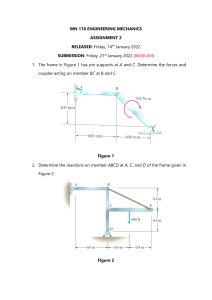
CHAPTER 10 (Rev 03) Electrical Properties of Materials (Callister Ch.19, p680 – 688, pg 693-699 ,p722-723 (Callister 10th ed Ch.18 pg.689-709; pg.729-731) 2/7/2022 1 10.1 Outcomes 2/7/2022 Formulate the relevant definitions of the terms and know and understand the terms used in this chapter Relate examples of typical practical applications of conductors, semiconductors and insulators Explain why electrical resistance (R) is not a materials property, whilst resistivity and conductivity are considered as materials properties Explain and indicate the similarity between the microscopic and macroscopic form of Ohm's law .Calculate the resistivity and conductivity of a material at a specific temperature if its temperature resistivity coefficient is given Predict the effect of impurities and alloying elements on the resistivity and the conductivity of a conductor, by applying graphs with data Explain why the resistivity of a conductor like copper is increased by: (1) a temperature increase, (2) alloy additions and (3) by cold working of the material 2 Outcomes apply the energy-band model to explain why some elements are good conductors and why others are poor conductors of electricity Explain the mechanism by which electrical conduction occurs in intrinsic semiconductors like silicon and germanium Derive an expression for the conductivity of an intrinsic semiconductor calculate the resistivity and the conductivity of intrinsic semiconductors. Explain why the conductivity of semiconductors is increased by an increase of the temperature, whilst the conductivity of good conductors is decreased by an increase of the temperature. Explain by using a typical example of each, the difference in the mechanism of electrical conduction between n-type extrinsic semiconductors and p-type extrinsic semiconductors. Derive expressions for the conductivity of n-type and p-type extrinsic semiconductors and calculate the resistivity and the conductivity of extrinsic semiconductors 2/7/2022 3 10.2 Use of electricity and materials Which areas are we going to cover? Materials are vital for conducting, for insulating, for semi-conductors and for capacitors. Without these, our civilisation would still be very primitive. 2/7/2022 4 Use of Electricity and Materials What is the fundamental difference between these? The differences lie in the electron arrangements and in particular in the valence electrons 2/7/2022 5 Use of Electricity and Materials Valence electrons and the band gap The valence electrons need to be lifted into the conduction band to conduct electricity. In conductors this requires no energy, and in insulators it requires a very large energy input. In semi-conductors one can supply enough energy to become a conductor or to “switch it off”. 2/7/2022 6 10.3 Electrical Conduction in Metals How do metals become good conductors? No electric field Electric field: electrons directed 1. In the absence of an electric potential, the motion of the valence electrons is at random and there is no net current or current flow. 2. When an electric potential is applied, the electrons attain a directed drift velocity, which results in directed flow of electrons (or current). 2/7/2022 7 Electrical Conduction and Resistivity i) Ohm's Law: ∆V = I R voltage drop (volts = J/C) resistance (Ohms) current (amps = C/s) C = Coulomb A (cross sect. area) e- I ∆V L Note the geometry dependence: A and L ii) Resistivity ρ and Conductivity σ: -- geometry-independent forms of Ohm's Law -- Resistivity is a matl. property, is independent of sample E: electric field intensity iii) Resistance: 2/7/2022 ∆V I = ρ A L ρL L R= = A Aσ resistivity (Ohm-m) J: flux or current density conductivity 1 σ= ρ 8 Electrical Conduction and Resistivity Ohm's law can also be expressed in its microscopic form, which is independent of the shape or size of the conductor: J = E/ρ or J = Eσ J = current density [A / m2 ] E = electrical field [V / m ] ρ = electrical resistivity [Ω.m] σ = electrical conductivity [Ω.m] -1 2/7/2022 9 Electrical Conduction and Resistivity 2/7/2022 10 Conductivity: Comparison Room Temp. values for σ (Ohm-m)-1 METALS Silver Copper Iron conductors 6.8 x 10 7 6.0 x 10 7 1.0 x 10 7 CERAMICS -10 Soda-lime glass 10 -10-11 Concrete 10 -9 Aluminum oxide <10-13 POLYMERS SEMICONDUCTORS Polystyrene Silicon 4 x 10 -4 