g) If this amount of fuel is burned completely to CO (g) and H yO(l) at 25a ◦25 C, °C, the el calor If this amount of fuel is burned completely to CO22 (g) and H22 O(l) at 25◦ C, the de combustión es: heat of combustion is: heat of combustion is: ◦ = (−43,515)(156.2) = −6,797,040 J �H298 ◦ = (−43,515)(156.2) = −6,797,040 J �H298 However, the reaction actually occurring not represent complete combustion, Sin embargo, la reacción que en realidad ocurre does no representa la combustión completa, y el H2O However, the reaction actually occurring does not represent complete combustion, and the H O is formed as vapor rather than as liquid. The 156.2 g of fuel,que consist2 se formaand másthe bien como vapor as que como líquido. 156.2 gThe de 156.2 combustible, están formaH2 O is formed vapor rather thanLos as liquid. g of fuel, consist, is represented by the empirical formula ing of 11.6 mol of C and 8.5 mol of H 2 dos por ing 11.6ofmoles y 8.5 moles , se por labyfórmula empírica C11.6H17. Al , is represented the empirical formula 11.6 de molCof C and 8.5 de molH2of H2representan C11.6 the 6.2 mol O2de andN282.2 mol N2 entran which enter anddel leave the reactor 17 .. Omit omitir las 6.2H de O y las 82.2 , las cuales y salen reactor sin cambio, la 2 C11.6 Hmoles Omit the 6.2 mol O and 82.2 mol N which enter and leave the reactor 17 2 2 write the reaction: reacciónunchanged, se escribe:and unchanged, and write the reaction: C11.6 H17 (l) + 15.65O2 (g) → 11.2CO2 (g) + 0.4CO(g) + 8.5H2 O(g) C11.6 H17 (l) + 15.65O2 (g) → 11.2CO2 (g) + 0.4CO(g) + 8.5H2 O(g) This result is obtained by addition of the following reactions, for each of which This result is obtained by addition the following each of which ◦ Cof Este resultado se obtiene agregando las25 siguientes reacciones,reactions, para cadafor una de las cuales se cothe standard heat of reaction at known: ◦ C is the standard heat of reaction at 25 is known: noce el calor de reacción estándar a 25 °C: 04-SmithVanNess.indd 148 8/1/07 13:09:14 4.7. Heat Heat Effects of Industrial Industrial Reactions 4.7. Efectos térmicos de las reacciones industriales 4.7. Effects of Reactions 4.7. Heat Effects of Industrial Reactions 149 149 149 149 149 C H (l) ++ 15.85O 15.85O (g) → 11.6CO (g) ++ 8.5H 8.5H O(l) 11.6 17 C C11.6 H17 (l) + 15.85O22 (g) (g) → → 11.6CO 11.6CO22 (g) (g) + 8.5H22 O(l) O(l) 11.6H 17(l) 11.6 17 C11.6 H17 (l) + 15.85O222(g) → 11.6CO222(g) + 8.5H222O(l) O(l) → 8.5H O(g) 8.5H 8.5H 8.5H22 O(l) O(l) → → 8.5H 8.5H22 O(g) O(g) 8.5H222O(l) → 8.5H222O(g) 0.4CO (g) → 0.4CO(g) ++ 0.2O 0.2O (g) 0.4CO 0.4CO22 (g) (g) → → 0.4CO(g) 0.4CO(g) + 0.2O22 (g) (g) 0.4CO222(g) → 0.4CO(g) + 0.2O222(g) ◦◦◦◦ The sum of these reactions yields the actual reaction, and the sum of the �H The The sum sum of of these these reactions reactions yields yields the the actual actual reaction, reaction, and and the the sum sum of of the the �H �H298 298 ◦298 298 La sumaThe de estas reacciones da como resultado la reacción efectiva, y la suma de los valores de ◦ sum of these reactions yields the actual reaction, and the sum of the �H ◦ ◦ values gives the standard heat of the reaction occurring at 25 C: 298 values values gives gives the the standard standard heat heat of of the the reaction reaction occurring occurring at at 25 25◦◦C: C: ∆H° values nos da gives el calor la reacción cuandooccurring ocurre a at 2525 °C:C: theestándar standardde heat of the reaction 298 ◦ ◦◦◦ = = −6,797,040 + (44,012)(8.5) + (282,984)(0.4) = −6,309,740 �H �H = −6,797,040 −6,797,040 + + (44,012)(8.5) (44,012)(8.5) + + (282,984)(0.4) (282,984)(0.4) = = −6,309,740 −6,309,740 JJJ �H298 298 ◦298 298 = −6,797,040 + (44,012)(8.5) + (282,984)(0.4) = −6,309,740 J �H298 ◦ C to products at 300◦◦◦C is The actual process leading from reactants at 25 The C to to products products at at 300 300◦◦C C is is The actual actual process process leading leading from from reactants reactants at at 25 25◦◦◦◦C Cdiagram. to products at 300 is The actual process leading from reactants at 25 El proceso real que conduce de los reactivos a 25 °C a los productos a 300 °C seC representa represented by the dashed line in the accompanying For purposes of represented For purposes of represented by by the the dashed dashed line line in in the the accompanying accompanying diagram. diagram. For purposes of represented bydel the dashed line in the accompanying diagram. For por la línea punteada siguiente diagrama. Con el any propósito de calcular ∆Hpurposes para esteofproceso, calculating �H for this process, we may use any convenient path. The one drawn calculating �H for this we use path. one calculating �H for this process, process, we may may use any convenient convenient path. The The one drawn drawn ◦ ◦ is es una calculating �H for this process, we may use any convenient path. The one drawn puede emplearse cualquier trayectoria conveniente. La línea dibujada de forma continua ◦ ◦ with solid lines is logical one: �H has already been calculated and �H ◦ has with already with solid solid lines lines is is aaa logical logical one: one: �H �H298 has already been been calculated calculated and and �H �HPP◦◦P◦◦ is is 298 ◦298 298 has o already P solid lines is a logical one: �H been calculated and �H elecciónwith lógica: ∆H° ya ha sido calculada y ∆H se evalúa con facilidad. 298 P easily evaluated. P is 298 easily easily evaluated. evaluated. easily evaluated. H �H �H �H �H �H Reactivos aat11bar Reactants bar Reactants Reactants at at 11 1 bar bar Reactants at bar y 25 °C Reactants at 1 bar and 25�C and and 25�C 25�C and 25�C 156.2 fuel andgg 156.2 gg25�C fuel 156.2 156.2 fuel 156.2 g fuel fuel 21.85 mol O 156.2 g fuel 21.85 mol O 21.85 mol 21.85 mol O22222 21.85 mol O O 82.2 mol 21.85 O 82.2 mol N 82.2 N 82.2 mol mol N N2222 82.2 mol N 82.2 mol N22 HP �H� �H� �H�PPP �H� �H�PP Productos a 11 bar bar Products at Products Products at at 1 1 bar bar Products at 1 bar y 300 °C Products at 1 bar and 300�C and and 300�C 300�C and 300�C 11.2 mol CO andmol 300�C 11.2 mol CO 11.2 CO 11.2 mol CO22222 11.2 mol CO 0.4 mol mol CO CO2 11.2 0.4 0.4 0.4 mol mol CO CO 0.4 CO 8.5 mol mol H HO O 0.4 8.5 8.5 mol H 8.5 mol mol CO H22222O O 8.5 mol H O 6.2 mol mol H O22O 8.5 6.2 O 6.2 6.2 mol mol O O2222 6.2 mol O 82.2 mol N N 6.2 mol 82.2 82.2 mol N 82.2 mol O N2222 82.2 mol N 82.2 mol N22 H �� �H 298 �298 298 �H �H298 � �H �298 �H298 El cambio entalpíachange causadocaused por elby calentamiento de los productos de la reacción The enthalpy change caused by heating the products of reaction from 25 to The heating of from 25 Thedeenthalpy enthalpy change caused by heating the the products products of reaction reaction from 25 to tode 25 a ◦◦C is: The enthalpy change caused by heating the products of reaction from 25 to ◦ 300 °C 300 es: 300 ◦ � � C is: is: 300 C � � � � 300◦ C is: � � ◦◦ ◦ ◦ ◦ (573.15 − 298.15) = �C �H nnnii �C �H (573.15 − − 298.15) 298.15) = �CP◦◦ ���HH (573.15 �HP◦ = (573.15 − 298.15) n ii�C P◦PPiiii�HH �H P◦PP = donde � � i Pi H iiii i ◦ ◦ ◦ o � /R values are: where the �C ◦ ��HHP/R where the /R values values are: are: where the �C �CPP◦P⟨C los valores de Hi⟩ H /R son: Piii H where the �C Pii�H /R values are: MCPH(298.15,573.15;5.457,1.045E-3,0.0,-1.157E+5) == 5.2352 5.2352 CO CO MCPH(298.15,573.15;5.457,1.045E-3,0.0,-1.157E+5) = 5.2352 CO2222::: MCPH(298.15,573.15;5.457,1.045E-3,0.0,-1.157E+5) CO CO:2 : MCPH(298.15,573.15;5.457,1.045E-3,0.0,-1.157E+5) MCPH(298.15,573.15;3.376,0.557E-3,0.0,-0.031E+5) = = 5.2352 3.6005 P CO: CO: CO: H O: H H2222O: O: H 22O: O O O222::: O : N N N222 ::: N222: Whence, Whence, Whence, De donde, Whence, MCPH(298.15,573.15;3.376,0.557E-3,0.0,-0.031E+5) MCPH(298.15,573.15;3.376,0.557E-3,0.0,-0.031E+5) == 3.6005 3.6005 MCPH(298.15,573.15;3.376,0.557E-3,0.0,-0.031E+5) 3.6005 MCPH(298.15,573.15;3.470,1.450E-3,0.0,0.121E+5) ===4.1725 4.1725 MCPH(298.15,573.15;3.470,1.450E-3,0.0,0.121E+5) MCPH(298.15,573.15;3.470,1.450E-3,0.0,0.121E+5) = 4.1725 MCPH(298.15,573.15;3.470,1.450E-3,0.0,0.121E+5) ==4.1725 MCPH(298.15,573.15;3.639,0.506E-3,0.0,-0.227E+5) 3.7267 MCPH(298.15,573.15;3.639,0.506E-3,0.0,-0.227E+5) MCPH(298.15,573.15;3.639,0.506E-3,0.0,-0.227E+5) = = 3.7267 3.7267 MCPH(298.15,573.15;3.639,0.506E-3,0.0,-0.227E+5) =3.5618 3.7267 MCPH(298.15,573.15;3.280,0.593E-3,0.0,0.040E+5) = MCPH(298.15,573.15;3.280,0.593E-3,0.0,0.040E+5) MCPH(298.15,573.15;3.280,0.593E-3,0.0,0.040E+5) == 3.5618 3.5618 MCPH(298.15,573.15;3.280,0.593E-3,0.0,0.040E+5) = 3.5618 ◦ = (8.314)[(11.2)(5.2352) + (0.4)(3.6005) + (8.5)(4.1725) �H �H = (8.314)[(11.2)(5.2352) (8.314)[(11.2)(5.2352) + + (0.4)(3.6005) (0.4)(3.6005) + + (8.5)(4.1725) (8.5)(4.1725) �HP◦◦◦ = �H PP◦PP = (8.314)[(11.2)(5.2352) + (0.4)(3.6005) + (8.5)(4.1725) + (6.2)(3.7267) + (82.2)(3.5618)](573.15 − 298.15) + + (6.2)(3.7267) (6.2)(3.7267) + + (82.2)(3.5618)](573.15 (82.2)(3.5618)](573.15 − − 298.15) 298.15) + (6.2)(3.7267) + (82.2)(3.5618)](573.15 − 298.15) = 940,660 = = 940,660 940,660 JJJ = 940,660 J 04-SmithVanNess.indd 149 8/1/07 13:09:26 CHAPTER 4. Effects CAPÍTULO 4. Efectos CHAPTER 4. Heat Heat Effectstérmicos 150 150 150 y and and ◦◦ + �H ◦◦ = −6,309,740 + 940,660 = −5,369,080 J �H P �H = = �H �H298 298 + �H P = −6,309,740 + 940,660 = −5,369,080 J Because the process is of steady flow which the work kineticPuesto que se trata un proceso estable, para el cual trabajo deand flecha y los térmiBecause thede process is one onede offlujo steady flow for for which theelshaft shaft work and kineticand potential-energy terms in the energy balance [Eq. (2.32)] are zero or negliginos de las energías cinética y potencial en el balance energético [ecuación (2.32)] son cero o and potential-energy terms in the energy balance [Eq. (2.32)] are zero or negligible, �H = Q. Thus, Q = −5,369.08 kJ, and this amount of heat is transferred to despreciables, ∆H = Q. Así, Q = –5 369.08 kJ, y esta cantidad de calor se transfiere a ble, �H = Q. Thus, Q = −5,369.08 kJ, and this amount of heat is transferred la to caldera the boiler for every 100 mol dry flue gases formed. This represents para cada 100 moles de gases secos que son productos de la chimenea. Esto representa the boiler for every 100 mol dry flue gases formed. This represents 5,369,080 5,369,080 (100) = 79.0% (100) = 79.0% 6,797,040 6,797,040 of the heat of heat of of combustion combustion of the the fuel. fuel. del calorofdethe combustión del combustible. In In the the foregoing foregoing examples examples of of reactions reactions that