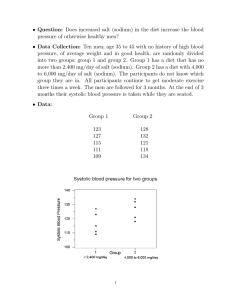
Sodium Chloride Introduction Considered to be the salt, although there are several salts and sodium chloride is just one of these salts it is the most commonly used salt out of all of them. Sodium Chloride's formula - NaCl Melting point - 801 °C Density - 2.16 g/cm³ 11 NA The Usage Used asseasoning in food /preserve foods electrolyte replenisher (help prevent heat cramps) treat patients that have lost salt and water in their body deice roads that have been frozen Used in many industrial ways healthcare to help dehydrated patients How is it made? Made when sodium atoms interact with chlorine atoms. Chlorine atoms will be given an electron by the sodium , there is one sodium atom for every chloride atom in Sodium Chloride . Manufacturing sodium chloride involves injecting water into a halite vein in the earth, extracting the brine, and sending it to a refinery for evaporation and processing into granulated salt. Other methods involve evaporating seawater via the sun and wind. More Facts Sodium Chloride is often referred to as Table Salt or Sea Salt. Mixing Sodium Chloride in with the water of cut flowers in a vase will help the flowers live longer. Sodium Chloride is in every cell in the body. It is often used as a binding and/or thickening ingredient in skin and hair care products like shampoos, and as an abrasive in salt scrub products on occasion. Thank you ^ ^ Done byBhone Myat Thu Grace Fay Kaitlyn Kristen