Polyethylene Germanium 2 x 10 0 GaAs 10 -6 semiconductors 2/7/2022 -14 <10 10 -15-10-17 insulators 11 10.4 Factors that affect the Conductivity Some metals are good conductors and others not? i) Pure defect-free metals at low temperatures have low resistivity or high conductivity, like pure and annealed Cu at room temperature ii) In general a metal’s total resistivity is governed by a combination of: External component – a thermal (ρt) component – an impurity (ρi) component Material – a deformation (ρd) component component ρTot = ρt + ρi + ρd 2/7/2022 12 Factors that affect the Conductivity The material and external components ρTot = ρt + ρi + ρd The Material component ρr = (ρi+ρr) is known as the residual resistivity with the total becoming ρTot = ρt + ρr i) The thermal ρt increases almost linearly with temperature. ii) At higher temperatures the metal ions at the lattice positions vibrate more vigorously, by which the conduction (valence) electrons are increasingly scattered. iii) Thus, the free path of electrons are decreased and resistivity is increased 2/7/2022 13 Factors that affect the Conductivity The thermal component of resistivity/conductivity ρT = ρ0°C (1 + αTT) [Ω.m] or σ = σ0°C (1 + αTT)-1 [Ω.m]-1 ρ0°C = electrical resistivity at 0°C σ0°C= conductivity at 0°C αT = temperature coefficient of resistivity (αT is a function of the type of metal) T = temperature of conductor (°C) An increase in temperature as well as impurities increases Copper’s resistivity, in many cases almost linearly 2/7/2022 14 Factors that affect the Conductivity The deformation and alloying components i) Alloying Elements Many alloying elements form solid solutions. This strains the crystal lattice and causes additional scattering of the conduction electrons. Resistivity increases ii) Crystal Defects Crystal defects like dislocations and grain boundaries also cause additional scattering of the conducting electrons. By recrystallisation the conductivity may be restored. 2/7/2022 15 Factors that affect the Conductivity The resistivity of Cu – Ni alloys 1. Note the very sharp increase from adding Ni to Cu 2. Secondly a temperature increase has a moderate effect. 3. Deformation has the smallest effect. 2/7/2022 16 10.5 Energy band model of conduction The fundamental model of conduction and insulation The energy band model explains conductivity by: i. The number of electrons available for conduction The arrangement of these electrons in energy levels How these levels are filled with electrons ii. According to the Pauli-exclusion principle each electron has an unique energy and finds itself on energy levels according to the electron configuration of that element iii. Electrons fill the lowest energy levels with two electrons with opposite spins per energy level. 2/7/2022 17 Energy band model of conduction The fundamental model of conduction and insulation 1. The electrical properties of any material is governed by its electron structure, i.e. the arrangement of the outer electron levels and whether they are filled or not 2. Electrons in lower levels are more strongly bound to the core of the atom but at higher levels they are more free to move around and become available for electron pairing. . 2/7/2022 18 Energy band model of conduction Example of Sodium Sodium (Na) Atomic Number: 11 Electron configuration = 1s2 2s2 2p6 3s1 Note that the 3s1 electron is only loosely bound and is available for conduction 2/7/2022 19 Energy band structure Metals (Cu etc) Magnesium (e.g.) Insulators Semi-conductors Ef = Highest filled level at 0 K (Fermi-energy). E.g. Mg no band gap. In 2/7/2022 metals (a) and (b) electrons are easily elevated into the conduction band.20 Energy band structure Valence band: – filled by highest energy electrons Conduction band: - empty, lowest unfilled energy band In Mg the valence band and conduction band already overlap Conduction band Metals have small band gap valence band Adapted from Fig. 18.3, Callister 7e. 2/7/2022 21 10.6 Energy band model: insulators All the electrons are strongly bound by covalent or ionic interactions. No free electrons are available for conduction The valence electrons are separated by a large energy gap Eg from the conduction band For conduction a very large amount of energy is required to “tear loose” an electron into the conduction band 2/7/2022 22 10.7 Semi-conductors: band model Electrical conduction in semi-conductors is possible by the activation of conducting electrons over the band gap This requires a “switch-on” excitation energy of >Eg but it can also be “switched off” by removing this activation. Semi-conductors are, therefore, basically nothing else than an “on-off switch”, from there the binary system. A conducting electron can be excited at will into the conduction band or de-excited to become a nonconductor 2/7/2022 23 Semi-conductors: types Intrinsic semi-conductors: these consist of pure Silicon or Germanium and have no additive alloying elements Extrinsic semi-conductors: these consist of Si and Ge but alloyed with dilute solid solution quantities of elements from either group 3 or group 5 atoms 3 2/7/2022 5 24 10.8 Intrinsic Semi-conductors i. ii. iii. iv. v. Silicon and Germanium are both covalent bonded These form directional sp3 hybrid covalent bonds A Diamond cubic structure is formed Each atom’s four valence electrons are therefore strongly bound to the core of the atom These valence electrons therefore need a high exciting potential Eg to become conductive 2/7/2022 25 Intrinsic Semi-conductors Conduction in Si and Ge semi-conductors 1. Where an electron is excited from its energy level, it leaves a positively charged “hole” behind 2. Conduction will occur under the applied electric field due to the movements of the positive "holes" to the negative pole and the negative electrons to the positive pole 2/7/2022 Electric field 26 Intrinsic Semi-conductors Relationships for Conduction in Si and Ge 1. There are two charge carriers contributing to electrical conduction in intrinsic semiconductors i.e. free electrons and positive holes 2. The conduction is expressed as: σ = nqµn + pqµp (Ω.m)-1 n = number of conduction electrons/m3 p = number of conduction holes/m3 q = absolute value of electron or hole charge (q = 1.6 x 10 -19 coulombs) µn = mobility of electrons [m2/V.s] µp = mobility of holes [m2/V.s] But n = p = ni ni = intrinsic carriers/m3 Thus: 2/7/2022 σ = ni q (µn + µp ) (Ωm)-1 27 Conduction in Intrinsic Semi-conductors Concept of electrons and holes: valence electron no applied electric field electron hole pair migration electron hole pair creation Si atom + applied electric field + applied electric field n = p = ni Electrical Conductivity given by: -1 σ = n q (µ + µ ) (Ωm) i n p 2/7/2022 28 Conduction in Intrinsic Semi-conductors Effect of temperature on intrinsic semi-conductivity 1. At zero Kelvin the valence bands of intrinsic semiconductors are completely filled and their conduction bands are completely empty. 2. In contrast to metals, the conductivity of semiconductors increase with an increase in temperature. σ = σo e-Eg/2kT σo = temp. independent constant k = Boltzman’s constant = 8.620x10-5 (eV/K) E = Energy gap (eV) 2/7/2022g 29 Conduction in Intrinsic Semi-conductors Typical values for Si and Ge semiconductors σ = ni q (µn + µp ) Note the lower band gap Eg for Ge and its higher mobilities for both electrons and holes, which provides a much higher electrical conductivity at RT than in Si 2/7/2022 30 Conduction in Intrinsic Semi-conductors Effect of temperature on intrinsic semi-conductivity Room temperature σ = ni q (µn + µp ) Increase in ni with temperature as thermal activation also assists in exciting valence electrons Here Ge has a much larger number of electrons or holes at room temperature than Si Si: ni = 1.5 × 1016 carriers/m3 Ge: ni = 2.4 × 1019 carriers/m3 2/7/2022 31 Conduction in Intrinsic Semi-conductors Example 1: For an intrinsic Ga-As semi-conductor at room temperature electrical conductivity σ = 10-6 (Ωm)-1. The electron/hole mobilities are 0.85 en 0.04 m2/V.s respectively. Calculate the intrinsic carrier concentration at room temperature. q = 1.6x10-19 Coulomb or Ampere.seconds σ = ni q (µn + µp ) 2/7/2022 32 10.9 Extrinsic Semi-conductors 1. Highly dilute substitutional solid solution atoms or solutes, are added to the parent semiconductor Si or Ge by doping 2. The valence electron’s arrangement of these donor atoms differ from those of the parent or solvent Si or Ge There are two types of extrinsic semi-conductors: • n-type extrinsic semi-conductors: negative • p-type extrinsic semi-conductors: positive 2/7/2022 33 n- and p-type Extrinsic semi-conductors Differences between n- and p- types: From the periodic