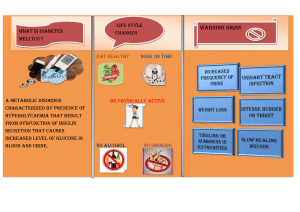
RAJKUMARI AMRIT KAUR COLLEGE OF NURSING NEW DELHI SUBJECT: MEDICAL SURGICAL NURSING (SEM: III) SEMINAR ON: TOPIC MANAGEMENT OF PATIENT WITH HYPOGLYCEMIA AND KETOACIDOSIS SUBMITTED TO MRS.MADHUMITA DEY ASSISTANT PROFESSOR RAKCON SUBMITTED BY MS.ANUGRAH MSC.FINAL YEAR RAKCON 1 MANAGEMENT OF HYPOGLYCEMIA: INTRODUCTION: Hypoglycaemia, a condition where the body experiences low blood sugar levels, is often a great cause of concern for people diagnosed with diabetes. Symptoms associated with low blood sugar can greatly impact Patient’s dayto-day life, which makes management of hypoglycaemia a commonly addressed topic during routine check-ups. What is Hypoglycaemia “Although Patient’sblood glucose levels can vary throughout the day, certain factors can cause them to reduce significantly. This dip in the levels, usually recorded below 70 mg/dl, is called hypoglycemia. It can be dangerous for patients if it is not treated appropriately and on time. Hypoglycemia is a medical condition where blood glucose levels fall below normal levels. If left untreated it can lead to severe health problems like a coma. One in every 25 patients suffering from type 1 diabetes dies due to severe hypoglycemia. But sugar levels can be managed naturally and one can lead a healthy life by the Diabetes Reversal Method When the blood sugar levels fall below 70mg//dL it is an indication of Hypoglycemia. Classifications of Hypoglycemia ❑ 1) Severe hypoglycemia ❑ 2) Documented symptomatic hypoglycemia ❑ 3) Probable symptomatic hypoglycemia ❑ 4) Asymptomatic hypoglycemia(or hypoglycemic Unawareness) ❑ 5) Relative hypoglycemia ❑ ❑ ❑ • Without diabetes mellitus the hypoglycemia is classified as: 1) reactive(sometimes called "postprandial") 2) and nonreactive(sometimes called "fasting"). Classifications of Hypoglycemia 1. Severe hypoglycemia: which requires the assistance of another person to administer a carbohydrate (preferably glucose sublingually or intravenously) or subcutaneous glucagon 2. Documented symptomatic hypoglycemia : which occurs when a patient feels typical hyperadrenergic hypoglycemic symptoms and verifies the blood glucose level is less than 70 mg/dL (3.9 mmol/L) before self treating with 15 grams of a carbohydrate 3. Probable symptomatic hypoglycemia : Typical hypoglycemia symptoms not accompanied by plasma glucose determination but likely caused by plasma glucose ≤70 mg/dL (≤3.9 mmol/L) 4. Asymptomatic hypoglycemia (or hypoglycemic unawareness): in which a patient does not develop typical hyperadrenergic symptoms but has a measured plasma glucose level of less than 70 mg/dL (3.9mmol/L). this situation occurs most often in type 1 diabetes in patients striving for excellent glycemic control 5. (hemoglobin A1c value <7.0%) who have chronic, frequent episodes of hypoglycemia. The body's ability to recognize hypoglycemia and secrete counterregulatory hormones in response to hypoglycemia deteriorates and leaves these patients vulnerable to further episodes of severe hypoglycemia. In diabetic patients, if severe neuropathy is present, the autonomic response (epinephrine) to hypoglycemia is not activated. This leads to neuroglycopenic symptoms. 6. Relative hypoglycemia: in which a patient experiences hyperadrenergic hypoglycemic symptoms but has a measured plasma glucose level greater than 70 mg/dL (3.9 mmol/L). • this situation occurs most often in 2 patients who have had months (or longer) of hyperglycemia (plasma glucose levels >200 mg/dL [11.1mmol/L] at all times) whose plasma glucose levels are then lowered by medication or lifestyle changes closer to the normal range. 7. Hyperadrenergic hypoglycemic symptoms can occur when the plasma glucose level in these patients is 120 mg/dL (6.7 mmol/L) or even higher. • If these patients continue to keep their plasma glucose level substantially less than 200 mg/dL (11.1 mmol/L), the threshold at which they manifest hypoglycemic symptoms will fall to more typical levels (<70 mg/dL [3.9mmol/L]). 8. Reactive or postprandial hypoglycemia : develops in response to a nutrient challenge. see it in some post-GI surgical patients, when gastric contents get dumped into the small intestine too quickly. 9. Idiopathic reactive hypoglycemia requires fulfillment of the Whipple triad to be a true diagnosis. • Some patients have symptoms but normal blood glucoses; they need no further workup, in spite of their insistence. 10. Nonreactive or fasting hypoglycemia can be further subdivided into: 1) iatrogenic (most common overall cause) 2) and fasting/factitious. • In the fasting/factitious type, the patient is unable to maintain glucose levels with fasting. • Most common causes: alcohol abuse, drugs (oral hypoglycemics, pentamidine), sepsis, and renal failure. “WHAT IS THE FIRST SIGN OF HYPOGLYCEMIA” “Our body’s initial response to low blood sugar levels is to stimulate the adrenaline hormone (adrenergic system) secretion, also known as the fright hormone. Patient may experience Patient body’s reaction to the excess adrenaline in the form of symptoms such as: • Palpitations or irregular heartbeats • Tremors • Sweats • A tingling sensation in or around Patient’s mouth However, the brain is incapable of tolerating reduced levels of blood sugar for long periods of time. This is why, if not treated promptly, Patient’s brain starts sending out warning signals such as: • An increased sense of hunger or cravings • Feeling cloudy • Blurred vision • Dizziness or light-headedness • Blackouts or loss of consciousness • Seizures • Coma 3 Hypoglycemia signs can be understood through 2 body responses these are: Adrenergic responses: The counter-regulatory hormones like norepinephrine control this process of Insulin decreasing the glucose levels in the blood. Any change in these hormone levels can decrease the regulation of insulin. Body responds to these changes and these responses serve as the early warning signs of hypoglycemia. These signs are: ▪ Diaphoresis (too much sweating) ▪ Dizziness ▪ Hand tremor ▪ Anxiety ▪ Hunger ▪ Tachycardia (increased heart rate), Along with Neuroglycopenic response which occurs due to the deficiency of glucose levels within the brain. This results in rapid dysfunction of the central nervous system. The initial signs of this response are: Neuroglycopenic response ▪ Headache ▪ Lethargy ▪ Confusion ▪ Disorientation ▪ Blurred vision ▪ Speech impairment Signs of Serious Condition/ Emergency: Ignoring initial signs and symptoms can lead to a bad situation. It is found that it can impact the medullary phase and can cause brain damage. The signs of this phase are: ▪ Seizures ▪ Deep coma, ▪ Bradycardia (slow heart rate), ▪ Shallow breathing, and ▪ Pupillary dilatation. ▪ Please note that these symptoms can vary from person-to-person. As Patient’s brain is deprived of blood glucose, the symptoms gradually worsen as well until management of hypoglycemia is carried out on time. ▪ Have Patient come across a situation of uneasiness or confusion, anxiety with irritation, increased heart rates, or tingling sensation in Patient’shands? If yes, this could mean Patient’sblood sugar levels are low. This could lead to a condition called Hypoglycemia. Patient need to eat a sugar-rich diet to bring the sugar levels back. Rmember it can be fatal if left untreated. 4 Glucose In Our Body: Blood sugar known commonly as Glucose is the main source of energy. Our body generates glucose after decomposing the food we eat. Our bloodstream absorbs Glucose. It is through the bloodstream it travels to the body cells. Pancreas produces a hormone known as Insulin in our body.It is the Insulin that helps our body cells to use glucose and give us energy to perform various activities like walking, running, etc. WHAT CAUSES HYPOGLYCEMIA: CAUSES: • The main function of medication, either orals or injectables, is to regulate Patient’s blood glucose levels. This is why an inadvertent overdose of medicine often causes hypoglycaemia amongst patients. • A proper treatment plan is pre-determined by doctor and customized to patient body’s requirements. Any adjustments to be made to this plan should be discussed with them beforehand as any discrepancy in medication timings can adversely affect Patient’s blood sugars. • Due to the fact that Patient’s medication is going to complete its function whether Patient eat or not, • • • skipping or delaying meals can cause Patient’s blood sugars to reach a level lower than recommended. Strenuous exercise or unintended physical activity that is out of Patient’s usual daily routine Alcohol consumption. Any reduction of carb content from Patient’s meals that is different from the usual. Can hypoglycemia happen to patients who don’t have diabetes: Uncommon causes of hypoglycemia that also require timely management can be caused due to the following factors: • If Patient have been experiencing hypoglycemia recurrently, it might be time for Patient to rule out the possibility of underlying issues in Patient’s kidneys or liver and treat them before Patient’s symptoms • • • worsen. Mistakenly ingesting diabetes medication not intended for Patient can also induce hypoglycemia. People who have undergone gastric bypass surgery (bariatric surgery) tend to have a reduced appetite and absorption as the food transits to the intestines faster, not leaving the body enough time to take in adequate amount of blood glucose. Abnormal pancreatic conditions where insulin secretions are produced at a higher rate than normal and cause Patient’s blood glucose levels to fall. 5 Hypoglycemia Causes, Without Diabetes: People without diabetes can also face the condition of Hypoglycemia. The main causes of hypoglycemia in such situation are: • • • • • • Due to medicines: It happens when Patient consume some medicines accidentally. Some medicines like Quinine which is used to treat malaria can cause hypoglycemia. One should always Consult doctor before consuming such medicines. Alcohol consumption: Heavy alcohol consumption on an empty stomach affects the liver. It then affects our blood glucose levels. Certain illnesses: Diseases like liver disease, kidney disorders; all impact the blood glucose levels. Therefore such diseases can lead to hypoglycemia. Eating disorder: People who eat less or suffer from anorexia nervosa have eating disabilities. Consequently, such people can suffer from hypoglycemia. Insulinoma: It is a condition where a tumor grows in the pancreas because of which insulin increases in our body. More insulin causes more glucose absorption and thus decreases glucose levels resulting in hypoglycemia. Hormone disorders: Due to disorders in adrenal glands, pituitary glands there is a decrease in glucose production. This leads to low blood sugar levels, causing Hypoglycemia HOW DO YOU ASSESS THE PATIENT: A key component of the nursing management of hypoglycemia is your assessment of the patient and recognition that you need to intervene. Assess all patients at risk for blood glucose abnormalities for: • Confusion • Level of consciousness • Presence of other signs/symptoms as outlined above • Risk factors for developing hypoglycemia such as reduced oral intake, increased exercise, or inappropriate use of anti-diabetic medications • • • Use of culprit medications Who should be evaluated Only those patients in whom Whipple's triad is documented require evaluation and management of hypoglycemia. • In patients with symptoms of hypoglycemia but normal plasma glucose concentrations at the same time, no further evaluation is needed. 1.Clinical Evaluation • The first step is to review the patient's history in detail, including the nature and timing of symptoms (particularly in relationship to meals), existence of underlying illnesses or conditions, surgical history ,medications taken by the individual and by family members, and social history. • In a patient with documented hypoglycemia, the cause may be apparent from the history and physical examination. • In a seemingly well individual, the cause is less apparent and may be due to hyperinsulinism or factitious hypoglycemia. • When the cause of hypoglycemia is not evident, detailed laboratory evaluation is needed. 6 2. Fasting evaluation: There are patients in whom symptoms occur after only a short period of food withdrawal. Plasma glucose should be measured repeatedly during the period of observation. • If symptoms occur and hypoglycemia is documented (plasma glucose <55 mg/dL [3 mmol/L]), the other tests should be performed( Insulin, C-peptide,Beta-hydroxybutyrate,Proinsulin Sulfonylurea and meglitinide screen) • If this approach causes neither symptoms nor hypoglycemia and if clinical suspicion remains high, the patient should undergo a 72-hour fast. 3.Postprandial evaluation: If symptoms of hypoglycemia typically occur within five hours after eating, patients should be evaluated in the postprandial state (mixed meal test). 4.Mixed meal diagnostic test: the patient consumes a meal that usually leads to symptoms and is then observed for up to five hours . • Samples are collected for plasma glucose, insulin, C-peptide, and proinsulin prior to ingestion of the meal and every 30 minutes thereafter for five hours. If severe symptoms occur prior to five hours, samples for the above lab tests should be collected before the administration of carbohydrates (to assess for correction of symptoms). All glucose samples are sent for analysis. • The samples for insulin, C-peptide, and proinsulin should be analyzed only in those samples in which plasma glucose is <60 mg/dL (3.3 mmol/L). • If Whipple's triad is demonstrated, sulfonylureas, meglitinides, and antibodies to insulin should also be measured. 5. 72-hour fast : • Normal subjects do not have symptomatic hypoglycemia after a prolonged fast because of a hormonally mediated increase in glucose production. • Gluconeogenesis accounts for approximately 50% of glucose production after an overnight fast and for almost all glucose production after 42 hours or more of fasting . • The prolonged fast will result in hypoglycemia only if there is a defect in the ability to maintain normoglycemia due, for example, to an excess of insulin. PROCEDURE 1) Date the onset of the fast at the time of the last intake of calories. Discontinue all nonessential medications. 2) Allow the patient to drink beverages that are calorie and caffeine free. 3) Ensure that the patient is active during waking hours. 