that occur occur at at approximately approximately 11 bar, bar, we we have have tacitly tacitly assumed that the heat effects of reaction are the same whether gases are mixed assumed that the heat effects of reaction are the same whether gases are mixed or or pure, pure, an an Enacceptable los ejemplos anteriores de reacciones a 1 bar, supuesto de procedure for pressures. For reactions at this may be acceptable procedure for low low pressures. que For ocurren reactionsaproximadamente at elevated elevated pressures, pressures, this hemos may not not be manera the tácita que los efectos térmicos de reacción son los mismos sin importar si son mezclados o puros, un case, and it may be necessary to account for the effects of pressure and of mixing on the the case, and it may be necessary to account for the effects of pressure and of mixing on the procedimiento aceptable para presiones reacciones a elevadas presiones, éste no es el caso, y pueheat these effects are small. heat of of reaction. reaction. However, However, these bajas. effectsPara are usually usually small. de ser necesario tomar en cuenta los efectos de la presión y el mezclado sobre el calor de reacción. De cualquier modo, estos efectos son generalmente pequeños. PROBLEMS PROBLEMS PROBLEMAS 4.1. For steady flow in a heat exchanger at approximately atmospheric pressure, what is the 4.1. 4.2. 4.3. 4.4. 4.1. For steady flow in a heat exchanger at approximately atmospheric pressure, what is the heat heat transferred: transferred: Para un flujo estable en un intercambiador de calor aproximadamente a presión atmosférica, ¿cuál ◦ (a) 10 is heated heated from from 200 200 to to 1,100 1,100◦C? C? (a) When When 10 mol mol of of SO SO22 is es el calor transferido? ◦ (b) (b) When When 12 12 mol mol of of propane propane is is heated heated from from 250 250 to to 1,200 1,200◦C? C? a) ¿Cuando 10 moles de SO2 son calentadas de 200 a 1 100 °C? For flow aa heat exchanger at pressure, b)4.2. 12 moles de propano calentadas de 250 a 1 200atmospheric °C? 4.2.¿Cuando For steady steady flow through through heatson exchanger at approximately approximately atmospheric pressure, what what is the final temperature, is the final temperature, Para un flujo estable a través de un intercambiador de calor aproximadamente a presión atmosféri(a) When heat amount of 800 added (a) es When heat in in the thefinal amount 800 kJ kJ is iscasos? added to to 10 10 mol mol of of ethylene ethylene initially initially at at ca, ¿cuál la◦temperatura en losofsiguientes 200 C? 200◦C? (b) heat the amount of kJ added to mol initially (b) When When heat in in800 the kJ amount of a2,500 2,500 kJ is is de added to 15 15 mol of of 1-butene 1-butene initially at at a) Cuando se◦ agregan de calor 10 moles etileno inicialmente a 200 °C. 260 ◦ C? 260 C? b) Cuando se añaden 2 500 kJ de calor a 15 moles de 1-buteno inicialmente a 260 °C. 6 6(Btu) (c) heat in the amount of added to mol) initially c) Cuando se agregan de calor 40(lb is mol) de etileno a 500(°F). (c) When When heat in10 the amount of 10 10a6(Btu) (Btu) is added to 40(lb 40(lbinicialmente mol) of of ethylene ethylene initially ◦ ◦ at 500( F)? at 500( F)? Si 250(pie)3(s)–1 de aire a 122(°F) y aproximadamente a presión atmosférica se precalientan para un 33(s)−1 ◦◦F) and approximately atmospheric pressure is preheated at 4.3. proceso de combustión 932(°F), es approximately la rapidez requerida de transferencia calor? (s)−1 of ofaair air at 122( 122(¿cuál F) ◦and