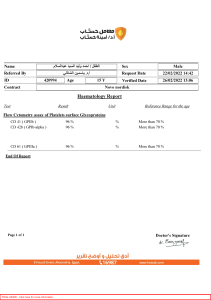
Energy Balance 1 • Most food processes require input of energy in one form or another • Energy takes many forms such as heat, kinetic energy, chemical energy, potential energy but, because of inter-conversions, it is not always easy to isolate separate constituents of energy balances. • However, under some circumstances certain aspects predominate. In many heat balances, other forms of energy are insignificant. • In the food industries, large proportion of energy leaving or entering the system is in the form of heat 2 • In some chemical situations, mechanical energy is insignificant and in some mechanical energy situations, as in the flow of fluids in pipes, the frictional losses appear as heat but the details of the heating need not be considered. • Energy balances can be calculated on the basis of external energy used per kilogram of product, or raw material processed, or on dry solids, or some key component. • In the SI system there is only one energy unit, the joule. However, kilocalories are still used by some nutritionists, and British thermal units (Btu) in some heat-balance work. 3 • The most common important energy form is heat energy and the conservation of this can be illustrated by considering operations such as heating and drying. • In these, enthalpy (total heat) is conserved and as with the mass balances so enthalpy balances can be written round the various items of equipment, or process stages, or round the whole plant, and it is assumed that no appreciable heat is converted to other forms of energy such as work. 4 Heat Balances Heat to surrounding Heat out in product. Heat from electricity Heat from fuel combustion Heat out in waste Heat stored Heat from mechanical sources Heat in raw materials Heat in from surrounding 5 Heat Balance Cont’d • Sensible heat……………….. • Latent heat ………………… • First law of thermodynamics; ΔE=Q-W 6 Heat Transfer Theory • Heat transfer: Dynamic process where heat is transferred spontaneously from one source to another cooler source • The rate of heat transfer depends upon temperature difference between the sources. • Increase in temperature difference increases driving force in heat transfer • Resistance affects rate of heat transfer • Rate of transfer = driving force/resistance 7 Heat Transfer Cont’d • Rate of heat transfer= ΔT / R • Where R= heat flow resistance of medium • Unsteady state heat transfer : Temperatures may change during processing and therefore the rate of heat transfer will change. • Steady state heat transfer: Temperature doesn’t change during processing. 