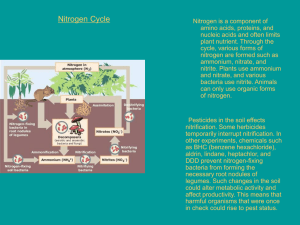
Environment & Safety Engineering (CH1400) Ecosystems Dr. Adhidesh S. Kumawat Ecosystem • Ecosystems are communities of organisms that interact with one another and with their physical environment, including sunlight, rainfall and soil nutrients. – Organisms within an ecosystem interact with one another to a higher extent compared to those between different ecosystems – Examples – a tidal pool (even as small as 2 m diameter), a tropical rainforest • Habitats – a place where a population of organisms lives • Characteristics of ecosystem: 1. Matter flows – flow into and out of ecosystem exists but its extent is very small compared to the amount contained in the ecosystem 2. Changes with time – for ex. Lakes change (naturally or anthropogenic over time. Several phenomenon such as flooding, droughts, extreme temperature changes and extreme environmental conditions (e.g. volcanic activity or forest fires) 3. Can be natural or artificial – lakes, tidal pools = natural; constructed wetlands for reducing storm runoff, agricultural land = artificial ecosystems. 2 Human Influence on Ecosystems – Environmental engineers and scientists have a responsibility to protect ecosystems and the life residing within them. – Activities such as release of pesticides, fertilizers and greenhouse gases to environment – Dams provide a clean, renewable energy source. Further, dam construction can have detrimental effects on river ecosystems related to fish population, soil erosion and vegetation. • Human activities can cause ecosystem changes resulting in destruction of species – Destruction of rainforest in Mexico threatens existence of Monarch butterfly. If forests are destroyed to the extent that it looses its winter roosting grounds than global extinction may result. However, localized destruction of milkweed plants have deprived of its nesting environment, resulting in local extinction. – Toxic chemical threaten wildlife such as DDT, petroleum compounds or heavy metals. – Acid rain is a result of emission from power plants, automobiles and industrial operations. 3 There is a strong relationship between oxides of sulfur and nitrogen Human Influence on Ecosystems – Agricultural operations in California resulted in increase of concentration of selenium to an extent that several species of water birds including black – necked stilts became threatened. – Species could also be threatened by introduction of nonnative (exotic) species in ecosystems. The introduction of rabbits onto Norfolk Island in 1830 resulted in loss of 13 species of vascular plants by 1967. – The zebra musel in inland waters of Ontario and Quebec is assumed to be responsible for the reduction of about 80 % of mass of phytoplankton in Lake Erie. Because It is an efficient water filterer of water, its presence significantly increase water clarity, allowing light to penetrate deeper into the water column, increasing the density of aquatic vegetation, algae and other organisms. – Another method by which species can become extinct is excessive hunting. Fr ex. Rhinoceros is threatened due to poaching, mainly for its horns. 4 Energy and Mass Flow • Primary producers: Sunlight – using organisms; Autotrophic. – – – – Trophic is the term to describe the level of nourishment Plants are photoautotrophic – obtain carbon from inorganic sources 6CO2 + 6H2O + 2800 kJ energy from sun (in presence of chlorophyll) –> C6H12O6 + 6O2 Net Primary Productivity (NPP) – rate of production of biomass glucose, cells and other organic chemicals by primary producers. For ex. Swamps and tropical forests have high NPP but deserts and arctic tundra does not. – Photoheterotrophs – they can obtain energy through photosynthesis but are not capable of reducing CO2. they obtain carbon from reduced carbon compounds generated by other organisms. 5 Ecological and Energy Pyramid 6 Energy and Mass Flow Ex.1. A deer eats 25 kg of herbaceous material per day. The herbaceous matter is approximately 20% dry matter (DM) and has an energy content of 10 MJ (kg/DM). Of the total energy ingested per day, 25% is excreted as undigested material. Of the 75% that is digested, 80% is lost to metabolic waste products and heat. The remaining 20% is converted to body tissue. How many megajoules are converted to body tissue on a daily basis? Calculate the percentage of energy consumed that is converted to body tissue. Solution. The percentage of consumed energy used for body tissue is = = (75 MJ/day)/(50 MJ/day) * 100 = 15 % 7 Energy and Mass Flow Ex.2. For every megajoule of energy used by the phytoplankton in Lake Michigan, how many joules of energy are used in building cell tissue in the lake trout? How many in humans? Use the following food web path: Phytoplankton −→ zooplankton −→ alewife −→ lake trout −→ humans Solution. • Given the rule of thumb that only 10% of the energy consumed is converted to biomass, then Phytoplankton −→ zooplankton −→ alewife −→ lake trout −→ humans 1 MJ 0.1 MJ 