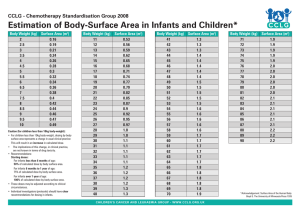
Induction I Drug Dose for patients ≥10kg and ≥1 month, not born significantly premature Dose for patients <10kg and or <3 month and born significantly premature Route No. doses Schedule Azacitidine AZA 75 mg/m2/day 2.5mg/kg/day 5 Day -4 to 0 100mg/m2/q12h 500mg/m2/day 3.3mg/kg/q12h 16.7mg/kg/day IV over 10-40 mins IV over 30m IV over 15m immediately prior to daunorubincin IV over 6h IV over 15m IV over 4h 20 3 Day 1 -10 Day 1,3,5 3 3 5 Day 1,3,5 Day 3-5 Day 1-5 100mg/via/, $15233 Cytarabine Dexrazoxane Daunorubicin 50mg/m2/day 1.67mg/kg/day 2 Idarubicin 8mg/m /day 0.27 mg/kg/day Etoposide 100mg/m2/day 3.3mg/kg/day Emetogenicity of drugs used in Induction I. Moderate Azacitidine, Daunorubicin, Idarubicin Cytarabine( ≤1000 mg/m2), Etoposide Low Induction II Drug Dose for patients ≥10kg and ≥1 month, not born significantly premature Dose for patients <10kg and or <3 month and born significantly premature Route No. doses Schedule Azacitidine AZA 75 mg/m2/day 2.5mg/kg/day 5 Day -4 to 0 Fludarabine Cytarabine Dexrazoxane 30 mg/m2/day 2000mg/m2/day 400mg/m2/day 1 mg/kg/day 66.7mg/kg/day 13.5mg/kg/day 5 5 3 Day 1 -5 Day 1 -5 Day 3-5 Idarubicin Filgrastim 8mg/m2/day 5mcg/kg/day 0.27 mg/kg/day 5mcg/kg/day 3 7 Day 3-5 Day 1-7 Sofarenib2 200mg/m2 QD NA IV over 10-40 mins IV over 30m IV over 4h1 IV over 15m immediately prior to ida IV over 15m SC or IV 15-30m PO 21 For 21 days 1: Cytarabine should be given 4 hours after fludarabine 2: Sorafenid will be given for 21 days, as tolerated, for FLT3 ITD and NTP98-NSD1 or FLT3 and WT1 mutation after completion of other agents. Emetogenicity of drugs used in Induction II. Moderate Azacitidine, Cytarabine >1000 mg/m2, , Idarubicin Minimal Fludarabine Intensification I Drug Dose for patients ≥10kg and ≥1 month, not born significantly premature Dose for patients <10kg and or <3 month and born significantly premature Cytarabine 1000mg/m2/q12h 33.3mg/kg/q12h Etoposide 150mg/m2/day 5mg/kg/day Emetogenicity of drugs used in Intensification I. Moderate Cytarabine >1000 mg/m2 Low Etoposide Route No. doses Schedule IV over 2h IV over 2h 10 5 Day 1 -5 Day 1-5 Intensification II Drug Dose for patients ≥10kg and ≥1 month, not born significantly premature Dose for patients <10kg and or <3 month and born significantly premature Route No. doses Schedule Dexrazoxane 480mg/m2/day 16mg/kg/day IV over 15m immediately prior to mitoxantrone 3 Day 3-5 Mitoxantrone 12 mg/m2/day 0.4 mg/kg/day 2 Cytarabine 1000mg/m /q12h 33.3mg/kg/q12h Emetogenicity of drugs used in Intensification II. Moderate Cytarabine >1000 mg/m2 Low Mitoxantrone IV over 1h IV over 2h 3 8 Day 3-5 Day 1 -4 Intensification III Drug Dose for patients ≥10kg and ≥1 month, not born significantly premature Dose for patients <10kg and or <3 month and born significantly premature Route No. doses Schedule Cytarabine Erwinase1 Leunase Sofarenib2 3000mg/m2/q12h 25000 U/m2/day 7500 U/m2/day* 200mg/m2 QD 100mg/kg/q12h 833 U/kg/day 250 U/kg/day* NA IV > 3h IV >1h or IM 8 2 2 21 Day 1,2,8,9 Day 2, 9 Day 2, 9 For 21 days PO 1: Erwinia asparaginase should be administered 3 hours after 4th and 8th dose of cytarabine. 2: Sorafenid will be given for 21 days, as tolerated, for FLT3 ITD and NTP98-NSD1 or FLT3 and WT1 mutation after completion of other agents. *: Leunase dose = Erwinase÷3.33 (from TPOG-ALL-2013, for patients hypersensitive to Leunase 6000 U/m2, switch to the Erwinia 20000 U/m2). But in Dana-Farber Cancer InstituteALL Consortium Protocol 95-01, they used 1:1 conversion (Blood. 