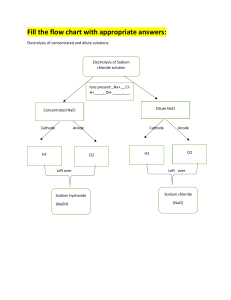
12 88 22 1 99 Mg Ra Ti H Es Hydrogen Fuel Cell And their consequences By: Arhum, Jabir, Sohman and Nithin 56 84 40 3 100 Ba Po Zr Li Fm Process by which ions in an electrolyte are separated by the usage of electricity. What is Electrolysis? How to Manipulate Electrolysis We first need to make sure what sort of solution we shall use, will it be molten, aqueous, dilute or concentrated. Next we shall inquire upon which gas or metal do we need to extract out of the process. 104 88 22 4 72 85 90 Rf Ra Ti Be Hl At Th 01. 01. How to get Hydrogen From a electrolytic cell The methods… There are many methods to get Hydrogen out of electrolytic cells ■ ■ ■ Water Electrolysis Alkaline electrolysis Dilute Sulfuric Acid electrolysis There are many more, but these are the more common methods 84 Po 40 72 100 Zr Hl Fm Water Electrolysis The most common method of obtaining hydrogen from electrolytic cells is through the electrolysis of water. In this process, water is split into its constituent elements, hydrogen and oxygen, using an electric current. The hydrogen gas is then collected at the cathode, while oxygen gas is collected at the anode. 22 88 Ti Ra Alkalineolysis In this method, a solution of potassium hydroxide or sodium hydroxide is used as the electrolyte instead of pure water. This makes the process more efficient as the hydroxide ions in the solution help to conduct the electric current. 56 84 40 3 100 Ba Po Zr Li Fm Sulfuric Acid Electrolysis In this method, we use DILUTE sulfuric acid, this acid has 3 ions, they are H+, OH-, SO42- the anode receives OH- making oxygen gas, and the CATHODE makes a wonderfully flammable HYDROGEN gas. 22 88 Ti Ra Dangers Burning Toxic fumes Corrosion Hydrogen is flammable Sulfur is toxic Sulfuric acid is corrosive Danke! You people got any questions?? 88 Ra CREDITS: Me, Myself and I 90 Th