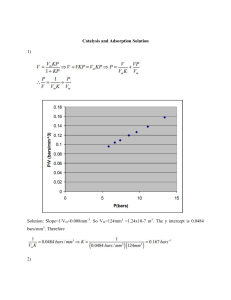
Group:08 SEPARATION PROCESS LAB Adsorption apparatus Department Chemical Engineering Group Members: • • • • • Muhammad Hammad Asghar Muhammad Faisal Afridi Muhammad Ramzan Laiba Shahbaz Manahil Eman Pakistan Institute of Engineering and Applied Sciences Objective: To understand the phenomenon of adsorption on activated carbon. Abstract: The aim of this experiment was to study and demonstrate the phenomenon of adsorption on activated carbon. First of all, 500 ppm solution of a colored dye was prepared using distilled water and the solution was poured into the tank. Distilled water was added to the recirculation water tank. The circulation pump was turned on to circulate the water through the bed of activated carbon and after sometime adsorbate pump was also turned on. At last, samples were taken from the different points of the column. Apparatus and Chemicals required: Adsorption column, Activated carbon, Beakers, Stirrer, Stopwatch, Distilled water, Dye Experimental Setup: Figure 1: Adsorption Column Theory: Adsorption is a process that involves the accumulation of a substance in molecular species in higher concentrations on the surface. For the adsorption process, two components are required, • Adsorbate: Substance that is deposited on the surface of another substance. For example, H2, N2 and O2 gases. • Adsorbent: Surface of a substance on which adsorbate adsorbs. For example, Charcoal, Silica gel, Alumina. On the basis of interaction forces between adsorbate and adsorbent, adsorption is of two types. 1. Physical adsorption: This type of adsorption is also known as physisorption. It is due to weak Van der Waals forces between adsorbate and adsorbent. For example, H2 and N2 gases adsorb on coconut charcoal. 2. Chemical adsorption: This type of adsorption is also known as chemisorption. It is due to strong chemical forces of bonding type between adsorbate and adsorbent. We can take the example involving the formation of iron nitride on the surface when the iron is heated in N2 gas at 623 K. Applications of Adsorption 1) Air pollution masks: These consist of silica gel or activated charcoal powder, when dust or smoke are paused through them, those particles get adsorbed on the surface of these materials. 2) Separation of noble gases by Dewar’s flask process: A mixture of noble gases of Ne, Ar, Kr is passed through Dewar’s flask in presence of heated coconut charcoal. Argon and Krypton gels adsorbed leaving Neon. 3) Purification of water: By the addition of alum stone to the water, impurities get adsorbed on the alum and water gets purified. 4) Removal of moisture and humidity: Moisture in the air is removed by placing silica gel on which water molecular gets adsorbed. 5) Adsorption chromatography: It is used to separate pigments and hormones. 6) Ion exchange method: In this method of removing the hardness of water, calcium and magnesium ions get adsorbed on the surface of ion exchange resin 7) In metallurgy: In the froth floatation process of concentration of ore, the particle gets adsorbed on the froth. Activated Carbon Adsorption: Activated carbon is used for the removal of odors, tastes, colors, or even poisons in either a liquid or gas state. The ability for activated carbon to remove contaminants is not based on how much carbon one uses, but rather the capacity of carbon to adsorb the contaminant. The higher the capacity, the greater amount of contaminants the carbon adsorbs per unit volume. Because natural carbon is not able to treat every contaminant, there are different carbon products which remove specific contaminants. In a specific case, activated carbon can remove certain chemicals or contaminants from water. It is most effective in the removal of organics due to their composition of hydrogen and carbon. Therefore, the activated carbon has a much greater attraction to the contaminant than the contaminant has with the water. This causes the activated carbon to adsorb the contaminant, leaving the water pure and contamination free. However, there are still organics that are poorly adsorbed by activated carbon. Procedure: • Prepare 5 ppm dye solution using distilled water. • Fill the feed tank with distilled water. • Fill the second tank with adsorbate that is dye solution. • Turn on the breaker and the power switch. • Turn on the circulation pump and adjust its speed using knob. • Adjust the flow rate of water to 1.5 LPM using rotameter. • Turn on the adsorbate pump and adjust its speed. • Take samples of purified water at different points using beaker. Shutdown Procedure: • Turn off the adsorbate pump and recirculation pump. • Turn off the breaker and main power supply. • Drain all the tanks. Observation and Calculations: Sample calculation: For preparing 500 ppm dye solution 1ppm= 1 mg/L So, 500 ppm= (500) (1 mg/L) = 0.5 g/L For preparing 15 L dye solution (0.5 g/L)(15 L) = 7.5 grams In this way, 15 L dye solution was prepared by adding 7.5 grams of dye in water and making total volume of solution up to 15 L. Discussion and Conclusion: In this experiment, the dye solution was purified using adsorption. The adsorbent used to purify the dye solution was activated carbon that was filled in a column. The dye solution was allowed to pass through the bed of activated carbon. It was observed that as the dye solution was moving down the column, the concentration of dye was decreasing with the increasing distance from the top portion of bed. This decrease in concentration with increasing distance from the top of bed was due to the reason that as the solution moved down, the number of active sites available for adsorption of dye increased so some dye from the solution which could not get adsorbed on the top portion of bed adsorbed on the bottom portion and a more clear water was obtained from the sample taken from bottom portion of the column. On the other hand, the sample taken from upper portion of column contained more dye as compared to the sample taken from bottom portion. Results: • The sample taken from the upper portion of column (valve 1) contained highest concentration of dye. • The sample taken from the middle portion (valve 5) of column contained moderate concentration of dye. • The sample taken from the bottom portion (valve 9) of column contained almost clear water. Samples after adsorption: Figure 2: Adsorption Samples (taken from valves 1, 5, and 9)