4) Collect blood specimens for measurement of plasma glucose, insulin, C-peptide, proinsulin, and beta hydroxybutyrate (BHOB) every six hours until the glucose concentration is below 60 mg/dL (3.3 mmol/L); at this point, the frequency of sampling should be increased to every one to two hours. Although blood is collected repeatedly, we measure insulin, C- peptide, and proinsulin only in those specimens in which the plasma glucose concentration is ≤60 mg/dL (3.3 mmol/L). Test end points and duration : 1) when the plasma glucose concentration is ≤45 mg/dL 2) the patient has symptoms or signs of hypoglycemia 3) 72 hours have elapsed 4) or when the plasma glucose concentration is ≤55 mg/dL if Whipple's triad was documented on a prior occasion Ending the fast: Three steps are performed at the end of the fast: Collect samples for plasma glucose, insulin, C-peptide, proinsulin, BHOB, and oral hypoglycemic agents 1 mg of glucagon is given intravenously and the plasma glucose measured 10, 20, and 30 minutes later The patient is fed Laboratory tests • CBC • Glucose • Insulin 7 • • • • • • • • C-peptide Beta-hydroxybutyrate Proinsulin Antibodies for insulin and its receptors Sulfonylurea and meglitinide screen Electrolytes, BUN/Cr, UA liver function tests, cortisol and thyroid levels , growth hormone level Other tests: ECHO, ECG, CXR , CT and MRI Diagnosis of Hypoglycemia For diabetes patients: 1. Check the blood glucose levels whenever Patient feel any of the symptoms listed above. 2. If the blood glucose reading is below 70 mg/dL, eat fast-acting carbohydrates. For non-diabetes patients: 1. If Patient don’t have diabetes but still have the symptoms of hypoglycemia, consult a doctor. 2. The doctor will try to find out the cause by examining Patient’smedical history and symptoms. He will also consider Patient’sfamily history and look out for the possibility of Heredity. 3. Blood tests are done if necessary. Blood tests may include glucose levels, hepatitis, deficiency of hormones. Treatment Hypoglycemia requires quick attention as low blood sugar levels can cause problems like seizures and brain failure. Therefore, general treatment aims to bring the blood sugar levels to normal through food or medicines. The treatment for hypoglycemia varies as per the patient’s condition. Treatment For Early Symptoms Of Hypoglycemia: Early Hypoglycemia treatment is based on the 15-15 rule. Follow these simple steps to normalise Patient’sblood sugar levels▪ When Patient have early signs of hypoglycemia, eat 15 grams of food like a fast-acting carbohydrate. Due to fast-acting carbohydrates, blood glucose levels increase. ▪ After 15 minutes check the blood glucose levels again. ▪ If blood glucose levels are still below the normal, eat again 15 grams of carbohydrate. Repeat the above simple steps 1 to 3 times till Patient’s blood sugar levels are normal. Eat normal meals to maintain the blood sugar levels. ▪ ▪ ▪ Some fast-acting carbohydrates are Glucose tablets, raisins, 1 tablespoon honey, jelly beans, 1 tablespoon corn. Don’t eat complex carbohydrates or chocolates as they take time to digest. Due to which the rise of blood sugar levels is very slow. If the condition remains the same even after repeating the above steps 3 times, consult the doctor. Treatment For Severe Hypoglycemia: Emergency: ▪ ▪ ▪ In an emergency condition, glucagon treatment is given. Glucagon helps in releasing stored glucose from the liver into the bloodstream. Due to this blood sugar levels rise rapidly. Glucagon 1 mg is given through intramuscular (IM), or subcutaneous (SC) injection. Patient should be trained in using the Glucagon emergency kit. 8 Treatment If the Patient Is Unconscious: ▪ ▪ ▪ If the patient is unconscious intravenous glucose administration is done as per the recommended dose As the patient recovers from unconsciousness, oral glucose can be given. It is recommended to admit the patient in severe hypoglycemia condition. HYPOGLYCEMIA PREVENTION: Specific Prevention If Patient Have Diabetes: ▪ ▪ ▪ ▪ Follow the diabetes management plan, Patient and Patient’s doctor have developed. Have a personal Diet Coach who can monitor Patient’s food and help Patient regularise blood sugar levels naturally. Before changing any medicines, or food, or exercise, consult Patient’s doctor Make use of a continuous glucose monitor (CGM) kit and monitor glucose levels daily. Maintain a chart or install an app to make things easier. Always keep fast-acting carbohydrate or glucose tablets handy, to treat falling blood sugar levels Why is management of hypoglycemia important: -Hypoglycaemia can seriously affect Patient’s working capacity and reduce Patient’s performance or efficiency in Patient’s day-to-day life..“People who experience hypoglycemia persistently are often anxious about traveling or going to work for fear of getting low blood sugars at inconvenient moments. This fear can often have an effect on their day-to-day lives.”Recurrent episodes of hypoglycemia can also contribute to worsening of associated comorbidities such as retinopathy. 9 Hypoglycaemia Unawareness • • If Patient have been undergoing frequent episodes of hypoglycemia for a long period of time, then Patient’s body will slowly get used to it. Patient’s brain eventually stops emitting warning signals every time Patient’s blood sugars dip. This is called hypoglycemia unawareness and can prove dangerous. “This can be avoided by taking proper steps to prevent episodes of hypoglycemia right from the beginning of the disease.”There is no particular level at which every individual starts exhibiting symptoms of hypoglycemia as the body’s limits can vary from person to person. learn to recognize the signs in body, gives out when blood glucose levels fall. HOW IS HYPOGLYCAEMIA TREATED: It is a commonly believed notion that regulating Patient’s blood sugar levels to remain within a certain limit is a herculean task but this misconception needs to be addressed! Pateint should be properly counselled on how to treat hypoglycaemia at home in case of emergencies. “Untreated hypoglycemia can be harmful and should be managed appropriately. If Patient’s blood sugars fall below 70 mg/dl, then follow the ‘rule of 15’.” • • “Foods high in sugar content such as sugar, glucose tablets, or 1⁄2 a glass of fruit juice can be taken in order to treat hypoglycaemia in a conscious patient.” “If the person is, however, unconscious or not in a state to ingest sugar orally, then Patient will be advised to transport them to the Emergency Room (ER) immediately. They will be administered IV glucose or injected with IM glucose injections. Management of Hypoglycemia The management of hypoglycaemia can be divided into three phases: 1. acute intervention to prevent and minimize neurological damage . 2. maintenance therapy to prevent recurrence of hypoglycemia . 3. subsequent measures to search for and treat the underlying cause . Acute intervention 1. it is important if possible to obtain a blood sample for laboratory glucose measurement before glucose administration and to save serum for more sophisticated investigation if the cause of hypoglycaemia is not obvious (i.e. hypoglycaemia in a seemingly healthy, nondiabetic patient). 10 2. If the patient has a history of malnutrition or chronic alcohol abuse, intravenous (IV) thiamine at a bolus dose of 12 mg/kg should be given before initiation of glucose treatment, to avoid precipitating Wernicke’s encephalopathy. 3. The treatment for all hypoglycaemia events is the administration of glucose. 4. The route and amount of administration will depend on the glucose level as well as the patient’s level of consciousness and available access. 5. Where possible an oral carbohydrate load should be administered urgently, followed by careful blood glucose monitoring. 6. Consider the ‘rule of 15s’ during therapy (i.e. 15 g of carbohydrate will raise the glucose level about 15 mg/dl in about 15 minutes). 7. If the oral route is not possible, such as in cases of impaired consciousness or an uncooperative patient, IV glucose intramuscular (IM) or subcutaneous (SC) glucagon can be considered Oral carbohydrate 1. It is ideally provided in the form of 15-20 g (or 20-30 g if blood glucose 50 mg/dl) glucose tablets. 2. However, any form of carbohydrate that contains glucose can be used. 3. This dose of glucose will typically maintain euglycaemia for up to 2 hours, and therefore a complex carbohydrate (e.g. snack or meal) should be administered as soon as it is safe to do so. 4. Failure of the hypoglycaemia to correct within 15 minutes following one dose of glucose should lead to administration of a second dose, and occasionally a third, But failure thereafter should prompt the clinician to consider other interventions 5. The patient should be warned not to drive for at least 45 minutes after correction of hypoglycaemia. Oral treatments • are clearly inappropriate in the unresponsive patient or those who are unable to take oral medications. • In this situation, the presence of IV access allows 25-50 ml (i.e.12.5-25 g) 50% dextrose (D50) to be administered and is adequate in most circumstances. • D50 is highly irritating and should be administered through a large gauge needle into a large vein if possible and followed by a saline flush. • Larger volumes of less concentrated dextrose in IV infusions (e.g. 125 ml of 20% dextrose D20 or 250 ml of 10% dextrose [D10]) may be used to minimize irritation. • For inpatients with hypoglycaemia, D50 mixed with equal parts of water can also be given through a feeding tube if available. • • • • If IV access is not available, or is delayed, glucagon 1mg IM (or SC) can be administered, but its action is short lived. However, glucagon may not be effective in cases where gluconeogenesis is defective, such as in cases of extreme fasting, liver failure, alcohol induced hypoglycaemia or adrenal insufficiency because of glycogen depletion. Glucocorticoid replacement (after plasma is saved for cortisol testing) is required for patients with suspected adrenal insufficiency. Owing to these various issues, IV glucose remains the treatment of choice for severe hypoglycaemia. Maintenance therapy • The clinical response of hypoglycaemia to IV glucose administration should be rapid and dramatic. • Patients with hypoglycaemic coma are expected to regain consciousness and become coherent within 5-10 minutes. • However, complete cognitive recovery may be delayed for 30-60 minutes after restoration of normoglycaemia. 11 • If there is no obvious improvement in symptoms or consciousness within 10-15 minutes, alternative diagnoses (e.g. stroke or drug overdose) should be reconsidered. • A common mistake is to assume that once the glucose has been corrected, it will maintain itself. • Depending on the initial cause for hypoglycaemia, a concurrent source of glucose may need to be administered for some time. • As the effect of IV glucose is relatively transient, patients should receive an additional form of glucose along with protein (e.g. milk, cheese and crackers) to replenish hepatic glycogen stores. • If the hypoglycaemic episode is expected to be prolonged or recurrent (e.g. due to long acting insulin or sulfonylurea), an IV infusion of 5-10% dextrose (D5 or D10) should be commenced and continued as necessary. • For example sulfonylurea-induced hypoglycaemia, which due to long duration of action can cause prolonged hypoglycaemic events (especially in the elderly or in patients with renal impairment). • Octreotide, a synthetic somatostatin analogue, inhibits insulin release and has been used to treat hypoglycaemia in this context. • Various regimens are described but 50 mcg 6-8 hourly administered IV or SC are commonly used • The use of this agent should be considered in any situation where endogenous hyperinsulinaemia is apparent. Subsequent Measures • After initial stabilization, subsequent management should be directed at searching for the underlying etiology of hypoglycemia and preventing further attacks . • Once the underlying cause is established, definitive therapy should be offered. • Repeated hypoglycemia in an otherwise stable diabetic patient should alert the healthcare provider of the onset of nephropathy, concomitant Addison’s disease, hypothyroidism, hypopituitarism or interfering medications Treatment of Non-Diabetes Related Hypoglycemia • Non diabetic hypoglycemia definitive management depends on the underlying etiology. • Hypoglycemia induced by medications improves promptly once the medication is removed. • Correction of sepsis and improvement in hepatic and renal function improves hypoglycemia of the critical illness. • Deficiencies of counterregulatory hormones can be corrected with replacement of relevant hormone. Dietary changes are importance in the context of hyperinsulinaemic hypoglycemia, and the frequency and severity of episodes can be significantly reduced with frequent smaller volume meals. • Complex carbohydrates such as bread, rice and pasta should be consumed frequently. • Wherever possible, surgery to remove an insulinoma should be employed, although patient preference and significant comorbidities may preclude the use of surgery. • In these cases and in the context of NIPHS (where partial pancreatectomy can also be offered if diet and/or medical treatment fails, although may be ineffectual if diffuse nesidioblastosis is present), medical therapies should be used in the knowledge that each has significant limitations or side effects • Give additional maintenance glucose by mouth or IV. IV dextrose infusion should ensure delivery of 6 to 9 mg/kg per minute of glucose. Amounts needed vary depending upon the cause and severity of the symptomatic hypoglycemia. Once the patient is able to ingest carbohydrate safely, providing a mixed meal (including carbohydrates, such as a sandwich) is the preferred means of maintaining glucose levels. • Measure a blood glucose 10 to 15 minutes after the initial IV bolus and monitor every 30 to 60 minutes thereafter until stable (minimum of 4 hours). 