table: Group IIIA Dopant added (valence 3+) B Al Ga In p-type 2/7/2022 Group IVA Parent metal (valence 4+) Si Ge Group VA Dopant added (valence 5+) P As Sb n-type 34 10.10 n- Type Extrinsic semi-conductors 1. If a Group 5 atom (P, As or Sb) replaces a Si atom (Group 4), one extra valence electron is available on that atom 2. This extra valence atom is loosely bound and requires only Eg=0.044 eV to cross the energy gap 3. Compare this with the excitation energy gap of Eg=1.1 eV that would have been necessary in the intrinsic semi-conductor of pure Si 4. The donor impurities from Group 5 (P, As, Sb) therefore provide conducting electrons and are known as “impurity donor atoms” 2/7/2022 35 n- Type Extrinsic semi-conductors Note that the donor electron from the Group 5 atoms require very little excitation energy to become conducting. For a pure Si atom in an intrinsic semi-conductor, excitation over the full E2/7/2022 36 g would have been necessary. n- Type Extrinsic semi-conductors Free electron Note the “free” electron at the donor atom that can be excited to the conducting level relatively easy 2/7/2022 37 10.11 p-Type Extrinsic semi-conductors 1. If a parent Si or Ge atom with valence 4+ is replaced by a Group III atom (B, Al or Ga) with a valence of 3+, there is a shortage of one valence electron. 2. This creates a positive hole at one of the covalent bonds that surrounds this Group III atom 3. With the introduction of an electric excitation energy on the Si crystal, one of the electrons from a nearby Si atom can be excited to neutralise the hole. 4. Note that the hole has now moved to another electron position which made the hole also mobile 5. Conduction, therefore, talks place by the movement of the holes to the negative pole of the applied electric field 2/7/2022 38 p-Type Extrinsic semi-conductors 6. The excitation energy associated with the movement of holes from B in Si, Eg =0.045 eV is small in comparison to the Eg = 1.1 eV for conductivity with only Si atoms present. 7. These atoms are known as “impurity acceptor donor” atoms 8. The majority carriers are therefore holes (p-type) while the minority carriers are electrons 2/7/2022 39 p-Type Extrinsic semi-conductors The excitation energy associated with the movement of holes from B in Si, Eg =0.045 eV is small in comparison to the Eg = 1.1 eV for conductivity with only Si atoms present. 2/7/2022 40 10.12 Intrinsic vs Extrinsic Conduction 1. Intrinsic: # electrons = # holes (n = p) --case for pure Si 2. Extrinsic: --n ≠ p --occurs when impurities are added with a different # valence electrons than the host (e.g., Si atoms) 2.1 n-type Extrinsic: (n >> p) 2.2 p-type Extrinsic: (p >> n) Phosphorus atom 4+ 4+ 4+ 4+ σ ≈ n e µe 4+ 5+ 4+ 4+ 4+ 4+ 4+ 4+ 2/7/2022 no applied electric field Boron atom hole conduction electron 4+ 4+ 4+ 4+ valence electron 4+ 4+ 4+ 4+ Si atom 4+ 3+ 4+ 4+ no applied electric field σ ≈ p e µh 41 10.13 Finding carrier concentrations in extrinsic semiconductors A. n-type semi-conductors σ = ni q (µn + µp ) In n-type semiconductors nn >> pn as electrons are the majority carriers and holes as minority carriers make only a minor contribution 1. Therefore σ ≈ nnqµn as nn >> pn 2. Resistivity ρ ≈ 1 / n qµ n n 3. And the concentration of the donor in atoms/m3 (P, As or Sb) N =n d 2/7/2022 n 42 Finding carrier concentrations in extrinsic semiconductors A. n-type semi-conductors (cont’d) 4. At constant temperature, the product of the negative free electron concentration (n) and the positive hole concentration (p) is a constant where ni is the intrinsic concentration of carriers in a semiconductor and is constant at a given temperature. 5. Charge densities in extrinsic semiconductors: The material must be electrically neutral Charge carriers in extrinsic semiconductors are donor ions (Nd), acceptor ions (Na), free electrons (n) and holes (p). In general then, sum of negative carriers = sum of positive carriers, or: 2/7/2022 Na + n = Nd + p 43 Finding carrier concentrations in extrinsic semiconductors A. n-type semi-conductors (cont’d) 6. Charge carriers in extrinsic semiconductors are donor ions (Nd), acceptor ions (Na), free electrons (n) and holes (p). 