0.01 MJ 1000 J 100 J 8 Bioaccumulation • Bioaccumulation has serious implications for the movement of chemicals in the environment. – Chemicals that are hydrophobic will tend to be lipophilic. As a result, these chemicals will tend to partition (move into) into the fat tissue of animals. This process results in bioaccumulation. • Bioaccumulation is the total uptake of chemicals by an organism from food items (benthos, fish prey, sediment ingestion, etc.) as well as via mass transport of dissolved chemicals through the gills and epithelium (Schnoor, 1996). • When chemical bioaccumulate, the concentration of a chemical increases over time in an organisms body relative to the surrounding environment. This occurs because chemical retain in the living tissues more than the extent to which they are broken down (metabolized) or excreted. 9 Biomagnification • • Biomagnification is the process that results in the accumulation of a chemical in an organism at higher levels than are found in its own food. It occurs when a chemical becomes more and more concentrated as it moves up through a food chain. – • For ex. Algae eats chemical… small fish eats that algea… bigger fish eats that small fish ad finally human eats that big fish. If each step results in bioaccumulation, then an animal at the top of the food chain, through its regular diet, will accumulate a much greater concentration of chemical than was present in organisms lower in the food chain, and biomagnification will occur. 10 Ref. blue-growth.com Bioaccumulation vs Biomagnification 11 Bio-Concentration • • • bioconcentration—the uptake of chemicals from the dissolved phase. Through bioconcentration, the concentration of a chemical in an organism becomes greater than its concentration in the air or water in which the organism lives. Although the process is the same for both natural and synthetically made chemicals, the term bioconcentration usually refers to chemicals foreign to the organism. – • • For fishes, bioconcentration after uptake through the gills (or skin) is the most important bioaccumulation process. Bioconcentration factors, the ratio of the concentration of the chemical of interest in the organism to the concentration in water, are used to measure the tendency of a chemical to accumulate in lipid tissue and to relate pollutant concentrations in the water column with that in fish using the following equation: Concentration in fish = (concentration in water) × (bioconcentration factor) These factors are important in performing risk assessment calculations for predicting the effect of a chemical on a target species. Ex.3. The concentration of the pesticide DDT was found to be 5 g ・ L−1 in the water of a pond. The bioconcentration factor for DDT is 54,000 L ・ kg−1 (U.S. EPA, 1986). What is the expected concentration of DDT in the fish living in the pond? Solution. Concentration in fish = (5 g ・ L−1)(54,000 L ・ kg−1) = 270,000 g ・ kg−1, or 270 mg ・ kg−1 12 Nutrient Cycles • The basic elements of which all organisms are composed are carbon, nitrogen, phosphorus, sulfur, oxygen, and hydrogen. – – – • The first four of these elements are much more limited in mass and easier to trace than are oxygen and hydrogen. Because these elements are conserved, they can be recycled indefinitely (or cycled through the environment). Because the pathways used to describe the movement of these elements in the environment are cyclic, they are referred to as the carbon, nitrogen, phosphorus, and sulfur cycles. In any particular environment, the nutrient cycle must be balanced and stable if the organisms that live in that environment are to flourish and be maintained in a constant population (MARTIN 2010). 13 Nutrient Cycles disturbance • population growth and resulting human activities such as large-scale farming have caused some significant changes in nutrient cycles – – • with the introduction of water-borne sewage, this cycle was interrupted and replaced by a linear system that transports nutrients away from soils and into watercourses agriculture accelerates land erosion — because ploughing and tilling disturb and expose the soil — so more nutrients drains away with runoff In any particular environment, the nutrient cycle must be balanced and stable if the organisms that live in that environment are to flourish and be maintained in a constant population (MARTIN 2010). – – – – Depletion of soils Depletion of nutrient sources Affordability and food security Eutrophication of waterways and dead zones 14 Carbon Cycle • • • Ocean is the greatest reservoir of carbon (about 85%) Photosynthesis is the major driving force for the carbon cycle Ocean is the major sink of carbon – – – – Uptake of CO2 is driven by: (1) the solubility and (2) biological pumps Solubility pump – dissolution of CO2 into waters such as polar waters are colder than deeper regions and have greater capacity of dissolving CO2. Phytoplankton, Zooplanktons and bacteria make up the biological pump. They take up carbon