12 Diazoxide is a potassium channel activator, • first developed as an antihypertensive agent, but now more commonly used in the context of hypoglycaemia due to inhibition of insulin secretion. • It is administered at a dose of 5 mg/kg/day (with higher doses in refractory cases up to 15 mg/kg/day) in two or three divided oral doses (e.g. 200-1200 mg/day) . somatostatin inhibits insulin production, and analogues can be used in any state of chronic hyperinsulinaemia. Octreotide therapy • is effective in reducing hypoglycaemia in over 50% of patients with an insulinoma and can be administered as a long-acting formulation. • Octreotide is commenced at a dose of 50 mcg three times daily by SC injection, and can be titrated to a maximum dose of 500 mcg three times daily. Many other medications have been used in the management of hyperinsulinaemic hypoglycemia including: • verapamil (i.e. because calcium influx is required for insulin secretion, calcium channel blockers have been tried for treatment of hyperinsulinaemia), • a-glucosidase inhibitors (e.g. acarbose or miglitol) and glucocorticoids, but evidence supporting their efficacy is based primarily on case reports only 3. Glucocorticoid and/or GH therapy have been used with some success in patients with NICTH, for tumors which cannot be resected completely. Treatment Of Diabetes Related Hypoglycemia Evaluation of hypoglycemia in a diabetic patient is focused on intensity of glycemic control and the treatment regimen. • If hypoglycemic agents (e.g. insulin, sulfonylureas or meglitinides) are prescribed, adjustments may be required to prevent further episodes. • Patient education is vital with respect to diet, exercise, timing of medications, insulin injection sites and frequent self-monitoring of blood glucose (SMBG) or continuous glucose monitoring (CGM), to try to avoid hypoglycemia. • The patient should be concerned about the possibility of developing hypoglycaemia when the SMBG is falling rapidly or is ≤70 mg/dl. • In the UK, it has been recommended that ‘four is the floor’ for blood glucose targets in order to limit hypoglycaemia frequency. • For non critically ill patients, glycemic goals are premeal and random blood glucose levels <140 mg/dl and<180 mg/dl respectively If intensive insulin therapy is used in ICU patients a target blood glucose range of 140-200 mg/dl Summary Of Management • Obtain blood glucose concentration as soon as possible (usually with a meter and strips, if available): • For symptomatic patient known to have diabetes and with a low glucose value, <70 mg/dL, administer treatment. If a glucose test cannot be performed, do not delay. Treat as if hypoglycemia has been confirmed. • If the glucose is low (<55 mg/dL) and the patient is a not a diabetic, draw blood for glucose, insulin, Cpeptide, and an oral hypoglycemic agent screen and then treat • Do not delay treatment if symptomatic hypoglycemia is suspected but rapid blood glucose measurement is not available or blood for diagnostic studies cannot be collected • If the patient is conscious and able to drink and swallow safely (ie, alert enough to do so and with gag reflex intact), administer a rapidly- absorbed carbohydrate (eg, 3 to 4 glucose tablets or a tube of gel with 15 grams, 4 to 6 oz. fruit juice or non-diet soda, or a teaspoon of honey or table sugar). 13 • • • • • • If the patient has altered mental status, is unable to swallow, or does not respond to oral glucose administration within 15 minutes, give an IV bolus of 12.5 to 25 g of glucose (25 to 50 mL of 50 percent dextrose). Measure a blood glucose 10 to 15 minutes after the IV bolus. Readminister 12.5 to 25 grams of glucose as needed to maintain the blood glucose above 80 mg/dL. If glucose cannot be given by parenteral or oral routes, give glucagon 1 mg IM or SQ. Response may be transient and should be followed by careful glucose monitoring and oral or intravenous glucose administration Give additional maintenance glucose by mouth or IV. IV dextrose infusion should ensure delivery of 6 to 9 mg/kg per minute of glucose. Amounts needed vary depending upon the cause and severity of the symptomatic hypoglycemia. Once the patient is able to ingest carbohydrate safely, providing a mixed meal (including carbohydrates, such as a sandwich) is the preferred means of maintaining glucose levels. Measure a blood glucose 10 to 15 minutes after the initial IV bolus and monitor every 30 to 60 minutes thereafter until stable (minimum of 4 hours). Complications 1-recurrent/persistent psychosocial morbidity(Emotional liability , irritability, depression). 2-Fear of hypoglycemia-barrier for diabetic control. 3-Seizure 4-permanent neurologic deficit (including cognitive impairment) 5-Coma 6-Death • • • If Patient’s blood sugars fall below 70 mg/dl, then follow the ‘rule of 15’.” “Foods high in sugar content such as sugar, glucose tablets, or 1⁄2 a glass of fruit juice can be taken in order to treat hypoglycaemia in a conscious patient.” “If the person is, however, unconscious or not in a state to ingest sugar orally, then Patient will be advised to transport them to the Emergency Room (ER) immediately. They will be administered IV glucose or injected with IM glucose injections.” NURSING MANAGEMENT: Nursing Diagnosis: Unstable Blood Glucose Level related to insufficient checking of blood sugar levels and lack of compliance to proper diabetes management secondary to hypoglycemia as evidenced by fatigue and tremors. Desired Outcome: The patient must have a blood sugar level reading of less than 180 mg/dL, fasting blood glucose levels of less than 140 mg/dL, and a hemoglobin A1C (HbA1C) level of less than 7%. Nursing Interventions for Hypoglycemia Rationale Assist the patient in recognizing eating habits that should be changed. This intervention serves as the foundation for highly personalized diet recommendations related to the clinical health status contributing to blood glucose fluctuations. Consult a registered nutritionist and dietician A personalized meal plan depends on the patient’s body, muscle mass, blood sugar levels, activity levels, and health 14 for personalized diet recommendations. status. Adjustments in the patient’s diet will help to control blood glucose levels. Educate the patient who is enduring hypoglycemia about raising blood glucose levels. Throughout most instances of hypoglycemia, consuming foods can raise blood glucose levels. People who have diabetes should consume an incredibly quickly absorbed source of carbohydrates. Discuss to the patient the significance of a balanced diet and regular exercise. Strength training helps in balancing glucose levels by expediting glucose uptake into cells. The patient must comprehend the correlation between workouts, food consumption, and blood sugar levels. Refer the patient to an exercise physiologist, physiotherapist, or cardiac rehabilitation nurse for particular exercise guidelines. Specialized workouts can be prescribed based on the patient’s physical capability. This intervention will help the patient know the appropriate workout for his condition. Hypoglycemia Nursing Care Plan 2 Fatigue • Nursing Diagnosis: Fatigue related to severely diminished metabolic energy production and changes in body chemistry, such as a deficiency of insulin secondary to hypoglycemia as evidenced by immense exhaustion, failure to sustain daily routines, reduced productivity, deficient ability to focus, lethargy, and reduced interest in surroundings. • Desired Outcome:The patient will learn to demonstrate an increase in energy and prove an enhanced ability to engage in desired daily activities. Nursing Interventions for Hypoglycemia Rationale Explain the need for activity with the patient. Strategize the patient’s routine and identify activity that causes exhaustion—however, alternate activity intervals with rest periods and uninterrupted sleep. Even if the patient typically feels too weak, proper education about the essential things can motivate them to increase their activity level. On the other hand, the patient must rest between activities to avoid excessive fatigue. Discuss energy-saving techniques for bathing, transferring, and other activities to the patient. The patient will be able to achieve more with minimal energy consumption. Advise the patient to do deep breathing techniques and help ensure they have enough air circulation. This intervention aids in stress relief and adequate oxygenation. Make provisions for comfort and safety for the This intervention aims to avoid injury while participating 15 patient. in physical activity. As considered acceptable, boost the patient’s involvement in activities of daily living (ADLs). This approach increases self-esteem, assertiveness, and tolerance. Hypoglycemia Nursing Care Plan 3 Risk for Disturbed Sensory Perception • Nursing Diagnosis: Risk for Disturbed Sensory Perception related to intrinsic chemical changes such as glucose, insulin, or electrolyte insufficiency secondary to hypoglycemia as evidenced by leg or foot hyperesthesia, discomfort, or loss of sensation. • Desired Outcome: This intervention aims to maintain normal mentation levels while also acknowledging and trying to compensate for emerging sensory impairments. Nursing Interventions for Hypoglycemia Rationale When the level of consciousness (LOC) is impaired, ensure the protection of the patient by preventing or reducing restraints as needed. Lower the bed and pad the bed rails if the patient is highly prone to convulsions. Patients with impaired sensory perceptions are more vulnerable to injury, particularly at night, and safety measures must be taken as needed. Seizure precautions must be followed as required to prevent severe trauma, aspiration, and falls. Keep an eye on the following laboratory values: Blood glucose levelsSerum osmolalityHb/Hct (Hemoglobin or Hematocrit Test)BUN/Cr (Blood Urea Nitrogen or Creatinine Test) Imbalances can hinder judgment. It should be noted that if the fluid is replaced too rapidly, excess water may fill brain cells and cause an alteration in the level of consciousness. Educate the patient about the treatment regimen for Diabetic Ketoacidosis (DKA) as advised. . Once the hyperosmolar state is rectified, any modifications in thought processes typically ameliorate the possibilities for seizure activity Examine the patient and look into reports of hyperesthesia, discomfort, or loss of sensation in the feet or legs. Examine for lesions, inflamed areas, trigger points, and a loss of pedal pulses. Peripheral neuropathies can produce catastrophic discomfort, disturbance of sensory perception, increased risk of subcutaneous injury, and loss of balance. Maintain as much consistency as possible in the patient’s daily routine. As much as probable, encourage active participation of the patient in activities of daily living (ADLs). This method aids in keeping the patient in touch with reality and oriented to his or her surroundings. • Hypoglycemia Nursing Care Plan 4 16 Deficient Knowledge • Nursing Diagnosis: Deficient Knowledge related to unfamiliarity with proper insulin injection and lack of comprehension of the signs and symptoms of the condition secondary to hypoglycemia as evidenced by the emergence of avoidable complications. • Desired Outcome: The patient will learn about the proper adherence to the treatment regimen and avoid acquiring severe complications. Nursing Interventions for Hypoglycemia Rationale Confirm that the patient understands and knows the procedure and frequency of home monitoring of blood glucose. Monitoring gives information on the extent of glucose control and reveals the necessity for adjustments in insulin dosage. Educate the patient to strictly follow a diet low in refined sugar, low in fat, and high in fiber and whole grains. . A low-fat and high-fiber diet minimizes cholesterol and triglyceride levels. Three daily meals and a midnight snack are advised. Simple and refined sugars should be minimized, and substantial carbohydrates, such as grains and rice, should be maintained Educate the patient on managing hypoglycemia with crackers, a snack, or administering glucagon. A carbohydrate snack should be consumed to treat hypoglycemia. Examples are teaspoons of sugar, hard candies, soda, fruit juice, or commercially prepared glucose tablets. If the patient is unconscious, a nurse or caregiver should administer glucagon intramuscularly (IM) or subcutaneously (SC). Teach the patient how to acknowledge hypoglycemia symptoms. Tremors, perspiration, anxiousness, lack of strength, hunger, and alteration in level of consciousness are all manifestations of hypoglycemia. Hypoglycemia happens when blood glucose levels fall below 60 mg/dL. It is beneficial to explain to patients that hypoglycemia occurs with too much insulin, not enough food, too many oral hypoglycemic medications, or too much physical activity. Provide the patient with written diabetes management information that he or she can review. This method fortifies knowledge about the condition while conveying the most important information possible. 17 Hypoglycemia Nursing Care Plan 5 Risk for Injury • Nursing Diagnosis: Risk for Injury related to neurological effects, sensory loss, and mobility impairment secondary to hypoglycemia as evidenced by changes in muscle control and gait, as well as seizures and alterations in level of consciousness. • Desired Outcome: These interventions aim to prevent the patient from any injury brought about by the health condition. Nursing Interventions for Hypoglycemia Rationale Examine the patient for factors that increase the risk of injury. This intervention aims to provide the baseline information necessary for the growth of individualized care. The variety of factors identified increases the risk of injury. Examine the patient for signs of infection. Infection may be the catalyst for inevitable amputation. Pain and tenderness symptoms may be absent due to neuropathy. Therefore, examine the area for erythema, drainage, and inflammation. Examine the presence of edema. Edema is a significant risk factor for ulceration. The decline of vasomotor reflexes and swelling in the foot are symptoms of autonomic neuropathy. Advise the patient to wear safety footwear at all times and never go barefoot. This approach is beneficial since keeping the feet covered prevents foot injuries. Take note of any callus formation or corns. Pressure on bony prominences causes callus formation, resulting in skin breakdown formation. Prevention In General: To prevent hypoglycemia, follow these steps: ▪ ▪ ▪ ▪ ▪ Monitor Patient’s blood glucose level several times a week or twice a day. Avoid postponing or skipping a meal or snack. Follow a proper meal plan. Avoid using supplements to fulfill Patient’s nutrient requirements. Take help from a Diet Coach who can provide Patient with a personalised Diet Plan that can be followed for life. Measure the dose of medicines carefully, and take it on time. As we read above, skipping an Insulin dose or overdose of the same can lead to serious health problems. In case of increased physical activity, adjust Patient’smedications and meals depending on the requirement. In the Diabetes Reversal Program, Diet Coach and Fitness Coach work together to give Patient the best plan that helps in getting Patient’sblood sugar to normal levels. Carry a medical identification bracelet to help others know that Patient have diabetes, in case of emergency. 