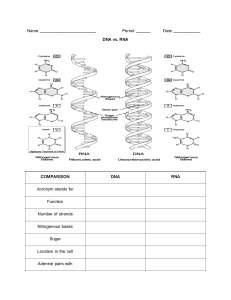
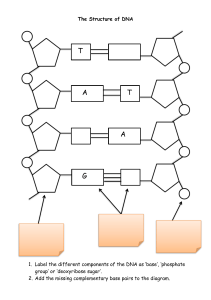
Introduction to Biological Sciences BSCI 1510 Section 01 Have you . . . Lecture 3: Monomers to macromolecules DNA Submitted your questions to Brightspace discussions? Read assigned reading for today? Started a small study group? Downloaded the bonds practice questions (in lecture 2 Atoms and Bonds folder)? These materials are for the sole purpose of assisting 2021 BSCI1510-01 students to learn and study for this course. No material from this course may be reprinted for any other purpose or redistributed. RNA Protein Lecture 3: Macromolecules | Chemical composition of cells (by weight) Monome E. coli 4% 4 small organic molecule 26% E. coli (not shown) Fatty acids ---> Fats and membrane lipid Chemical composition of cells Monomer (E. coli) Polymer 4 SMALL, 4 classes of organic LARGE molecules organic MMs 1. 2. (not shown) Fatty acids & glycerol 3. DNA 4. RNA Chemical composition of cells Monomer (E. coli) Polymer 4 SMALL, 4 classes of organic LARGE molecules organic MMs 1. 2. DNA or RNA (not shown) Fatty acids & glycerol (not shown) Lipids Formation of all macromolecules Nucleotide monomer Monomers Condensation Reaction drives linkages of monomers Polymer Cell schematic Macromolecules Carbohydrates Lipids Nucleic Acids: DNA, RNA Protein mRNA Nucleus Mitochondra DNA RNA Formation of nucleic acid macromolecules (DNA, RNA) Nucleotide Monomer Nucleic acid Polymer (4 Nucleotide MONOMERS of DNA) Condensation reaction makes a Phosphodiester bond Phosphate bound to two O’s (Phospho), in turn bound to two carbons (diester bond, 2 C-O bonds) All DNA and RNA nucleotides have 3 parts 1 ring C, T (U) 2 rings A, G 3 building blocks to make a nucleotide monomer 1. nitrogenous base (i.e., adenine) nucleoside nucleotide 2. sugar (ribose in RNA, deoxyribose in DNA) 3. phosphate (s). This is adenosine monophosphate (AMP), which can also become adenosine triphosphate (ATP) and diphosphate (ADP). RNA sugar RNA is single stranded. DNA is double stranded (how?). PUrines H bond to Pyrimidines to make dsDNA How did nucleotides, the chemicals of a cell’s genome, originate? Evidence: Nucleotides and their precursors are in meteorites Guanine Adenine 12 meteorites Carbonaceous meteorites contain a wide range of extraterrestrial nucleobases Michael P. Callahana et al (2011), PNAS Laser Formamide Gas Red Clay Evidence: Lab studies specify nucleotides can form from simple Earth-like conditions and high energy from a meteorite impact Nucleic acids are polymers of nucleotides DNA - monophosphates phosphate 3. sugar DNA nucleotide shown with P group is deoxyadenosine monophosphate ( AMP) phosphate sugar phosphate 2. DNA (ds) 4. sugar phosphate RNA nucleotide shown with P group is adenosine monophosphate ( AMP) sugar OH Outside Inside 2. RNA (ss) Carbon positions in sugar have specific features DNA phosphate sugar Deoxyribose + Base phosphate sugar phosphate DNA sugar DNA nucleotide shown with P group is deoxyadenosine monophosphate (dAMP) Deoxyribose + Base +P phosphate sugar OH RNA nucleotide shown with P group is adenosine monophosphate ( AMP) 2. RNA Why are DNA and RNA called nucleic acids? DNA phosphate DNA nucleotide shown with P group is deoxyadenosine monophosphate (dAMP) sugar phosphate sugar phosphate DNA sugar phosphate sugar OH RNA nucleotide shown with P group is adenosine monophosphate ( AMP) RNA RNA vs DNA – Which one originated first in cells? The “RNA World Hypothesis” = Cells started with RNA, DNA came later • ssRNA is a simpler start. • mRNA carries the genetic message between DNA and protein • Some viruses have RNA genomes, and