atmospheric pressure isdepreheated 4.3. If If 250(ft) 250(ft) for for aa combustion combustion process process to to 932( 932(◦F), F), what what rate rate of of heat heat transfer transfer is is required? required? ¿Cuánto calor se necesita cuando 10 000 kg de CaCO3 son calentados de 50 °C a 880 °C a presión 4.4. is atmosférica? is heated heated at at atmospheric atmospheric prespres4.4. How How much much heat heat is required required when when 10,000 10,000 kg kg of of CaCO CaCO33 is ◦◦C to 880◦◦C? sure from 50 sure from 50 C to 880 C? 04-SmithVanNess.indd 150 8/1/07 13:09:29 Problemas Problems 151 151 4.5. Si la capacidad calorífica de una sustancia se representa de manera correcta por una ecuación de la 4.5. If the heat capacity of a substance is correctly represented by an equation of the form, forma, CC BT ++CT C 2T 2 P == A A+ + BT P show thatque theelerror �C P⟨C �HP⟩isH se assumed evaluated demuestre errorresulting resultantewhen cuando suponeequal igual to a CCPPevaluado enatla the media aritmética de las temperaturas final, C(T2 – T1)2/12. arithmetic mean of theinicial initial yand finalestemperatures is C(T2 − T1 )2 /12. 4.6. capacidad calorífica de una sustancia se representa de manera correctaofpor ecuación de la 4.6.SiIflathe heat capacity of a substance is correctly represented by an equation theuna form, forma, C P = A + BT + DT −2 CP = A + BT + DT-2 show that the error resulting when �C � is assumed equal to C evaluated at the demuestre que el error resultante cuando P⟨CHP⟩ H se supone igual a CPPevaluado en la media aritméarithmetic mean of the initial and final temperatures is: tica de las temperaturas inicial y final, es: � � T2 − T1 2 D T1 T2 T2 + T1 4.7. Calcule la capacidad calorífica de una muestra de gas a partir de la información siguiente: la mues4.7. Calculate the heat capacity of a gas sample from the following information: The samtra llega al equilibrio dentro de un frasco a 25 °C y 121.3 kPa. Una llave de paso se abre por corto ple comes to equilibrium in a flask at 25◦ C and 121.3 kPa. A stopcock is opened tiempo, permitiendo que la presión disminuya a 101.3 kPa. Con la llave cerrada, el frasco se calienta, briefly, allowing the pressure to drop to 101.3 kPa. With the stopcock closed, the flask regresando a 25 °C, y la presión medida es de 104.0 kPa. Determine CP en J mol–1 K–1 suponiendo warms, returning to 25◦ C, and the pressure is measured as 104.0 kPa. Determine C P que el gas−1 es ideal y que la expansión del gas restante en el frasco es reversible y adiabática. in J mol K−1 assuming the gas to be ideal and the expansion of the gas remaining in the flask to be reversible and adiabatic. 4.8. Se calienta una corriente de proceso como un gas de 25 a 250 °C a presión constante. Una estimación rápida del requerimiento energético se obtiene de la ecuación (4.3), con una CP tomada como 4.8. A process stream is heated as a gas from 25◦ C to 250◦ C at constant P. A quick esticonstante e igual a su valor a 25 °C. ¿El valor estimado de Q probablemente será más alto o más mate of the energy requirement is obtained from Eq. (4.3), with C P taken as constant bajo? ¿Por qué? and equal to its value at 25◦ C. Is the estimate of Q likely to be low or high? Why? 4.9. a) Para uno de los compuestos que se listan en la tabla B.2 del apéndice B, evalúe el calor latente 4.9. (a) For one of the compounds listed in Table B.2 of App. B, evaluate the latent heat de vaporización ∆Hn mediante la ecuación (4.12). ¿Cómo se compara