18 Last But Not Least To safeguard Patient self from the unpleasant symptoms and negative consequences of hypoglycemia, follow the two simple rules: Practice good diabetes management plan. Reverse Diabetes naturally at the comfort of Patient’s home. ▪ Monitor Patient’s blood sugar level as often as possible. ▪ Keep a simple carbs rich diet like glucose tablets, an apple, a banana, an orange, etc. with Patient. Consume it whenever Patient experience any symptom of Hypoglycemia. -There are patients who have successfully reversed diabetes by making simple modifications that ensure sustained results. Sticking to a healthy regimen has helped them reduce or completely eliminate Insulin and other medications, which looked impossible before they got on a call with us. “It’s never too early nor too late to work towards being the healthiest Patient.” ▪ How will you EDUCATE the patient/family? Since hypoglycaemia often occurs with diabetes, you want to ensure all patients with diabetes receive adequate education on their disease process, dietary modifications, how to test their blood sugar, and how to take medications. In addition: • Teach the patient/family the signs of hypoglycaemia. If the patient takes a beta blocker, ensure they understand their signs/symptoms may not be as noticeable. • Teach the family not to place anything into the mouth of an unconscious patient. • Teach the patient/family the importance of carrying a glucagon kit. • Teach the patient to always carry a fast-acting carbohydrate such as glucose tablets. • Teach the patient/family which foods/beverages to use in cases of hypoglycemia and the carbohydrate counts of each. For example, 15 g carbohydrate is equal to 4 oz juice, 8 oz skim milk or 6 saltine crackers. 30 g carbohydrate is equal to 8 oz juice or 2 tablespoons jelly or sugar. • If the patient is taking acarbose, ensure they understand to use the glucose gel for treatment as sugary foods/beverages will not be effective. • Teach the patient to keep a log of their blood sugar levels and symptoms. Over time, with repeated incidences of hypoglycemia, the body will stop producing the classic signs/symptoms making the risk for life-threatening hypoglycemia very real. If the patient’s symptoms decrease or are absent even with low levels, this is called hypoglycemia unawareness and is cause for serious concern. These individuals may benefit from a continuous glucose monitor or even a diabetic alert dog. 19 Your key takeaways for the nursing management of hypoglycemia are: • Symptoms are shakiness, irritability, diaphoresis and tachycardia • Patients taking beta blockers may not show outward signs of hypoglycemia • Treatment is glucose (modality will vary based on blood glucose level, alertness of patient, and facility protocol) • 15 g carbohydrate is equal to 4 oz juice, 8 oz skim milk, or 6 saltine c rackers • Recheck blood sugar after 15 minutes and repeat as necessary • Patients should carry glucagon kit and fast-acting carbohydrates at all times. MANAGEMENT OF PATIENT WITH KETOACIDOSIS Introduction Diabetic ketoacidosis (DKA) is not a rare presentation to hospital, despite being an entirely preventable condition. DKA is caused by a decrease in effective circulating insulin associated with elevations in counter-regulatory hormones. This potentially life-threatening complication of type 1 diabetes mellitus (T1DM) is frequently mismanaged, leading to morbidity and increased length of stay. Mortality rates have fallen significantly in the past 20 years to <1%.1 Advances in near-patient testing technology have improved patient care, by facilitating rapid diagnosis and closer monitoring of treatment response. National Diabetes Inpatient Audits also found that a concerning number of people develop DKA while already in hospital.2 Causes of DKA • New diagnosis of T1DM. • Poor concordance with insulin treatment. • Inadequate insulin therapy in hospital. • Infection: commonly chest, urinary tract, skin. • Acute coronary/vascular event. New diagnosis of diabetes presenting with DKA: is it T1DM It is fortunately becoming less common to see DKA as the first presentation of T1DM, although it remains a frequent occurrence in the paediatric population. In most cases, if the first presentation of diabetes is with DKA, it is likely that the underlying diagnosis is T1DM, especially if the patient has a short history of symptoms, young age and normal BMI. However, some individuals with type 2 diabetes mellitus (T2DM) may develop DKA, socalled ketosis-prone type 2 or ‘Flatbush’ diabetes. This may relate to genetic factors: it is more commonly seen in those of African-Caribbean ethnicity, plus triggers such as acute intercurrent illness or metabolic stress. The absence of typical autoantibodies associated with a diagnosis of T1DM (glutamic acid decarboxylase, insulin autoantibody 2, zinc transporter 8) raises the suspicion of T2DM; however, results typically take >1 week so are not useful in acute management. Any patient presenting with DKA should initially be assumed to have T1DM and be treated accordingly. 20 DKA management In 2010 (updated 2013), the Joint British Diabetes Societies published a national guideline for managing DKA, in an effort to reduce variation in DKA management guidelines between hospitals, and thus hopefully improve standard of care.4 Readers are directed to this document for full details; the key points are summarised below. Key components of DKA management The most important initial therapeutic intervention in DKA is appropriate fluid replacement followed by insulin administration. The main aims for fluid replacement are: • restoration of circulatory volume • clearance of ketones • correction of electrolyte imbalance. -Insulin should be administered intravenously and given at a fixed rate using a weight-based formula: 0.1 units per kilogram body weight per hour. It may be necessary to estimate the patient’s weight; treatment should not be delayed waiting for an accurate weight. -Fixed rate intravenous insulin infusion (FRIII) not only reduces blood glucose levels, but just as importantly, suppresses further ketogenesis, as well as correcting the electrolyte disturbance. Frequent biochemical monitoring is required: glucose, capillary ketones, venous pH, and serum potassium. It is not necessary to use arterial blood to assess acid-base status; venous sampling is sufficient as the difference between arterial and venous pH/HCO3 is not significant enough to influence diagnosis or management of DKA. -Metabolic treatment targets • Reduction of the blood ketone concentration by 0.5 mmol/L/hour. • Increase the venous bicarbonate by 3.0 mmol/L/hour. • Reduce capillary blood glucose by 3.0 mmol/L/hour. • Maintain potassium between 4.0 and 5.5 mmol/L. If these targets are not achieved, then the FRIII rate should be increased. As clearing ketones is as important as normalising blood glucose, it is often necessary to give intravenous 10% dextrose, to avoid hypoglycaemia and allow continued FRIII to suppress ketogenesis; start 10% dextrose when the blood glucose falls below 14.0 mmol/L. It is important to continue 0.9% sodium chloride solution to correct circulatory volume, ie it is quite often necessary to infuse these solutions concurrently. Resolution of DKA • pH >7.3 units • Bicarbonate >15.0 mmol/L • Blood ketone level <0.6 mmol/L Troubleshooting If DKA is not resolving and the treatment targets are not being achieved, check cannula patency and placement. Confirm the correct rate of intravenous infusions (FRIII and fluids) has been administered. Look again for 21 concomitant pathology, eg intra-abdominal sepsis, myocardial infarction. Consider if insulin resistance is likely, eg obesity, concurrent steroid therapy; increase rate of FRIII. Reassess fluid status, and consider increasing rate of intravenous (IV) fluids. Continuing long acting subcutaneous insulin The patient’s basal (long acting) analogue insulin (eg Levemir/detemir, Lantus/glargine, Tresiba/degludec) should be continued alongside the FRIII to prevent rebound hyperglycaemia when IV treatment is stopped. Individuals with a new diagnosis of T1DM presenting with DKA should be started on basal insulin as soon as possible, and IV insulin continued until there is some basal subcutaneous (SC) insulin on board. For conversion to SC insulin in newly diagnosed patients, seek specialist input. Stopping FRIII Ideally all patients presenting with DKA will have early specialist review. Depending on the cause of the DKA episode, it may be appropriate to simply restart the pre-DKA SC insulin regimen. However, if pre-admission glycaemic control was suboptimal (high HbA1c, recurrent hypoglycaemia) then a medication review is advisable. Once DKA has resolved and patient is able to eat and drink, SC insulin therapy can be restarted. It is important that the intravenous insulin infusion is not discontinued until at least 30–60 minutes after the administration of the SC insulin dose taken with a meal. The patient’s basal insulin should have been continued alongside the FRIII. If the basal insulin had been stopped in error, the insulin infusion should not be stopped until some form of background insulin has been given, eg a stat dose of Insulatard at half the patient’s usual daily dose of basal insulin. If the patient was on twice daily fixed-mix insulin (eg NovoMix 30), restart their usual SC insulin either before breakfast or before the evening meal. Continue IV insulin infusion until 30–60 minutes after the SC insulin was given. Decisions regarding SC insulin treatment (what regimen/which insulin/what doses) to use in the newly diagnosed patient should be on the advice of the diabetes specialist team. If DKA has resolved but patient is not yet ready to eat and drink, then switch to a variable rate insulin infusion and IV fluids according to fluid status. DKA in specific situations Pregnancy DKA in pregnancy is a threat to both mother and fetus, with a risk of fetal demise as ketones are toxic to fetus. Women should be managed on the delivery suite or the high dependency unit with joint medical and obstetric care. Remember that DKA may present as abdominal pain and can occur with only very modest elevation of glucose levels in women. Manage as per standard DKA protocol, with early obstetrics review and fetal monitoring if appropriate to stage of pregnancy.5 Use booking weight for initial FRIII dose calculation; however, as pregnancy is a state of insulin resistance, the insulin infusion rate may need upwards titration. Occasionally, pregnancy is the precipitant for DKA, and all women of reproductive age presenting with DKA should have a pregnancy test. Pump users People using insulin pump therapy, also known as CSII (continuous subcutaneous insulin infusion) do NOT take any long acting insulin. Hence, if there is any interruption to insulin delivery (eg if the cannula is blocked or dislodged) hyperglycaemia and then ketoacidosis can develop very quickly, unless the problem is identified and rectified, eg by re-siting the cannula, changing the tubing, or starting alternative insulin such as an intravenous 22 infusion. If a pump user develops DKA, CSII becomes unreliable because of altered tissue perfusion affecting insulin absorption. Pump therapy should be temporarily discontinued and standard DKA treatment started. Once DKA has resolved, CSII can be restarted at the patient’s usual basal rate, but continue intravenous insulin infusion until a meal bolus has been given. DKA in association with sodium-glucose cotransporter-2 inhibitors Sodium-glucose cotransporter-2 inhibitors (SGLT2is) such as empagliflozin are the newest class of oral therapies used for the treatment of T2DM, licensed in the UK since 2013. SGLT2is reversibly inhibit sodium-glucose cotransporter 2 in the renal proximal convoluted tubule, which reduces glucose reabsorption and thus increases urinary glucose excretion. By 2015, post-marketing surveillance identified cases of serious DKA (in some cases, fatal) in patients taking an SGLT2 inhibitor. Some cases related to off-label use, or occurred in people with T1DM incorrectly diagnosed as T2DM. Presentation of DKA may be atypical with patients having so called euglycemic DKA: only moderately elevated blood glucose levels yet marked ketonaemia/acidosis. If diagnosed with DKA, SGLT2i treatment should immediately be discontinued, and not restarted unless another cause for DKA has been found and resolved. To reduce the risk of DKA, any patient admitted for major surgery or acute serious illnesses should discontinue SGLT2i until recovered. Preventing recurrent DKA A large percentage of acute hospital admissions with DKA occur in a small cohort of individuals with T1DM. These are often young people with psychosocial challenges, and poor concordance with insulin therapy is common. Support and education are the cornerstones of management rather than a need for a different insulin regimen. Access to psychology services for people with T1DM is suboptimal in many areas, despite NICE guidance. Referral to diabetes specialist teams should happen within 24 hours of any admission with DKA, with specialist review prior to discharge. Treatment The therapeutic goals for diabetic ketoacidosis consist of improving circulatory volume and tissue perfusion, reducing blood glucose and serum osmolality toward normal levels, clearing ketones from serum and urine at a steady rate, correcting electrolyte imbalances and identifying precipitating factors. A suggested flow sheet for monitoring therapeutic response is provided in Figure 3. FIGURE 3. 