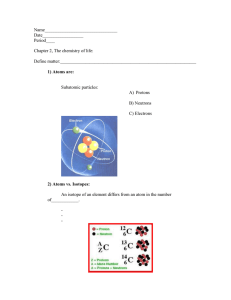
SiO2 is naturally found in sand & quartz Each carbon atom is covalently bonded to 4 other carbon atoms Each carbon atom is covalently bonded to 3 other carbon atoms Each oxygen atom is covalently bonded to 2 silicon atoms. Each silicon atom is covalently bonded to 4 oxygen atoms. Si2O4 → SiO2 Repeated tetrahedral structure Form a hexagonal structure in layers Made up entirely of STRONG covalent bonds with no weak intermolecular forces. High melting point and boiling point as large amount of energy is required to break the strong covalent bonds Extremely hard and dense (makes good cutting tools) STRONG covalent bonds within the layers 1 silicon & 4 oxygen atoms form a repeated tetrahedral structure Made up entirely of STRONG covalent bonds with no weak intermolecular forces. High melting point and boiling point as large amount of energy is required to break the strong covalent bonds Similar properties as diamond Hard & insoluble in water Do not conduct electricity due to lack of free mobile ions or electrons High melting point and boiling point as large amount of energy is required to break the strong covalent bonds Weak intermolecular forces (van der waals) hold the layers together ⸫ Layers can slide over each other and graphite is slippery (pencil/lubricants) Each carbon atom has a free electron (not shared) ⸫ these free mobile electrons between the layers can carry charges and conduct electricity Do not conduct electricity due to lack of free mobile ions or electrons