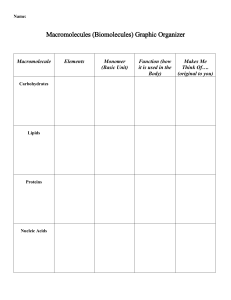
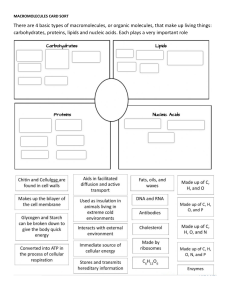
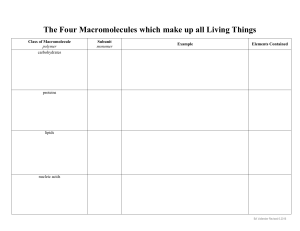
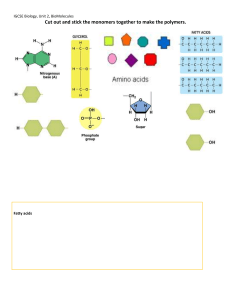
BL1002 Cells, Biomolecules, Genetics and Evolution Carbohydrates Lipids (fats) Protein Nucleic Acid: DNA and RNA Biomolecules are all carbon-based chains that vary by the functional groups attached General chemical principles of biomolecules • ALL biomolecules are made up of a few number of atoms – carbon (18.5%), hydrogen (9.5%), oxygen (65%), nitrogen (3.3%), phosphorous (1%). • Carbon is key to making a large variety of molecules • Macromolecules are made from smaller pieces Carbon is central to all living organisms • Because of carbon’s ability to form four bonds, these building blocks can be used to make a huge variety of organic molecules. • Valence electrons are those in the outermost shell, or valence shell • The number of unpaired electrons in the valence shell of an atom is generally equal to its valence, the number of covalent bonds it can form • Carbon chains form the skeletons of most organic molecules and all biomolecules • Carbon chains vary in length and shape The chemical properties of biomolecules are largely determined by their functional groups What are functional groups • An arrangement of certain atoms in a particular configuration • Vary based on which atoms are involved • In combination with carbon-based chains, they allow biomolecules to have unique structures AND function What are functional groups? • Functional groups are KEY to understanding molecular diversity, chemical reactions and all of biochemistry! Carbohydrates Lipids Proteins Nucleic acids ARE ALL EXAMPLES OF MACROMOLECULES... THESE ARE THE MAJOR CONSTITUENTS OF CELLS KEY POINT: Cells must make the macromolecules that they need from smaller molecules. HOWEVER, one important difference among the macromolecules is that... • Carbohydrates, Proteins, and Nucleic acids are made from the stepwise polymerization of monomers. • Lipids are NOT polymers but are generally classified as macromolecules due to their high molecular weight. Carbohydrates • Are the most abundant biomolecules on Earth! • Have the general chemical formula of (CH2O)n • The most common monosaccharide is glucose (C6H12O6) • Monosaccharides are simple sugars that are either aldehydes or ketones • Carbohydrates can be polysaccharides, which are long polymers of repeating monosaccharide units •Carbohydrates (CH2O)n •The suffix -ose is used to form the names of sugars •Line drawings of carbohydrates (angle is a carbon and each carbon make 4 bonds): Carbohydrates have several functions •Structural (e.g., chitin, cellulose) •Source of carbon and energy The architecture and functions of a polysaccharide are determined by its sugar monomers and the positions of its glycosidic linkages STRUCTURE = FUNCTION Lipids •Lipids are the one class of biomolecules that does not include true polymers •The unifying feature of lipids is that they mix poorly, if at all, with water HYDROPHOBIC •Lipids consist mostly of hydrocarbon regions •The most biologically important lipids are fats, phospholipids, and steroids Several cellular functions •Components of membranes Phospholipid bilayer •Can act as signalling molecules •Energy storage