23 Flow Sheet for Monitoring Diabetic Ketoacidosis 24 FLUID REPLACEMENT The severity of fluid and sodium deficits (Table 1) is determined primarily by the duration of hyperglycemia, the level of renal function and the patient's fluid intake. Dehydration can be estimated by clinical examination and by calculating total serum osmolality and the corrected serum sodium concentration. Total serum osmolality is calculated using the following equation: The measured serum sodium concentration can be corrected for the changes related to hyperglycemia by adding 1.6 mEq per L (1.6 mmol per L) to the measured sodium value for every 100 mg per dL (5.6 mmol per L) of glucose over the normal baseline of 100 mg per dL.1,8 Corrected serum sodium concentrations of greater than 140 mEq per L (140 mmol per L) and calculated total osmolalities of greater than 330 mOsm per kg of water are associated with large fluid deficits.4,6 Calculated total osmolalities are correlated with mental status, in that stupor and coma typically occur with an osmolality of greater than 330 mOsm per kg of water.1,6 The initial priority in the treatment of diabetic ketoacidosis is the restoration of extra-cellular fluid volume through the intravenous administration of a normal saline (0.9 percent sodium chloride) solution. This step will restore intravascular volume, decrease counterregulatory hormones and lower the blood glucose level.9 As a result, insulin sensitivity may be augmented. In patients with mild to moderate volume depletion, infusion rates of 7 mL per kg per hour have been as efficacious as infusion rates of 14 mL per kg per hour.10 The subsequent administration of a hypotonic saline (0.45 percent sodium chloride) solution, which is similar in composition to the fluid lost during osmotic diuresis, leads to gradual replacement of deficits in both intracellular and extracellular compartments. When the blood glucose concentration is approximately 250 mg per dL (13.9 mmol per L), glucose should be added to the hydration fluid (i.e., 5 percent dextrose in hypotonic saline solution). This allows continued insulin administration until ketonemia is controlled and also helps to avoid iatrogenic hypoglycemia. Another important aspect of rehydration therapy in patients with diabetic ketoacidosis is the replacement of ongoing urinary losses. INSULIN THERAPY Modern management of diabetic ketoacidosis has emphasized the use of lower doses of insulin. This has been shown to be the most efficacious treatment in both children and adults with diabetic ketoacidosis.11–14 The current recommendation is to give low-dose (short-acting regular) insulin after the diagnosis of diabetic ketoacidosis has been confirmed by laboratory tests and fluid replacement has been initiated. It is prudent to withhold insulin therapy until the serum potassium concentration has been determined. In the rare patient who presents with hypokalemia, insulin therapy may worsen the hypokalemia and precipitate lifethreatening cardiac arrhythmias.1,2 Standard low-dose insulin therapy consists of an initial intravenous bolus of 0.15 unit of regular insulin per kg followed by the continuous intravenous infusion of regular insulin prepared in normal saline or hypotonic saline solution at a rate of 0.1 unit per kg per hour. 25 In clinical situations in which continuous intravenous insulin cannot be administered, the recommended initial insulin dose is 0.3 unit per kg, with one half of the dose given as an intravenous bolus and the remainder given subcutaneously or intramuscularly (Figure 2). Subsequently, regular insulin should be given in a dosage of 0.1 unit per kg per hour until the blood glucose level is approximately 250 mg per dL. If the blood glucose concentration does not fall by 50 to 70 mg per dL (2.8 to 3.9 mmol per L) in the first hour, the intravenous infusion rate should be doubled or additional intravenous 10-unit boluses of insulin should be given every hour (Figure 2). Either of these treatments should be continued until the blood glucose level falls by 50 to 70 mg per dL. Low-dose insulin therapy typically produces a linear fall in the glucose concentration of 50 to 70 mg per dL per hour.12 More rapid correction of hyperglycemia should be avoided because it may increase the risk of cerebral edema. This dreaded treatment complication occurs in approximately 1 percent of children with diabetic ketoacidosis.5 The typical presentation is onset of headache and decreased mental status occurring several hours after the start of treatment. Cerebral edema is associated with a mortality rate of up to 70 percent.15 When a blood glucose concentration of 250 mg per dL has been achieved, the continuous or hourly insulin dosage can be reduced to 0.05 unit per kg per hour. The insulin and fluid regimens are continued until ketoacidosis is controlled. This requires the achievement of at least two of these acid-base parameters: a serum bicarbonate concentration of greater than 18 mEq per L, a venous pH of 7.3 or greater and an anion gap of less than 14 mEq per L. POTASSIUM THERAPY Although the typical potassium deficit in diabetic ketoacidosis is 500 to 700 mEq (500 to 700 mmol), most patients are hyperkalemic at the time of diagnosis because of the effects of insulinopenia, hyperosmolality and acidemia.4 During rehydration and insulin therapies for diabetic ketoacidosis, the serum potassium concentration typically declines rapidly as potassium reenters the intracellular compartment. One protocol entails using insulin and intravenous fluids until the serum potassium concentration is less than 5.5 mEq per L (5.5 mmol per L). At this time, potassium chloride is added to intravenous fluids in the amount of 20 to 40 mEq per L. The exact amount of potassium that is administered depends on the serum potassium concentration. When the serum potassium level is less than 3.3 mEq per L (3.3 mmol per L), the administration of 40 mEq per L of potassium is appropriate. If the serum potassium is greater than 3.3 mEq per L but less than 5.5 mEq per L, 20 to 30 mEq per L of potassium can be administered. The goal is to maintain the serum potassium concentration in the range of 4 to 5 mEq per L (4 to 5 mmol per L).1,2,6 BICARBONATE THERAPY In general, supplemental bicarbonate therapy is no longer recommended for patients with diabetic ketoacidosis, because the plasma bicarbonate concentration increases with insulin therapy.1,4,8,16,17 Insulin administration inhibits ongoing lipolysis and ketone production and also promotes the regeneration of bicarbonate. Retrospective reviews and prospective randomized studies have failed to identify changes in morbidity or mortality with sodium bicarbonate therapy in patients who presented with a pH of 6.9 to 7.1. Therefore, the use of bicarbonate in a patient with a pH greater than 7.0 is not recommended. Furthermore, bicarbonate therapy carries some risks, including hypokalemia with overly rapid administration, paradoxic cerebrospinal fluid acidosis and hypoxia.6,15,17 Some authorities, however, recommend bicarbonate administration when the pH is less than 7.0, for the purpose of treating the possible adverse hemodynamic effects of profound acidemia. If bicarbonate is used, it should be 26 given as a nearly isotonic solution, which can be approximated by the addition of one ampule of sodium bicarbonate in 300 mL of sterile water. The bicarbonate solution is administered over a one-hour period.1,2,8 A small percentage of patients who have diabetic ketoacidosis present with metabolic acidosis and a normal anion gap. Therefore, they have fewer ketones available for the regeneration of bicarbonate during insulin administration.18 Bicarbonate therapy may be warranted in this subset of patients. PHOSPHATE THERAPY Osmotic diuresis leads to increased urinary phosphate losses. During insulin therapy, phosphate reenters the intracellular compartment, leading to mild to moderate reductions in the serum phosphate concentration. Adverse complications of hypophosphatemia are uncommon and occur primarily in patients with severe hypophosphatemia (a serum phosphate concentration of less than 1.0 mg per dL [0.32 mmol per L]). Prospective studies have indicated no clinical benefit for phosphate replacement in the treatment of diabetic ketoacidosis, and excessive phosphate replacement may contribute to hypocalcemia and soft tissue metastatic calcification. Although the replacement of phosphate per se is not routinely recommended, it may be useful to replace some potassium as potassium phosphate. One protocol is to administer two thirds of the potassium as potassium chloride and one third as potassium phosphate. The use of phosphate for this purpose reduces the chloride load that might contribute to hyperchloremic acidosis and decreases the likelihood that the patient will develop severe hypophosphatemia during insulin therapy. Immediate Post Hyperglycemic Care When diabetic ketoacidosis has been controlled, subcutaneous insulin therapy can be started. The half-life of regular insulin is less than 10 minutes. Therefore, to avoid relapse of diabetic ketoacidosis, the first subcutaneous dose of regular insulin should be given at least one hour before intravenous insulin is discontinued.1,22 A protocol for the administration of subcutaneous insulin is included in Figure 2. In patients who are unable to eat, 5 percent dextrose in hypotonic saline solution is continued at a rate of 100 to 200 mL per hour. Blood glucose levels are monitored every four hours, and regular insulin is given subcutaneously every four hours using a sliding scale (Figure 2). When patients are able to eat, multidose subcutaneous therapy with both regular (short-acting) and intermediate-acting insulin may be given. In patients with newly diagnosed diabetes, an initial total insulin dosage of 0.6 to 0.7 unit per kg per day is usually adequate to achieve metabolic control. A typical regimen is two thirds of the total daily dosage before breakfast and one third of the total daily dosage before dinner, with the insulin doses consisting of two-thirds NPH (intermediate-acting) insulin and one-third regular (short-acting) insulin. Patients with known diabetes can typically be given the dosage they were receiving before the onset of diabetic ketoacidosis. Complications of Therapy Symptomatic cerebral edema occurs primarily in pediatric patients, particularly those with newly diagnosed diabetes. No single factor predictive for cerebral edema has yet been identified. As noted previously, however, overly rapid rehydration or overcorrection of hyperglycemia appears to increase the risk of cerebral edema. Onset of headache or mental status changes during therapy should lead to consideration of this complication. Intravenous mannitol in a dosage of 1 to 2 g per kg given over 15 minutes is the mainstay of therapy. Prompt involvement of a critical care specialist is prudent. 27 Adult respiratory distress syndrome (ARDS) is a rare but potentially fatal complication of the treatment of diabetic ketoacidosis.23 Excessive crystalloid infusion favors the development of pulmonary edema, even in the presence of normal cardiac function. Patients with an increased alveolar to arterial oxygen gradient (AaO2) and patients with pulmonary rales on physical examination may be at increased risk for ARDS. Monitoring of oxygen saturation with pulse oximetry may assist in the management of such patients. Hyperchloremic metabolic acidosis with a normal anion gap typically persists after the resolution of ketonemia. This acidosis has no adverse clinical effects and is gradually corrected over the subsequent 24 to 48 hours by enhanced renal acid excretion.8,18 The severity of hyperchloremia can be aggravated by excessive chloride administration in hydration fluids. Resource Utilization in Diabetic Ketoacidosis No randomized prospective studies have evaluated the optimal site of care for patients with diabetic ketoacidosis. The response to initial therapy in the emergency department can be used as a guideline for choosing the most appropriate hospital site (i.e., intensive care unit, step-down unit, general medical ward) for further care. Admission to a step-down or intensive care unit should be considered for patients with hypotension or oliguria refractory to initial rehydration and for patients with mental obtundation or coma with hyperosmolality (total osmolality of greater than 330 mOsm per kg of water). Most patients can be treated in step-down units or on general medical wards in which staff members have been trained in on-site blood glucose monitoring and continuous intravenous insulin administration. Milder forms of diabetic ketoacidosis can be treated in the emergency department using the same treatment guidelines described in this review. Successful outpatient therapy requires the absence of severe intercurrent illness, an alert patient who is able to resume oral intake and the presence of mild diabetic ketoacidosis (pH of greater than 7.2 and a plasma bicarbonate concentration of greater than 10 mEq per L).24 With the use of standardized written treatment guidelines and flow sheets for monitoring therapeutic response, the mortality rate for patients with diabetic ketoacidosis is now less than 5 percent.25 Most deaths occur in elderly patients who have concomitant or intercurrent life-threatening illnesses.1–4,6 Similar outcomes for the treatment of diabetic ketoacidosis have been observed in both community and training hospitals. These outcomes have not been altered by the specialty of the primary treating physicians (e.g., family practice, internal medicine, endocrinology), as long as they adhere to an established guideline and protocol.26 Prevention An educational program should include sick-day management instructions (i.e., for any illness that alters routine care), including the use of short-acting insulin, blood glucose and urinary ketone monitoring, and the use of a liquid diet containing carbohydrates and salt. Patients should not discontinue insulin therapy when they are ill, and they should contact their physician early in the course of illness. Indications for hospitalization include greater than 5 percent loss of body weight, respiration rate of greater than 35 per minute, intractable elevation of blood glucose concentrations, change in mental status, uncontrolled fever and unresolved nausea and vomiting. Address correspondence to Abbas E. Kitabchi, Ph.D., M.D., Division of Endocrinology/Department of Medicine, University of Tennessee, Memphis, College of Medicine, 951 Court Ave., Room 335M, Memphis, TN 38163. Reprints are not available from the authors. 1. Kitabchi AE, Wall BM. Diabetic ketoacidosis. Med Clin North Am. 1995;79:9-37. 