and nutrient from the water resulting in carbon cycling. Human have affected the carbon cycle significantly through the combustion of fossil fuels, industrial activities and burning of materials. Pmfias.com 15 Carbon Cycle • • • Ocean is the greatest reservoir of carbon (about 85%) Photosynthesis is the major driving force for the carbon cycle Ocean is the major sink of carbon – – – – Uptake of CO2 is driven by: (1) the solubility and (2) biological pumps Solubility pump – dissolution of CO2 into waters such as polar waters are colder than deeper regions and have greater capacity of dissolving CO2. Phytoplankton, Zooplanktons and bacteria make up the biological pump. They take up carbon and nutrient from the water resulting in carbon cycling. Human have affected the carbon cycle significantly through the combustion of fossil fuels, industrial activities and burning of materials. Pmfias.com Pmfias.com 16 Nitrogen Cycle • Cycling of nitrogen content • Nitrogen in lakes as nitrate (NO3-) and taken up by algae and reduced to amino (NH2–R). Dead algae contains NH3, which dissolves to form NH4+. Nitrifying bacteria oxidized this to NO3- to a process called nitrification. 4NH4+4 + 6O2 = 4NO2− + 8H+ + 4H2O 4NO2− + 2O2 = 4NO3− • Nitrate is reduced back to N2 by denitrification. 2NO3- + organic carbon = N2 + CO2 + H2O • Nitrogen fixation is also accomplished in the soils by almost all legumes. Nitrogen fixation occurs in the root nodules that contain bacteria. Pmfias.com 17 Nitrogen Cycle • The basic elements of which all organisms are composed are carbon, nitrogen, phosphorus, sulfur, oxygen, and hydrogen. – – – The first four of these elements are much more limited in mass and easier to trace than are oxygen and hydrogen. Because these elements are conserved, they can be recycled indefinitely (or cycled through the environment). Because the pathways used to describe the movement of these elements in the environment are cyclic, they are referred to as the carbon, nitrogen, phosphorus, and sulfur cycles. Pmfias.com 18 Phosphorus Cycle • Cycling of phosphorus content • It enters unpolluted waters through dust in precipitate • Phosphorus is normally present in watersheds in extremely small amounts, usually existing dissolved as inorganic orthophosphate, suspended as organic colloids, adsorbed onto particulate organic and inorganic sediment, or contained in organic water. • Phosphorus available to plants is the amount soluble as well as in organic compounds • Largely, the permanent removal of phosphorus from overlying waters to the sediments depends on the amount of iron, aluminium, calcium ad clay entering the lake along with phosphorus • Excess use of phosphorus fertilizer also adds in disturbing ecosystem 19 Phosphorus Cycle • The basic elements of which all organisms are composed are carbon, nitrogen, phosphorus, sulfur, oxygen, and hydrogen. – – – The first four of these elements are much more limited in mass and easier to trace than are oxygen and hydrogen. Because these elements are conserved, they can be recycled indefinitely (or cycled through the environment). Because the pathways used to describe the movement of these elements in the environment are cyclic, they are referred to as the carbon, nitrogen, phosphorus, and sulfur cycles. Pmfias.com 20 Sulfur Cycle H. W. Assignment 21 Population Dynamics • It is the study of changes in nos. and composition of individuals in population and factors that affect these numbers. • It helps in several ways: (1) how environment changes affects populations; (2) determining resources needs; (3) predicting bacterial populating in systems; (4) using population as indicator of environmental quality. • Reduction in resources => inc. Mortality • Enables us to utilize mathematical models to understand and predict population dynamics – what, how and why are the changes in population of a particular species occurring? • Particular relevant in the studies of mathematical biology – more than 2 centuaries • Earliest studies – Thomas Robert Malthus • Malthusian growth model, sometimes called a simple exponential growth model, is essentially exponential growth based on the idea of the function being proportional to the speed to which the function grows – P(t) = P0 ert – dP/dt = rP or dP 22 Population Dynamics • Bacteria population Growth: • Model Growth of bacteria using geometric progression • Mixed culture growth becomes complicated • Bacterial growth in pure cultures: • Lag phase: when bacteria adjusts to their new environment • Accelerated growth phase: bacteria begins to divide. But all organisms do not divide at the same time. (production by binary fission) • nth generation growth: P = Po(2)n • log P = log Po + n log 2 (exponential or log growth) • Growth rate = straight line with slope “n” • Stationary phase: balance in death & reproduction rates due to limitation of resources • Death phase: die faster than reproduction • Carrying capacity: point at which decline occurs 23 Thank You 24