Environmental Chemistry Assignment: Eutrophication, Toxic Metals
advertisement

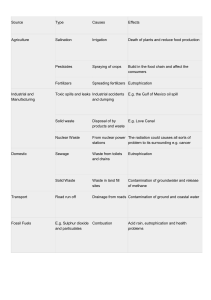
ENVIRONMENTAL CHEMISRTY ASSINGMENT Roll# CH-0119-115 Full Name: TARIQ ALI S/o MUHAMMAD ASHRAF MEMON Department: Chemistry Class: BS-II Semester: Third (3 rd ) Subject: Environmental Chemistry Teacher: Prof.Naseem-ul-Ghani Qureshi Batch: 2k19 0 SHAH ABDUL LATIF UNIVERSITY KHAIRPUR 1. What is mean by Eutrophication of nitrate and phosphate? Answer: The term eutrophication is defined as increased or excess amount of nutrients in water such as minerals, nitrates, sulphates, phosphates etc. OR The process in which water receives higher concentration of nutrients i.e. nitrates, sulphates and phosphates than their required limit. Therefore eutrophication of nitrate and phosphate means excess amount of phosphate and nitrates in water. Causes/Sources of Eutrophication of Nitrates and Phosphate Nitrogen and phosphorous are essential elements for growth of plants and also for animals. These substances used in fertilizers. When fertilizers are used in excess they cause the reason for deposition of nitrates and phosphate. Detergents also contain nitrogen and phosphate when these are drained into rivers, lakes they also causes eutrophication of nitrate and phosphate. Poor sewage treatment also causes of eutrophication of nitrate and phosphate. Dead plant and animals are the source of nitrogen fixation, this process increase the amount of nitrates in soil. Converging of dead organisms into nitrates as given as under; Dead Organism Decomposers (Fungi and bacteria) converts it into Ammonium by process of Ammunificatin Ammonium(NH4+) Nitrites (NO2-) Nitrification Nitrates (NO3-) Nitrification Major effects of Eutrophication of nitrate and phosphate Eutrophication also known as algal bloom; due to accumulation of nitrates and phosphates alga produce in water. Production of algae in large number causes reduction of photosynthesis because sun light can’t easily reach on plants due this aquatic living organisms’ food chain disturbed. It’s not only effect on plants but also other organisms life. Control on Eutrophication of nitrate and phosphate 1 SHAH ABDUL LATIF UNIVERSITY KHAIRPUR It can be reduced by minimizing pollution sources Less use of fertilizers containing nitrogen and phosphate. By using organisms that reduce the production of algae. Except these there are many other sources through which we control eutrophication of nitrate and phosphate. 2. What are toxic metals found in the water? What are the hazards one can expect from drinking contaminated water which contains heavy and toxic metals? Answer: Toxic Metals Founds in Water Water is the essential substance for all living organism. It is used for many purposes i.e. drinking, washing, fertilizing, manufacturing and so on. Due to anthropogenic and global activities, it has been continuously become harmful by toxic metals and other wastes. o Following are the major toxic metals found in water; o o o o o o o o o Zinc (Zn) Lead (Pb) Chromium (Cr) Mercury (Hg) Copper (Cu) Nickel (Ni) Cobalt (Co) Arsenic (As) Cadmium (Cd) Health Hazards due to drinking contaminated water contains heavy toxic metals Drinking contaminated water contains heavy metals causes following known diseases; Diseases caused by Cadmium Itai-Itai disease, liver/kidney lesions, osteoporosis, hypertension Diseases caused by Lead Calcium deficiency due to accumulation of lead in bones, coma, convulions, and many other diseases by disturbing neurological & musculoskeletal systems Diseases caused by Chromium Nose ulcer, Asthma, Hemolysis, and damage to DNA, liver, carcinoma and kidney Disease caused by Arsenic Anemia, hypertension, dementia, Alzheimer’s, bladder cancer and so on 2 SHAH ABDUL LATIF UNIVERSITY KHAIRPUR Diseases caused by Mercury Damage to central systema nervosum, visual disorders, nose and skin irritation, renal toxicity etc. Diseases caused by Cobalt Seizures, growth retardation, asthma, interstitial lung diseases, increase red blood cells, thyroid deficiency, emphysema, and paralysis of systema nervosum. Diseases caused by Zinc Chest tightness, headache, nausea, gastrointestinal problems, and immune toxicity Diseases caused by Copper Liver and Kidney infections, Wilson’s, anemia, damage animal tissues, melena, jaundice etc. 3. Sulphur is also one of the environmental pollutants found in land, rocks and water? What do you mean by rotten egg smell? Answer: Sulphur is also one of the Environmental Pollutants Found in land, rocks and water Introduction to sulphur: Sulphur is the second member of VIA group of periodic table. Group VIA also known as Oxygen Family or Chalcogen. It has symbol “S”. Its atomic number is 16 while atomic mass is 32 a.m.u. Occurrence Sulfur is fifth most common on earth and tenth most common by mass in the universe. Sulphur on earth usually occurs as sulfide and sulfate minerals. In air it usually occurs as combined state as Sulfur oxides etc. It is also found near hot springs and volcanic. Sulfur as Environmental Pollutant Sulfur itself is not cause of pollutant for environment but its compounds i.e. SO2, H2S, H2SO4 and other oxides, mineral ores and sulphur metalized become the causes of environmental pollution. Some of these causes are defied as under. Photochemical Smog (cause of air pollution) Photochemical smog is a complete pollutant mixture of partially oxidized hydrocarbon, ozone & other oxidant. A type of photochemical smog called as London or classical smog associate with use of traditional fuel and coal. These fuel and coal produces unburned carbon soot and elevated levels of atmospheric sulphur dioxide. Due to presence of a mild reducing agent and present of week acid, overall properties are reducing and acidic where the atmosphere is humid. This causes eye and other membrane irritation and also adversely affects plant growth and other serious ecotoxicological problems. 3 SHAH ABDUL LATIF UNIVERSITY KHAIRPUR Acid Rain (causes of air, water, rocks and soil pollution) Acid rain is the deposition an earth of dilute solutions acids (specially sulphuric and nitric oxide) with rain fall. Two predominant acids in acid rain are sulphuric and nitric acid. SO2 mainly comes from burning coal and oil power generating industries, industrial bowlers and smelters. During the day time and conditions of low humidity in the pressure of hydrocarbons and nitrogen oxides, the predominant reaction is oxidation of sulphur to sulphuric acid, sooth particles are strongly involved in catalyzing the oxidation of sulphur di oxide. o Formation of Sulphuric acid SO2 + hv → SO.2 SO2 + O2 → SO.4 SO.4 + O2 → SO3 + O3 SO3 + H2O → H2SO4 Acid rain is very destructive to lakes and rivers, killing fishes and destroying phosphate resources on which aquatic plants depend for nutrition. Soil is likewise affected as acidification due to acid deposition and certain biological processes with the soil erodes fertility, it also affect the originality statue. Rotten Egg Smell Rotten egg smell is commonly used for Hydrogen Sulphide gas H2S.Because it has an offensive rotten egg odor. It is highly flammable and respiratory inhibitor. It is created naturally by decaying organic matter and is released from sulphur hot springs, liquid manure, sewage sludge and other wastages. It may be harmful for health when inhaled in high concentration. It causes burning of skin and eyes, sensation, iratest and watering of eyes, coughing and aging nausea/vomiting, unconsciousness, coma or may be lethal to death. 4