28 Key points 1. Cerebral oedema is the key life-threatening complication of DKA 2. When managing DKA, hypoglycaemia and hypo/hyperkalaemia are the other main complications which must be monitored for and treated 3. Children with DKA are deplete in total body potassium regardless of the initial serum potassium level 4. Measured serum sodium may be low due to osmotic dilution with glucose. Corrected sodium levels should be calculated and monitored Background The biochemical criteria for diagnosis of DKA are: • Serum glucose >11 mmol/L • Venous pH <7.3 or Bicarbonate <15 mmol/L • Presence of ketonaemia/ketonuria Children with hyperglycaemia (Blood glucose level (BGL) >11 mmol/L) +/- ketosis who are not acidotic can be managed with subcutaneous insulin (see Diabetes mellitus, new presentation, mildly ill). Hyperglycaemic children with altered conscious state who are not acidotic (pH of ≥7.3) and have little to no ketonaemia may have hyperglycaemic-hyperosmolar non-ketotic coma. If this is a possibility, insulin should ONLY be given after discussion with local paediatric team and/or paediatric endocrinologist. Precipitants for DKA • Inadequate insulin in a child or adolescent with known diabetes (eg missed insulin doses, insulin pump failure). • First presentation of Type 1 diabetes mellitus. • Illness. Assessment History and examination are directed towards potential precipitants, assessment of severity, and detecting complications of DKA. Assessment of Dehydration Weigh child – compare to recent weight if available. Caution • The degree of dehydration is often over-estimated in DKA, this may be compounded by peripheral shutdown due to acidosis. • Excessive fluid replacement may increase the risk of cerebral oedema. Degree of dehydration Clinical signs Assessment Percentage 29 Mild <4% No clinical signs Moderate 4-7% Easily detectable dehydration – decreased tissue turgor, poor central capillary return Severe >7% Shock - Poor perfusion, rapid pulse, hypotension Management Children and adolescents with DKA should be managed in a unit that has: • Access to laboratory services for frequent and timely evaluation of biochemical variables • Experienced nursing staff trained in monitoring and management of DKA in children and adolescents • A paediatrician, endocrinologist, or critical care specialist with training and expertise in the management of paediatric DKA. Where such expertise is not available on-site, telephone advice should be sought from the appropriate specialists Investigations Bloods - Insert an IV when taking bloods • Serum glucose • Urea, creatinine and electrolytes (sodium, potassium, calcium, magnesium, phosphate) • Venous gas (including bicarbonate) • FBE (haematocrit may be elevated as a marker of dehydration, WCC may be elevated as a stress response) • Blood ketones (bedside test, normal <0.6 mmol/L) • Consider investigations for precipitating cause: if clinical signs of infection consider septic work up • The following bloods are part of a diagnostic work up for first presentation T1DM: Insulin antibodies, GAD antibodies, ZnT8 antibodies, celiac screen (total IgA, anti-gliadin Ab, tissue transglutaminase Ab) TSH and fT4. Collect these bloods with initial blood sampling if practical. Please handover to admitting team if not done Urine • Dipstick for ketones, glucose and FWT • Culture if clinical suspicion of UTI Consider ECG if potassium results will be delayed Once DKA is confirmed, the following biochemical monitoring should be put in place to guide ongoing management. These will continue until resolution of DKA: • Hourly – BGL, bedside ketone testing • At 2 hours and 2-4 hourly thereafter – VBG, UEC, Ca, Mg, PO4 30 Severity of DKA Severity of DKA Assessed based on the more severe of these parameters: Venous pH Bicarbonate (mmol/L) Mild <7.3 <15 Moderate <7.2 <10 Severe <7.1 <5 Treatment Goals of Treatment: Sections below include: 1. Correct dehydration 2. Reverse ketosis, correct acidosis and glucose 3. Monitor for complications of DKA and its treatment: Cerebral oedema, hypo/hyperkalaemia, hypoglycaemia 4. Identify and treat any precipitating cause Airway/Breathing/Circulation See Resuscitation Supportive measures and monitoring • Nurse head up • In children with reduced conscious state monitor airway safety. Consider inserting an NGT to prevent aspiration • Keep nil by mouth until child is alert and preferably until acidosis resolves. Children can be given ice to suck on for comfort • Insert second IVC to use as a blood sampling line, take initial diagnostic bloods if not drawn with initial IVC insertion • Supplemental oxygen for children with severe circulatory impairment or shock • Cardiac monitoring – for assessment of ECG changes related to potassium levels (hyperkalaemia: peaked T waves, widened QRS, hypokalaemia: flattened or inverted T waves, ST depression, PR prolongation). See ECG Interpretation • Consider antibiotics for febrile children after obtaining appropriate cultures • Consider urinary catheter for children who are unconscious to allow strict monitoring of fluid balance. Weighing nappies can be used for strict fluid balance for children who can’t urinate on demand 31 Timeline of monitoring and management Timeline Observation/Investigation Management Presentation Assessment and ABC IV access and initial bloods including VBG 0.9% sodium chloride +/potassium Weigh child Start rehydration fluids Nurse head up Document passage of urine (ask child to void) 1 hour Fluid balance Start insulin infusion Vital signs Neurological observations BGL and bedside ketones 2 hours Fluid balance Adjust fluids if required based on glucose and electrolytes Vital signs Neurological observations, VBG, VBG and bedside ketones UEC (must check serum potassium within 1 hour of starting insulin infusion) Serum calcium, magnesium, phosphate 3 hours Fluid balance Adjust fluids if required based on glucose Vital signs Neurological observations BGL and bedside ketones 4 hours Fluid balance Adjust fluids if required based on glucose and electrolytes Vital signs Neurological observations VBG and bedside ketones UEC Serum calcium, magnesium, phosphate 5 hours Fluid balance Adjust fluids if required based on glucose Vital signs Neurological observations BGL and bedside ketones 32 6 hours Continue the following hourly: Fluid balance Vital signs Neurological observations BGL and bedside ketones Continue the following 1-2 hourly: BGL Continue the following 2-4 hourly: UEC VBG Serum calcium, magnesium, phosphate 1) Correct Dehydration Children with DKA will be dehydrated. Clinical estimates of fluid deficits are subjective and often inaccurate thus most children can initially be commenced on the “mild” or “moderate” fluid rate (see below). A 10 mL/kg 0.9% sodium chloride bolus can be given to children who are tachycardic with delayed central capillary refill. • This should be followed by a reassessment • Acidosis results in poor peripheral perfusion so use central capillary refill with vital signs to assess response to fluids Initial Fluid Replacement Commence rehydration with isotonic fluid eg 0.9% sodium chloride. • Ask child to pass urine or remove nappy • Add 40 mmol/L potassium chloride to this fluid if the serum potassium ≤ 5.5mmol/L and the child is passing urine • If anuric or serum potassium >5.5 mmol/L, do not add potassium to the fluids until this has resolved Keep nil by mouth until child is alert and acidosis has resolved. • Children can be given ice to suck on for comfort. Intravenous or oral fluids that have been given at another facility may need to be factored into the assessment and calculation of fluid deficit and replacement. 33 Initial fluid rates (mL/hr) based on degree of dehydration Weight (kg) Mild/nil dehydration (mL/hr) Moderate Dehydration (mL/hr) Severe Dehydration (mL/hr) 40 105 130 160 42 105 135 170 44 110 135 175 46 115 140 180 48 115 145 185 50 120 150 190 52 120 155 195 54 125 160 205 56 125 160 210 58 130 165 215 60 133 171 220 62 136 175 226 64 139 179 232 66 140 185 240 68 145 185 245 70 150 190 250 Fluid Adjustments Frequent monitoring of electrolytes, glucose, and osmolality will guide fluid composition and infusion rates. Fluids containing 0.9% sodium chloride should be continued for at least the first 6 hours. The three key parameters to monitor and manage are: • Glucose • Potassium • Sodium Osmolality can be calculated using the following formula: Osmolality = 2 x (serum sodium + serum potassium) + glucose + urea 34 Glucose • BGL will decrease rapidly during initial rehydration / volume expansion and continue to decrease once the insulin infusion is started • Once BGL is ≤ 15 mmol/L change fluids to 0.9% sodium chloride with 5% glucose and potassium chloride (maximum 60 mmol/L) as required • Aim to keep the BGL between 5-10 mmol/L • If BGL falls below 5mmol/L or is falling rapidly (>5 mmol/L/hour) in the range between 5-15 mmol/L and the child remains acidotic, increase the glucose content to 10% • Insulin infusion rate should only be decreased if BGL continues to fall despite glucose concentration of 10% Potassium • Children with DKA have a deficit of total body potassium however serum potassium levels may be normal, high, or low • Treatment with insulin will shift potassium to the intracellular space rapidly resulting in hypokalaemia if potassium is not replaced • Start potassium chloride at a concentration of 40 mmol/L, increase to a maximum 60 mmol/L if required to maintain serum potassium in the normal range • Once insulin is commenced, a repeat serum potassium should be measured within one hour and two to four hourly thereafter Sodium Measured sodium is depressed by the dilutional effect of hyperglycaemia Corrected sodium can be calculated with this formula: Corrected sodium = measured sodium + 0.3 x (glucose – 5.5) mmol/L ie 3 mmol/L sodium to be added for every 10 mmol/L of glucose above 5.5 mmol/L. • 0.9% sodium chloride with glucose and potassium chloride (maximum 60 mmol/L) as required should generally be used for the duration of IV rehydration • If a hypotonic solution is later thought to be required, discuss this with the paediatric endocrinologist on call. The sodium chloride content should be at least 0.45% or greater • If measured sodium does not rise as the glucose falls during treatment or if hyponatraemia develops, this usually indicates excessive fluid correction which may increase the risk of cerebral oedema • Extremes of corrected sodium should be discussed with a senior doctor early IV fluids can be ceased once pH and bicarbonate have normalised and the child is able to tolerate oral intake (this usually coincides with insulin being changed to subcutaneous injections). Phosphate • Phosphate replacement is rarely required (due to intracellular phosphate stores usually being adequate) • If phosphate levels drop below 0.32 mmol/L and/or symptoms of hypophosphataemia are present, IV phosphate replacement may be required • Calcium levels will need to be monitored if IV phosphate is administered 35 2) Reverse ketosis and correct acidosis IV rehydration should be commenced prior to starting an insulin infusion. Insulin To make up the insulin infusion: • Add 50 units of clear/rapid acting insulin (Actrapid HM or Humulin R) to 49.5 mL of 0.9% sodium chloride to form a 1 unit/mL solution. Initial insulin infusion rates • Children with DKA should generally be commenced at 0.1 units/kg/hour • A reduced infusion rate of 0.05 units/kg/hour should be used for: o Children undergoing inter-hospital transfer (limited access to biochemical monitoring) o Children less than 5 years old o Children with BGL <15 mmol/L at the time of commencement of the insulin infusion Practical points • Insulin infusion can be run as a sideline with the rehydration fluids via a three-way tap provided a syringe pump is used. Ensure the insulin is clearly labelled • Adequate insulin must be continued to clear ketones and correct acidosis • Aim to keep the blood glucose between 5-10 mmol/L by increasing the fluid glucose concentration to 10% before adjusting the insulin infusion rate • Changing from an insulin infusion to subcutaneous insulin: o Can be changed when child is alert and metabolically stable (pH>7.3, HCO3 > 15) o Best time to change to subcutaneous insulin is just prior to a meal. Discuss the dose and regimen with the consultant on call o Continue the insulin infusion for 30 minutes after the first subcutaneous injection of rapid-acting insulin, then cease 3) Monitor for complications of DKA and its treatment The most important complications of DKA and its treatment are: • Cerebral oedema • Hypoglycaemia • Hypo/hyperkalaemia • Hypo/hypernatraemia • Aspiration (if obtunded) Cerebral Oedema Background • Some degree of subclinical brain swelling is present during most episodes of DKA • Clinical cerebral oedema occurs suddenly, usually between 6-12 hours after starting therapy (range 2-24 hours) 36 • Mortality and severe morbidity rates are very high without early treatment • If cerebral oedema is suspected, this should be immediately discussed with a consultant Risk Factors • First presentation diabetes • Long history of poor control • Age <5 years old Signs • Early: Headache, irritability, lethargy, vomiting • Later: depressed consciousness, incontinence, thermal instability • Very late: bradycardia, increased BP, respiratory impairment Treatment • Nurse head up • Reduce fluid infusion rate by one-third • Contact Paediatric, Infant and Perinatal Emergency Retrievals on 1300 137 650[KH14] • Give mannitol immediately if cerebral oedema suspected – do NOT wait for cerebral imaging. • Mannitol 20% (0.2 g/mL) dose: 0.5 g/kg IV over 20 minutes (dose range: 0.25-1 g/kg), repeat if no improvement within 30-60mins. • Discuss with consultant on call and liaise with intensive care or paediatric retrieval service to discuss transfer. Hypoglycaemia A BGL of <4.0 mmol/L should be treated with additional glucose as below. • 2 mL/kg 10% glucose IV bolus (repeat if required) and change rehydration fluids to include 10% glucose with 0.9% sodium chloride, with potassium chloride (maximum 60 mmol/L) as required • Do NOT discontinue the insulin infusion • If hypoglycaemia occurs despite use of 10% glucose in preceding 2 hours – decrease insulin infusion to 0.05 units/kg/hr (0.03 units/kg/hr if previously on 0.05 units/kg/hr) • Continue with 10% glucose in fluids until BGL is stable between 5-10 mmol/L • Oral treatment for hypoglycaemia can be used if pH≥7.3 and the child is alert and able to tolerate oral intake. Use 4-5 Jelly beans or a serve of juice: 60 mL (5g carbohydrate) for children <25kg, 120 mL (10g carbohydrate) for children ≥25kg • Recheck BGL after 15-20mins and give another serve of juice or jelly beans if BGL still <4.0 mmol/L. If oral treatment has been used discuss timing of transition to sub-cutaneous insulin and oral diet with endocrinology / consultant on call 4) Identify and Treat precipitating cause Assess for underlying infections and consider antibiotics after obtaining relevant cultures if appropriate. • Overall, infective precipitants are uncommon. 