8 Heat Transfer Cont’d Heat transfer can be transferred by: • Conduction • Convection • Radiation 9 • Conduction: The molecular energy is directly exchanged from the hotter to the cooler regions, the molecules with greater energy communicating some of this energy to neighbouring molecules with less energy. • Convection: The transfer of heat by the movement of groups of molecules in a fluid. The groups of molecules may be moved by either density changes or by forced motion of the fluid. • Radiation: The transfer of heat energy by electromagnetic waves, which transfer heat from one body to another. 10 Heat Conduction • Molecular energy is directly exchanged, from the hotter to the cooler regions. • Heat transfer of conduction in and fluids follows a basic equation known as Fourier’s law which states that heat transfer rate per unit area is proportional to the normal temperature gradient. • Heat flows from a hotter to a colder body, that is in the direction of the negative temperature gradient. • Thermal conductivity does change slightly with temperature, but in many applications it can be regarded as a constant for a given material. 11 Heat Conduction Cont’d • Generally, metals have a high thermal conductivity (50-400 J m-1 s-1 oC-1). • Most foodstuffs contain a high proportion of water. Water has a thermal conductivity of 0.7 J m-1 s-1 oC-1 above 0oC. • Thermal conductivities of foods are in the range 0.6-0.7 J m-1 s-1 oC-1. • Ice has a substantially higher thermal conductivity than water, about 2.3 Jm-1 s-1 oC-1. the thermal conductivity of frozen foods is therefore higher than foods at normal temperatures. 12 Heat Conduction Cont’d • Rate of heat transfer =driving force × conductance, where driving force is temperature gradient 𝑑𝑄 𝑑𝑇 = −k A 𝑑𝑡 𝑑𝑥 where, dQ/dt or q = the rate of heat transfer by conduction (W) A = the area of cross-section of heat flow path (m2) dT/dx = the temperature gradient k = constant = thermal conductivity of the medium (W/m °C) 13 Heat Conduction Cont’d • X =length (m) • T= Temperature (oC) Conduction through a slab; • 𝑑𝑄 𝑘𝐴∆𝑇 =𝑑𝑡 ∆𝑋 • 𝑑𝑄 𝑑𝑡 = q (constant) • q= • 𝑑𝑇 𝑑𝑥 = 𝑘𝐴 ∆𝑇 − ∆𝑋 𝑘𝐴 ∆𝑇 ∆𝑥 = 𝑇1−𝑇2 ; 𝑋 where x is thickness of the slab 14 • 𝑞=− 𝑘𝐴 (𝑇1−𝑇2) 𝑋 OR, q= - kA ∆T k = A ∆T x x 15 Example A cork slab 10 cm thick has one face at -12°C and the other face at 21 °C. If the mean thermal conductivity of cork in this temperature range is 0.042 J m-1 s-1 oC-1, what is the rate of heat transfer through 1 m2 of wall? 16 Solution T1 = 21 oC T2 = -12 oC DT = 33 oC A=1m2 K = 0.042 J m-1 s-1 oC-1 X= 0.1 m 0.042 q= × 1 × 33 0.1 q=13.9 J s-1 17 Heat Conductances • Heat conductance is the quantity of heat that will pass in unit time, through unit area of a specified thickness of material, under unit temperature difference. •C= 𝑘 𝑥 where, • C= heat conductance; x = thickness; k= thermal conductivity (Jm-1 s-1 oC-1) • Heat passes through several consecutive layers of different materials frequently in heat conduction. 