2007;109:896-904). Emetogenicity of drugs used in Intensification III. Moderate Cytarabine >1000 mg/m2 Supportive care requirements: prophylactic vancomycin, ciprofloxacin and voriconazole during times of neutropenia. Anthracycline cumulative dose after completing the treatment. Drug Daunorubicin Epirubicin Idarubicin – IV Mitoxantrone Maximum cumulative dose (mg/m2) 600 900 150 160 Received dose(mg/m2) 150 (25%) 225 (25%) 24 (16%) 36 (22.5%) AML16 Principal Investigator: Tanja Andrea Gruber, MD PhD SJCRH Confidential Document Overview Risk Stratification Low-risk (LR) criteria (4 courses of chemo, not eligible for SCT) .Inv16, t(8;21), NPM1 or CENPA and MRD < 0.1% after induction I Intermediate-risk (IR) criteria ( 5 courses of chemo, not eligible for SCT) .Absence of low or high risk features High-risk (HR) criteria (candidates for SCT) Presence of any of the following: .DEK-NUP214[t(6;9)],KAT6A-CREBBP [t(8;16)], -7, -5, -5q, KMT2A-MLLT10 [t(6;11)], KMT2A-MLLT4[t(10;11)], inv (3)(q21q26.2), CBFA2T3-GLIS2 [inv(16)(p13.3q24.3)], NUP98-KDM5A [t(11;12)(p15;p13)],ETV6-HLXB [t(7;12)(q36;p13)], NUP98-HOXA9 [t(7;11)(p15.4;p15)], NUP98-NSD1 .Patients carrying FLT3-ITD in combination with either NUP98-NSD1 fusion or WT1 mutation. .AML with minimal differentiation or acute erythroid leukemia .Acute megakaryoblastic leukemia with KMT2A rearrangements, CBFA2T3-GLIS2 [inv(16)(p13.3q24.3)], or NUP98-KDM5 [t(11;12)(p15;p13)]. All other acute megakaryoblasti leukemia subtypes will be considered intermediate risk. .Treatment-related (Secondary) AML. .Refractory anemia with excessive blasts and >10% bone marrow blasts (RAEB-2) or AML arising from prior MDS. All other patients with poor response to therapy (must have one of the following features) .MRD ≥ 1% after Induction I .MRD ≥ 0.1% after Induction II. Intrathecal Therapy All patients should undergo lumbar puncture and receive an age-appropriate dose of intrathecal therapy (IT) at the time of diagnosis. Patient age Methotrexate Hydrocortisone Cytarabine Volume < 1 year 6mg 12mg 18mg 6ml 1-2 years 8mg 16mg 24mg 8ml 2-3 years 10mg 20mg 30mg 10ml >3 years 12mg 24mng 36mg 12ml Leucovorin rescue (5mg/m2 per dose, 5mg maximum per dose) will be given orally or intravenously at 24 and 30 hours after each IT MHA treatment. Leucovorin dosing and intrathecal volume may be adjusted according to local institutional guideline. To reduce the risk of neurotoxicity, we recommend separating IT therapy from high-dose cytarabine (HDAC)(1g/m2 or greater) by at least 24 hours for induction I and Intensification I/II. Metrotrexate may be omitted from IT therapy for patients that have a history of MTX-induced leukoencephalopathy. CNS1 (no evidence of CNS disease, no leukemic blast cells on CSF cytospin.) .CNS1 participants will received 4 total doses of intrathecal therapy, given at approximately one month intervals or at the beginning of each of the 4 courses of chemotherapy. CNS2 (< 5 leukocytes per ul of CSF and the presence of leukemic blast cells on CSF cytospin.) .CNS2 participants will receive weekly intrathecal therapy until the CSF is free of blast cells. These patients will receive 3 additional doses of intrathecal therapy at approximately one month intervals (generally given with each subsequent course of chemotherapy.) Patients who are unable to undergo lumbar puncture and receive intrathecal therapy prior to starting Induction I should be treated as CNS2 unless they have overt CNS leukemia CNS3) CNS3 (overt CNS leukemia, ≥5 leukocytes per ul of CSF and the presence of leukemic blast cells on CSF cytospin or traumatic tap) .CNS3 participants will receive weekly intrathecal therapy until the CSF is free of blast cells (minimum number of doses:4) These patients will then receive 3 additional doses of intrathecal therapy (minimum number of doses: 7) at approximately 1-month intervals (generally given with each subsequent course of chemotherapy). AML16 Grade 4 non-hematologic events during induction I and II as of 12/11/2018 (N=31) Description Ind I Events Ind I patients Ind II Events Ind II patients Alanine aminotransferase↑ 1 1 2 2 Blood bilirubin↑ 0 0 1 1 Hypernatremia 1 1 0 0 Hypocalcemia 1 1 0 0 Hypokalemia 0 0 2 2 Respiratory failure 0 0 1 1 Sepsis 2 2 4 4 J Clin Oncol 33:3774-3780. Drug Daunorubicin Doxorubicin Epirubicin Idarubicin – IV Idarubicin – PO Mitoxantrone Maximum recommended cumulative dose (mg/m2) 600 450 900 150 400 160