37 In children with known T1DM the most common cause of DKA is omission or significant reduction in recent insulin doses. Other Things to Consider Persisting Acidosis If the acidosis is not correcting, consider the following: • Insufficient insulin to switch off ketosis (check insulin delivery) • Inadequate rehydration • Sepsis • Hyperchloraemic acidosis (secondary to IV fluids) • Salicylates or other substances that cause metabolic acidosis Rarely required Bicarbonate • Discuss with consultant endocrinologist prior to administration • ONLY appropriate for children with life threatening hyperkalaemia or requiring inotropic support • May cause paradoxical CNS acidosis • Associated with increased risk of cerebral oedema • Sodium bicarbonate dose (mmol) = 0.15 x body weight (kg) x base deficit (mmol/L) o Administer IV over 30-60 minutes with cardiac monitoring o Reassess acid-base status and beware of hypokalaemia Consider consultation with local paediatric team for All children with DKA All newly diagnosed diabetes mellitus Hyper/hyponatraemia Consider transfer when Intensive care monitoring is recommended for • Children <2 years of age • Coma • Cardiovascular compromise • Seizures • Signs of cerebral oedema • Severe acidosis (pH <7.1 or HCO3<5) The care required is beyond the level of comfort or resources of the local hospital. Children and adolescents with DKA should be managed in a unit that has: • Access to laboratory services for frequent and timely evaluation of biochemical variables • Experienced nursing staff trained in monitoring and management of DKA in children and adolescents 38 • A paediatrician, endocrinologist, or critical care specialist with training and expertise in the management of paediatric DKA. Where such expertise is not available on-site, telephone advice should be sought from the appropriate specialists • Diabetic Ketoacidosis: Diabetic ketoacidosis (DKA) results from absolute or relative deficiency of circulating insulin (Kitabchi AE, Umpierrez GE, Murphy MB, Kreisberg RA, 2006). DKA is characterized by severe depletion of water and electrolytes from both the intra and extracellular fluid compartments. Despite their dehydration, patients continue to maintain normal blood pressure and have considerable urine output until extreme volume depletion and shock occurs leading to a critical decrease in renal blood flow and glomerular filtration. Assessment: • • • • • • Perform a clinical evaluation to confirm the diagnosis and determine its cause. Carefully look for evidence of infection. Also weigh the patient. Assess clinical severity of dehydration. Assess level of consciousness using Glasgow coma scale [GCS]. Management: Laboratory blood glucose should be measured at diagnosis. An ECG monitor should be applied until the patient is stable. Consulate the Diabetes Team as soon as possible for a decision on continuing management. Transfer to subcutaneous (SC) insulin one the patient is able to eat and drink properly. Do not stop the IV insulin until SC insulin has been given. Patient with known diabetes should go back to their previous insulin regimen. If possible identify the precipitating cause of DKA. Always inform the Diabetes Team so that education can be given to reduce the risk of future episodes of DKA. Hyperosmolar non – ketotic coma (HONK): Type 2 diabetes, usually in patients over 60 years. This condition is characterized by hyperglycemia and high plasma osmolality without significant keton-uria or acidosis (Pinies JA, Cairo G, Gaztambide S, et al.1994). Assessment: Same as Diabetic ketoacidosis (DKA) but look for precipitating medical condition, e.g. sepsis, myocardial infarction etc • Management: • Management of Hyperosmolar non – ketotic coma is same as DKA . Also the insulin infusion rate should be halved as paradoxically these patients can be quite insulin sensitive. Elderly patients are more likely to need a CVP line to optimize fluid replacement. Risk of thromboembolic disease is high – anticoagulant fully if no contraindications. Most patient can be managed with oral hypoglycemic agents or diet, but recovery of insulin may take time and insulin may be required for few weeks. Hypoglycemia: Hypoglycemia is the result of a mismatch between insulin dose, food consumed, and recent exercise and is rarely, if ever, a spontaneous event (Kovatchev BP, Cox DJ, Farhy LS, Straume M, Gonder-Frederick L, Clarke WL,2003).Because it can be accompanied by unpleasant, embarrassing, and potentially dangerous symptoms and because it causes significant anxiety and fear in the patient and their caregivers, it’s occurrence is a major limiting factor in attempts to achieve near normal BG levels (Clarke WL, Gonder-Frederick A, Snyder AL, Cox DJ 1998,Cryer PE 2002). Assessment: • • • Each hypoglycaemic episode should be assessed carefully to determine its cause evaluating the insulin action profile (time of insulin administration, peak insulin action and intensity of insulin action). Check the recent food intake (timing and amount of carbohydrates eaten and peak BG effect on recent food. Also check recent physical activity (timing, duration and intensity). Also check and missed signs and symptoms of early hypoglycaemia. 39 • Management: • Management of hypoglycemia involves immediately raising the blood sugar to normal, determining the cause, and taking measures to hopefully prevent future episodes. Initially Glucose 10-20 g is given by mouth either in liquid form or as granulated sugar (2 teaspoons) or sugar lumps (Smeeks FC, 2006). If hypoglycemia causes unconsciousness, or patient is unco-operative, 50 mL of glucose intravenous (IV) infusion 20% can be given. Alternatively, 25 mL of glucose intravenous infusion 50% may be given, but this higher concentration is viscous, making administration difficult; it is also more irritant. Once the patient regains consciousness oral glucose should be administered as above. • Diabetic Ketoacidosis DKA Nursing Care Plan 1 Nursing Diagnosis: Fatigue related to decreased metabolic energy production as evidenced by overwhelming lack of energy, verbalization of tiredness, generalized weakness, blood sugar level of 11 mg/dL, and shortness of breath upon exertion Desired Outcome: The patient will demonstration active participation in necessary and desired activities and demonstrate increase in activity levels. DKA Nursing Interventions Rationales Assess the patient’s degree of fatigability by asking to rate his/her fatigue level (mild, moderate, or severe). Explore activities of daily living, as well as actual and perceived limitations to physical activity. Ask for any form of exercise that he/she used to do or wants to try. To create a baseline of activity levels, degree of fatigability, and mental status related to fatigue and activity intolerance. Encourage progressive activity through self-care and exercise as tolerated. Explain the need to reduce sedentary activities such as watching television and using social media in long periods. Alternate periods of physical activity with rest and sleep. To gradually increase the patient’s tolerance to physical activity. Teach deep breathing exercises and relaxation techniques. Provide adequate ventilation in the room. To allow the patient to relax while at rest. To allow enough oxygenation in the room. Refer the patient to physiotherapy / occupational therapy team as required. To provide a more specialized care for the patient in terms of helping him/her build confidence in increasing daily physical activity. Diabetic Ketoacidosis DKA Nursing Care Plan 2 Nursing Diagnosis: Risk for Fluid Volume Deficit due to osmotic diuresis secondary to DKA Desired Outcome: The patient will demonstrate adequate hydration and balanced fluid volume 40 DKA Nursing Interventions Rationales Assess vital signs and signs of dehydration. Hyperglycemia may cause Kussmaul’s respirations and/or acetone breath. Hypotension and tachycardia may result from hypovolemia, or low levels of intravascular volume. Commence a fluid balance chart, monitoring the input and output of the patient. To monitor patient’s fluid volume accurately and effectiveness of actions to monitor signs of dehydration. Start intravenous therapy as prescribed. Encourage oral fluid intake of at least 2500 mL per day if not contraindicated. To replenish the fluids lost from polyuria and to promote better blood circulation around the body. Educate the patient (or guardian) on how to fill out a fluid balance chart at bedside. To help the patient or the guardian take ownership of the patient’s care, encouraging them to drink more fluids as needed, or report any changes to the nursing team. Monitor patient’s serum electrolytes and recommend electrolyte replacement therapy (oral or IV) to the physician as needed. Sodium is one of the important electrolytes that are lost when a person is passing urine. Hyponatremia or low serum sodium level may cause brain swelling. Diabetic Ketoacidosis DKA Nursing Care Plan 3 Nursing Diagnosis: Risk for Infection Desired Outcome: The patient will be able to avoid the development of an infection. DKA Nursing Interventions Rationales Assess vital signs and observe for any signs of infection. Ketoacidotic state in diabetic patients may increase their risk for infection. Perform an initial head-to-toe assessment, particularly checking for the presence of any wounds and cuts. Diabetic and DKA patients suffer from slow wound healing. Any wound or cut needs to be managed early and appropriately to prevent infection which may spread and may lead to amputation of the affected toe, finger, or limb. Educate the patient for the need to monitor and report any signs of infection or new wounds and cuts. To facilitate early detection and management of infection and to provide proper wound management as needed. Teach the patient how to perform proper hand hygiene. To maintain patient safety and reduce the risk for cross contamination. Provide careful skin care. Massage the limbs and keep the skin dry. To reduce the risk of skin breakdown that may lead to infection. To 41 Provide wrinkle-free linens. facilitate a better peripheral blood circulation. Administer prescribed antibiotics if an infection is found. To immediately treat an infection as healing can be slow for DKA patients. Diabetic Ketoacidosis DKA Nursing Care Plan 4 Nursing Diagnosis: Imbalanced Nutrition: Less than Body Requirements related to insulin deficiency, as evidenced by unexplained weight loss, increased urinary output, dilute urine, high blood glucose levels, fatigue, and weakness Desired Outcome: The patient will be able to achieve a weight within his/her normal BMI range, demonstrating healthy eating patterns and choices. DKA Nursing Interventions Rationale Explain to the patient the relationship between diabetes and unexplained weight loss. To help the patient understand why unexplained weight loss is one of the signs of diabetes. Create a daily weight chart and a food and fluid chart. Discuss with the patient the short term and long-term goals of weight loss. To effectively monitory the patient’s daily nutritional intake and progress in weight loss goals. Help the patient to select appropriate dietary choices to follow 60% carbohydrates, 20% fats, 20% proteins. These proportions are ideal for diabetic ketoacidosis patients. Refer the patient to the dietitian. To provide a more specialized care for the patient in terms of nutrition and diet in relation to diabetic ketoacidosis. Diabetic Ketoacidosis DKA Nursing Care Plan 5 Nausea Nursing Diagnosis: Nausea related to depletion of sodium and potassium levels in the body secondary to diabetic ketoacidosis (DKA), as evidenced by gagging sensation, uremia, vomiting, presence of ketones in the blood or urine, and frequent urination. Desired Outcomes: • • • The patient will be able to report decreased severity or elimination of nausea. The patient will be able to maintain an adequate fluid and electrolyte balance. The patient will be able to identify different methods on how to minimize the severity of nausea. 42 Diabetic Ketoacidosis Nursing Interventions Rationale Assess the cause of the patient’s nausea. The choice of treatment will be guided by the patient’s assessment of the reasons for nausea. If the stimulus is removed, treatment may not be required. Assess the characteristics of the patient‘s nausea, including the history, duration, frequency, severity, precipitating factors, measures done to alleviate nausea, and medications. An in-depth assessment and evaluation of the characteristics of the patient’s nausea can aid in the development of solutions to lessen or resolve the problem. Monitor and record the patient’s hydration, weight, blood pressure, intake and output, and skin turgor. Nausea is frequently associated with vomiting and frequent urination can result in a change in a patient’s hydration status due to fluid loss that may cause dehydration. Advice the patient to take an adequate number of fluids and closely monitor that patient’s fluid and electrolyte balance. To avoid dehydration and complications such as low sodium, potassium, calcium, and magnesium. Electrolyte imbalances can develop from high blood glucose levels, which can produce nausea and vomiting. Further problems and heart arrhythmias can also result from electrolyte imbalance. Place an emesis basin within the patient’s reach. Vomiting and nausea are related closely. If the nausea is psychogenic, keep the emesis basin out of sight but within reach of the patient. Educate and help the patient in maintaining good dental hygiene. Anorexia and excessive salivation are linked to this condition. Oral hygiene aids in the treatment of nausea and makes it more comfortable. Assist the patient in preparing for diagnostic tests and explain to the patient the importance of diagnostic tests. To determine the contributing factor, a set of tests may be performed including upper gastrointestinal tract study, abdominal computed tomography scan, ultrasonography. Keep the patient’s room clean and well- ventilated and remove any strong odors from the area surrounding the patient such as perfumes, dressings, emesis. A well-ventilated room will help the patient to breath easily. Strong and unpleasant odors can contribute to the patient’s nausea. Allow the patient to use non-pharmaceutical nausea management methods including relaxation, guided visualization, music therapy, distraction, or deep breathing exercises. Non-pharmaceutical techniques have helped patients alleviate the nausea, but the methods should be used before the nausea occurs. Include cold water, ice chips, ginger items, and room temperature broth or bouillon to the patient’s diet if tolerated and acceptable. These help with hydration. Ginger, whether in the form of ginger ale, ginger tea, or candied ginger, helps to reduce nausea. Fluids that are excessively 43 cold or too hot might be uncomfortable to drink. Allow the patient to have a small amount of feedings with their own food preference, suggest including dry crackers, toast, broth, banana, rice or jelly to the diet. Small feedings with the patient’s own food preference will aid in the maintenance of nutritional status. An empty stomach can aggravate nausea in certain patients. Educate the patient or caregiver about nausea-relieving fluid and food options. By noting dietary factors to consider while nauseated, patients and caregivers can support proper hydration and nutritional status. Inform the patient to stay away from foods and scents which can aggravate nausea. Strong and unpleasant odors can trigger the nausea of the patient. 