18 q= 𝐴∆𝑇1𝑘1 𝑥1 = 𝐴∆𝑇2𝑘2 𝑥2 = 𝐴∆𝑇3𝑘3 𝑥3 Heat conductances in series At steady state, q= 𝑇1−𝑇2 𝑘1 𝐴1 = k2 ∆𝑋 𝑇2−𝑇3 A2 = ∆𝑋 (𝑇3−𝑇4) k3 A3 ∆𝑋 19 Heat Conductance conti… If the areas are the same, A1 =A2 =A3 =………=A ∆𝑥1 𝑞( ) 𝑘1𝐴1 = 𝑇1 − 𝑇2 ∆𝑥2 𝑞( ) 𝑘2 𝐴2 (1) (2) =𝑇2 − 𝑇3 ∆𝑥3 𝑞( )= 𝑘3𝐴3 (3) 𝑇3 − 𝑇4 (1) +(2) + (3) ∆𝑥1 𝑞( 𝑘1 𝐴1 𝑞= + ∆𝑋2 𝑘2 𝐴2 + 𝑇1−𝑇4 ∆𝑋3 ) 𝐾3𝐴3 ∆𝑋1 ∆𝑋2 ∆𝑋3 + + 𝑘1 𝐴1 𝐾2𝐴2 𝐾3𝐴3 = =𝑇1 − 𝑇4 𝑇1−𝑇4 𝑅1+𝑅2+𝑅3 20 Heat Conductance conti… 𝑘1 = conductance 𝑥1 𝑘2 = 𝑥2 𝑋1 𝐾1 of he material in the first layer , C1 conductance of the material in the second layer , C2 + 𝑋2 𝐾2 + 𝑋3 + 𝐾3 ….. = 1 𝐶1 + 1 1 + 𝐶2 𝐶3 1 = 𝑈 • Where U= overall conductance for combined layers (J m-2 s-1 oC-1) Therefore, So, 1 𝑈 𝐴∆𝑇 = 𝑞( ) 𝑞 = 𝑈𝐴∆𝑇 21 Example A cold store has a wall comprising of 11cm of brick on the outside, then 7.5cm of concrete and then 10cm of cork . The mean temperature within the store is maintained at -18°𝐶 and the mean temperature outside of the surface of the wall is 18°𝐶. Calculate the rate of heat transfer through the wall. The appropriate thermal conductivities are for the brick, concrete and cork respectively 0.69, 0.76, 0.043 Jm-1s-1 ℃. Determine also the temperature at the interfaces between the concrete and cork layers, and the brick and concrete layers. 22 Solution For brick 𝑥1 𝑘1 = 0.11 0.69 = 0.16 For concrete 𝑥2 = 0.075 = 0.10 𝑘2 For cork But 1/U 𝑥3 𝑘3 𝑥1 = 𝑘1 0.76 = + 0.10 = 0.043 𝑥2 𝑘2 + 2.33 𝑥3 𝑘3 =0.16 + 0.10 + 2.33 =2.59 23 U= 0.38 Jm−2s−1 oC-1 ∆T= 18−(−18) = 36 oC A= 1 m2 q = UA ∆ T =0.38 × 1 × 36 =13.7 Js−1 18/05/2022 24 A3∆T3k3 q= 𝑥3 13.7 = 1 × ∆T3 × 1/2.33 ∆T3 = 32 oC ∆t3 = difference between temp. of cork /concrete surface (Tc) and temp. of cork surface inside the cold store. Tc− (−18) = 32 Tc= 14 oC If ∆T1= difference between the temp. of the brick/ concrete surface (Tb), and the temp. of the external air. Then, 13.7 = 1 × ∆T1 × 1/0.16 = 6.25 ∆T1 25 18− Tb = T1 =13.7/ 6.25 = 2.2 Tb = 15.8 oC 18/05/2022 26 Heat Conductance in Parallel • In heat conductances in parallel, the surfaces of the material are parallel to the direction of heat transfer and to each other. • Heat passes through each material at the same time. qa= 𝐴𝑎∆𝑇𝑘𝑎 𝑥 qb= 𝐴𝑏∆𝑇𝑘𝑏 𝑥 qc= 𝐴𝑐∆𝑇𝑘𝑐 𝑥 27 Example The wall of a bakery oven is built of insulating brick 10 cm thick and thermal conductivity 0.22 J m-1 s-1 °C-1. Steel reinforcing members