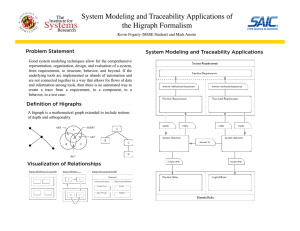
Areas to analyze: Specific Points of Analysis: Suppliers / Supplies Raw Materials Crisis Management Product Recalls Complaints Audit Traceability Document Traceability Allergens Microbial Control Sanitation Cross Contact / Contamination Packaging and Label Issues HACCP Organization Pest Control Safety All floors free of garbage, pests, debris, etc. Tools in appropriate areas Cleanliness of equipment, floors, sinks, QA critical equipment, etc. Operators following GMPs Traceability of Batch Records and floor protocols GDP of documents on floor Standing water Calibration dates Finished product specifications and raw material storage Propped doors, openings that are not allowed, broken windows/ doors/ equipment. Environment and equipment maintenance Glass and brittles CCP checks Safety all around Findings: Actionable items for operators to drive and follow up on! Building/ Area: ____________ Auditor(s): _________________ Date: _________ Production Attendee: _______________________ Finding Major/ Minor Resolved during audit? (Yes/ No) Comments/Follow Up Required Production sign & Date: ________________________________________________________________ Quality sign & Date: ___________________________________________________________________