14. Keep the patient upright while eating and for 1 to 2 hours afterward. Keeping the patient upright can assist to reduce the risk 15. Educate the patient on the importance of taking prescribed medications as directed. Taking drugs according to the healthcare provider instructions will help to prevent nausea. Diabetic Ketoacidosis DKA Nursing Care Plan 6 Risk for Unstable Blood Glucose Level Nursing Diagnosis: Risk for Unstable Blood Glucose Level related to non-adherence to therapeutic regimen and inadequate blood glucose monitoring, secondary to diabetic ketoacidosis (DKA). Desired Outcomes: • • The patient will be able to show willingness to participate in the therapeutic management plan. The patient will be able to demonstrate blood glucose levels less than 180 mg/dL, fasting blood glucose levels that are less than 140 mg/dL, and hemoglobin A1C less than 7%. DKA Nursing Interventions Rationale Assess the patient for symptoms of hypoglycemia or hyperglycemia. Hypoglycemia symptoms vary from person to person, although they are consistent in the same person. Increased adrenergic activity and reduced glucose transport to the brain cause the symptoms. Tachycardia, diaphoresis, tremors, dizziness, headache, weariness, hunger, and visual abnormalities may occur in the patient. When there is insufficient insulin to glucose, hyperglycemia occurs. An osmotic effect occurs when there is too much glucose in the blood, resulting in increased thirst (polydipsia), hunger (polyphagia), and urination (polyuria) (polyuria). Nonspecific symptoms such as anxiety and impaired vision may also be reported by the patient. 44 Monitor the patient’s fasting and postprandial blood glucose levels. An adult’s fasting blood glucose level should be between 70 and 105 mg/dL. Hypoglycemia has a critical value of less than 40 to 50 mg/dL. Hyperglycemia has a critical value of more than 400 mg/dL. Because the solution includes up to 50% dextrose, patients receiving total parenteral nutrition (TPN) may have a higher-than-normal blood glucose level. After eating, patients with reactive hypoglycemia will have a lower blood glucose level than normal. Determine the patient’s blood insulin levels regularly. Early on in the progression of type 2 diabetes, hyperinsulinemia develops. Insulin production from the pancreas is stimulated by obesity and insulin receptor dysfunction in peripheral organs. Insulinomas and some extra pancreatic tumors raise insulin levels, which can lead to hypoglycemia. Assess the patient’s feet and note the temperature, pulses, color, and feelings. This is to assess for peripheral perfusion and neuropathy. Assist the patient in recognizing and changing unhealthy eating habits. This information serves as the foundation for personalized dietary advice related to the clinical condition that causes blood glucose levels to fluctuate. Monitor the patient’s vital signs and report the blood pressure reading of more than 160 mm Hg (systolic). Administer hypertensive medication as directed. Diabetes and hypertension are frequently linked. Controlling blood pressure helps to prevent heart disease, stroke, retinopathy, and nephropathy. Educate the patient about the foods or other glucose sources that are allowed to be taken if hypoglycemia is present. To treat hypoglycemia, a fast absorbed type of glucose is recommended. Oral ingestion of hard candies or fruit juice is one of these sources of glucose. If a patient is unable to take something orally, an intravenous glucose infusion may be necessary. Instruct the patient on the need and importance of sticking to a meal plan. A meal plan advised by the doctor will assist the patient in maintaining stable blood glucose levels. Educate the patient on how to take the prescribed medications correctly. To lower blood glucose, the diabetic patient must learn how to use oral or parenteral hypoglycemic medications. Discuss with the patient the importance of a healthy diet and exercise routine. Exercise helps to keep a balanced glucose levels by boosting glucose uptake into cells. The relationship of exercise, food intake, and blood glucose levels must all be understood by the patient. Teach the patient on how to properly take capillary blood glucose readings. Capillary blood glucose monitoring gives the patient real-time blood glucose readings. Determine the patient’s physical activity Physical activity helps in the reduction of blood glucose levels. Exercise is an important aspect of diabetes care since it lowers the 45 pattern. risk of cardiovascular problems. Refer the patient to a licensed dietitian for individualized diet advice. The patient’s body, weight, blood glucose levels, exercise habits, and specific clinical condition all factor into a tailored food plan. Blood glucose levels will be stabilized as a result of changes in the patient’s diet. Diabetic Ketoacidosis DKA Nursing Care Plan 7 Risk for Injury Nursing Diagnosis: Risk for Injury related to drowsiness secondary to diabetic ketoacidosis (DKA). Desired Outcomes: • Within eight hours of receiving nursing intervention and treatment, the patient will be able to identify the elements that increase their risk of injury and display injury-avoidance behaviors. • The patient will be free of injuries after four hours of nursing interventions and training. DKA Nursing Interventions Rationale Determine the patient’s age, developmental stage, health status, lifestyle, impaired communication, sensory-perceptual impairment, mobility, cognitive awareness, and decision-making abilities of the client. These elements influence the patient’s ability to protect oneself from harm. When establishing a treatment plan or teaching patients about safety precautions, nurses must properly analyze each of these elements. Assess the patient’s health and cognitive awareness for any changes. A patient’s risk for injury may increase as the health status changes. A postoperative patient, for example, may have confusion, disorientation, and memory loss, placing the patient at risk of falling or injuring themselves. Certain drugs might also affect the patient’s judgment. Assess the patient’s lifestyle. When determining injury risk, consider the patient’s lifestyle. For example, hazardous working conditions, settling in a high-crime area, having access to guns or weapons, being unable to obtain safety equipment owing to a lack of cash, and misusing prescription medicines are all examples. Examine the patient for impairment in communication. Language barriers, as well as speech and hearing difficulties, may impair the patient’s capacity to comprehend information, putting the patient at risk of an adverse incident. Determine the patient’s ability to walk and identify the risks for fall by using the Morse Fall Falls are more likely when there are changes in mobility due to muscle weakness, paralysis, poor balance, or lack of coordination. The Morse Fall Scale (MFS) is a regularly 46 Scale. used fall risk assessment instrument in health care institutions. It evaluates six characteristics (history of falling in the previous three months, secondary diagnosis, use of assistive equipment, IV/heparin lock, gait/transferring, and mental status) using a point scale system. An MFS score of 0-24 (no risk) indicates that no interventions are required. Standard fall prevention treatments should be started if the score is 25-50 (low risk). A score of >51, which indicates a high risk of falling, necessitates the implementation of high-risk fall prevention treatments. Note of the patient’s age and observe for evidence of physical injuries such as bruises, burns or scalds, history of fractures, lacerations, bite marks, social withdrawal, fearfulness. Signs of suspected deliberate injury or abuse that must be properly evaluated to ensure that the patient receives medical attention, is referred for extra help, and is protected from further harm. All healthcare providers have a moral and legal obligation to report these types of injuries and abuse to social welfare or Child Protective Services as soon as possible (CPS). Assess the patient’s safety at home or in the hospital if admitted. Nurses do an environmental risk assessment to look for things or equipment such as cords or hooks that could be used in suicidal hanging. As a result, it should be removed to safeguard the safety of the patient. Assist the patient in becoming familiar with the surroundings and place the patient’s important items within reach. To prevent injury, the patient should be familiar with the layout of the area. Items kept too far away from the patient may pose a risk. Assess the patient’s vital signs and blood glucose levels regularly. An increase or decrease in blood glucose levels and blood pressure may cause dizziness, headache and blurring of vision that may increase the patient’s risk for injury. Advice the patient to avoid thermal extremes like heating pads, hot water for baths/showers. Patients with impaired intellect or sensory impairments are unable to distinguish between temperature extremes. Burn damage is more likely in people with age-related physiological changes (e.g., loss of dermal appendages, dermal atrophy, and loss of insulating subcutaneous fat) and cognitive diseases (e.g., dementia, peripheral artery disease, and diabetes) that compromise mobility and judgment. Validate the patient’s feelings and concerns about environmental hazards. Validation assures the patient that the nurse has heard and comprehended the concerns. It also aids in the development of the nurse-patient relationship. 47 References: • American Diabetes Association. (n.d.). Hyperglycemia (High blood glucose). American Diabetes Association. https://www.diabetes.org/healthy-living/medication-treatments/blood-glucose-testing-andcontrol/hyperglycemia • Busti, A. J. (2015, October). How diabetics taking beta blockers still sweat with hypoglycemia. EvidenceBased Medicine Consult. https://www.ebmconsult.com/articles/diabetics-beta-blockers-hypoglycemiasweating, sympathetic. • Davis’s Drug Guide. (n.d.). Acarbose (Precose) | davis’s drug guide. Davis’s Drug Guide. https://www.drugguide.com/ddo/view/Davis-Drug-Guide/51003/all/acarbose?refer=true • Diabetes Teaching Center at UCSF. (n.d.). The liver & blood sugar. Diabetes Teaching Center at UCSF. • Mayo Clinic. (2020, March 13). Hypoglycemia—Symptoms and causes. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/hypoglycemia/symptoms-causes/syc-20373685 • Medline Plus. (2020). Drug-induced low blood sugar. MedlinePlus Medical Encyclopedia. https://medlineplus.gov/ency/article/000310.htm • Wang J, Williams DE, Narayan KM, Geiss LS. Declining death rates from hyperglycemic crisis among adults with diabetes, US, 1985–2002. Diabetes Care 2006;29:2018–22. Health and Social Care Information Centre. National Diabetes Inpatient Audit (NaDIA) 2012. www.hscic.gov.uk/diabetesinpatientaudit [Accessed 12 August 2019]. Shalitin S, Fisher S, Yackbovitch-Gavan M, et al. Ketoacidosis at onset of type 1 diabetes is a predictor of long-term glycemic control. Pediatr Diabetes 2018;19:320–8. Joint British Diabetes Societies Inpatient Care Group. The management of diabetic ketoacidosis in adults. 2nd Edn. Joint British Diabetes Societies Inpatient Care Group, 2013. https://abcd.care/resource/management-diabetic-ketoacidosis-dka-adults [Accessed 12 August 2019]. Joint British Diabetes Societies for inpatient care. Management of glycaemic control in pregnant women with diabetes on obstetric wards and delivery units. JBDS-IP, 2017. Bonora BM, Avogaro A, Fadini GP. Sodium-glucose co-transporter-2 inhibitors and diabetic ketoacidosis: An updated review of the literature. Diabetes Obes Metab 2018;20:25–33. National Institute for Health and Care Excellence Type 1 diabetes in adults: diagnosis and management 2016 NICE guideline [NG17]. London: NICE. Ackley, B. J., Ladwig, G. B., Makic, M. B., Martinez-Kratz, M. R., & Zanotti, M. (2020). Nursing diagnoses handbook: An evidence-based guide to planning care. St. Louis, MO: Elsevier. Gulanick, M., & Myers, J. L. (2017). Nursing care plans: Diagnoses, interventions, & outcomes. St. Louis, MO: Elsevier. • • • • • • • • 48 Research inputs: [Hypoglycemia--a dreaded complication of diabetes] [Article in Norwegian] M Bjørgaas 1 • PMID: 11475234 Abstract Hypoglycaemia is a common side effect of insulin therapy in type 1 diabetes. In patients with type 2 diabetes treated with blood-glucose lowering agents of the sulphonylurea group, hypoglycaemia is less frequent than in insulin-treated patients. In most patients strict metabolic control increases the risk of hypoglycaemia, but this risk may be reduced if patients are offered individualised insulin treatment in combination with active support and education. Previously experienced hypoglycaemic episodes and lack of endogenous insulin production are risk factors for repeated episodes. Patients with longstanding diabetes and loss of warning symptoms have increased risk of severe hypoglycaemic episodes, which may lead to loss of consciousness or convulsions. Driving performance is significantly disrupted at relatively mild hypoglycaemia, and persons with diabetes should not start driving when their blood glucose is in the 4-5 mmol/l range without prophylactic treatment. They ought to have carbohydrate-rich snacks easily available in the car and should stop driving if they feel hypoglycaemic. Repeated episodes of severe hypoglycaemia seem to be associated with cognitive dysfunction. When deciding the targets of blood-glucose lowering therapy, the risk of severe hypoglycaemia must be weighed against the beneficial effects of good metabolic control. Clinical Profile of Diabetic Ketoacidosis in Adults in Dhulikhel Hospital B K Prajapati 1 • PMID: 29446358 Abstract Background Diabetic ketoacidosis is one of the life-threatening acute complications of diabetes mellitus. Despite the improvements in diabetic care, it remains a major clinical problem in clinical practice. Objective To assess the clinical and laboratory profile of adults with diabetic ketoacidosis in Dhulikhel hospital. Method This is a descriptive cross-sectional study including adult patients admitted in Dhulikhel hospital from July 2014 to July 2016 with the diagnosis of diabetic ketoacidosis according to the guidelines of American diabetes association. The hospital records of these patients were reviewed for their clinical and biochemical profiles. Result Forty eight patients fulfilled the criteria of diabetic ketoacidosis and were included in the study. Seventy three percent of patients had type 2 diabetes mellitus. Twenty three percent of the patients were cases of newly diagnosed diabetes mellitus. Polyuria and polydipsia as presenting complaint was more common in patients with type 1 diabetes mellitus (p=0.002) whereas fever was more common in type 2 diabetes mellitus patients (p=0.03). Majority of patients had normal serum sodium and potassium level. Forty two percent of the patients have high serum urea level and just over one third had high serum creatinine level. The most common precipitating factor of diabetic ketoacidosis for patients with type 1 diabetes mellitus was omission of insulin whereas in type 2 diabetic patients was infection. Conclusion Diabetic ketoacidosis is complication of both type 1 and type 2 diabetes mellitus. High degree of suspicion is needed for early detection of this life threatening condition especially in patients without history of diabetes mellitus. x 49