penetrate the brick, and their total area of cross-section represents 1% of the inside wall area of the oven. If the thermal conductivity of the steel is 45 J m-1 s-1 °C-1 calculate (a) the relative proportions of the total heat transferred through the wall by the brick and by the steel (b) the heat loss for each m2 of oven wall if the inner side of the wall is at 230 °C and the outer side is at 25 °C. 28 Solution (a) Considering the loss through an area of 1 m2 of wall (0.99 m2 of brick, and 0.01 m2 of steel) A ∆T kb For brick; qb = b x 0.99 230−25 0.22 = = 446 J s-1 0.10 For steel; qs = As ∆T ks x 0.01 230−25 = 0.10 45 = 923 J s-1 Therefore, qb/ qs = 0.48 29 (b) Total heat loss q= (qb + qs) per m2 of wall q= 446 + 923 q= 1369 J s−1 Percentage of heat carried by steel = 923 1369 × 100 = 67% 30 Hollow Cylinder • Most process materials are transported through hollow cylinder as pipes that may or may not be insulated. • Let the internal or inner radius be r where the temperature is T1 and outer radius r2 with temperature T2. The length of the pipe segment is L and heat flows radially from the inner surface to the outside. 𝑞 𝑑𝑇 = −𝑘 𝐴 𝑑𝑟 • For a cylinder A = 2𝜋𝑟𝐿 • 𝑞 2𝜋𝑟𝐿 = 18/05/2022 𝑑𝑇 −𝑘 𝑑𝑟 31 = 𝑞 = 2𝜋𝑙 ln = 𝑞 2𝜋𝐿 = 𝑞 2𝜋𝐿 = -k (T2 –T1) ln 𝑟2 − ln 𝑟1 = -k (T2 –T1) 𝑟 𝑟1 ln 2= k (T1 –T2) q= k. q=( 𝑟21 𝑟 2𝜋𝑙 ln 𝑟2 𝑟1 (T1 –T2) 𝑇1 −𝑇2 ) 𝑅 𝑟 R= ln 𝑟2 1 2𝜋𝑙𝑘 32 Multilayer Cylinder 33 At steady state; 𝑇 −𝑇2 q= (𝑟 −𝑟 1) 2 1 𝑘𝐴 where AA𝑙𝑚 = 𝑇 −𝑇3 AA = (𝑟 −𝑟 2) 3 2 A 𝐾𝐵 𝑙𝑚 𝑇 −𝑇4 = (𝑟 −𝑟 3) 4 𝐵𝑙𝑚 3 𝑘𝐶𝐴𝐶𝑙𝑚 𝐴2−𝐴1 ln 𝐴2 𝐴 1 A𝐵𝑙𝑚 = 𝐴3−𝐴2 ln(𝐴3 𝐴2 𝐴𝐶𝑙𝑚 = ) 𝐴4−𝐴3 ln(𝐴4 𝐴3) 34 𝑇1−𝑇4 q= (𝛾2−𝛾1) (𝛾3−𝛾2) (𝛾4−𝛾3) + + 𝑘 𝐴 𝑙𝑚 𝑘 AA 𝐾 A 𝐴 = 𝑙𝑚 𝐵 𝐵𝑙𝑚 𝐶 𝐶 𝑇1−𝑇4 𝑅𝐴+𝑅𝐵+𝑅𝐶 35 Example A thick walled cylindrical tubing material, having an inside radius of 5 mm and an outside radius of 20 mm is being used as a cooling coil in a bath. Iced waters is flowing rapidly inside it, and the inside wall temperature is 274.9 K. The outside wall temperature is 297.1 K. A total of 14.65 W must be removed from the bath by the cooling coil, how many meters of tubing are needed. [k = 0.151 W/mk]. 36 Solution Find the heat flow rate for 1m length of tubing r1 = 5 mm = 0.005m r2= 20 mm= 0.02m A1 = 2𝜋𝑟1𝐿 = 2𝜋 0.005 1.0 = 0.0314 m2 A2 = 2𝜋𝑟2𝐿 = 2𝜋(0.02)(1.0) = 0.1257 m2 q= 𝑘𝐴𝑙𝑚 𝐴𝑙𝑚 = 𝑇1−𝑇2 𝑟2−𝑟1 0.1257−0.0314 01.257 ln( 0.0314) 37 𝐴𝑙𝑚 = 0.0943 1.387 = 0.0688 q= 0.151 × 0.0688 274.9−297.1 0.02−0.005 q= -15.2 W (-ve means the heat is from r2 to r1) 1.52 1m 14.65 14.65 15.2 1 15.2 × 14.65𝑚 = 0.964m L= 0.964m 38 Trial Question A hollow cylinder has the inner radius, r1, and outer radius, r0. Inner and outside temperatures T1 and T0 respectively, made of material whose thermal conductivity is k for the length l. Derive the expression for the conducted heat loss in Watts. If r1 = 7.5cm, r0 = 4.5cm. k = 52 W/mk, where heat conducted out from the cylinder of length 1m is 465 W. If the inside temperature is 400 oC determine the outside temperature. 