7. In general then, sum of negative carriers = sum of positive carriers, or: Na + n = N d + p 8. For n-type extrinsic semiconductors: Where nn = concentration (-) electrons in n-type semiconductor and pn = concentration (+) holes in n-type semiconductor Na = 0 and nn>> pn and nn = Nd Nd = concentration of dopant atoms like P,As and Sb 2/7/2022 44 Finding carrier concentrations in extrinsic semiconductors B. p-type semi-conductors σ = nqµn + pqµp 1. Conductivity: (p-type) where pn >> n σ ≈ pp q µp pp ≈ Na 2/7/2022 45 10.14 Example on extr. semiconductors Example 1: A silicon wafer is doped with 1021 phosphorous atoms / m3. Calculate or find: (a) the majority carrier concentration. (b) the minority carrier concentration. (c) the electrical resistivity of the doped silicon at 300 K. Data for silicon: ni = 1.5 x 1016 [carriers / m3] µn = 0.135 [m2 / V s] µp = 0.048 [m2 / V s] 2/7/2022 46 10.15 Manufacture of semi-conductors 1. Grow Si or Ge single crystals of about 100 to 120 mm diameter and about 25 cm long by the Czochralski process 2/7/2022 47 Manufacture of semi-conductors 2. For Intrinsic semi-conductors, slice wafers off each single crystal and proceed with the manufacture of the semi-conducting chips 2/7/2022 48 Manufacture of semi-conductors 2. For Extrinsic semi-conductors, the Si or Ge single crystal needs to doped with, for instance P atoms. This can be done by one of two methods: i) By diffusion doping in a suitable P-containing atmosphere at a high temperature. This produces a Pconcentration gradient through the thickness which needs careful consideration in the final chips. ii) A more elegant process is to lightly radiate them in a research reactor as in SAFARI 1 where the correct amount of P atoms are produced by a neutron-Si reaction. Here no P-concentration gradient is found through the thickness. 2/7/2022 49 10.16 Dielectric (insulation) Behaviour A dielectric material is one that is electrically insulating and exhibits (or is made to exhibit) an electric dipole structure High and low voltage electrical insulators. i) Examples: ceramic materials used as electrical insulators: electrical porcelain , alumina (Al2O3) , steatite ii) Examples: polymer materials used as electrical insulators: phenol formaldehyde (bakelite) , nylon , polyethylene Inserts for capacitors. (A capacitor is an electrical device which can store electric charge.) i) Examples of ceramic materials used in capacitors: barium titanate (BaTiO3), or BaTiO3 mixed with other ceramics 2/7/2022 50 10.17 Piezoelectric ceramics Piezoelectric ceramics: these are some ceramics which can convert electrical pulses into mechanical vibrations and vice versa Utilized in transducers Examples of piezoelectric ceramics: barium titanate (BaTiO3) mixtures of lead zirconate (PbZrO3) and lead titanate (PbTiO3) Cochlear implant 2/7/2022 51 10.18 References Chapter 10 Slide no Reference 4 Thought Co: https://www.thoughtco.com/examples-of-electrical-conductors-and-insulators-608315; Toshiba: https://www.elprocus.com/different-types-of-transistor-and-their-functions/ 5 http://nextgenelectrical.blogspot.com/2015/03/dc-circuits-basics-introduction-circuit.html 6 Quora: https://www.quora.com/How-do-semiconductors-differ-from-conductors-and-insulators 7 Howard Univ: https://chem.libretexts.org/Courses/Howard_University/General_Chemistry%3A_An_Atoms_First_Approach/Unit_2%3A__Molecular_S tructure/Chapter_5%3A_Covalent_Bonding/Chapter_5.7%3A_Metallic_Bonding 10 NGK, Japan; 14 Medeliene et al, Galvanotechnik, 103(2012)p942 16 Material Science and Engineering Callister 9e Fig 19.8 pg 689 19 http://www.gcsescience.com/a4-sodium-ion.htm ; 20 Material Science and Engineering Callister 9e Fig 19.4 pg 685 21 Material Science and Engineering Callister 9e Fig 19.3 pg 684 24 Thought Co: https://www.thoughtco.com/examples-of-electrical-conductors-and-insulators-608315 26 Material Science and Engineering Callister 9e Fig 19.11pg 695 31 Material Science and Engineering Callister 9e Fig 19.16 pg 700 36 Material Science and Engineering Callister 9e Fig 19.13 pg 697 37 http://hobbygenius.co.uk/tutorials/physics/1456 39 http://hyperphysics.phy-astr.gsu.edu/hbase/Solids/dope.html 40 Material Science and Engineering Callister 9e Fig 19.15 pg 698 47 http://meroli.web.cern.ch/Lecture_silicon_floatzone_czochralski.html 48 http://maltiel-consulting.com/How_to_Make_a_Semiconductor_Chip.htm 51 CERAMTEC: https://www.ceramtec.com/ceramic-materials/piezo-ceramics/ ; Shin et al, Sensors: 18(2018)p1694 https://doi.org/10.3390/s18061694 2/7/2022 52