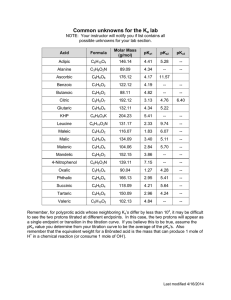
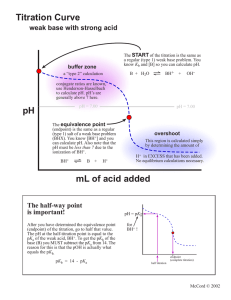
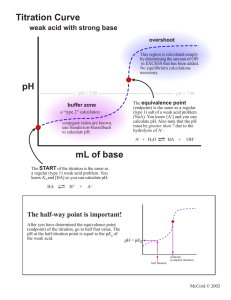
Danny Name: ______________________________ General Chemistry II, Spring 2021 . Data Sheet I Part II: Titration of a Weak Acid Unknown number #2 ________________ [NaOH], $ 0.200 M ________________ initial Volume (mL) pH - 3.02 pH Midpoint 3.25mL 4.76 EQP 6.5mL 8.69 Part III: Identifying an Unknown Diprotic Acid I Unknown number ________________ Mass of sample ________________ 0.0953g initial pH Volume (mL) pH 1st Midpoint 1.89mL 2.21 1st EQP 3.79mL 2.91 2nd Midpoint 5.68mL 3.84 2nd EQP 7.57mL 8.04 '- I -90 8 General Chemistry II, Spring 2021 Data Sheet II 9 General Chemistry II, Spring 2021 Data Sheet III I RBI 10 General Chemistry II, Spring 2021 Name: ______________________________ Post-Laboratory Questions 1. (5 points) Determine the concentration of the weak acid in Part II. 2 Unknown # ______ (0.200174-0065-1)=0.26 (o -005L) 2. (5 points) Determine the pKa of the acid in Part II and compare it to the actual value. pKa Experimental Actual 3. M pKa = = 4.76 4.75 (2 points) Why is the EQP of the titration in Part II at a pH > 7? (one sentence) Because we're titrating there's resulting to going in a be a more pH > 7 weak OH acid , - meaning than Hzot , . 11 General Chemistry II, Spring 2021 4. (4 points) Determine the molar mass of the unknown diprotic acid. 1 Unknown # ______ ( 0.200 M )(O . 00757L ) -2 0-095392 7-57×10 5. - 4 not = 126 = 7-57×10-4 mot glmol (4 points) Determine the pKa values of the unknown diprotic acid. pKa 1=2.21 pka 2=3.84 6. (4 points) Using your results and Table 1, identify the diprotic acid. oxakc Aced 7. (2 points) During the titration in Part III, what was the primary species of your unknown diprotic acid at pH 5? Use your titration curve from Part III to answer this question. Hint: Figure 4 shows the primary species in solution at different points of a titration of H2SO3. C. 042 - 12 General Chemistry II, Spring 2021 8. In another lab, students were asked to titrate solutions of NH3(aq) with standardized HCl(aq) solutions. a. (2 points) Sketch the curve for this titration. ¥¥¥py pit Volume of HCl b. (2 points) Is the equivalence point at a pH = 7, > 7 or < 7? Explain (one sentence). egp of is a weak the conjugate acid because the > present is base solution in . c. (4 points) Explain how to determine the Kb of NH3 using the titration curve. By finding the of volume find by 2 where , Pkb can fand the 14 - pKa raise the , the point equivalence IS new nrcdpotnt volume ill at be pkatpkb use so , - divide the the pH= pKa = midpoint and pH To midpoint the From there pkg to Kb value , lopkb . . -44 . you to . 13