39 Example A thick walled tube of stainless steel (A) having a k=21.63 W/mk, with dimensions of 0.0254m ID and 0.058m OD is covered with a 0.0254m layer of (B), insulation of k=0.2423 W/mK The inside wall temperature of the steel is 811K while the outside temperature of the insulation is 310.8 K, for 0.30m length pipe, calculate the heat loss and also the temperature at the interface between the metal and the insulation. 18/05/2022 40 Solution T1 811k, T2 interface temprature, T3 310.8k 18/05/2022 41 0.0254 r1= 2 =0.0127m 0.0508 r2= 2 = 0.02254m r3 = r2 + thicknesss of insulator= 0.20254 + 0.20254 = 0.0508m For l = 0.305m A1=2𝜋𝑟1𝑙= 2𝜋 0.0127 (0.305) =0.0243𝑚2 A2= 2𝜋𝑟2𝑙 = 2𝜋 0.0254 (0.305) =0.0487𝑚2 18/05/2022 42 A3 = 2𝜋𝑟3𝑙 = 2𝜋 0.0508 (0.305) = 0.0974𝑚2 A A/M = 𝐴2−𝐴1 𝐴 ln𝐴2 1 = 0.0487−0.0243 0.0487 ln0.0243 = 0.0351m2 A B/M = 𝐴3−𝐴2 ln 𝐴3 𝐴2 = 0.0974−0.0487 0.0974 ln0.0487 = 0.073m2 18/05/2022 43 𝑅𝐴 = 𝑟2−𝑟1 𝑘𝐴 AA𝑙𝑚 𝑅𝐵 = 𝑟3−𝑟2 𝑘B AB𝑙𝑚 = 0.0127 (0.2423)(0.0703) = 0.01673K/W = 0.0254 (0.2423)(0.0703) =1.491K/W The heat transfer rate q= q= 𝑇1−𝑇3 𝑅𝐴+𝑅𝐵 = 811−310.8 =331.7W 0.01673+1.491 𝑇1−𝑇2 𝑅𝐴 T2= 𝑞 𝑅𝐴 - T1 T2= 805.5K 18/05/2022 44 Hollow sphere For a hollow sphere, A = 4𝜋𝑟2 From the Fourier’s law 𝑞 𝑑𝑇 = −𝑘 𝐴 𝑑𝑟 𝑞 𝑑𝑇 = −𝐾 2 4𝜋𝑟 𝑑𝑟 𝑞 𝑟2 𝑑𝑟 = 4𝜋 𝑟1 𝑟2 𝑞 4𝜋 − 𝑞 1 4𝜋 𝑟1 1 𝑟 -k 𝑇2 𝑑𝑇 𝑇1 =k(T1-T2) 1 − 𝑟2 =k (T1-T2) 4𝜋𝑘(𝑇1 − 𝑇2) 𝑞= 1 1 − 𝑟1 𝑟2 45 (𝑇1 − 𝑇2) = 𝑘(4𝜋𝑟1𝑟2) (𝑟2 − 𝑟1) = 𝑇1−𝑇2 𝑟2−𝑟1 4𝜋𝑟1𝑟2𝑘 46 Trial Question A hollow sphere contains 100W electrical filament and the following data are known. The internal radius equals 24cm, external =32cm, k=4.16 W/mk. Assuming steady state, determine the internal temperature if the outside temperature is 22oC. 47 Trial Question A hollow steel sphere contain 100W electrical filament, and the following data re known: Internal radius = 24cm External radius = 32cm Inner surface film confident of heat = 30 W/m2K Outer surface film coefficient of heat = 10 W/m2-K Neglecting the thermal resistance of the metal and assuming steady conditions. Determine the internal temperature if the outside temperature is 22oC. Neglecting the thermal resistance of the metal and assuming steady conditions. Determine the internal temperature if the outside temperature is 22℃ 18/05/2022 48 Convective Heat Transfer Coefficient The rate of heat transfers from a fluid to a solid surface or vice versa is usually expressed