all cells make RNA. • Single-stranded nature enables 2D base pairing with itself. • Folding produces 3D RNA structures with wonderful variety and complexity. • The structure can give RNA catalytic potential (e.g., copying, cleaving) • Denoted ribozymes. Woese’s Tree of Life was based on rRNA sequences A B E q 16S and 18S rRNA are part of small subunit of ribosome with structural and catalytic RNAs q 16S in Bacteria and Archaea; 18S is divergent copy in Eukaryotes Ribosome RNA RNA Protein Macromolecules opro tein Carbohydrates Lipids Nucleic Acids Protein glyc Cell schematic Energy storage • glycogen • starch Recognition • Cell-cell contacts Structural support in bacteria • Peptidoglycan Carbohydrates are made up of simple sugar monomers: Linear form (Glucose) Carbohydrates Ring form (Sugars with at least 5C’s can circularize with themselves): most common General monosaccharide formula (CH2O) n = typically 3-6 Cells can produce many sugars with the same chemical formula, but different structures, e.g. (CH2O)6 = Stereoisomers Sugar ring forms bond in condensation reactions to produce polymers: Glucose Disaccharide (Table sugar) Carbohydrates Fructose High Fructose Corn Syrup – made by hydrolyzing sucrose and converting glucose to fructose ~60:40 ratio Sugar ring forms bond in condensation reactions to produce polymers: Carbohydrates Disaccharide: Lactose - principal sugar in milk Sugar ring forms bond in condensation reactions to produce polymers: Oligosaccharide (3-10) Carbohydrates Oligo (tri-, tetrasaccharides, etc) When covalently linked to other macromolecules, they form glycoprotein and glycolipids Glycogen, Starch – glucose polysaccharide Polysaccharide (>10) some of the many possible sugar monosaccharides Triose tetroses pentoses hexoses Cell schematic Macromolecules Carbohydrates Lipids Nucleic Acids Protein Organic molecules that are insoluble in water. Fatty acids are the monomers that make up lipids. 3. Hydrophilic carboxyl end • Polar, Chemically reactive Why is this a fatty acid? Amphipathic Molecules Hydrophobic hydrocarbon tail • Chains of C,H • Nonpolar • Not very chemically reactive • Energy stored here 4. 2 Major Types of Fatty Acids Often stored as food reserves (TAGs) - 3 linked fatty acids with glycerol; essentially stored fat droplets; energy-rich Fatty acids have about 6X more chemical energy than glucose. Fatty acids : The tale of two tails 2 Major Types of Fatty Acids Lipids are formed from FAs attached to a glycerol (TAGs) Fatty acids can be stored as food reserves (TAGs) ready for energizing the cell TAGs or Tryglycerides are 3 linked fatty acids to a 3 Carbon (tri) glycerol; TAGs in cell form fat droplet(s) The Wonders of Fatty Acids and Lipids in Water Cell membranes Fatty acids alone Phospholipids (phosphate attached to glycerol attached to FAs) – most recognizable example of lipids and major constituent of membranes Glycolipids (carbohydrate attached to glycerol) Origin of Life: Can Fatty Acids and NT’s Spontaneously Form Into Cells? Protocells are an experimental model of autonomous structures synthesized from chemical components (fatty acids and RNA nt’s); self-assembling, spherical or cell-like Origin of Life: Can Fatty Acids and NT’s Spontaneously Form Into Cells? Lecture 3 learning objectives • Know the four classes of macromolecules found in living systems • Describe condensation and hydrolysis reactions and how these are involved in forming DNA, RNA, carbohydrates, and lipids • Know the precursors for each class of biological macromolecules and how they are linked together • Know in general terms the function of the classes of biological macromolecules