este resultado con el valor of vaporization �Hn by Eq. (4.12). How does this result compare with the value mostrado en la tabla B.2? listed in Table B.2? b) Los valores del manual para los calores latentes de vaporización a 25 °C de cuatro compuestos (b) Handbook values for the latent heats of vaporization at 25◦ C of four compounds se proporcionan en la tabla. Para cada uno de ellos calcule ∆Hn mediante la ecuación (4.13), y are given in the table. For one of these calculate �H by Eq. (4.13), and compare compare el resultado con el correspondiente valor dadon en la tabla B.2. the result with the value given in Table B.2. –1 Calores de vaporización Latent heatslatentes of vaporization at 25◦ Cain25J °C g−1en J g Benceno 433.3 n-Pentano 366.3 n-Pentane 366.3 Benzene 433.3 Ciclohexano n-Hexano n-Hexane 366.1366.1 Cyclohexane 392.5 392.5 4.10.LaTable thelas thermodynamic properties of saturated liquid and vapor tetrafluo4.10. tabla9.1 9.1lists indica propiedades termodinámicas del líquido y vapor saturados del tetrafluoretaroethane. use of thedevapor as a function of temperature and of the de líquino. Haga usoMaking de las presiones vaporpressures como función de la temperatura y de los volúmenes andpara saturated-vapor volumes, calculate the latentmediante heat of vaporization dosaturated-liquid y vapor saturados, calcular el calor latente de vaporización la ecuación (4.11) para by de Eq.las(4.11) at onetemperaturas of the following temperatures compare the withcalculado the una siguientes y después compareand el resultado conresult el valor de los value calculated from theseenthalpy valores de la entalpía que dan en lavalues tabla.given in the table. ◦ F), (d) 80(◦ F), (e) 105(◦ F). 5(◦b) F),30(°F), (b) 30(c)◦ F), (c) 55( a) (a) 5(°F), 55(°F), d) 80(°F), e) 105(°F). 04-SmithVanNess.indd 151 8/1/07 13:09:31 152 CAPÍTULO 4. Efectos térmicos 4.11. Los valores en los manuales para los calores latentes de vaporización en J g–1 están dados en la tabla para diferentes líquidos puros a 0 °C. ∆H a 0 °C Cloroformo 270.9 Metanol 1 189.5 Tetraclorometano 217.8 Para una de estas sustancias, calcule: a) El valor del calor latente a Tn mediante la ecuación (4.13), dado el valor a 0 °C. b) El valor del calor latente a Tn mediante la ecuación (4.12). ¿En qué porcentajes difieren estos valores de los listados en la tabla B.2 del apéndice B? 4.12. La tabla B.2 del apéndice B proporciona los parámetros para una ecuación que da Psaturado como una función de T para un número de compuestos puros. Para uno de éstos, determine el calor de vaporización en su punto de ebullición normal mediante el uso de la ecuación de Clapeyron [ecuación (4.11)]. Evalúe d Psaturado/dT con una ecuación de presión de vapor que se proporciona, y use las correlaciones generalizadas del capítulo 3 para estimar ∆V. Compare el valor computado con el valor de ∆Hn presentado en la tabla B.2. Advierta que el punto de ebullición normal se encuentra en la última columna de la tabla B.2. 4.13. Un método para la determinación del segundo coeficiente virial de un gas puro se basa en la ecuación de Clapeyron y en mediciones del calor latente de vaporización ∆H lν, el volumen molar del líquido saturado V l, y la presión de vapor P saturado. Determine B en cm3 mol–1 para metil etil cetona a 75 °C a partir de la información siguiente a esta temperatura: ∆H lν = 31 600 J mol–1 V l = 96.49 cm3 mol–1 1n Psaturado/kPa = 48.157543 – 5 622.7/T – 4.70504 1n T [T = K] 4.14. 100 kmol/hora de un líqui