as q = h A(Tw – Tr) q = heat transfer rate A= Area of heat transfer (m2) TW = Temperature of solid wall surface Tr = Average of bulk temperature of the fluid (k) h = convective heat transfer coefficient (W/m2k) The coefficient h is a function of many variable including system geometry, fluid properties, and flow velocity and temperature differences. It is often predicted by empirical relations. h is also called the film coefficient 18/05/2022 49 𝑞 = ℎ𝐴(𝑇𝑊 − 𝑇𝑓) = q= (𝑇𝑊−𝑇𝑓) 1 ℎ𝐴 𝑇1−𝑇4 1 ∆𝑋 1 + + ℎ𝑖𝐴 𝑘𝐴 ℎ𝑜𝐴 50 Trial Question A flat composite plate is made of two layers of aluminum and steel of 5cm and 2cm thick respectively. The aluminum side is in contact with a fluid at 200oC. The heat transfer coefficient of liquid film being 14 W/m2K. The cold surface of steel side is in contact with liquid at 25oC. The heat transfer coefficient being 29 W/m2K Determine (i) heat transfer rate from the hot liquid to the cold liquid, through a surface of interface temperatures (ii) Thermal conductivities of Aluminum and steel are 205 and 45 (W/mK) respectively 18/05/2022 51 SOLUTION (i) Roverall = = 1 14𝑥10 q= 1 ℎ𝑖𝐴 + ∆𝑋𝐴 𝐾𝐴𝐴 + ∆𝑋𝐵 𝐾𝐵𝐴 0.05 205𝑥10 + 0.02 45𝑥10 + 𝑇𝑖 −𝑇0 𝑅𝑜𝑣𝑒𝑟𝑎𝑙𝑙 18/05/2022 = 200−25 0.01066 + 1 ℎ𝑜𝐴 + 1 29𝑥10 =0.01066k/W = 16416.5 = 200−𝑇1 1 14𝑥10 52 (16416.5 x 1 ) 14𝑋10 =200-T1 T1= ? (solve) (ii) q= 𝑇𝑖 −𝑇1 1 ℎ𝐴𝑖 = 𝑇1−𝑇2 ∆𝑋𝐴 𝐾𝐴𝐴𝐴 = 𝑇2−𝑇3 ∆XB/KBAB 𝑇 −𝑇0 = 13 ℎ0𝐴0 calculate for T2 and T3 temperature 18/05/2022 53 Example A cold storage room is constructed of an inner layer of 12.7mm of pine, a middle layer of 101.6mm of cork board and an outer layer of 76.2mm of concrete. The wall surface temperature is (255.4K inside the cold room and 297.1 K at the surface of the concrete. Calculate the heat loss in W for 1 m2 and the temperature at the interface between the wood and cork board. Thermal conductivities :pine – 0.151 W/mk cork – 0.0433 W/mk concrete - 0.762 W/mk 18/05/2022 54 SOLUTION ∆𝑋𝐴 = 0.0127𝑚 ∆𝑋𝐵 = 0001016𝑚 ∆𝑋𝐶 = 0.0762𝑚 18/05/2022 55 ∆𝑋𝐴 RA= 𝑘𝐴𝐴 = 0.0127 (0.151)(1.0) ∆𝑋𝐵 RB= 𝑘𝐵𝐴 = 0.1016 (0.0433)(1.0) ∆𝑋𝐶 𝑘 𝐶𝐴 = 0.0762 (0.762)(1.0) RC= q= 𝑇1−𝑇4 𝑅𝐴+𝑅𝐵+𝑅𝐶 = =0.0841K/W = 2.346K/W = 0.100K/W 255.4−297.1 0.0841+2.346+0.100 = −41.7 2.530 = -16.49W (-ve means heat flow is from outside). To calculate the temperature T2 at the interface between the pine and cork: 18/05/2022 56 q= 𝑇1−𝑇2 𝑅𝐴 -16.48= 255.4−𝑇2 0.0841 T2= 256.79K Calculate T3 18/05/2022 57 EXAMPLE A cold room has one of the walls 5m x 2.5m made of bricks 12cm thick insulated externally by cork slabbing 8cm thick. Cork is protected externally by 2.5cm wood. Estimate the heat infiltration through the wall in 24 hours if the interior of the cold room is maintained at a temperature of 0 oC and the outside temperature is 20 oC. Thermal conductivities for brick, cork and wood are 0.93, 0.044 and 0.175 W/m.k respectively. What will be the interface temperatures? 18/05/2022 58 Solution RA = ∆𝑋𝐴 𝐾𝐴𝐴 A = 5 X 25 = 12.5m2 KA = 0.93 18/05/2022 KB = 0.044 KC = 0.175W/mk 59 ∆𝑋𝐴 = 0.12𝑚 ∆𝑋𝐵 = 0.08𝑚 ∆𝑋𝐶 = 0.025𝑚 0.12 RA = X 12.5 = 0.01K/W 0.93 0.08 RB = X 12.5 = 0145K/W 0.044 0.025 RC= X 0.175 12.5=0.01143K/W RTOTAL = 0.01 + 0.145 + 0.01143 = 0.16643 K/W 18/05/2022 60 𝑇1−𝑇2 q= 𝑅𝑇𝑂𝑇𝐴𝐿 20−0 = = 0.16643 q 120.17W 120.17 𝐽 3600 𝑠 = . 𝑠 ℎ 𝐽 𝑠 𝑠 ℎ = 120.17 .3600 𝐽 = 120.17𝑥 3600 ℎ If 1 hr = 120.17 x 3600 J 24 hrs = 120.17 𝑥 3600 𝑥24 𝐽 24ℎ𝑟𝑠 103820. 𝑘𝐽 = 24ℎ𝑟𝑠 18/05/2022 61 Heat transfer through each layer is the same 𝑇1−𝑇2 = 𝑅𝐶 = 𝑇3−𝑇4 𝑅𝐴 20−𝑡2 0.01143 q= 120.17 = = t2= 20 – (120.17)(0.01143) = 18.63 oC Also, 120.17 = 𝑡3−𝑡4 0.01 =0 t3 = 1.2 oC (t4 - 0) 18/05/2022 62 Question A composite insulating wall has three layers of material held together by 3cm diameter aluminium rivet per 0.1m2 surface. The layer of materials composite are 10cm thick brick with hot surface at 200oC, 1cm thick timber with cold surface at 10oC. These two layers are interposed by a third layer of insulating wall 25cm thick. The conductivities of walls are; K(brick) = 0.95 w/m.k k (wood) = 0.175 w/mk k(insulation) = 0.115w/mk k(aluminium) =200w/mk If the heat transfer is assumed to take place only in one direction normal to the layers, determine the percentage increase in heat transfer rate due to rivets. 18/05/2022 63 Trial question A flat composite plate made of two layers of aluminium and steel of 5cm and 2cm thickness respectively. The thermal conductivities are 205 and 45 W/mk for aluminium and steel respectively. Aluminium side is in contact with hot liquid at 200oC. The heat transfer coefficient of liquid film being 14 W/mk . The cold surface of steel side is in contact with liquid at 25 oC. The heat transfer coefficient of liquid film being 29 W/m oK. Determine the : (i) heat transfer rate from hot liquid to cold liquid through a surface of 10m2 (ii) overall heat transfer coefficient (iii) interface temperatures. 18/05/2022 64 Solution ti t1 t2 t3 to q 𝑅 𝑜𝑣𝑒𝑟𝑎𝑙𝑙 𝑅𝑜𝑣𝑒𝑟𝑎𝑙𝑙 18/05/2022 1 ∆𝑋1 ∆𝑋2 1 = + + + ℎ𝑖𝐴 𝐾1𝐴 𝐾2𝐴 ℎ𝑂𝐴 1 0.05 0.02 1 = + + + 14 𝑥10 205 𝑥10 45 𝑥10 29 𝑥10 65 𝑡𝑖−𝑡𝑜 𝑅𝑜𝑣𝑒𝑟𝑎𝑙𝑙 (i) q= = 200−25 0.01066 = 16416.5𝑊 = 16.4165𝑘𝑊 ii. Q = 𝑈𝐴∆𝑇 iii. U = 𝑄 𝐴∆𝑇 Or U = = 16416.5 10 𝑋 175 1 1 ∆𝑋1 ∆𝑋2 1 = 0.1066 + + 𝑘 +ℎ ℎ𝑖 𝑘1 2 𝑜 18/05/2022 1 = 9.38 W/m2 = 9.38 𝑊/𝑂𝐾 66



![The Power of NOW[3741]](http://s2.studylib.net/store/data/025690828_1-687eebfb243c6979cdc2ac319fc077fd-300x300.png)